The Central Carbon and Energy Metabolism of Marine Diatoms
Abstract
:1. Introduction
2. Distinctive Carbon Metabolism in Diatoms
2.1. Light Harvesting Machinery to Cope with High Light
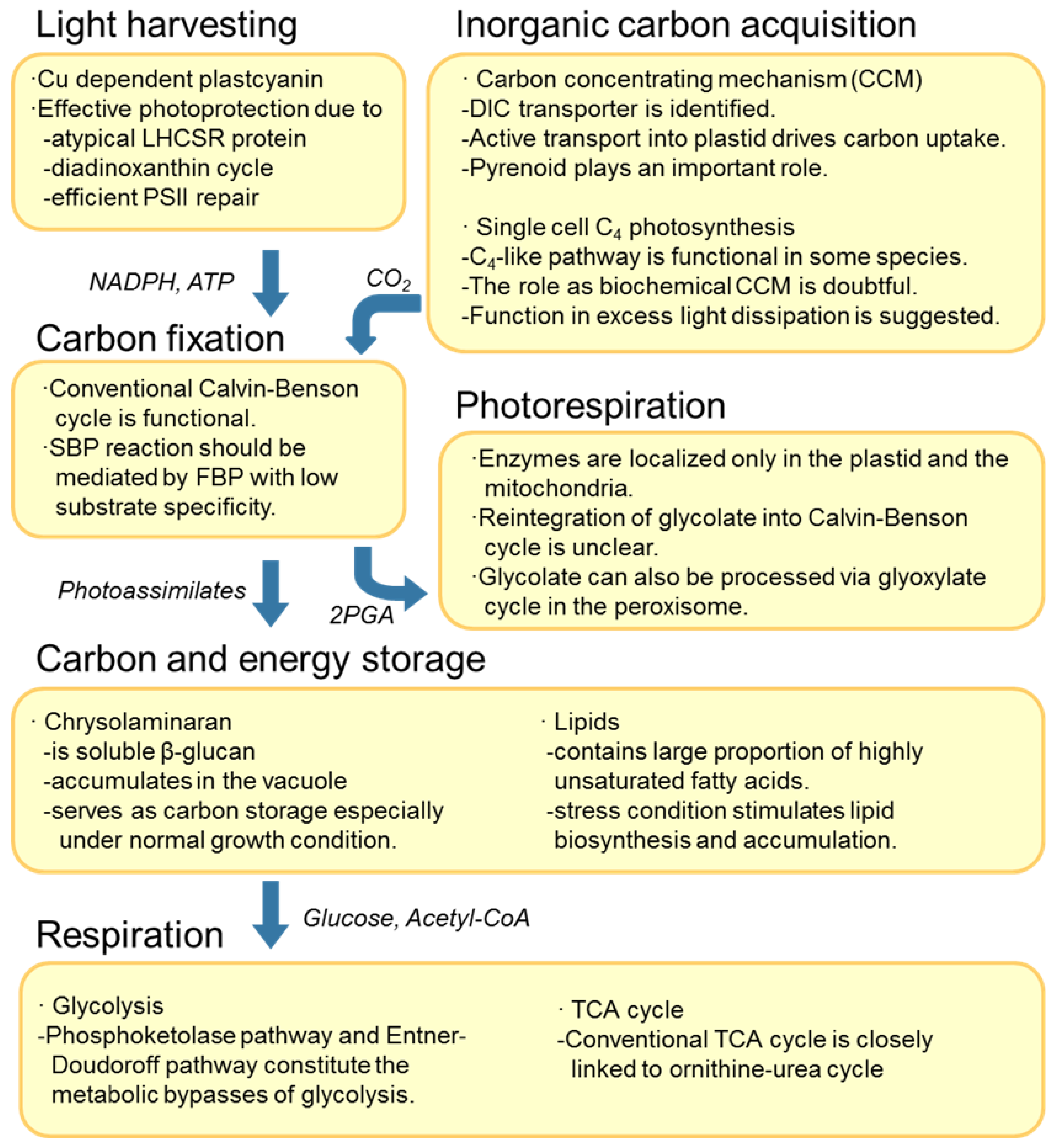
2.2. Inorganic Carbon Acquisition—Biophysical or Biochemical Carbon Concentrating Mechanism?
2.2.1. Inorganic Carbon Concentrating Mechanism and Ion Pumping
2.2.2. Role of C4-like Pathway
2.3. The Calvin-Benson Cycle and Photoassimilate Conversion to the Chrysolaminaran Storage Form
2.4. Photorespiration in Two Cellular Compartments
2.5. Respiration Closely Linked to Other Metabolic Pathways
2.5.1. Presence of Glycolytic Bypasses
2.5.2. The TCA Cycle is Intimately Linked to the Ornithine-Urea Cycle
3. Adaptation of Diatom Carbon Metabolism to a Changing Environment
3.1. Elevated CO2 Level and Ocean Acidification
3.2. Iron Limitation
3.3. Chemical Pollution
4. Lipid Production and Potential Use of Diatom as a Source of Biofuel
4.1. Outlook of the Industrial Use of Microalgae
4.2. Lipid Biosynthesis in Diatom Cells
4.2.1. Pathways for Fatty Acid and Lipid Metabolism
4.2.2. Influences of Nutrient Deprivation on Lipid Accumulation

5. Conclusions
Acknowledgments
Conflict of Interest
References
- Hasle, G.R.; Syvertsen, E.E. Marine diatoms. In Identifying Marine Diatoms and Dinoflagellates; Elsevier: Amsterdam, The Netherland, 1996; pp. 5–385. [Google Scholar]
- Villareal, T.A. Buoyancy properties of the giant diatom Ethmodiscus. J. Plankton Res. 1992, 14, 459–463. [Google Scholar] [CrossRef]
- Bertrand, M. Carotenoid biosynthesis in diatoms. Photosynth. Res. 2010, 106, 89–102. [Google Scholar] [CrossRef]
- Kröger, N.; Poulsen, N. Diatoms-from cell wall biogenesis to nanotechnology. Annu. Rev. Genet. 2008, 42, 83–107. [Google Scholar] [CrossRef]
- Schubert, H.; Sagert, S.; Forster, R.M. Evaluation of the different levels of variability in the underwater light field of a shallow estuary. Helgol. Mar. Res. 2001, 55, 12–22. [Google Scholar] [CrossRef]
- Anderson, L.W.J.; Sweeney, B.M. Diel changes in sedimentation characteristics of ditylum brightwelli: changes in cellular lipid and effects of respiratory inhibitors and ion-transport modifiers. Limnol. Oceanogr. 2007, 22, 539–552. [Google Scholar] [CrossRef]
- Bowler, C.; Vardi, A.; Allen, A.E. Oceanographic and biogeochemical insights from diatom genomes. Annu. Rev. Mar. Sci. 2010, 2, 333–365. [Google Scholar] [CrossRef]
- Bowler, C.; Karl, D.M.; Colwell, R.R. Microbial oceanography in a sea of opportunity. Nature 2009, 459, 180–184. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Fenchel, T.; Delong, E.F. The microbial engines that drive Earth’s biogeochemical cycles. Science 2008, 320, 1034–1039. [Google Scholar] [CrossRef]
- Coleman, M.L.; Sullivan, M.B.; Martiny, A.C.; Steglich, C.; Barry, K.; Delong, E.F.; Chisholm, S.W. Genomic islands and the ecology and evolution of Prochlorococcus. Science 2006, 311, 1768–1770. [Google Scholar] [CrossRef]
- Keeling, P.J.; Palmer, J.D. Horizontal gene transfer in eukaryotic evolution. Nat. Rev. Genet. 2008, 9, 605–618. [Google Scholar]
- Curtis, B.A.; Tanifuji, G.; Burki, F.; Gruber, A.; Irimia, M.; Maruyama, S.; Arias, M.C.; Ball, S.G.; Gile, G.H.; Hirakawa, Y.; et al. Algal genomes reveal evolutionary mosaicism and the fate of nucleomorphs. Nature 2012, 492, 59–65. [Google Scholar]
