Gas-Chromatography Mass-Spectrometry (GC-MS) Based Metabolite Profiling Reveals Mannitol as a Major Storage Carbohydrate in the Coccolithophorid Alga Emiliania huxleyi
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of Sample Preparation Protocol for E. huxleyi

| Metabolite | Cell number |
|---|---|
| Amino acids | |
| Isoleucine | 2 |
| Valine | 2 |
| Leucine | 5 |
| Threonine | 5 |
| Serine | 5 |
| Alanine | 10 |
| Aspartate | 10 |
| Glutamate | 10 |
| Glycine | 10 |
| Lysine | 10 |
| O-acetylserine | 10 |
| Phenylalanine | 10 |
| Organic acids | |
| Dehydroascorbate | 1 |
| Citrate | 5 |
| Glycerate | 5 |
| Threonate | 5 |
| Fatty acids | |
| Dodecanoate* | 2 |
| Decanoate | 5 |
| Sugars and sugar alcohols | |
| Mannitol* | 1 |
| Glucose | 2 |
| myo-inositol | 2 |
| Ribose | 2 |
| Sucrose | 2 |
| Maltotriose | 5 |
| Fructose | 10 |
| Tocopherol | |
| α-tocopherol | 10 |
| Metabolite | Recovery in % |
|---|---|
| Alanine | 87.79 ± 1.55 |
| Aspartate | 34.11 ± 1.27 |
| Decanoate | 149.30 ± 4.71 |
| Dehydroascorbate | 86.38 ± 1.91 |
| Dodecanoate | 121.54 ± 11.23 |
| Fructose | 82.47 ± 24.30 |
| Glucose | 166.95 ± 14.08 |
| Glutamate | 53.93 ± 3.49 |
| Glycerate | 94.042 ± 3.86 |
| Glycine | 91.64 ± 2.84 |
| Isoleucine | 94.63 ± 2.93 |
| Leucine | 89.90 ± 1.94 |
| Lysine | 73.82 ± 3.48 |
| Maltotriose | 65.45 ± 0.93 |
| Mannitol | 102.87 ± 3.97 |
| Myoinositol | 88.40 ± 2.60 |
| Phenylalanine | 71.02 ± 2.53 |
| Ribose | 129.13 ± 5.13 |
| Serine | 64.95 ± 2.19 |
| Sucrose | 106.86 ± 4.52 |
| Threonate | 88.34 ± 4.52 |
| Threonine | 81.14 ± 2.17 |
| Valine | 97.26 ± 1.98 |
| alpha-tocopherol | 71.97 ± 3.40 |
2.2. Metabolite Profiling Revealed Metabolic Features of E. huxleyi
2.2.1. High Mannitol Content and Faint Amounts of Fructose and Sucrose
2.2.2. Detection of Antioxidants
2.2.3. Low Contents of Malate and Fumarate
2.3. 13C-Label Accumulation Analysis Suggested Large Metabolic Flux into C3 Pathway and Mannitol Synthesis

2.3.1. The C3 Cycle is Dominant for CO2 Fixation in E. huxleyi
