Cu Purification Using an Extraction Resin for Determination of Isotope Ratios by Multicollector ICP-MS
Abstract
:1. Introduction
2. Samples and Analytical Methods
2.1. Reagents and Silicate Samples
2.2. TEVA™ Resin Column and Silicate Sample Solution
2.3. Copper Purification by the TEVA™ Resin Column
| Washing | ||
| 2 mol·L−1 HNO3 | 6.4 mL | |
| water | 1.6 mL | |
| 0.05 mol·L−1 HBr | 3.2 mL | |
| Conditioning | ||
| 0.05 mol·L−1 HBr | ||
| with 0.15% ascorbic acid | 3.2 mL | |
| Loading the sample (leave 30 min) | 0.05 mol·L−1 HBr | |
| with 0.15% ascorbic acid | 0.1 mL | |
| Removing major elements | ||
| 0.05 mol·L−1 HBr | ||
| with 0.15% ascorbic acid | 4.8 mL | |
| water | 0.8 mL | |
| Collecting Cu | ||
| 2 mol·L−1 HNO3 | 6.4 mL |
2.4. Measurements of Cu, Distribution Coefficients, and Major Elements (Elution Curves)
2.5. Measurement of Cu Isotope Ratios
| 1. Sample introduction and ICP conditions | |
| Nebulizer | Micro-flow PFA nebulizer, PFA-50 (Elemental Scientific, Omaha, NE, USA), self-aspiration (flow rate: ~50 μL·min−1) |
| Plasma power | 1.2 kW (27.12 MHz) |
| Torch | Quartz glass torch with a sapphire injector |
| Plasma Ar gas flow rate | 15 L·min−1 |
| Auxiliary Ar gas flow rate | 0.80 L·min−1 |
| Nebulizer Ar gas flow rate | 0.90 L·min−1 |
| 2. Desolvator conditions | |
| Desolvator | ARIDUS II (CETAC Technologies, Omaha, NE, USA) |
| Spray chamber temperature | 110 °C |
| Desolvator temperature | 160 °C |
| Sweep gas (Ar) | 8~9 L·min−1 |
| 3. Interface | |
| Sampling cone | Made of Ni |
| Skimmer cone | Made of Ni (X-skimmer) |