- Armbrust, E.V.; Berges, J.A.; Bowler, C.; Green, B.R.; Martinez, D.; Putnam, N.H.; Zhou, S.; Allen, A.E.; Apt, K.E.; Bechner, M.; et al. The genome of the diatom Thalassiosira pseudonana: ecology, evolution, and metabolism. Science 2004, 306, 79–86. [Google Scholar] [CrossRef]
- Bowler, C.; Allen, A.E.; Badger, J.H.; Grimwood, J.; Jabbari, K.; Kuo, A.; Maheswari, U.; Martens, C.; Maumus, F.; Otillar, R.P.; et al. The Phaeodactylum genome reveals the evolutionary history of diatom genomes. Nature 2008, 456, 239–244. [Google Scholar]
- Kroth, P.G.; Chiovitti, A.; Gruber, A.; Martin-Jezequel, V.; Mock, T.; Parker, M.S.; Stanley, M.S.; Kaplan, A.; Caron, L.; Weber, T.; et al. A model for carbohydrate metabolism in the diatom Phaeodactylum tricornutum deduced from comparative whole genome analysis. PloS One 2008, 3, e1426. [Google Scholar]
- Fabris, M.; Matthijs, M.; Rombauts, S.; Vyverman, W.; Goossens, A.; Baart, G.J.E. The metabolic blueprint of Phaeodactylum tricornutum reveals a eukaryotic Entner-Doudoroff glycolytic pathway. Plant. J. 2012, 70, 1004–1014. [Google Scholar]
- Maheswari, U.; Jabbari, K.; Petit, J.-L.; Porcel, B.M.; Allen, A.E.; Cadoret, J.-P.; De Martino, A.; Heijde, M.; Kaas, R.; La Roche, J.; et al. Digital expression profiling of novel diatom transcripts provides insight into their biological functions. Genome Biol. 2010, 11, R85. [Google Scholar] [CrossRef] [Green Version]
- Chauton, M.S.; Winge, P.; Brembu, T.; Vadstein, O.; Bones, A.M. Gene regulation of carbon fixation, storage and utilization in the diatom Phaeodactylum tricornutum acclimated to light/dark cycles. Plant. Physiol. 2012, 161, 1034–1048. [Google Scholar]
- Nunn, B.L.; Aker, J.R.; Shaffer, S.A.; Tsai, S.; Strzepek, R.F.; Boyd, P.W.; Freeman, T.L.; Brittnacher, M.; Malmström, L.; Goodlett, D.R. Deciphering diatom biochemical pathways via whole-cell proteomics. Aquat. Microb. Ecol. 2009, 55, 241–253. [Google Scholar] [CrossRef]
- Hockin, N.L.; Mock, T.; Mulholland, F.; Kopriva, S.; Malin, G. The response of diatom central carbon metabolism to nitrogen starvation is different from that of green algae and higher plants. Plant. Physiol. 2012, 158, 299–312. [Google Scholar]
- Murata, N.; Takahashi, S.; Nishiyama, Y.; Allakhverdiev, S.I. Photoinhibition of photosystem II under environmental stress. Biochimi. Biophys. Acta 2007, 1767, 414–421. [Google Scholar]
- Goss, R.; Jakob, T. Regulation and function of xanthophyll cycle-dependent photoprotection in algae. Photosynth. Res. 2010, 106, 103–122. [Google Scholar] [CrossRef]
- Lavaud, J.; Rousseau, B.; Van Gorkom, H.J.; Etienne, A.-L. Influence of the diadinoxanthin pool size on photoprotection in the marine planktonic diatom Phaeodactylum tricornutum. Plant. Physiol. 2002, 129, 1398–1406. [Google Scholar] [CrossRef]
- Bailleul, B.; Rogato, A.; De Martino, A.; Coesel, S.; Cardol, P.; Bowler, C.; Falciatore, A.; Finazzi, G. An atypical member of the light-harvesting complex stress-related protein family modulates diatom responses to light. Proc. Natl. Acad. Sci. USA. 2010, 107, 18214–18219. [Google Scholar] [CrossRef]
- Domingues, N.; Matos, A.R.; Marques da Silva, J.; Cartaxana, P. Response of the diatom Phaeodactylum tricornutum to photooxidative stress resulting from high light exposure. PloS One 2012, 7, e38162. [Google Scholar]
