In Vitro Assessment of Migratory Behavior of Two Cell Populations in a Simple Multichannel Microdevice
Abstract
:1. Introduction
2. Experimental Section
2.1. Cell Isolation and Culture
2.2. Device Fabrication
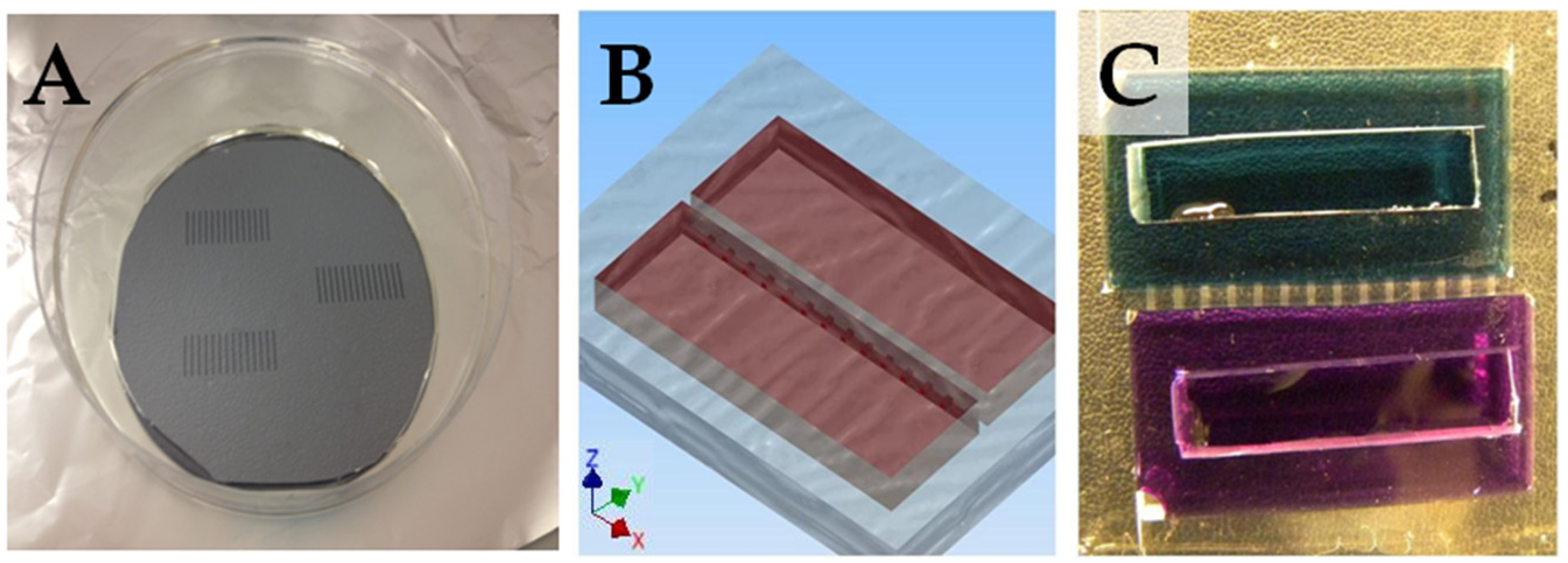
2.3. Loading the Cell Migration Device
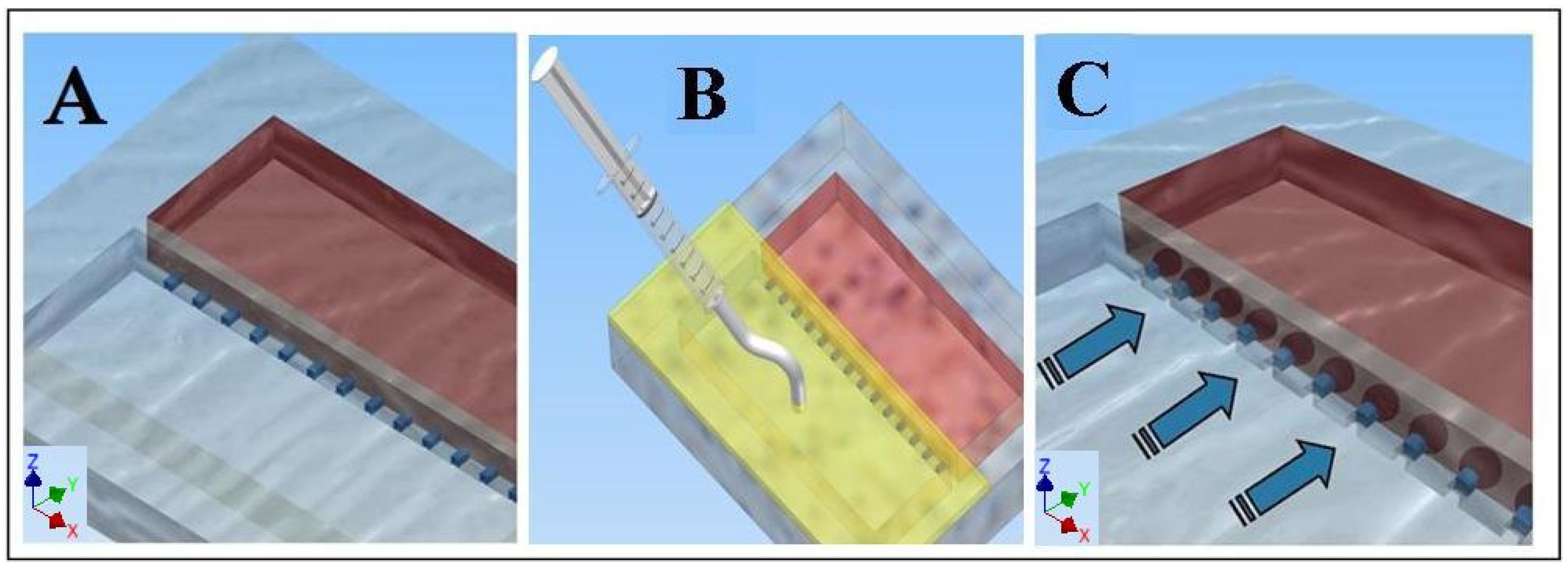
2.4. Assessing Cell Migration
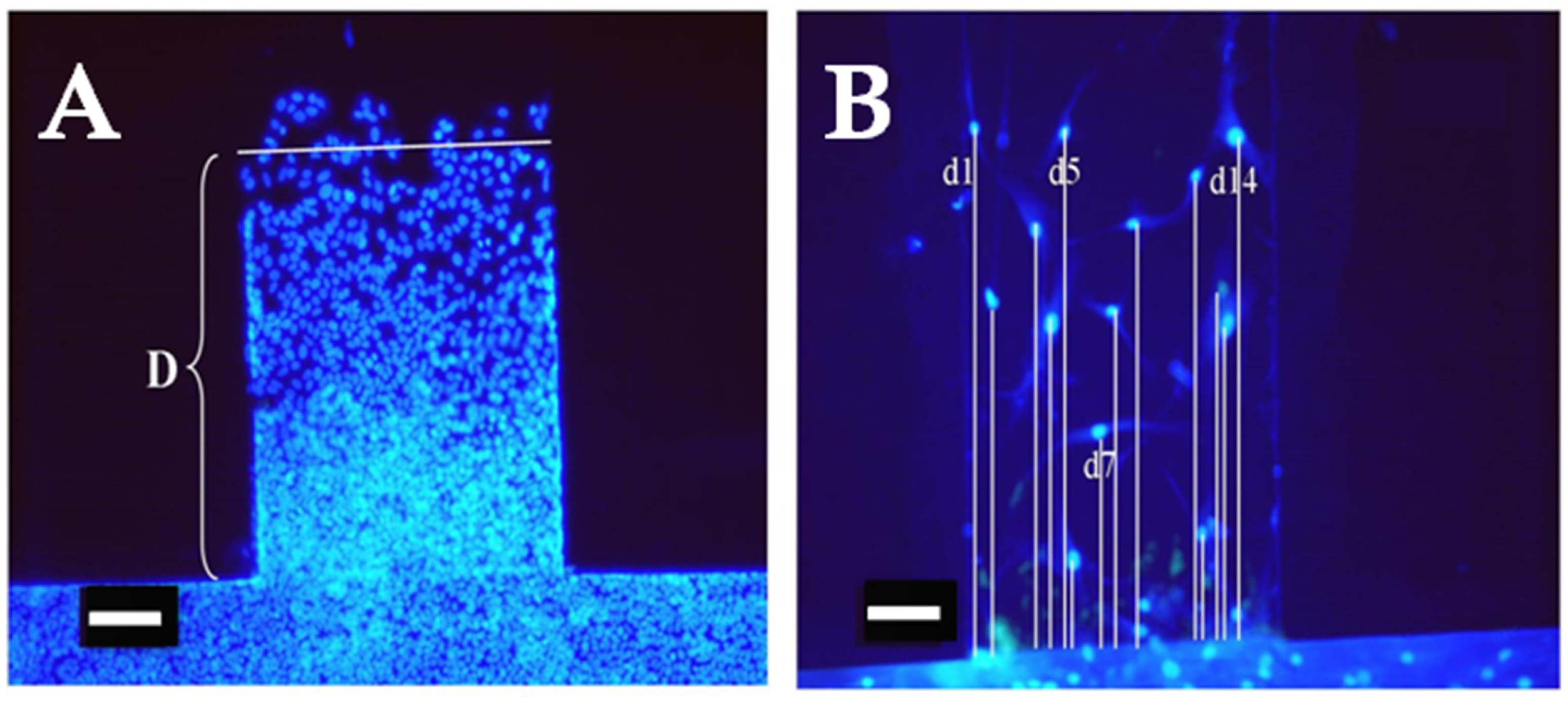
2.5. Cell Migration Study Design
3. Results and Discussion
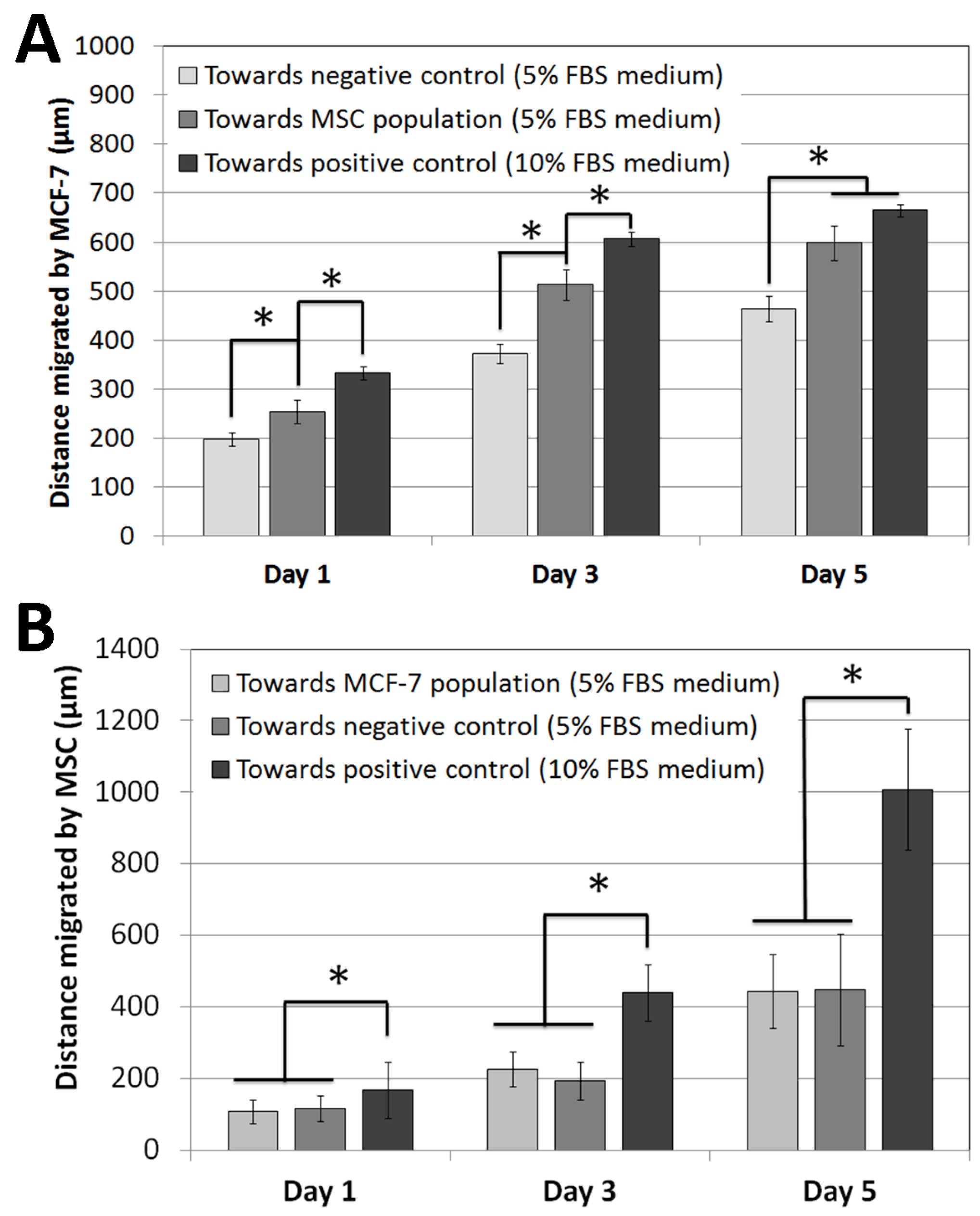
3.1. Cell Migration Experiments
4. Conclusions
Conflicts of Interest
References
- Lima, F.R.; Kahn, S.A.; Soletti, R.C.; Biasoli, D.; Alves, T.; da Fonseca, A.C.C.; Garcia, C.; Romão, L.; Brito, J.; Holanda-Afonso, R.; et al. Glioblastoma: Therapeutic challenges, What lies ahead. Biochim. Biophys. Acta 2012, 1826, 338–349. [Google Scholar]
- Huang, T.T.; Sarkaria, S.M.; Cloughesy, T.F.; Mischel, P.S. Targeted therapy for malignant glioma patients: Lessons learned and the road ahead. Neurotherapeutics 2009, 6, 500–512. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef]
- English, K.; French, A.; Wood, K.J. Mesenchymal stromal cells: Facilitators of successful transplantation? Cell Stem Cell 2010, 7, 431–442. [Google Scholar] [CrossRef]
- Si, Y.L.; Zhao, Y.L.; Hao, H.J.; Fu, X.B.; Han, W.D. MSCs: Biological characteristics, clinical applications and their outstanding concerns. Ageing Res. Rev. 2011, 10, 93–103. [Google Scholar] [CrossRef]
- Herzog, E.L.; van Arnam, J.; Hu, B.; Zhang, J.; Chen, Q.; Haberman, A.M.; Krause, D.S. Lung-specific nuclear reprogramming is accompanied by heterokaryon formation and Y chromosome loss following bone marrow transplantation and secondary inflammation. FASEB J. 2007, 21, 2592–2601. [Google Scholar] [CrossRef]