2.3.2. Extremely High Flux into Mannitol
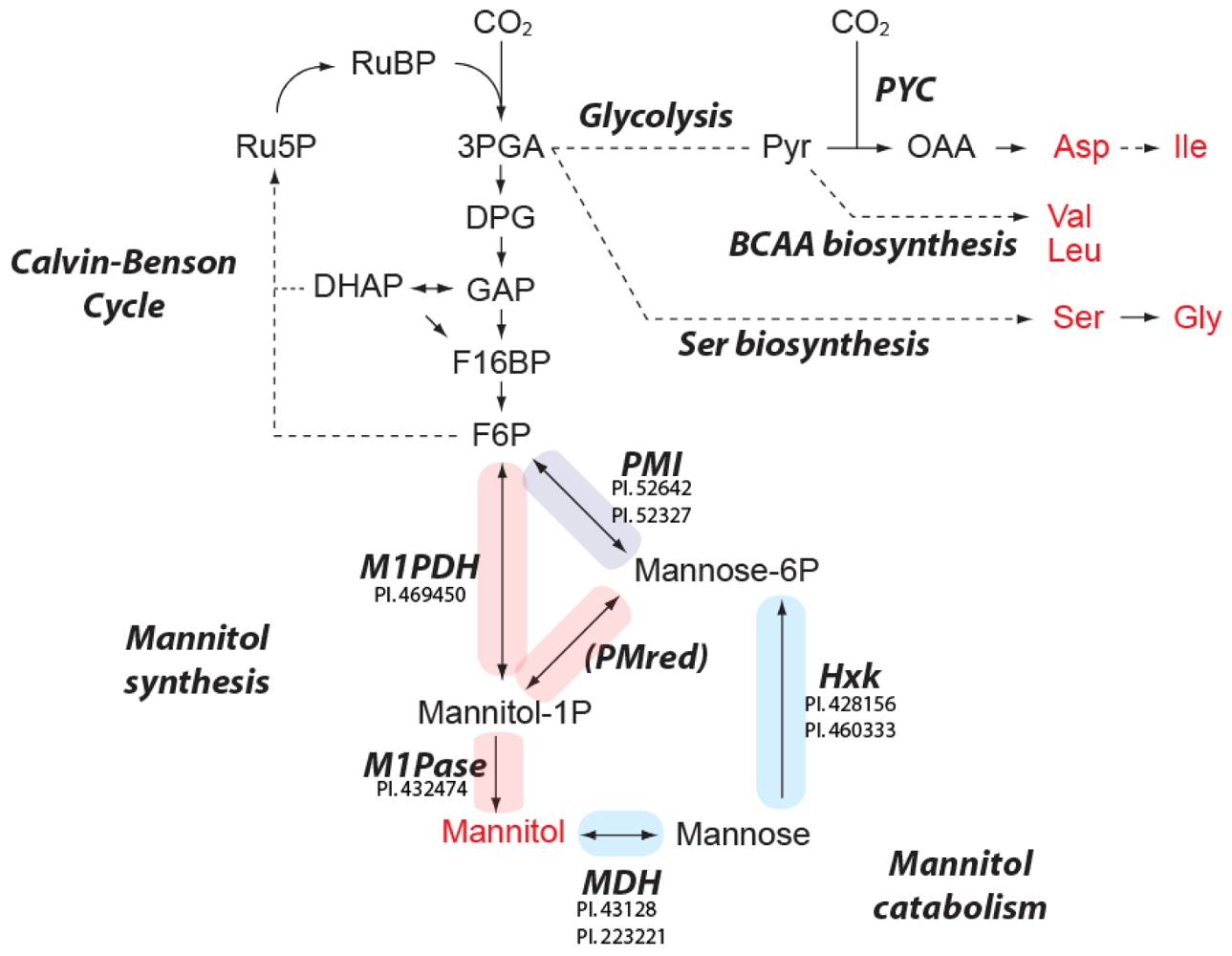
2.4. Predicted Pathways for Mannitol Metabolism in E. huxleyi
3. Experimental
3.1. Algal Strain and Culture Condition
3.2. Sampling and Extraction Procedure for Metabolite Profiling
3.3. GC-MS Based Metabolite Profiling
3.4. H13CO3− Feeding and Label Accumulation Analysis
3.5. Identification of Enzymes Involved in Mannitol Biosynthesis and Catabolism
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Falkowski, P.G.; Katz, M.E.; Knoll, A.H.; Quigg, A.; Raven, J.A.; Schofield, O.; Taylor, F.J.R. The evolution of modern eukaryotic phytoplankton. Science 2004, 305, 354–360. [Google Scholar] [CrossRef]
- Keeling, P.J.; Palmer, J.D. Horizontal gene transfer in eukaryotic evolution. Nat. Rev. Genet. 2008, 9, 605–618. [Google Scholar] [CrossRef]
- Bowler, C.; Allen, A.E.; Badger, J.H.; Grimwood, J.; Jabbari, K.; Kuo, A.; Maheswari, U.; Martens, C.; Maumus, F.; Otillar, R.P.; et al. The Phaeodactylum genome reveals the evolutionary history of diatom genomes. Nature 2008, 456, 239–244. [Google Scholar]
- Fernie, A.R.; Obata, T.; Allen, A.E.; Araújo, W.L.; Bowler, C. Leveraging metabolomics for functional investigations in sequenced marine diatoms. Trends Plant. Sci. 2012, 17, 395–403. [Google Scholar] [CrossRef]
- Raven, J.A. Cellular location of starch synthesis and evolutionary origin of starch genes. J. Phycol. 2005, 41, 1070–1072. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Raven, J.A. Aquatic Photosynthesis; Blackwell Science: Malden, MA, USA, 1997; p. 375. [Google Scholar]
- Obata, T.; Fernie, A.R. The use of metabolomics to dissect plant responses to abiotic stresses. Cell. Mol. Life Sci. 2012, 69, 3225–3243. [Google Scholar] [CrossRef]
- Huege, J.; Krall, L.; Steinhauser, M.C.; Giavalisco, P.; Rippka, R.; Tandeau de Marsac, N.; Steinhauser, D. Sample amount alternatives for data adjustment in comparative cyanobacterial metabolomics. Anal. Bioanal. Chem. 2011, 399, 3503–3517. [Google Scholar] [CrossRef]