- Wu, H.; Cockshutt, A.M.; McCarthy, A.; Campbell, D.A. Distinctive photosystem II photoinactivation and protein dynamics in marine diatoms. Plant. Physiol. 2011, 156, 2184–2195. [Google Scholar] [CrossRef]
- Brunet, C.; Lavaud, J. Can the xanthophyll cycle help extract the essence of the microalgal functional response to a variable light environment? J. Plankton Res. 2010, 32, 1609–1617. [Google Scholar] [CrossRef]
- Roberts, K.; Granum, E.; Leegood, R.C.; Raven, J.A. Carbon acquisition by diatoms. Photosynth. Res. 2007, 93, 79–88. [Google Scholar] [CrossRef]
- Reinfelder, J.R. Carbon concentrating mechanisms in eukaryotic marine phytoplankton. Ann. Rev. MarI. Sci. 2011, 3, 291–315. [Google Scholar] [CrossRef]
- Wang, Y.; Duanmu, D.; Spalding, M.H. Carbon dioxide concentrating mechanism in Chlamydomonas reinhardtii: inorganic carbon transport and CO2 recapture. Photosynth. Res. 2011, 109, 115–122. [Google Scholar] [CrossRef]
- Matsuda, Y.; Nakajima, K.; Tachibana, M. Recent progresses on the genetic basis of the regulation of CO2 acquisition systems in response to CO2 concentration. Photosynth. Res. 2011, 109, 191–203. [Google Scholar] [CrossRef]
- Raven, J.A. Inorganic carbon acquisition by eukaryotic algae: four current questions. Photosynth. Res. 2010, 106, 123–134. [Google Scholar] [CrossRef]
- Trimborn, S.; Lundholm, N.; Thoms, S.; Richter, K.-U.; Krock, B.; Hansen, P.J.; Rost, B. Inorganic carbon acquisition in potentially toxic and non-toxic diatoms: the effect of pH-induced changes in seawater carbonate chemistry. Physiol. Plant 2008, 133, 92–105. [Google Scholar] [Green Version]
- Martin, C.L.; Tortell, P.D. Bicarbonate transport and extracellular carbonic anhydrase in marine diatoms. Physiol. Plant 2008, 133, 106–116. [Google Scholar] [CrossRef]
- Satoh, D.; Hiraoka, Y.; Colman, B.; Matsuda, Y. Physiological and molecular biological characterization of intracellular carbonic anhydrase from the marine diatom Phaeodactylum tricornutum. Plant Physiol. 2001, 126, 1459–1470. [Google Scholar] [CrossRef]
- Nakajima, K.; Tanaka, A.; Matsuda, Y. SLC4 family transporters in a marine diatom directly pump bicarbonate from seawater. Proc. Natl. Acad. Sci. USA. 2013, 110, 1767–1772. [Google Scholar] [CrossRef]
- Muto, M.; Fukuda, Y.; Nemoto, M.; Yoshino, T.; Matsunaga, T.; Tanaka, T. Establishment of a genetic transformation system for the marine pennate diatom Fistulifera sp. strain JPCC DA0580—A high triglyceride producer. Mar. Biotechnol 2013, 15, 48–55. [Google Scholar] [CrossRef]
- Falciatore, A.; Casotti, R.; Leblanc, C.; Abrescia, C.; Bowler, C. Transformation of nonselectable reporter genes in marine diatoms. Mar. Biotechnol. 1999, 1, 239–251. [Google Scholar] [CrossRef]
- Apt, K.E.; Kroth-Pancic, P.G.; Grossman, A.R. Stable nuclear transformation of the diatom Phaeodactylum tricornutum. Mol. Gen. Genet. 1996, 252, 572–579. [Google Scholar]
- Hopkinson, B.M.; Dupont, C.L.; Allen, A.E.; Morel, F.M.M. Efficiency of the CO2-concentrating mechanism of diatoms. Proc. Natl. Acad. Sci. USA. 2011, 108, 3830–3837. [Google Scholar]
- Reinfelder, J.R.; Kraepiel, A.M.; Morel, F.M. Unicellular C4 photosynthesis in a marine diatom. Nature 2000, 407, 996–999. [Google Scholar] [CrossRef]
- McGinn, P.J.; Morel, F.M.M. Expression and inhibition of the carboxylating and decarboxylating enzymes in the photosynthetic C4 pathway of marine diatoms. Plant. Physiol. 2008, 146, 300–309. [Google Scholar] [CrossRef]