| 4. Data acquisition conditions | |
| Resolution | M/∆M = ~500 |
| Washing time | 480 s after measurement |
| Uptake time | 90 s |
| Background data integration | 4 s for 1 scan, 20 scans in one run |
| Background was measured at the same mass | |
| with aspirating HNO3 | |
| Sample data integration | 4 s for 1 scan, 20 scans in one run |
| 5. Cup configuration | |
| L4 L3 L2 L1 C H1 H2 H3 H4 | |
| 63Cu 65Cu | |
3. Results and Discussion
3.1. Kinetic Effects of Adsorption of Cu(I)

3.2. Elution Curves for Cu, Mg, and Zn Using the TEVA™ Column and the Effects of Coexisting Zn
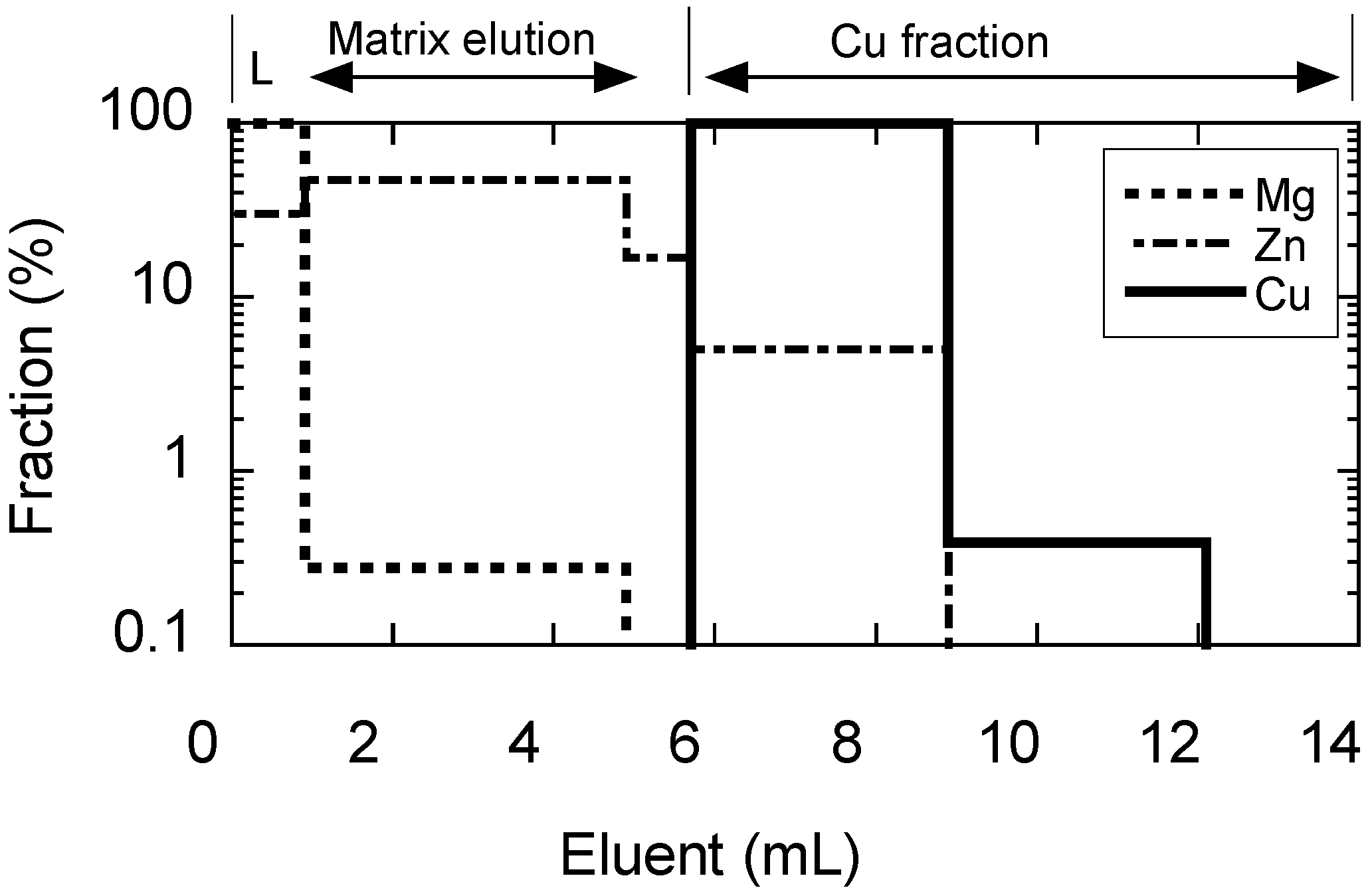
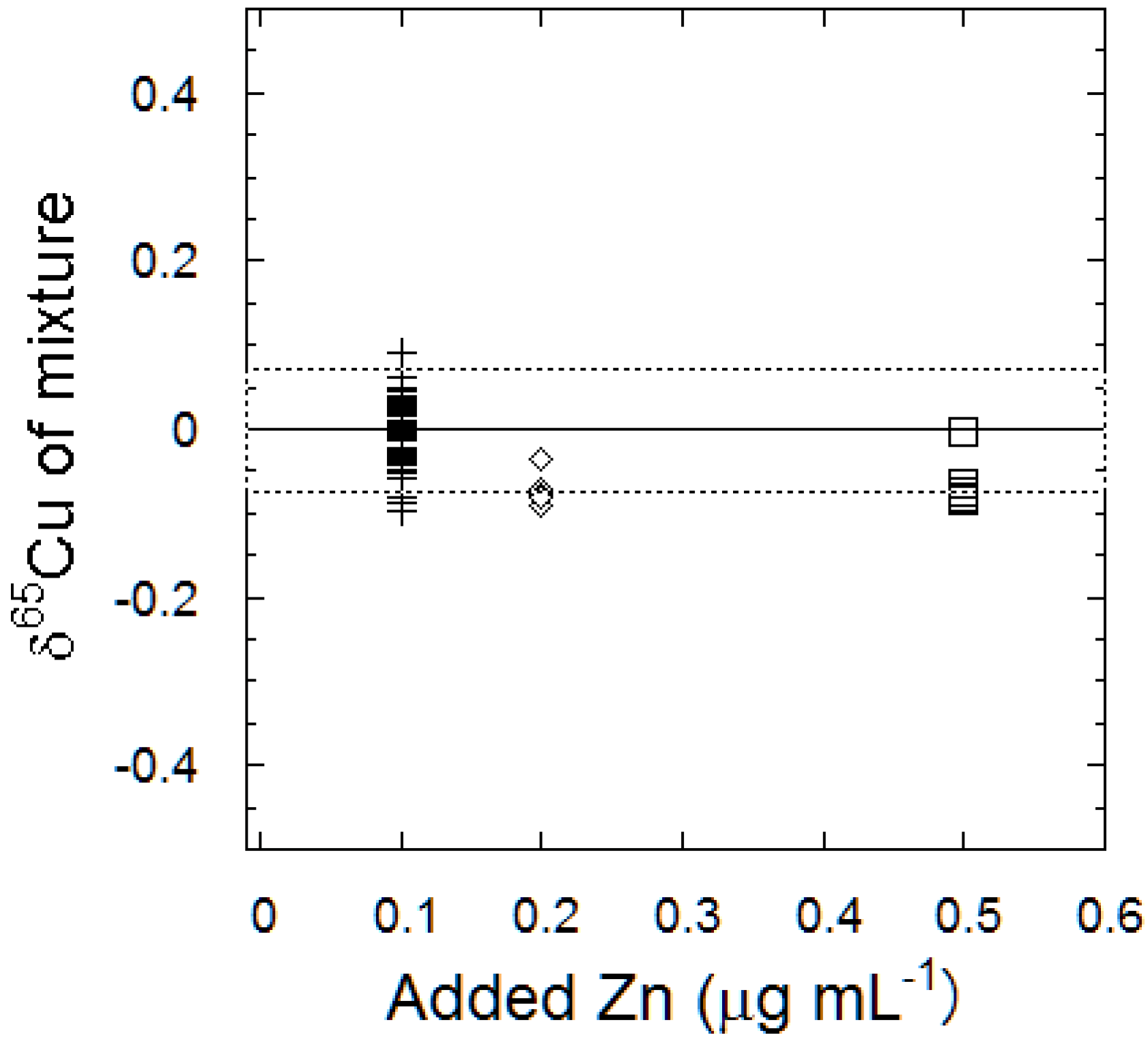
3.3. Mass Fractionation, Recovery Yield, and Blank of the TEVA™ Resin Column Chemistry
3.4. Merits of the New Cu Column Chemistry
3.5. Evaluation of the Accuracy of the Cu Isotope Ratio Measurements
| Calculated | Observed | ||||
|---|---|---|---|---|---|
| δ65Cu | SD (‰) | δ65Cu | SD (‰) | n | |
| Mixture #1 | −0.14 | 0.01 | −0.14 | 0.09 | 4 |
| Mixture #2 | −0.91 | 0.06 | −0.92 | 0.16 | 4 |
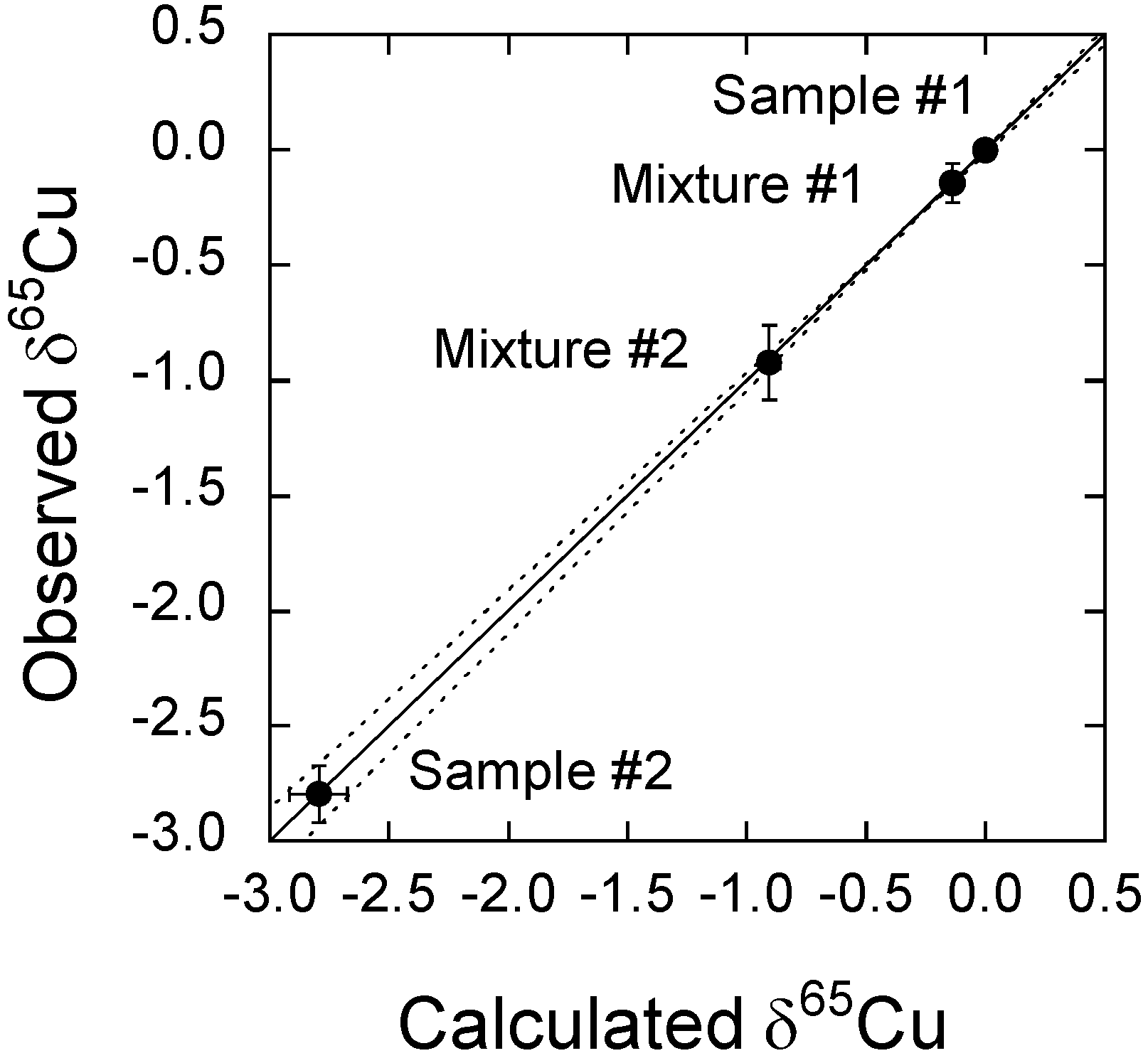
3.6. Repeatability of Cu Isotope Ratios in Silicate Reference Materials
| Sample | δ65Cu | SD | n |
|---|---|---|---|
| Average | (‰) | ||
| BHVO-1 | −0.44 | 0.07 | 3 |
| AGV-1 | −0.64 | 0.47 | 3 |
| PCC-1 | −0.71 | 0.50 | 3 |
| JB-1 | 0.83 | 0.06 | 5 |
| JB-2 | 1.01 | 0.01 | 3 |
| JB-3 | −0.75 | 0.43 | 3 |
| JA-1 | 0.86 | 0.17 | 3 |
| JA-2 | −0.33 | 0.04 | 3 |