- Weiss, D.J.; Bertoncello, I.; Borok, Z.; Kim, C.; Panoskaltsis-Mortari, A.; Reynolds, S.; Rojas, M.; Stripp, B.; Warburton, D.; Prockop, D.J. Stem cells and cell therapies in lung biology and lung diseases. Proc. Am. Thorac. Soc. 2011, 8, 223–272. [Google Scholar]
- Albera, C.; Polak, J.; Janes, S.; Griffiths, M.; Alison, M.; Wright, N.; Navaratnarasah, S.; Poulsom, R.; Jeffery, R.; Fisher, C.; et al. Repopulation of human pulmonary epithelium by bone marrow cells: A potential means to promote repair. Tissue Eng. 2005, 11, 1115–1121. [Google Scholar] [CrossRef]
- Barbash, I.M.; Chouraqui, P.; Baron, J.; Feinberg, M.S.; Etzion, S.; Tessone, A.; Miller, L.; Guetta, E.; Zipori, D.; Kedes, L.H.; et al. Systemic delivery of bone marrow—Derived mesenchymal stem cells to the infarcted myocardium. Circulation 2003, 108, 863–868. [Google Scholar] [CrossRef]
- Lee, R.H.; Seo, M.J.; Reger, R.L.; Spees, J.L.; Pulin, A.A.; Olson, S.D.; Prockop, D.J. Multipotent stromal cells from human marrow home to and promote repair of pancreatic islets and renal glomeruli in diabetic NOD/scid mice. Proc. Natl. Acad. Sci. USA 2006, 103, 17438–17443. [Google Scholar] [CrossRef]
- Hara, M.; Murakami, T.; Kobayashi, E. In vivo bioimaging using photogenic rats: Fate of injected bone marrow-derived mesenchymal stromal cells. J. Autoimmun. 2008, 30, 163–171. [Google Scholar] [CrossRef]