- Malin, G.; Steinke, M. Dimethyl sulfide production: What is the contribution of coccolithopores? In Coccolithophores: From Molecular Processes to Global Impact; Thierstein, H.R., Young, J.R., Eds.; Springer-Verlag: Berlin Heidelberg, Germany, 2004; pp. 127–164. [Google Scholar]
- Tyrrell, T.; Taylor, A. A modelling study of Emiliania huxleyi in the NE atlantic. J. Mar. Syst. 1996, 9, 83–112. [Google Scholar] [CrossRef]
- Joassin, P.; Delille, B.; Soetaert, K.; Harlay, J.; Borges, A.V.; Chou, L.; Riebesell, U.; Suykens, K.; Grégoire, M. Carbon and nitrogen flows during a bloom of the coccolithophore Emiliania huxleyi: Modelling a mesocosm experiment. J.Mar. Syst. 2011, 85, 71–85. [Google Scholar] [CrossRef]
- Tsuji, Y.; Suzuki, I.; Shiraiwa, Y. Photosynthetic carbon assimilation in the coccolithophorid Emiliania huxleyi (Haptophyta): Evidence for the predominant operation of the C3 cycle and the contribution of β-carboxylases to the active anaplerotic reaction. Plant. Cell. Physiol. 2009, 50, 318–329. [Google Scholar] [CrossRef]
- Tsuji, Y.; Suzuki, I.; Shiraiwa, Y. Enzymological evidence for the function of a plastid-located pyruvate carboxylase in the Haptophyte alga Emiliania huxleyi: A novel pathway for the production of C4 compounds. Plant. Cell. Physiol. 2012, 53, 1043–1052. [Google Scholar] [CrossRef]
- Obata, T.; Araie, H.; Shiraiwa, Y. Bioconcentration mechanism of selenium by a coccolithophorid, Emiliania huxleyi. Plant. Cell. Physiol. 2004, 45, 1434–1441. [Google Scholar] [CrossRef]
- Obata, T.; Shiraiwa, Y. A novel eukaryotic selenoprotein in the haptophyte alga Emiliania huxleyi. J. Biol. Chem. 2005, 280, 18462–18468. [Google Scholar] [CrossRef]
- Araie, H.; Shiraiwa, Y. Selenium utilization strategy by microalgae. Molecules 2009, 14, 4880–4891. [Google Scholar] [CrossRef]
- Emiliania huxleyi CCMP1516 main genome assembly v1.0 in the Joint Genome Institute (JGI) Genome Portal website. Available online: http://genome.jgi-psf.org/Emihu1/Emihu1.home.html/ (accesed on 23 January 2013).
