Biology of Bony Fish Macrophages
Abstract
:1. Introduction
2. Macrophage Development
2.1. Teleost Embryonic Monopoiesis
2.2. Teleost Adult Monopoiesis
2.3. Roles of Colony-Stimulating Factor-1 in Teleost Monopoieis
3. Classical Macrophage Activation
3.1. Molecular Mechanisms of Classical Activation
3.1.1. Type II Interferons
3.1.2. Tumor Necrosis Factor-Alpha
3.1.3. Granulocyte-Macrophage Colony-Stimulating Factor
3.2. M1 Macrophage Cytokine Profiles
4. Antimicrobial Roles of Teleost M1 Macrophages
4.1. Respiratory Burst Response
4.2. Nitric Oxide Response
4.3. Phagolysosome Fusion
4.4. Nutrient Deprivation
4.4.1. Solute Carrier 11 Member 1
4.4.2. Ferroportin
4.4.3. Indoleamine 2,3-Dioxygenase
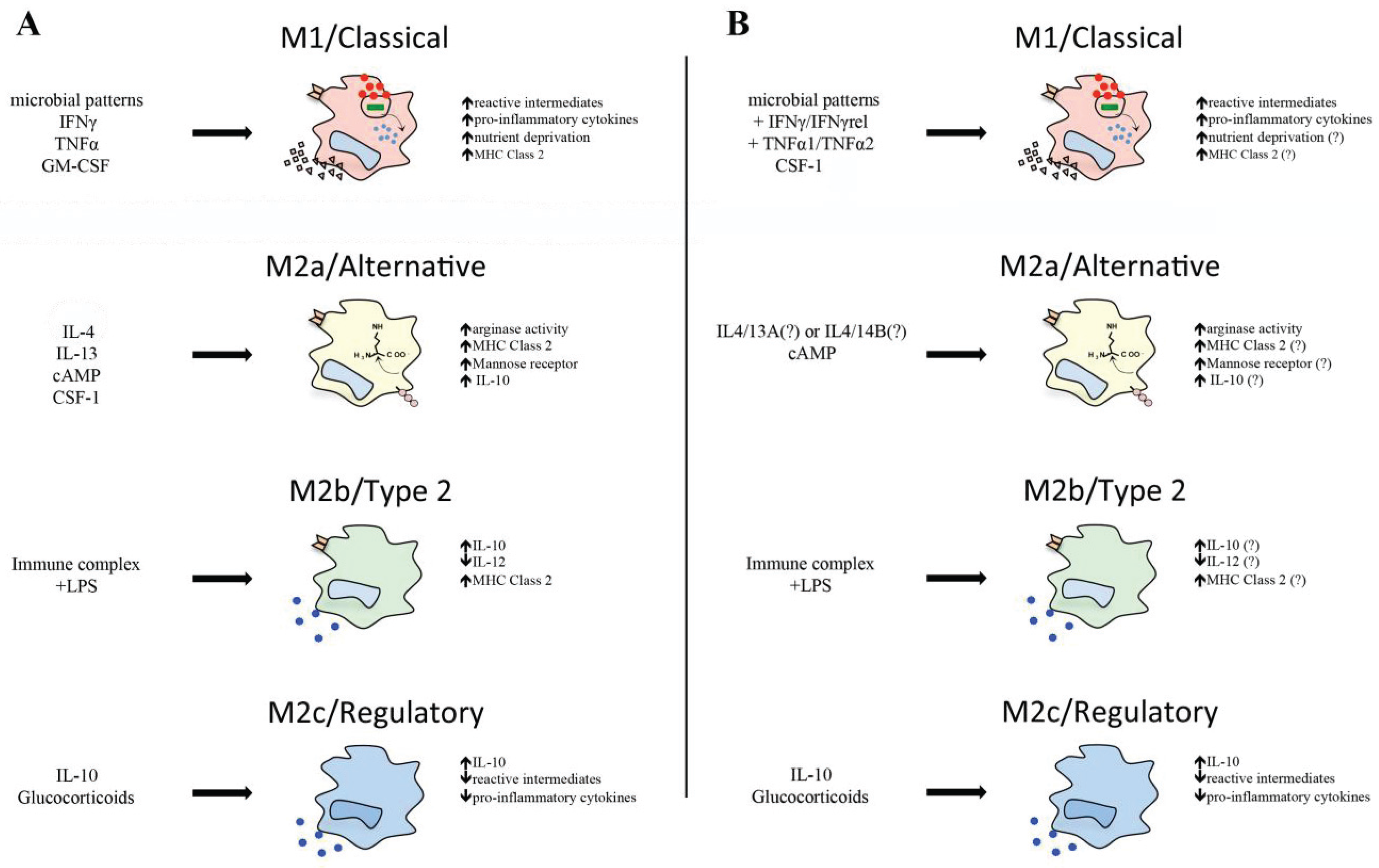
5. Alternative Teleost Macrophage Activation
5.1. Interleukin-4/13
5.2. Arginase
5.3. Immune Complex and Lipopolysaccharides
5.4. Glucocorticoids and Interleukin-10
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
References
- Metchnikoff, E. Immunity in Infective Diseases; Cambridge University Press: Cambridge, UK, 1905. [Google Scholar]
- Zhou, D.; Huang, C.; Lin, Z.; Zhan, S.; Kong, L.; Fang, C.; Li, J. Macrophage polarization and function with emphasis on the evolving roles of coordinated regulation of cellular signaling pathways. Cell. Signal. 2014, 26, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Neumann, N.F.; Stafford, J.L.; Belosevic, M. Biochemical and functional characterisation of macrophage stimulating factors secreted by mitogen-induced goldfish kidney leucocytes. Fish Shellfish Immunol. 2000, 10, 167–186. [Google Scholar] [CrossRef] [PubMed]
- Rieger, A.M.; Hall, B.E.; Barreda, D.R. Macrophage activation differentially modulates particle binding, phagocytosis and downstream antimicrobial mechanisms. Dev. Comp. Immunol. 2010, 34, 1144–1159. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Hodgkinson, J.W.; Belosevic, M. Antimicrobial responses of teleost phagocytes and innate immune evasion strategies of intracellular bacteria. Dev. Comp. Immunol. 2014, 43, 223–242. [Google Scholar] [CrossRef] [PubMed]
- Joerink, M.; Savelkoul, H.F.J.; Wiegertjes, G.F. Evolutionary conservation of alternative activation of macrophages: Structural and functional characterization of arginase 1 and 2 in carp (Cyprinus carpio L.). Mol. Immunol. 2006, 43, 1116–1128. [Google Scholar] [CrossRef] [PubMed]
