A Novel Micro Pressurized Liquid Extraction Method for Rapid Sample Preparation of Polycyclic Aromatic Hydrocarbons in Various Solids
Abstract
:1. Introduction
2. Experimental Section
2.1. μPLE Apparatus
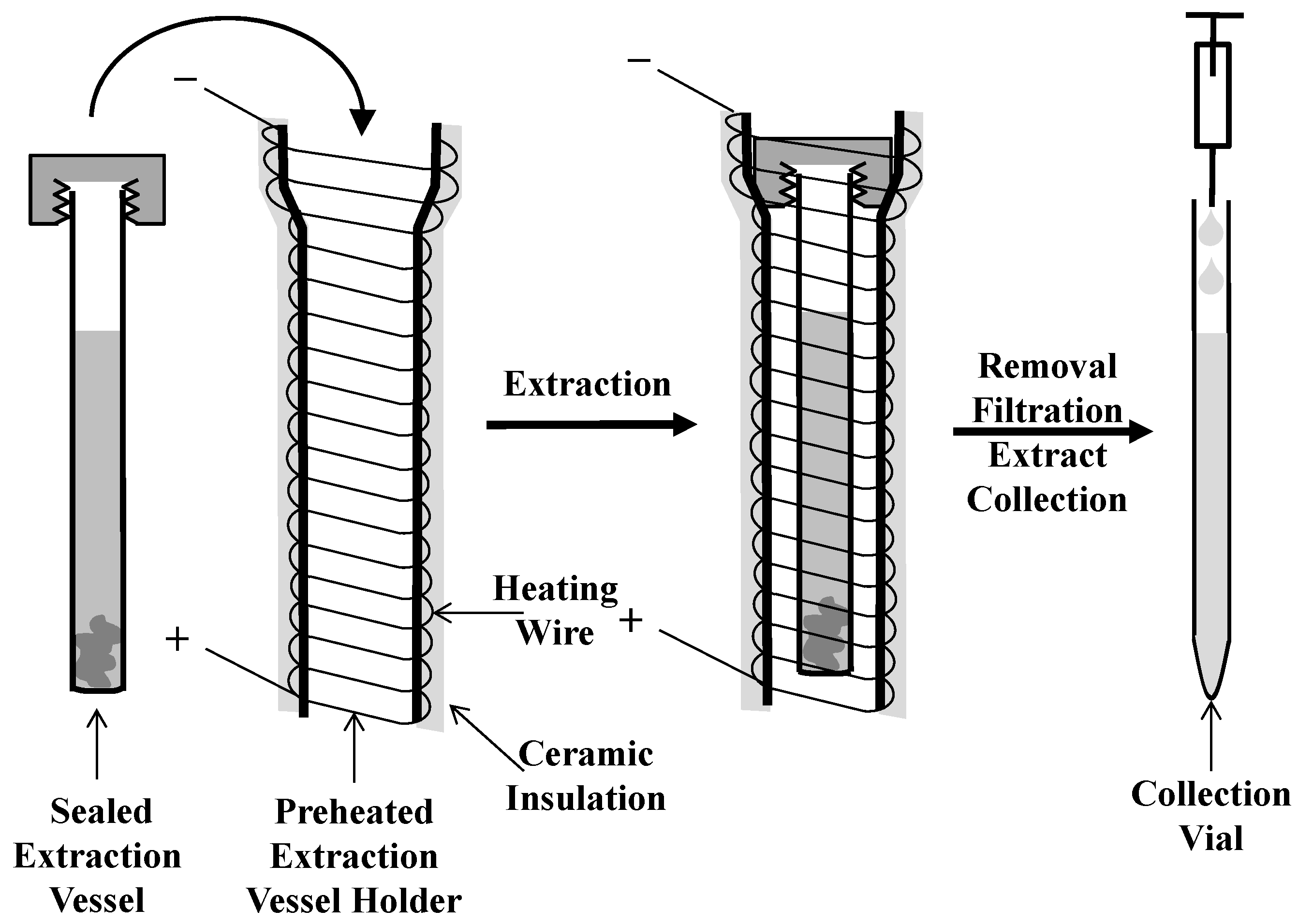
2.2. Extraction and Analysis Procedures
2.3. Quantification of Extracts

2.4. Matrix Preparation
2.5. Materials
3. Results and Discussion
3.1. Ottawa Sand
| Analyte | % Recovery | % RSD |
|---|---|---|
| Naphthalene | 112 | 9 |
| Acenaphthene | 114 | 6 |
| Acenaphthylene | 115 | 5 |
| Fluorene | 116 | 4 |
| Phenanthrene | 114 | 5 |
| Anthracene | 113 | 2 |
| Fluoranthene | 110 | 2 |
| Pyrene | 103 | 4 |
| Benzo[a]anthracene | 113 | 7 |
| Chrysene | 112 | 10 |
| Benzo[b]/[k] fluoranthene | 114 | 12 |
| Benzo[a]pyrene | 104 | 11 |
| Indeno[1,2,3-cd]pyrene | 97 | 11 |
| Dibenz[a,h]anthracene | 104 | 15 |
| Benzo[g,h,i]perylene | 100 | 14 |
| Average | 109 | 8 |
3.2. Soil
| Matrix | Soil | Chicken Breast | Charred Toast | |||
|---|---|---|---|---|---|---|
| Analyte | % Recovery | % RSD | % Recovery | % RSD | % Recovery | % RSD |
| Naphthalene | 92 | 8 | 114 | 4 | 93 | 6 |
| Acenaphthene | 90 | 3 | 103 | 3 | 87 | 4 |
| Acenaphthylene | 93 | 4 | 97 | 7 | 92 | 1 |
| Fluorene | 92 | 4 | 96 | 4 | 88 | 3 |
| Phenanthrene | 92 | 6 | 93 | 6 | 87 | 2 |
| Anthracene | 95 | 6 | 104 | 3 | 87 | 3 |
| Fluoranthene | 93 | 6 | 91 | 11 | 82 | 2 |
| Pyrene | 109 | 20 | 88 | 13 | 83 | 5 |
| Benzo[a]anthracene | 113 | 9 | 93 | 18 | 81 | 7 |
| Chrysene | 113 | 11 | 94 | 16 | 90 | 5 |
| Benzo[b]/[k]fluoranthene | 119 | 9 | 100 | 16 | 78 | 4 |
| Benzo[a]pyrene | 116 | 9 | 110 | 17 | 78 | 3 |
| Indeno[1,2,3-cd]pyrene | 130 a | - | 108 | 13 | 80 | 3 |
| Dibenz[a,h]anthracene | 122 a | - | 110 | 18 | 79 | 7 |
| Benzo[g,h,i]perylene | 127 | 12 | 107 | 10 | 82 | 3 |
| Average | 103 | 8 | 101 | 11 | 84 | 4 |
3.3. Food
3.4. Biochar-Amended Soil
| Matrix | Biochar-amended soil | Biochar | ||
|---|---|---|---|---|
| Analyte | % Recovery | % RSD | % Recovery | % RSD |
| Naphthalene | 102 | 5 | 61 | 7 |
| Acenaphthene | 104 | 3 | 60 | 13 |
| Acenaphthylene | 106 | 6 | 66 | 11 |
| Fluorene | 101 | 7 | 66 | 18 |
| Phenanthrene | 101 | 4 | 65 | 18 |
| Anthracene | 97 | 4 | 65 | 14 |
| Fluoranthene | 92 | 3 | 59 | 17 |
| Pyrene | 98 | 8 | 58 | 20 |
| Benzo[a]anthracene | 77 | 3 | 55 | 17 |
| Chrysene | 79 | 2 | 58 | 14 |
| Benzo[b]/[k]fluoranthene | 76 | 6 | 47 | 17 |
| Benzo[a]pyrene | 73 | 10 | 45 | 15 |
| Indeno[1,2,3-cd]pyrene | 51 | 8 | 33 | 25 |
| Dibenz[a,h]anthracene | 53 | 10 | 35 | 17 |
| Benzo[g,h,i]perylene | 53 | 5 | 38 | 28 |
| Average | 84 | 6 | 54 | 17 |
3.5. Biochar
3.6. Comparisons with Other Methods