| JA-3 | 0.11 | 0.06 | 3 |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Zhu, X.K.; O’Nions, R.K.; Guo, Y.; Belshaw, N.S.; Richard, D. Determination of naural Cu-isotope variation by plasma-source mass spectrometry: Implications for use as geochemical tracers. Chem. Geol. 2000, 163, 139–149. [Google Scholar] [CrossRef]
- Archer, C.; Vance, D. Mass discrimination correction in multiple-collector plasma source mass spectrometry: an example using Cu and Zn isotopes. J. Anal. At. Spectrom. 2004, 19, 656–665. [Google Scholar]
- Weinstein, C.; Moynier, F.; Wang, K.; Paniello, R.; Foriel, J.; Catalano, J.; Pichat, S. Isotopic fractionation of Cu in plant. Chem. Geol. 2011, 286, 266–271. [Google Scholar]
- Marechal, C.N.; Albarede, F. Ion-exchange fractionation of copper and zinc isotopes. Geochim. Cosmochim. Acta 2002, 66, 1499–1509. [Google Scholar] [CrossRef]
- Strelow, F.W.E. Improved separation of iron from copper and other elements by anion-exchange chromatography on a 4-percent cross-linked resin with high-concentrations of hydrochloric-acid. Talanta 1980, 27, 727–731. [Google Scholar] [CrossRef]
- Dauphas, N.; Rouxel, O. Mass spectrometry and natural variations of iron isotopes. Mass Spectrom. Rev. 2006, 25, 515–550. [Google Scholar] [CrossRef]
- Chapman, J.B.; Mason, T.F.D.; Weiss, D.J.; Coles, B.J.; Wilkinson, J.J. Chemical separation and isotopic variations of Cu and Zn from five geological reference materials. Geostand. Geoanal. Res. 2006, 30, 5–16. [Google Scholar] [CrossRef]
- Borrok, D.M.; Wanty, R.B.; Ridley, W.I.; Wolf, R.; Lamothe, P.J.; Adams, M. Separation of copper, iron, and zinc from complex aqueous solutions for isotopic measurement. Chem. Geol. 2007, 242, 1345–1352. [Google Scholar]