- Takizawa, F.; Koppang, E.O.; Ohtani, M.; Nakanishi, T.; Hashimoto, K.; Fischer, U.; Dijkstra, J.M. Constitutive high expression of interleukin-4/13A and GATA-3 in gill and skin of salmonid fishes suggests that these tissues form Th2-skewed immune environments. Mol. Immunol. 2011, 48, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Castro, R.; Zou, J.; Secombes, C.J.; Martin, S.A.M. Cortisol modulates the induction of inflammatory gene expression in a rainbow trout macrophage cell line. Fish Shellfish Immunol. 2011, 30, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Hodgkinson, J.W.; Hitchen, S.J.; Belosevic, M. Characterization and functional analysis of goldfish (Carassius auratus L.) interleukin-10. Mol. Immunol. 2011, 48, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Joerink, M.; Forlenza, M.; Ribeiro, C.M.S.; de Vries, B.J.; Savelkoul, H.F.J.; Wiegertjes, G.F. Differential macrophage polarisation during parasitic infections in common carp (Cyprinus carpio L.). Fish Shellfish Immunol. 2006, 21, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Glasauer, S.M.K.; Neuhauss, S.C.F. Whole-genome duplication in teleost fishes and its evolutionary consequences. Mol. Genet. Genom. 2014, 289, 1045–1060. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, J.Y.; Kim, A.D.; Violette, E.P.; Stachura, D.L.; Cisson, J.L.; Traver, D. Definitive hematopoiesis initiates through a committed erythromyeloid progenitor in the zebrafish embryo. Development 2007, 134, 4147–4156. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.T.; Zon, L.I. Zebrafish blood stem cells. J. Cell. Biochem. 2009, 108, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Lieschke, G.J.; Oates, A.C.; Paw, B.H.; Thompson, M.A.; Hall, N.E.; Ward, A.C.; Ho, R.K.; Zon, L.I.; Layton, J.E. Zebrafish SPI-1 (PU.1) marks a site of myeloid development independent of primitive erythropoiesis: Implications for axial patterning. Dev. Biol. 2002, 246, 274–295. [Google Scholar] [CrossRef] [PubMed]
- Willett, C.E.; Cortes, A.; Zuasti, A.; Zapata, A.G. Early hematopoiesis and developing lymphoid organs in the zebrafish. Dev. Dyn. 1999, 214, 323–336. [Google Scholar] [CrossRef]
- Carroll, K.J.; North, T.E. Oceans of opportunity: Exploring vertebrate hematopoiesis in zebrafish. Exp. Hematol. 2014, 42, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, I.; Kuniyoshi, S.; Saito, K.; Moritomo, T.; Takahashi, T.; Nakanishi, T. Long-term hematopoietic reconstitution by transplantation of kidney hematopoietic stem cells in lethally irradiated clonal ginbuna crucian carp (Carassius auratus langsdorfii). Dev. Comp. Immunol. 2008, 32, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Traver, D.; Winzeler, A.; Stern, H.M.; Mayhall, E.A.; Langenau, D.M.; Kutok, J.L.; Look, A.T.; Zon, L.I. Effects of lethal irradiation in zebrafish and rescue by hematopoietic cell transplantation. Blood 2004, 104, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Hanington, P.C.; Tam, J.; Katzenback, B.A.; Hitchen, S.J.; Barreda, D.R.; Belosevic, M. Development of macrophages of cyprinid fish. Dev. Comp. Immunol. 2009, 33, 411–429. [Google Scholar] [CrossRef] [PubMed]