| Sample | µPLE | Conventional Methods | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Time (s) | Solvent Volume (µL) | Recovery Range (%) | AVG ± RSD (%) | Method | Reference | Time | Solvent Volume (mL) | Recovery Range (%) | AVG ± RSD (%) | |
| Ottawa sand | 15 | 125 | 97–116 | 109 ± 8 | PLE | 27 | 7 min | 30 | 28–109 | 96 ± 12 |
| Soil | 30 | 125 | 90–130 | 103 ± 8 | PLE | 29 | 10 min | 50 | 75–103 | 88 ± 10 |
| Chicken breast | 30 | 125 | 88–114 | 101 ± 11 | PLE | 30 | 15 min | 20 | 60–100 | 80 ± 7 |
| Charred toast | 30 | 125 | 78–93 | 84 ± 4 | Soxhlet | 31 | 16 h | 150 | 81–95 | 89 ± 8 |
| Ultrasound | 19 | 30 min | 30 | 44–81 | 64 ± 3 | |||||
| Biochar-amended soil | 60 | 125 | 51–106 | 84 ± 6 | Soxhlet | 16 | 36 h | 160 | 49–136 | 99 ± 50 |
| Biochar | 60 | 125 | 33–66 | 54 ± 17 | Soxhlet | 35 | 36 h | 160 | 16–64 | 42 ± 18 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Luque de Castro, M.D.; Priego-Capote, F. Soxhlet extraction: Past and present panacea. J. Chromatogr. A 2010, 1217, 2383–2389. [Google Scholar] [CrossRef] [PubMed]
- Luque de Castro, M.D.; García-Ayuso, L.E. Soxhlet extraction of solid materials: An outdated technique with a promising innovative future. Anal. Chim. Acta 1998, 369, 1–10. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Grabanski, C.B.; Martin, E.; Miller, D.J. Comparisons of Soxhlet extraction, pressurized liquid extraction, supercritical fluid extraction and subcritical water extraction for environmental solids: recovery, selectivity and effects on sample matrix. J. Chromatogr. A 2000, 892, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Sporring, S.; Bøwadt, S.; Svensmark, B.; Björklund, E. Comprehensive comparison of classic Soxhlet extraction with Soxtec extraction, ultrasonication extraction, supercritical fluid extraction, microwave assisted extraction and accelerated solvent extraction for the determination of polychlorinated biphenyls in soil. J. Chromatogr. A 2005, 1090, 1–9. [Google Scholar] [PubMed]
- Nieto, A.; Borrull, F.; Pocurull, E.; Marcé, R.M. Pressurized liquid extraction: A useful technique to extract pharmaceuticals and personal-care products from sewage sludge. Trac-Trend. Anal. Chem. 2010, 29, 752–764. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Duarte, R.M.B.O.; Duarte, A.C. Considerations on the application of miniaturized sample preparation approaches for the analysis of organic compounds in environmental matrices. Cent. Eur. J. Chem. 2012, 10, 433–449. [Google Scholar] [CrossRef]
- Ramos, L.; Vreuls, J.J.; Brinkman, U.A.T. Miniaturised pressurised liquid extraction of polycyclic aromatic hydrocarbons from soil and sediment with subsequent large-volume injection-gas chromatography. J. Chromatogr. A 2000, 891, 275–286. [Google Scholar] [CrossRef]