- Nakai, T.; Yajima, S.; Fujii, I.; Okada, M. Activation analysis of impurities in silicon for semiconduction material. Jpn. Anal. 1959, 8, 367–372. [Google Scholar] [CrossRef]
- Korkisch, J.K. Handbook of Ion Exchange Resins: Their Application to Inorganic Analytical Chemistry; CRC Press: Boca Raton, FL, USA, 1989; Volume III; p. 288. [Google Scholar]
- Larner, F.; Rehkӓmper, M.; Coles, B.J.; Kreissig, K.; Weiss, D.J.; Sampson, B.; Unsworth, C.; Strekopyotov, S. A new separation procedure for Cu prior to stable isotope analysis by MC-ICP-MS. J. Anal. At. Spectrom. 2011, 26, 1627–1631. [Google Scholar]
- Horwitz, E.P.; Dietz, M.L.; Chiarizia, R.; Diamond, H.; Maxwell, S.L., III; Nelson, M.R. Separation and preconcentration of actinides by extraction chromatography using a supported liquid anion-exchanger—Application to the characterization of high-level nuclear waste solutions. Anal. Chim. Acta 1995, 310, 63–78. [Google Scholar] [CrossRef]
- Yang, X.-J.; Pin, C. Separation of hafnium and zirconium from Ti and Fe-rich geological materials by extraction chromatography. Anal. Chem. 1999, 71, 1706–1711. [Google Scholar] [CrossRef]
- Grahek, Z.; Macefat, M.R. Isolation of iron and strontium from liquid samples and determination of Fe-55 and Sr-89, Sr-90 in liquid radioactive waste. Anal. Chim. Acta 2004, 511, 339. [Google Scholar] [CrossRef]
- Makishima, A.; Nakamura, E. Low-blank chemistry for Zn stable isotope ratio determination using extraction chromatographic resin and double spike-multiple collector-ICP-MS. J. Anal. At. Spectrom. 2013, 28, 127–133. [Google Scholar] [CrossRef]