- Katzenback, A.B.; Katakura, F.; Belosevic, M. Regulation of teleost macrophage and neutrophil cell development by growth factors and transcription factors. In New Advances and Contributions to Fish Biology; Turker, H., Ed.; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Van Furth, R.; Cohn, Z.A.; Hirsch, J.G.; Humphrey, J.H.; Spector, W.G.; Langevoort, H.L. Mononuclear phagocytic system: New classification of macrophages, monocytes and of their cell line. Bull. World Health Organ. 1972, 47, 651–658. [Google Scholar] [PubMed]
- Ginhoux, F.; Jung, S. Monocytes and macrophages: Developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 2014, 14, 392–404. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, D.; Chow, A.; Noizat, C.; Teo, P.; Beasley, M.B.; Leboeuf, M.; Becker, C.D.; See, P.; Price, J.; Lucas, D.; et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 2013, 38, 792–804. [Google Scholar] [CrossRef] [PubMed]
- Yona, S.; Kim, K.W.; Wolf, Y.; Mildner, A.; Varol, D.; Breker, M.; Strauss-Ayali, D.; Viukov, S.; Guilliams, M.; Misharin, A.; et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 2013, 38, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Soza-Ried, C.; Hess, I.; Netuschil, N.; Schorpp, M.; Boehm, T. Essential role of c-myb in definitive hematopoiesis is evolutionarily conserved. Proc. Natl. Acad. Sci. USA 2010, 107, 17304–17308. [Google Scholar] [CrossRef] [PubMed]
- Garceau, V.; Smith, J.; Paton, I.R.; Davey, M.; Fares, M.A.; Sester, D.P.; Burt, D.W.; Hume, D.A. Pivotal Advance: Avian colony-stimulating factor 1 (CSF-1), interleukin-34 (IL-34), and CSF-1 receptor genes and gene products. J. Leukoc. Biol. 2010, 87, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Hanington, P.C.; Wang, T.; Secombes, C.J.; Belosevic, M. Growth factors of lower vertebrates: Characterization of goldfish (Carassius auratus L.) macrophage colony-stimulating factor-1. J. Biol. Chem. 2007, 282, 31865–31872. [Google Scholar] [CrossRef] [PubMed]