- Pena-Abaurrea, M.; Ramos, J.J.; Gonzalez, M.J.; Ramos, L. Miniaturized selective pressurized liquid extraction of polychlorinated biphenyls and polybrominated diphenyl ethers from feedstuffs. J. Chromatogr. A 2013, 1273, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Hyötyläinen, T.; Andersson, T.; Hartonen, K.; Kuosmanen, K.; Riekkola, M.-L. Pressurized hot water extraction coupled on-line with LC-GC: Determination of polyaromatic hydrocarbons in sediment. Anal. Chem. 2000, 72, 3070–3076. [Google Scholar] [CrossRef] [PubMed]
- Alkhateeb, F.L.; Thurbide, K.B. A novel micro pressurized liquid extraction method for very rapid solid sample preparation. Anal. Methods 2015, 7, 1509–1516. [Google Scholar] [CrossRef]
- Purcaro, G.; Moret, S.; Conte, L.S. Overview on polycyclic aromatic hydrocarbons: Occurrence, legislation and innovative determination in foods. Talanta 2013, 105, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Viegas, O.; Novo, P.; Pinho, O.; Ferreira, I.M.P.L.V.O. A comparison of the extraction procedures and quantification methods for the chromatographic determination of polycyclic aromatic hydrocarbons in charcoal grilled meat and fish. Talanta 2012, 88, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Lü, H.; Cai, Q.-Y.; Jones, K.C.; Zeng, Q.-Y.; Katsoyiannis, A. Levels of organic pollutants in vegetables and human exposure through diet: A review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1–33. [Google Scholar] [CrossRef]
- Brändli, R.C.; Hartnik, T.; Henriksen, T.; Cornelissen, G. Sorption of native polyaromatic hydrocarbons (PAH) to black carbon and amended activated carbon in soil. Chemosphere 2008, 73, 1805–1810. [Google Scholar] [CrossRef] [PubMed]
- Boström, C.-E.; Gerde, P.; Hanberg, A.; Jernström, B.; Johansson, C.; Kyrklund, T.; Rannug, A.; Törnqvist, M.; Victorin, K.; Westerholm, R. Cancer risk assessment, indicators, and guidelines for polycyclic aromatic hydrocarbons in the ambient air. Environ. Health Persp. 2002, 110, 451–488. [Google Scholar] [CrossRef]
- Fabbri, D.; Rombolà, A.G.; Torri, C.; Spokas, K.A. Determination of polycyclic aromatic hydrocarbons in biochar and biochar amended soil. J. Anal. Appl. Pyrol. 2013, 103, 60–67. [Google Scholar] [CrossRef]
- Barreca, S.; Mazzola, A.; Orecchio, S.; Tuzzolino, N. Polychlorinated biphenyls in sediments from Sicilian coastal area (Scoglitti) using automated soxhlet, GC-MS, and principal component analysis. Polycycl. Aromat. Comp. 2014, 34, 237–262. [Google Scholar] [CrossRef]