- Chapel, A.; Bertho, J.M.; Bensidhoum, M.; Fouillard, L.; Young, R.G.; Frick, J.; Demarquay, C.; Cuvelier, F.; Mathieu, E.; Trompier, F.; et al. Mesenchymal stem cells home to injured tissues when co-infused with hematopoietic cells to treat a radiation-induced multi-organ failure syndrome. J. Gene Med. 2003, 5, 1028–1038. [Google Scholar] [CrossRef]
- Plytycz, B.; Seljelid, R. From inflammation to sickness: Historical perspective. Arch. Immunol. Ther. Exp. (Warsz) 2003, 51, 105–109. [Google Scholar]
- Houghton, J.M.; Morozov, A.; Smirnova, I.; Wang, T.C. Stem cells and cancer. Semin. Cancer Biol. 2007, 17, 191–203. [Google Scholar] [CrossRef]
- Loebinger, M.R.; Eddaoudi, A.; Davies, D.; Janes, S.M. Mesenchymal stem cell delivery of TRAIL can eliminate metastatic cancer. Cancer Res. 2009, 69, 4134–4142. [Google Scholar]
- Kanehira, M.; Xin, H.; Hoshino, K.; Maemondo, M.; Mizuguchi, H.; Hayakawa, T.; Matsumoto, K.; Nakamura, T.; Nukiwa, T.; Saijo, Y. Targeted delivery of NK4 to multiple lung tumors by bone marrow-derived mesenchymal stem cells. Cancer Gene Ther. 2007, 14, 894–903. [Google Scholar] [CrossRef]
- Chen, X.; Lin, X.; Zhao, J.; Shi, W.; Zhang, H.; Wang, Y.; Kan, B.; Du, L.; Wang, B.; Wei, Y.; et al. A tumor-selective biotherapy with prolonged impact on established metastases based on cytokine gene-engineered MSCs. Mol. Ther. 2008, 16, 749–756. [Google Scholar] [CrossRef]
- Ren, C.; Kumar, S.; Chanda, D.; Chen, J.; Mountz, J.D.; Ponnazhagan, S. Therapeutic Potential of Mesenchymal Stem Cells Producing Interferon-α in a Mouse Melanoma Lung Metastasis Model. Stem Cells 2008, 26, 2332–2338. [Google Scholar] [CrossRef]
- Tang, C.; Russell, P.J.; Martiniello-Wilks, R.; Rasko, J.E.; Khatri, A. Concise review: Nanoparticles and cellular carriers-allies in cancer imaging and cellular gene therapy? Stem Cells 2010, 28, 1686–1702. [Google Scholar] [CrossRef]