- Roessner-Tunali, U. Metabolic profiling of transgenic tomato plants overexpressing hexokinase reveals that the influence of hexose phosphorylation diminishes during fruit development. Plant. Physiol. 2003, 133, 84–99. [Google Scholar] [CrossRef]
- Rumpho, M.E.; Edwards, G.E.; Loescher, W.H. A pathway for photosynthetic carbon flow to mannitol in celery leaves: activity and localization of key enzymes. Plant. Physiol. 1983, 73, 869–873. [Google Scholar]
- Iwamoto, K.; Shiraiwa, Y. Salt-regulated mannitol metabolism in algae. Mar. Biotechnol. 2005, 7, 407–415. [Google Scholar] [CrossRef]
- Stoop, J.M.H.; Williamson, J.D.; Pharr, M.D. Mannitol metabolism in plants: a method for coping with stress. Trends Plant. Sci. 1996, 1, 139–144. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Ikawa, T.; Nishizawa, K. Pathway of mannitol formation during photosynthesis in brown algae. Plant. Cell. Physiol. 1969, 10, 425–440. [Google Scholar]
- Schmitz, K.; Srivastava, L.M. On the fine structure of sieve tubes and the physiology of assimilate transport in Alaria marginata. Can.J. Bot. 1975, 53, 861–876. [Google Scholar] [CrossRef]
- Kremer, B.P.; Willenbrink, J. CO2-Fixierung und Stofftransport in benthischen marinen Algen. Planta 1972, 103, 55–64. [Google Scholar] [CrossRef]
- Simpson, G.L.W.; Ortwerth, B.J. The non-oxidative degradation of ascorbic acid at physiological conditions. Biochim. Biophys. Acta 2000, 1501, 12–24. [Google Scholar] [CrossRef]
- Munné-Bosch, S. The role of α-tocopherol in plant stress tolerance. J. Plant. Physiol. 2005, 162, 743–748. [Google Scholar] [CrossRef]
- Martinoia, E.; Rentsch, D. Malate compartmentation-responses to a complex metabolism. Annu. Rev. Plant. Physiol. Plant. Mol. Biol. 1994, 45, 447–467. [Google Scholar] [CrossRef]
- Fernie, A.R.; Martinoia, E. Malate. Jack of all trades or master of a few? Phytochemistry 2009, 70, 828–832. [Google Scholar] [CrossRef] [Green Version]
- Pracharoenwattana, I.; Zhou, W.; Keech, O.; Francisco, P.B.; Udomchalothorn, T.; Tschoep, H.; Stitt, M.; Gibon, Y.; Smith, S.M. Arabidopsis has a cytosolic fumarase required for the massive allocation of photosynthate into fumaric acid and for rapid plant growth on high nitrogen. Plant. J. 2010, 62, 785–795. [Google Scholar]
- Chia, D.W.; Yoder, T.J.; Reiter, W.-D.; Gibson, S.I. Fumaric acid: an overlooked form of fixed carbon in Arabidopsis and other plant species. Planta 2000, 211, 743–751. [Google Scholar] [CrossRef]
- Steinhauser, D.; Fernie, A.R.; Araújo, W.L. Unusual cyanobacterial TCA cycles: not broken just different. Trends Plant. Sci. 2012, 17, 503–509. [Google Scholar] [CrossRef]
- Hockin, N.L.; Mock, T.; Mulholland, F.; Kopriva, S.; Malin, G. The response of diatom central carbon metabolism to nitrogen starvation is different from that of green algae and higher plants. Plant. Physiol. 2012, 158, 299–312. [Google Scholar] [CrossRef] [Green Version]
- Allen, A.E.; Dupont, C.L.; Oborník, M.; Horák, A.; Nunes-Nesi, A.; McCrow, J.P.; Zheng, H.; Johnson, D.A.; Hu, H.; Fernie, A.R.; et al. Evolution and metabolic significance of the urea cycle in photosynthetic diatoms. Nature 2011, 473, 203–207. [Google Scholar]