- León, V.M.; Moreno-González, R.; González, E.; Martínez, F.; García, V.; Campillo, J.A. Interspecific comparison of polycyclic aromatic hydrocarbons and persistent organochlorines bioaccumulation in bivalves from a Mediterranean coastal lagoon. Sci. Total Environ. 2013, 463–464, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Rey-Salgueiro, L.; García-Falcón, M.S.; Martínez-Carballo, E.; Simal-Gándara, J. Effects of toasting procedures on the levels of polycyclic aromatic hydrocarbons in toasted bread. Food Chem. 2008, 108, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Viegas, O.; Yebra-Pimentel, I.; Martinez-Carballo, E.; Simal-Gandara, J.; Ferreira, I.M.P.L.V.O. Effect of beer marinades on formation of polycyclic aromatic hydrocarbons in charcoal-grilled pork. J. Agric. Food Chem. 2014, 62, 2638–2643. [Google Scholar] [CrossRef] [PubMed]
- Irvine, G.M.; Blais, J.M.; Doyle, J.R.; Kimpe, L.E.; White, P.A. Cancer risk to First Nations’ people from exposure to polycyclic aromatic hydrocarbons near in-situ bitumen extraction in Cold Lake, Alberta. Environ. Health 2014, 13, 7. [Google Scholar] [CrossRef] [PubMed]
- Lorenzi, D.; Cave, M.; Dean, J.R. An investigation into the occurrence and distribution of polycyclic aromatic hydrocarbons in two soil size fractions at a former industrial site in NE England, UK using in situ PFE–GC–MS. Environ. Geochem. Health 2010, 32, 553–565. [Google Scholar] [CrossRef] [PubMed]
- Lund, M.; Duedahl-Olesen, L.; Christensen, J.H. Extraction of polycyclic aromatic hydrocarbons from smoked fish using pressurized liquid extraction with integrated fat removal. Talanta 2009, 79, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Aguinaga, N.; Campillo, N.; Viñas, P.; Hernández-Córdoba, M. Determination of 16 polycyclic aromatic hydrocarbons in milk and related products using solid-phase microextraction coupled to gas chromatography-mass spectrometry. Anal. Chim. Acta 2007, 596, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Cacho, J.I.; Campillo, N.; Viñas, P.; Hernández-Córdoba, M. Use of headspace sorptive extraction coupled to gas chromatography-mass spectrometry for the analysis of volatile polycyclic aromatic hydrocarbons in herbal infusions. J. Chromatogr. A 2014, 1356, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Campíns-Falcó, P.; Verdú-Andrés, J.; Sevillano-Cabeza, A.; Molins-Legua, C.; Herráez-Hernández, R. New micromethod combining miniaturized matrix solid-phase dispersion and in-tube in-valve solid-phase microextraction for estimating polycyclic aromatic hydrocarbons in bivalves. J. Chromatogr. A 2008, 1211, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Haskins, S.D.; Duval, J.M.; Kelly, D.G.; Lundgreen-Nielsen, S.L.; Weir, R.D. Pressurized fluid extraction of polycyclic aromatic hydrocarbons using silanized extraction vessels. Microchim. Acta 2012, 178, 187–193. [Google Scholar] [CrossRef]
- Wilcke, W. Polycyclic aromatic hydrocarbons (PAHs) in soil—A review. J. Plant Nutr. Soil Sci. 2000, 163, 229–248. [Google Scholar] [CrossRef]