- Bacus, S.S.; Kiguchi, K.; Chin, D.; King, C.R.; Huberman, E. Differentiation of cultured human breast cancer cells (AU-565 and MCF-7) associated with loss of cell surface HER-2/neu antigen. Mol. Carcinogen. 1990, 3, 350–362. [Google Scholar] [CrossRef]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 2007, 449, 557–563. [Google Scholar] [CrossRef]
- Chung, B.G.; Choo, J. Microfluidic gradient platforms for controlling cellular behavior. Electrophoresis 2010, 31, 3014–3027. [Google Scholar] [CrossRef]
- Kabiri, M.; Kul, B.; Lott, W.; Futrega, K.; Ghanavia, P.; Upton, Z.; Doran, M. 3D mesenchymal stem/stromal cell osteogenesis and autocrine signalling. Biochem. Biophys. Res. Commun. 2012, 419, 142–147. [Google Scholar] [CrossRef]
- Markway, B.D.; Tan, G.K.; Brooke, G.; Hudson, J.E.; Cooper-White, J.J.; Doran, M.R. Enhanced chondrogenic differentiation of human bone marrow-derived mesenchymal stem cells in low oxygen environment micropellet cultures. Cell Transplant. 2010, 19, 29–42. [Google Scholar] [CrossRef]
- Doran, M.R.; Mills, R.J.; Parker, A.J.; Landman, K.A.; Cooper-White, J.J. A cell migration device that maintains a defined surface with no cellular damage during wound edge generation. Lab Chip. 2009, 9, 2364–2369. [Google Scholar] [CrossRef]
- Bodas, D.; Khan-Malek, C. Hydrophilization and hydrophobic recovery of PDMS by oxygen plasma and chemical treatment—An SEM investigation. Sens. Actuators B 2007, 123, 368–373. [Google Scholar] [CrossRef]