- Roberts, K.; Granum, E.; Leegood, R.C.; Raven, J.A. C3 and C4 pathways of photosynthetic carbon assimilation in marine diatoms are under genetic, not environmental, control. Plant Physiol. 2007, 145, 230–235. [Google Scholar] [CrossRef]
- Haimovich-Dayan, M.; Garfinkel, N.; Ewe, D.; Marcus, Y.; Gruber, A.; Wagner, H.; Kroth, P.G.; Kaplan, A. The role of C4 metabolism in the marine diatom Phaeodactylum tricornutum. New Phytol. 2013, 197, 177–185. [Google Scholar] [CrossRef]
- Wilhelm, C.; Büchel, C.; Fisahn, J.; Goss, R.; Jakob, T.; Laroche, J.; Lavaud, J.; Lohr, M.; Riebesell, U.; Stehfest, K.; et al. The regulation of carbon and nutrient assimilation in diatoms is significantly different from green algae. Protist 2006, 157, 91–124. [Google Scholar] [CrossRef]
- Shimonaga, T.; Konishi, M.; Oyama, Y.; Fujiwara, S.; Satoh, A.; Fujita, N.; Colleoni, C.; Buléon, A.; Putaux, J.-L.; Ball, S.G.; et al. Variation in storage alpha-glucans of the Porphyridiales (Rhodophyta). Plant Cell. Physiol. 2008, 49, 103–116. [Google Scholar] [CrossRef]
- Granum, E.; Kirkvold, S.; Myklestad, S. Cellular and extracellular production of carbohydrates and amino acids by the marine diatom Skeletonema costatum: diel variations and effects of N depletion. Mar. Ecol. Prog. Ser. 2002, 242, 83–94. [Google Scholar] [CrossRef]
- Roessler, P.G. UDP-glucose pyrophosphorylase activity in the diatom Cyclotella cryptica. Pathways of chrysolaminarin biosynthesis. J. Phycol. 1987, 23, 494–498. [Google Scholar] [CrossRef]
- Vårum, K.M.; Østgaard, K.; Grimsrud, K. Diurnal rhythms in carbohydrate metabolism of the marine diatom Skeletonema costatum (Grev.) Cleve. J. Exp. Mar. Bio. Ecol. 1986, 102, 249–256. [Google Scholar] [CrossRef]
- Parker, M.S.; Armbrust, E.V.; Piovia-Scott, J.; Keil, R.G. Induction of photorespiration by light in the centric diatom Thalassiosira weissflogii (Bacillariophyceae): Molecular characterization and physiological consequences. J. Phycol. 2004, 40, 557–567. [Google Scholar] [CrossRef]
- Timm, S.; Nunes-Nesi, A.; Pärnik, T.; Morgenthal, K.; Wienkoop, S.; Keerberg, O.; Weckwerth, W.; Kleczkowski, L.A.; Fernie, A.R.; Bauwe, H. A cytosolic pathway for the conversion of hydroxypyruvate to glycerate during photorespiration in Arabidopsis. Plant. Cell. 2008, 20, 2848–2859. [Google Scholar] [CrossRef]
- Tortell, P.D. Evolutionary and ecological perspectives on carbon acquisition in phytoplankton. Limonol. Oceanogr. 2000, 45, 744–750. [Google Scholar]
- Fernie, A.R.; Obata, T.; Allen, A.E.; Araújo, W.L.; Bowler, C. Leveraging metabolomics for functional investigations in sequenced marine diatoms. Trends Plant. Sci. 2012, 17, 395–403. [Google Scholar]
- Allen, A.E.; Dupont, C.L.; Oborník, M.; Horák, A.; Nunes-Nesi, A.; McCrow, J.P.; Zheng, H.; Johnson, D.A.; Hu, H.; Fernie, A.R.; et al. Evolution and metabolic significance of the urea cycle in photosynthetic diatoms. Nature 2011, 473, 203–207. [Google Scholar]
- Martinez, E.; Antoine, D.; D’Ortenzio, F.; Gentili, B. Climate-driven basin-scale decadal oscillations of oceanic phytoplankton. Science 2009, 326, 1253–1256. [Google Scholar]
- Weber, T.; Deutsch, C. Oceanic nitrogen reservoir regulated by plankton diversity and ocean circulation. Nature 2012, 489, 419–422. [Google Scholar] [CrossRef]