- Makishima, A. A simple and fast separation method of Fe employing extraction resin for isotope ratio determination by multicollector ICP-MS. Int. J. Mass Spectrom. Chromatogr. 2013, 1, 195–203. [Google Scholar]
- Makishima, A.; Nakanishi, M.; Nakamura, E. A group separation method of ruthenium, palladium, rhenium, osmium, iridium and platinum using their bromo complexes and an anion exchange resin. Anal. Chem. 2001, 73, 5240–5248. [Google Scholar] [CrossRef]
- Marechal, C.N.; Telouk, P.; Albarede, F. Precise analysis of copper and zinc isotopic compositions by plasma-source mass spectrometry. Chem. Geol. 1999, 156, 251–273. [Google Scholar] [CrossRef]
- Luck, J.M.; Ben Othman, D.; Barrat, J.D.; Albarede, F. Coupled Cu-63 and O-16 excesses in chondrites. Geochim. Cosmochim. Acta 2003, 67, 143–151. [Google Scholar] [CrossRef]
- Nakamura, E.; Makishima, A.; Moriguti, T.; Kobayashi, K.; Sakaguchi, C.; Yokoyama, T.; Tanaka, R.; Kuritani, T.; Takei, H. Comprehensive geochemical analyses of small amounts (100 mg) of extraterrestrial samples for the analytical competition related to the sample-return mission, MUSES-C. Inst. Space Astronaut. Sci. Rep. Special 2003, 16, 49–101. [Google Scholar]
- Yokoyama, T.; Makishima, A.; Nakamura, E. Evaluation of the coprecipitation of incompatible trace elements with fluoride during silicate rock dissolution by acid digestion. Chem. Geol. 1999, 157, 175–187. [Google Scholar] [CrossRef]