- Egeblad, M.; Jaattela, J. Cell death induced by TNF or serum starvation is independent of ErbB receptor signaling in MCF-7 breast carcinoma cells. Int. J. Cancer 2000, 86, 617–625. [Google Scholar] [CrossRef]
- Chen, S.-H.; Hung, W.-C.; Wang, P.; Paul, C.; Konstantopoulos, K. Mesothelin Binding to CA125/MUC16 Promotes Pancreatic Cancer Cell Motility and Invasion via MMP-7 Activation. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef]
- Irimia, D.; Toner, M. Spontaneous migration of cancer cells under conditions of mechanical confinement. Integrat. Biol. 2009, 1, 506–512. [Google Scholar] [CrossRef]
- Sordi, V.; Malosio, M.L.; Marchesi, F.; Mercalli, A.; Melzi, R.; Giordano, T.; Belmonte, N.; Ferrari, G.; Leone, B.E.; Bertuzzi, F.; et al. Bone marrow mesenchymal stem cells express a restricted set of functionally active chemokine receptors capable of promoting migration to pancreatic islets. Blood 2005, 106, 419–427. [Google Scholar] [CrossRef]
- Ryu, C.H.; Park, S.A.; Kim, S.M.; Lim, J.Y.; Jeong, C.H.; Jun, J.A.; Oh, J.H.; Park, S.H.; Oh, W.I.; Jeun, S.S. Migration of human umbilical cord blood mesenchymal stem cells mediated by stromal cell-derived factor-1/CXCR4 axis via Akt, ERK, and p38 signal transduction pathways. Biochem. Biophys. Res. Commun. 2010, 398, 105–110. [Google Scholar] [CrossRef]
- Bachelder, R.E.; Wendt, M.A.; Mercurio, A.M. Vascular endothelial growth factor promotes breast carcinoma invasion in an autocrine manner by regulating the chemokine receptor CXCR4. Cancer Res. 2002, 62, 7203–7206. [Google Scholar]
- Lee, B.C.; Lee, T.H.; Avraham, S.; Avraham, H.K. Involvement of the Chemokine Receptor CXCR4 and Its Ligand Stromal Cell-Derived Factor 1α in Breast Cancer Cell Migration Through Human Brain Microvascular Endothelial Cells. Mol. Cancer Res. 2004, 2, 327–338. [Google Scholar]