- Weber, T.S.; Deutsch, C. Ocean nutrient ratios governed by plankton biogeography. Nature 2010, 467, 550–554. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC), Climate Change 2001; Houghton, J.T.; Ding, Y.; Griggs, D.J.; Noguer, M.; van der Linden, P.J.; Dai, X.; Maskell, K.; Johnson, C.A. (Eds.) Oxford University Press: Oxford, UK, 2001.
- Field, C.B. Primary Production of the Biosphere: Integrating Terrestrial and Oceanic Components. Science 1998, 281, 237–240. [Google Scholar]
- Hein, M.; Sand-Jensen, K. CO2 increases oceanic primary production. 1997; 388, 526–527. [Google Scholar]
- Yang, G.; Gao, K. Physiological responses of the marine diatom Thalassiosira pseudonana to increased pCO2 and seawater acidity. Mar. Environ. Res. 2012, 79, 142–151. [Google Scholar]
- Gao, K.; Xu, J.; Gao, G.; Li, Y.; Hutchins, D.A.; Huang, B.; Wang, L.; Zheng, Y.; Jin, P.; Cai, X.; et al. Rising CO2 and increased light exposure synergistically reduce marine primary productivity. Nat. Clim. Change 2012, 2, 519–523. [Google Scholar]
- Crawfurd, K.J.; Raven, J.A.; Wheeler, G.L.; Baxter, E.J.; Joint, I. The response of Thalassiosira pseudonana to long-term exposure to increased CO2 and decreased pH. PloS one 2011, 6, e26695. [Google Scholar]
- Boyd, P.W.; Watson, A.J.; Law, C.S.; Abraham, E.R.; Trull, T.; Murdoch, R.; Bakker, D.C.; Bowie, A.R.; Buesseler, K.O.; Chang, H.; et al. A mesoscale phytoplankton bloom in the polar Southern Ocean stimulated by iron fertilization. Nature 2000, 407, 695–702. [Google Scholar]
- Coale, K.H.; Johnson, K.S.; Chavez, F.P.; Buesseler, K.O.; Barber, R.T.; Brzezinski, M.A.; Cochlan, W.P.; Millero, F.J.; Falkowski, P.G.; Bauer, J.E.; et al. Southern Ocean iron enrichment experiment: carbon cycling in high- and low-Si waters. Science 2004, 304, 408–414. [Google Scholar]
- Boyd, P.W.; Jickells, T.; Law, C.S.; Blain, S.; Boyle, E.A.; Buesseler, K.O.; Coale, K.H.; Cullen, J.J.; De Baar, H.J.W.; Follows, M.; et al. Mesoscale iron enrichment experiments 1993–2005: synthesis and future directions. Science 2007, 315, 612–617. [Google Scholar]
- Cao, L.; Caldeira, K. Can ocean iron fertilization mitigate ocean acidification? Clim. Change 2010, 99, 303–311. [Google Scholar]
- Strzepek, R.F.; Harrison, P.J. Photosynthetic architecture differs in coastal and oceanic diatoms. Nature 2004, 431, 689–692. [Google Scholar]
- Peers, G.; Price, N.M. Copper-containing plastocyanin used for electron transport by an oceanic diatom. Nature 2006, 441, 341–344. [Google Scholar]
- Kustka, A.B.; Allen, A.E.; Morel, F.M.M. Sequence analysis and transcriptional regulation of iron acquisition genes in two marine diatoms. J. Phycol. 2007, 43, 715–729. [Google Scholar]
- Allen, A.E.; Laroche, J.; Maheswari, U.; Lommer, M.; Schauer, N.; Lopez, P.J.; Finazzi, G.; Fernie, A.R.; Bowler, C. Whole-cell response of the pennate diatom Phaeodactylum tricornutum to iron starvation. Proc. Natl. Acad. Sci. USA 2008, 105, 10438–10443. [Google Scholar]
- Schirmer, K.; Fischer, B.B.; Madureira, D.J.; Pillai, S. Transcriptomics in ecotoxicology. Anal. Bioanal. Chem. 2010, 397, 917–923. [Google Scholar]
- Diggs, D.L.; Huderson, A.C.; Harris, K.L.; Myers, J.N.; Banks, L.D.; Rekhadevi, P. V.; Niaz, M.S.; Ramesh, A. Polycyclic aromatic hydrocarbons and digestive tract cancers: a perspective. J. Environ. Sci. Heal. C 2011, 29, 324–357. [Google Scholar]