- Zhang, Q.; Liang, T.; Wang, L.; Cao, H. Determination of polycyclic aromatic hydrocarbons from soil samples using selective pressurized liquid extraction. Anal. Methods 2012, 4, 2441–2446. [Google Scholar] [CrossRef]
- Wang, G.; Lee, A.S.; Lewis, M.; Kamath, B.; Archer, R.K. Accelerated solvent extraction and gas chromatography/mass spectrometry for determination of polycyclic aromatic hydrocarbons in smoked food samples. J. Agric. Food Chem. 1999, 47, 1062–1066. [Google Scholar] [CrossRef] [PubMed]
- Al-Rashdan, A.; Helaleh, M.I.H.; Nisar, A.; Ibtisam, A.; Al-Ballam, Z. Determination of the levels of polycyclic aromatic hydrocarbons in toasted bread using gas chromatography mass spectrometry. Int. J. Anal. Chem. 2010, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Manyà, J.J. Pyrolysis for biochar purposes: A review to establish current knowledge gaps and research needs. Environ. Sci. Technol. 2012, 46, 7939–7954. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Yuan, M. Enhanced sorption of polycyclic aromatic hydrocarbons by soil amended with biochar. J. Soil. Sediment. 2011, 11, 62–71. [Google Scholar] [CrossRef]
- Jonker, M.T.O.; Koelmans, A.A. Sorption of polycyclic aromatic hydrocarbons and polychlorinated biphenyls to soot and soot-like materials in the aqueous environment: Mechanistic considerations. Environ. Sci. Technol. 2002, 36, 3725–3734. [Google Scholar] [CrossRef] [PubMed]
- Hilber, I.; Blum, F.; Leifeld, J.; Schmidt, H.-P.; Bucheli, T.D. Quantitative determination of PAHs in biochar: A prerequisite to ensure its quality and safe application. J. Agric. Food Chem. 2012, 60, 3042–3050. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frantz, J.J.; Alkhateeb, F.L.; Thurbide, K.B. A Novel Micro Pressurized Liquid Extraction Method for Rapid Sample Preparation of Polycyclic Aromatic Hydrocarbons in Various Solids. Chromatography 2015, 2, 488-501. https://doi.org/10.3390/chromatography2030488
Frantz JJ, Alkhateeb FL, Thurbide KB. A Novel Micro Pressurized Liquid Extraction Method for Rapid Sample Preparation of Polycyclic Aromatic Hydrocarbons in Various Solids. Chromatography. 2015; 2(3):488-501. https://doi.org/10.3390/chromatography2030488
Chicago/Turabian StyleFrantz, Jackson J., Fadi L. Alkhateeb, and Kevin B. Thurbide. 2015. "A Novel Micro Pressurized Liquid Extraction Method for Rapid Sample Preparation of Polycyclic Aromatic Hydrocarbons in Various Solids" Chromatography 2, no. 3: 488-501. https://doi.org/10.3390/chromatography2030488
APA StyleFrantz, J. J., Alkhateeb, F. L., & Thurbide, K. B. (2015). A Novel Micro Pressurized Liquid Extraction Method for Rapid Sample Preparation of Polycyclic Aromatic Hydrocarbons in Various Solids. Chromatography, 2(3), 488-501. https://doi.org/10.3390/chromatography2030488