- Hall, J.M.; Korach, K.S. Stromal cell-derived factor 1, a novel target of estrogen receptor action, mediates the mitogenic effects of estradiol in ovarian and breast cancer cells. Mol. Endocrinol. 2003, 17, 792–803. [Google Scholar] [CrossRef]
- Müller, A.; Homey, B.; Soto, H.; Ge, N.; Catron, D.; Buchanan, M.E.; McClanahan, T.; Murphy, E.; Yuan, W.; Wagner, S.N.; et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001, 410, 50–56. [Google Scholar] [CrossRef]
- Spaeth, E.L.; Dembinski, J.L.; Sasser, A.K.; Watson, K.; Klopp, A.; Hall, B.; Andreeff, M.; Marini, F. Mesenchymal stem cell transition to tumor-associated fibroblasts contributes to fibrovascular network expansion and tumor progression. PLoS One 2009, 4, e4992. [Google Scholar] [CrossRef]
- Stoicov, C.; Li, H.; Carlson, J.; Houghton, J.M. Bone marrow cells as the origin of stomach cancer. Future Oncol. 2005, 1, 851–862. [Google Scholar] [CrossRef]
- Corsten, M.F.; Shah, K. Therapeutic stem-cells for cancer treatment: Hopes and hurdles in tactical warfare. Lancet Oncol. 2008, 9, 376–384. [Google Scholar] [CrossRef]
- Hall, B.; Dembinski, J.; Sasser, A.K.; Studeny, M.; Andreeff, M.; Marini, F. Mesenchymal stem cells in cancer: Tumor-associated fibroblasts and cell-based delivery vehicles. Int. J. Hematol. 2007, 86, 8–16. [Google Scholar] [CrossRef]
- Kim, S.M.; Kim, D.S.; Jeong, C.H.; Kim, D.H.; Kim, J.H.; Jeon, H.B.; Kwon, S.J.; Jeun, S.S.; Yang, Y.S.; Oh, W.; et al. CXC chemokine receptor 1 enhances the ability of human umbilical cord blood-derived mesenchymal stem cells to migrate toward gliomas. Biochem. Biophys. Res. Commun. 2011, 407, 741–746. [Google Scholar] [CrossRef]