- Makishima, A.; Nakamura, E. Determination of major, minor and trace elements in silicate samples by ICP-QMS and ICP-SFMS applying isotope dilution-internal standardization (ID-IS) and multi-stage internal standardization. Geostands. Geoanal. Res. 2006, 30, 245–271. [Google Scholar] [CrossRef]
- Pribil, M.J.; Wanty, R.B.; Ridley, R.I.; Borrok, D.M. Influence of sulfur-bearing polyatomic species on high precision measurements of Cu isotopic composition. Chem. Geol. 2010, 272, 49–54. [Google Scholar] [CrossRef]
- Makishima, A.; Nakamura, E. Determination of total sulfur at microgram per gram levels in geological materials by oxidation of sulfur into sulfate with in situ generation of bromine using isotope dilution high resolution ICPMS. Anal. Chem. 2001, 73, 2547–2553. [Google Scholar] [CrossRef]
- Nielsen, S.G.; Rehkämper, M.; Baker, J.; Halliday, A.N. The precise and accurate determination of thallium isotope compositions and concentrations for water samples by MC-ICPMS. Chem. Geol. 2004, 204, 109–124. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Makishima, A. Cu Purification Using an Extraction Resin for Determination of Isotope Ratios by Multicollector ICP-MS. Chromatography 2014, 1, 96-107. https://doi.org/10.3390/chromatography1030096
Makishima A. Cu Purification Using an Extraction Resin for Determination of Isotope Ratios by Multicollector ICP-MS. Chromatography. 2014; 1(3):96-107. https://doi.org/10.3390/chromatography1030096
Chicago/Turabian StyleMakishima, Akio. 2014. "Cu Purification Using an Extraction Resin for Determination of Isotope Ratios by Multicollector ICP-MS" Chromatography 1, no. 3: 96-107. https://doi.org/10.3390/chromatography1030096