- Tobiszewski, M.; Namieśnik, J. PAH diagnostic ratios for the identification of pollution emission sources. Environ. Pollut. 2012, 162, 110–119. [Google Scholar]
- Carvalho, R.N.; Bopp, S.K.; Lettieri, T. Transcriptomics responses in marine diatom Thalassiosira pseudonana exposed to the polycyclic aromatic hydrocarbon benzo[a]pyrene. PloS One 2011, 6, e26985. [Google Scholar]
- Becker, E.W. Microalgae: Biotechnology and Microbiology; Cambridge University Press: Cambridge, UK, 1994; p. 293. [Google Scholar]
- Lee, Y.K. Microalgal mass culture systems and methods: Their limitation and potential. J. Appl. Phycol. 2001, 13, 307–315. [Google Scholar]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar]
- Harun, R.; Singh, M.; Forde, G.M.; Danquah, M.K. Bioprocess engineering of microalgae to produce a variety of consumer products. Renew. Sust. Energ. Rev. 2010, 14, 1037–1047. [Google Scholar]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar]
- Schenk, P.M.; Thomas-Hall, S.R.; Stephens, E.; Marx, U.C.; Mussgnug, J.H.; Posten, C.; Kruse, O.; Hankamer, B. Second generation biofuels: High-efficiency microalgae for biodiesel production. Bioenerg. Res. 2008, 1, 20–43. [Google Scholar]
- Sheehan, J.; Dunahay, T.; Benemann, J.; Roessler, P. Look Back at the U.S. Department of Energy’s Aquatic Species Program: Biodiesel from Algae. Close-Out Report, NREL/TP-580-24190. 1998. [Google Scholar]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [Green Version]
- Khozin-Goldberg, I.; Cohen, Z. Unraveling algal lipid metabolism: Recent advances in gene identification. Biochimie 2011, 93, 91–100. [Google Scholar]
- Wijffels, R.H.; Barbosa, M.J. An outlook on microalgal biofuels. Science 2010, 329, 796–799. [Google Scholar]
- Caldana, C.; Li, Y.; Leisse, A.; Zhang, Y.; Bartholomaeus, L.; Fernie, A.R.; Willmitzer, L.; Giavalisco, P. Systemic analysis of inducible target of rapamycin mutants reveal a general metabolic switch controlling growth in Arabidopsis thaliana. Plant J. 2013. [Google Scholar] [CrossRef]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar]
- Derelle, E.; Ferraz, C.; Rombauts, S.; Rouzé, P.; Worden, A.Z.; Robbens, S.; Partensky, F.; Degroeve, S.; Echeynié, S.; Cooke, R.; et al. Genome analysis of the smallest free-living eukaryote Ostreococcus tauri unveils many unique features. Proc. Natl. Acad. Sci. USA 2006, 103, 11647–11652. [Google Scholar]
- Merchant, S.S.; Prochnik, S.E.; Vallon, O.; Harris, E.H.; Karpowicz, S.J.; Witman, G.B.; Terry, A.; Salamov, A.; Fritz-Laylin, L.K.; Maréchal-Drouard, L.; et al. The Chlamydomonas genome reveals the evolution of key animal and plant functions. Science 2007, 318, 245–250. [Google Scholar]
- Price, D.C.; Chan, C.X.; Yoon, H.S.; Yang, E.C.; Qiu, H.; Weber, A.P.M.; Schwacke, R.; Gross, J.; Blouin, N.A.; Lane, C.; et al. Cyanophora paradoxa genome elucidates origin of photosynthesis in algae and plants. Science 2012, 335, 843–847. [Google Scholar]
- Tirichine, L.; Bowler, C. Decoding algal genomes: tracing back the history of photosynthetic life on Earth. Plant J. 2011, 66, 45–57. [Google Scholar]
- Gong, Y.; Zhang, J.; Guo, X.; Wan, X.; Liang, Z.; Hu, C.J.; Jiang, M. Identification and characterization of PtDGAT2B, an acyltransferase of the DGAT2 acyl-coenzyme A: diacylglycerol acyltransferase family in the diatom Phaeodactylum tricornutum. FEBS Lett. 2013, 587, 481–487. [Google Scholar]