- Szecowka, M.; Heise, R.; Tohge, T.; Nunes-Nesi, A.; Huege, J.; Feil, R.; Lunn, J.; Nikoloski, Z.; Stitt, M.; Fernie, A.R.; et al. Metabolic fluxes of an illuminated Arabidopsis thaliana rosette. Plant Cell 2013, in press. [Google Scholar]
- Keys, A.J. The re-assimilation of ammonia produced by photorespiration and the nitrogen economy of C3 higher plants. Photosynth. Res. 2006, 87, 165–175. [Google Scholar] [CrossRef]
- Michel, G.; Tonon, T.; Scornet, D.; Cock, J.M.; Kloareg, B. Central and storage carbon metabolism of the brown alga Ectocarpus siliculosus: insights into the origin and evolution of storage carbohydrates in Eukaryotes. New Phytol. 2010, 188, 67–81. [Google Scholar]
- Dittami, S.M.; Aas, H.T.N.; Paulsen, B.S.; Boyen, C.; Edvardsen, B.; Tonon, T. Mannitol in six autotrophic stramenopiles and Micromonas. Plant. Signal. Behav. 2011, 6, 1237–1239. [Google Scholar] [CrossRef]
- Kroth, P. G.; Chiovitti, A.; Gruber, A.; Martin-Jezequel, V.; Mock, T.; Parker, M. S.; Stanley, M. S.; Kaplan, A.; Caron, L.; Weber, T.; et al. A model for carbohydrate metabolism in the diatom Phaeodactylum tricornutum deduced from comparative whole genome analysis. PloS one 2008, 3, e1426. [Google Scholar] [CrossRef]
- Rokitta, S. D.; John, U.; Rost, B. Ocean acidification affects redox-balance and ion-homeostasis in the life-cycle stages of Emiliania huxleyi. PloS one 2012, 7, e52212. [Google Scholar] [CrossRef] [Green Version]
- Loescher, W.H.; Everard, J.D.; Cantini, C.; Grumet, R. Sugar alcohol metabolism in source leaves. Curr. Top. Plant. Physiol. 1995, 13, 170–179. [Google Scholar]
- Pharr, D.M.; Stoop, J.M.H.; Williamson, J.D.; Feusi, M.E. S.; Massel, M.O.; Conkling, M.A. The dual role of mannitol as osmoprotectant and photoassimilate in celery. HortScience 1995, 30, 1182–1188. [Google Scholar]
- Vårum, K.M.; Kvam, B.J.; Myklestad, S. Structure of a food-reserve β-d-glucan produced by the haptophyte alga Emiliania huxleyi (Lohmann) Hay and Mohler. Carbohydr. Res. 1986, 152, 243–248. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Ikawa, T.; Nisizawa, K. Incorporation of radioactive carbon from H14CO3− into sugar constituents by a brown alga, Eisenia bicyclis, during photosynthesis and its fate in the dark. Plant. Cell. Physiol. 1966, 7, 217–229. [Google Scholar]
- Read, S.M.; Currie, G.; Bacic, A. Analysis of the structural heterogeneity of laminarin by electrospray-ionisation-mass spectrometry. Carbohydr.Res. 1996, 281, 187–201. [Google Scholar] [CrossRef]
- Gravot, A.; Dittami, S.M.; Rousvoal, S.; Lugan, R.; Eggert, A.; Collén, J.; Boyen, C.; Bouchereau, A.; Tonon, T. Diurnal oscillations of metabolite abundances and gene analysis provide new insights into central metabolic processes of the brown alga Ectocarpus siliculosus. New Phytol. 2010, 188, 98–110. [Google Scholar] [CrossRef]
- Schneider, K.H.; Giffhorn, F.; Kaplan, S. Cloning, nucleotide sequence and characterization of the mannitol dehydrogenase gene from Rhodobacter sphaeroides. J. Gen. Microbiol. 1993, 139, 2475–2484. [Google Scholar]
- Sunda, W.; Price, N.; Morel, F. Trace metal ion buffers and their use in culture studies. In Algal Culturing Techniques; Andersen, R.A., Ed.; Elsevier: Amsterdam, The Neatherland, 2005; pp. 35–63. [Google Scholar]