- Gehmert, S.; Prantl, L.; Vykoukal, J.; Alt, E.; Song, Y.H. Breast cancer cells attract the migration of adipose tissue-derived stem cells via the PDGF-BB/PDGFR-β signaling pathway. Biochem. Biophys. Res. Commun. 2010, 398, 601–605. [Google Scholar] [CrossRef]
- Ben-Baruch, A. The tumor-promoting flow of cells into, within and out of the tumor site: Regulation by the inflammatory axis of TNFα and chemokines. Cancer Microenviron. 2012, 1–14. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kabiri, M.; Lott, W.B.; Kabiri, E.; Russell, P.J.; Doran, M.R. In Vitro Assessment of Migratory Behavior of Two Cell Populations in a Simple Multichannel Microdevice. Processes 2013, 1, 349-359. https://doi.org/10.3390/pr1030349
Kabiri M, Lott WB, Kabiri E, Russell PJ, Doran MR. In Vitro Assessment of Migratory Behavior of Two Cell Populations in a Simple Multichannel Microdevice. Processes. 2013; 1(3):349-359. https://doi.org/10.3390/pr1030349
Chicago/Turabian StyleKabiri, Mahboubeh, William B. Lott, Ehsan Kabiri, Pamela J. Russell, and Michael R. Doran. 2013. "In Vitro Assessment of Migratory Behavior of Two Cell Populations in a Simple Multichannel Microdevice" Processes 1, no. 3: 349-359. https://doi.org/10.3390/pr1030349
APA StyleKabiri, M., Lott, W. B., Kabiri, E., Russell, P. J., & Doran, M. R. (2013). In Vitro Assessment of Migratory Behavior of Two Cell Populations in a Simple Multichannel Microdevice. Processes, 1(3), 349-359. https://doi.org/10.3390/pr1030349