- Guihéneuf, F.; Leu, S.; Zarka, A.; Khozin-Goldberg, I.; Khalilov, I.; Boussiba, S. Cloning and molecular characterization of a novel acyl-CoA:diacylglycerol acyltransferase 1-like gene (PtDGAT1) from the diatom Phaeodactylum tricornutum. FEBS J. 2011, 278, 3651–3666. [Google Scholar]
- Merchant, S.S.; Kropat, J.; Liu, B.; Shaw, J.; Warakanont, J. TAG, you’re it! Chlamydomonas as a reference organism for understanding algal triacylglycerol accumulation. Curr. Opin. Biotechnol. 2012, 23, 352–363. [Google Scholar]
- Riekhof, W.R.; Sears, B.B.; Benning, C. Annotation of genes involved in glycerolipid biosynthesis in Chlamydomonas reinhardtii: discovery of the betaine lipid synthase BTA1Cr. Eukaryot. Cell 2005, 4, 242–252. [Google Scholar]
- Liu, B.; Benning, C. Lipid metabolism in microalgae distinguishes itself. Curr. Opin. Biotechnol. 2012. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Fats, oils and hydrocarbons. In Microalgal biotechnology; Borowitzka, M.A., Borowitzka, L.J., Eds.; Cambridge University Press: Cambridge, UK, 1988; pp. 257–287. [Google Scholar]
- Dunstan, G.A.; Volkman, J.K.; Barrett, S.M.; Leroi, J.-M.; Jeffrey, S.W. Essential polyunsaturated fatty acids from 14 species of diatom (Bacillariophyceae). Phytochemistry 1993, 35, 155–161. [Google Scholar]
- Yu, E.T.; Zendejas, F.J.; Lane, P.D.; Gaucher, S.; Simmons, B.A.; Lane, T.W. Triacylglycerol accumulation and profiling in the model diatoms Thalassiosira pseudonana and Phaeodactylum tricornutum (Baccilariophyceae) during starvation. J. Appl. Phycol. 2009, 21, 669–681. [Google Scholar]
- Rezanka, T.; Lukavský, J.; Nedbalová, L.; Kolouchová, I.; Sigler, K. Effect of starvation on the distribution of positional isomers and enantiomers of triacylglycerol in the diatom Phaeodactylum tricornutum. Phytochemistry 2012, 80, 17–27. [Google Scholar]
- Wang, Z.T.; Ullrich, N.; Joo, S.; Waffenschmidt, S.; Goodenough, U. Algal lipid bodies: stress induction, purification, and biochemical characterization in wild-type and starchless Chlamydomonas reinhardtii. Eukaryot. Cell. 2009, 8, 1856–1868. [Google Scholar]
- Moellering, E.R.; Benning, C. RNA interference silencing of a major lipid droplet protein affects lipid droplet size in Chlamydomonas reinhardtii. Eukaryot. Cell 2010, 9, 97–106. [Google Scholar]
- Miller, R.; Wu, G.; Deshpande, R.R.; Vieler, A.; Gärtner, K.; Li, X.; Moellering, E.R.; Zäuner, S.; Cornish, A.J.; Liu, B.; et al. Changes in transcript abundance in Chlamydomonas reinhardtii following nitrogen deprivation predict diversion of metabolism. Plant. Physiol. 2010, 154, 1737–1752. [Google Scholar]
- Mock, T.; Samanta, M.P.; Iverson, V.; Berthiaume, C.; Robison, M.; Holtermann, K.; Durkin, C.; Bondurant, S.S.; Richmond, K.; Rodesch, M.; et al. Whole-genome expression profiling of the marine diatom Thalassiosira pseudonana identifies genes involved in silicon bioprocesses. Proc. Natl. Acad. Sci. USA. 2008, 105, 1579–1584. [Google Scholar]
- Stehfest, K.; Toepel, J.; Wilhelm, C. The application of micro-FTIR spectroscopy to analyze nutrient stress-related changes in biomass composition of phytoplankton algae. Plant Physiol. Biochem. 2005, 43, 717–726. [Google Scholar]