- De Marsac, N.T.; Houmard, J. Complementary chromatic adaptation: Physiological conditions and action spectra. Methods Enzymol. 1988, 167, 318–328. [Google Scholar] [CrossRef]
- Lisec, J.; Schauer, N.; Kopka, J.; Willmitzer, L.; Fernie, A.R. Gas chromatography mass spectrometry-based metabolite profiling in plants. Nat. Protoc. 2006, 1, 387–396. [Google Scholar] [CrossRef]
- Luedemann, A.; von Malotky, L.; Erban, A.; Kopka, J. TagFinder: preprocessing software for the fingerprinting and the profiling of gas chromatography-mass spectrometry based metabolome analyses. Methods Mol. Biol. 2012, 860, 255–286. [Google Scholar]
- Kopka, J.; Schauer, N.; Krueger, S.; Birkemeyer, C.; Usadel, B.; Bergmüller, E.; Dörmann, P.; Weckwerth, W.; Gibon, Y.; Stitt, M.; Willmitzer, L.; Fernie, A.R.; Steinhauser, D. [email protected]: The Golm Metabolome Database. Bioinformatics 2005, 21, 1635–1638. [Google Scholar] [CrossRef]
- Fernie, A.R.; Aharoni, A.; Willmitzer, L.; Stitt, M.; Tohge, T.; Kopka, J.; Carroll, A.J.; Saito, K.; Fraser, P.D.; DeLuca, V. Recommendations for reporting metabolite data. Plant. Cell. 2011, 23, 2477–2482. [Google Scholar] [CrossRef]
- Huege, J.; Sulpice, R.; Gibon, Y.; Lisec, J.; Koehl, K.; Kopka, J. GC-EI-TOF-MS analysis of in vivo carbon-partitioning into soluble metabolite pools of higher plants by monitoring isotope dilution after 13CO2 labelling. Phytochemistry 2007, 68, 2258–2272. [Google Scholar] [CrossRef]
- Basic Local Alignment Search Tool in the National Center for Biotechnology Information (NCBI) Website. Available online: http://blast.ncbi.nlm.nih.gov/Blast.cgi/ (accessed on 23 January 2013).
- Lohbeck, K.T.; Riebesell, U.; Reusch, T.B.H. Adaptive evolution of a key phytoplankton species to ocean acidification. Nat. Geosci. 2012, 5, 346–351. [Google Scholar] [CrossRef]
- Paasche, E. A review of the coccolithophorid Emiliania huxleyi (Prymnesiophyceae), with particular reference to growth, coccolith formation, and calcification-photosynthesis interactions. Phycologia 2001, 40, 503–529. [Google Scholar] [CrossRef]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Obata, T.; Schoenefeld, S.; Krahnert, I.; Bergmann, S.; Scheffel, A.; Fernie, A.R. Gas-Chromatography Mass-Spectrometry (GC-MS) Based Metabolite Profiling Reveals Mannitol as a Major Storage Carbohydrate in the Coccolithophorid Alga Emiliania huxleyi. Metabolites 2013, 3, 168-184. https://doi.org/10.3390/metabo3010168
Obata T, Schoenefeld S, Krahnert I, Bergmann S, Scheffel A, Fernie AR. Gas-Chromatography Mass-Spectrometry (GC-MS) Based Metabolite Profiling Reveals Mannitol as a Major Storage Carbohydrate in the Coccolithophorid Alga Emiliania huxleyi. Metabolites. 2013; 3(1):168-184. https://doi.org/10.3390/metabo3010168
Chicago/Turabian StyleObata, Toshihiro, Steffi Schoenefeld, Ina Krahnert, Susan Bergmann, André Scheffel, and Alisdair R. Fernie. 2013. "Gas-Chromatography Mass-Spectrometry (GC-MS) Based Metabolite Profiling Reveals Mannitol as a Major Storage Carbohydrate in the Coccolithophorid Alga Emiliania huxleyi" Metabolites 3, no. 1: 168-184. https://doi.org/10.3390/metabo3010168