- Quigg, A.; Beardall, J. Protein turnover in relation to maintenance metabolism at low photon flux in two marine microalgae. Plant. Cell. Environ. 2003, 26, 693–703. [Google Scholar]
- Jiang, Y.; Yoshida, T.; Quigg, A. Photosynthetic performance, lipid production and biomass composition in response to nitrogen limitation in marine microalgae. Plant. Physiol. Biochem. 2012, 54, 70–77. [Google Scholar]
- Lombardi, A.; Wangersky, P. Influence of phosphorus and silicon on lipid class production by the marine diatom Chaetoceros gracilis grown in turbidostat cage cultures. Mar. Ecol. Prog. Ser. 1991, 77, 39–47. [Google Scholar]
- McGinnis, K.M.; Dempster, T.A.; Sommerfeld, M.R. Characterization of the growth and lipid content of the diatom Chaetoceros muelleri. J. Appl. Phycol. 1997, 9, 19–24. [Google Scholar]
- Roessler, P.G. Changes in the activities of various lipid and carbohydrate biosynthetic enzymes in the diatom Cyclotella cryptica in response to silicon deficiency. Arch. Biochem. Biophys. 1988, 267, 521–528. [Google Scholar]
- Huang, A.; He, L.; Wang, G. Identification and characterization of microRNAs from Phaeodactylum tricornutum by high-throughput sequencing and bioinformatics analysis. BMC Genomics 2011, 12, 337. [Google Scholar]
- Reitan, K.I.; Rainuzzo, J.R.; Olsen, Y. Effect of nutrient limitation of fatty acid and lipid content of marine microalgae. J. Phycol. 1994, 30, 972–979. [Google Scholar]
- Van Mooy, B.A.S.; Fredricks, H.F.; Pedler, B.E.; Dyhrman, S.T.; Karl, D.M.; Koblízek, M.; Lomas, M.W.; Mincer, T.J.; Moore, L.R.; Moutin, T.; et al. Phytoplankton in the ocean use non-phosphorus lipids in response to phosphorus scarcity. Nature 2009, 458, 69–72. [Google Scholar]
- Martin, P.; Van Mooy, B.A.S.; Heithoff, A.; Dyhrman, S.T. Phosphorus supply drives rapid turnover of membrane phospholipids in the diatom Thalassiosira pseudonana. ISME J. 2011, 5, 1057–1060. [Google Scholar]
- Dyhrman, S.T.; Jenkins, B.D.; Rynearson, T.A.; Saito, M.A.; Mercier, M.L.; Alexander, H.; Whitney, L.P.; Drzewianowski, A.; Bulygin, V.V; Bertrand, E.M.; et al. The transcriptome and proteome of the diatom Thalassiosira pseudonana reveal a diverse phosphorus stress response. PloS One 2012, 7, e33768. [Google Scholar]
- Alonso, D.L.; Belarbi, E.H.; Fernández-Sevilla, J.M.; Rodríguez-Ruiz, J.; Molina Grima, E. Acyl lipid composition variation related to culture age and nitrogen concentration in continuous culture of the microalga Phaeodactylum tricornutum. Phytochemistry 2000, 54, 461–471. [Google Scholar]
- Liang, Y.; Beardall, J.; Heraud, P. Changes in growth, chlorophyll fluorescence and fatty acid composition with culture age in batch cultures of Phaeodactylum tricornutum and Chaetoceros muelleri (Bacillariophyceae). Bot. Mar. 2006, 49, 165–173. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Obata, T.; Fernie, A.R.; Nunes-Nesi, A. The Central Carbon and Energy Metabolism of Marine Diatoms. Metabolites 2013, 3, 325-346. https://doi.org/10.3390/metabo3020325
Obata T, Fernie AR, Nunes-Nesi A. The Central Carbon and Energy Metabolism of Marine Diatoms. Metabolites. 2013; 3(2):325-346. https://doi.org/10.3390/metabo3020325
Chicago/Turabian StyleObata, Toshihiro, Alisdair R. Fernie, and Adriano Nunes-Nesi. 2013. "The Central Carbon and Energy Metabolism of Marine Diatoms" Metabolites 3, no. 2: 325-346. https://doi.org/10.3390/metabo3020325