- Pixley, F.J.; Stanley, E.R. CSF-1 regulation of the wandering macrophage: Complexity in action. Trends Cell Biol. 2004, 14, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Hanington, P.C.; Belosevic, M.; Secombes, C.J. Two macrophage colony-stimulating factor genes exist in fish that differ in gene organization and are differentially expressed. J. Immunol. 2008, 181, 3310–3322. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.-M.; Ryan, G.R.; Hapel, A.J.; Dominguez, M.G.; Russell, R.G.; Kapp, S.; Sylvestre, V.; Stanley, E.R. Targeted disruption of the mouse colony-stimulating factor 1 receptor gene results in osteopetrosis, mononuclear phagocyte deficiency, increased primitive progenitor cell frequencies, and reproductive defects. Blood 2002, 99, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Guilbert, L.J.; Stanley, E.R. Specific interaction of murine colony-stimulating factor with mononuclear phagocytic cells. J. Cell Biol. 1980, 85, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Lichanska, A.M.; Browne, C.M.; Henkel, G.W.; Murphy, K.M.; Ostrowski, M.C.; McKercher, S.R.; Maki, R.A.; Hume, D.A. Differentiation of the mononuclear phagocyte system during mouse embryogenesis: The role of transcription factor PU.1. Blood 1999, 94, 127–138. [Google Scholar] [PubMed]
- Manos, M.M. Expression and processing of a recombinant human macrophage colony-stimulating factor in mouse cells. Mol. Cell. Biol. 1988, 8, 5035–5039. [Google Scholar] [CrossRef] [PubMed]
- Rettenmier, C.W.; Roussel, M.F. Differential processing of colony-stimulating factor 1 precursors encoded by two human cDNAs. Mol. Cell. Biol. 1988, 8, 5026–5034. [Google Scholar] [CrossRef] [PubMed]
- Hanington, P.C.; Belosevic, M. Interleukin-6 family cytokine M17 induces differentiation and nitric oxide response of goldfish (Carassius auratus L.) macrophages. Dev. Comp. Immunol. 2007, 31, 817–829. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Hanington, P.C.; Belosevic, M. Macrophage colony-stimulating factor (CSF-1) induces pro-inflammatory gene expression and enhances antimicrobial responses of goldfish (Carassius auratus L.) macrophages. Fish Shellfish Immunol. 2009, 26, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, T.A.; Zhao, C.; Pavicic, P.G.; Datta, S. Myeloid colony-stimulating factors as regulators of Macrophage polarization. Front. Immunol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Rieger, A.M.; Hanington, P.C.; Belosevic, M.; Barreda, D.R. Control of CSF-1 induced inflammation in teleost fish by a soluble form of the CSF-1 receptor. Fish Shellfish Immunol. 2014, 41, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Stanley, E.R.; Berg, K.L.; Einstein, D.B.; Lee, P.S.; Pixley, F.J.; Wang, Y.; Yeung, Y.G. Biology and action of colony—Stimulating factor-1. Mol. Reprod. Dev. 1997, 46, 4–10. [Google Scholar] [CrossRef]
- Bober, L.A.; Grace, M.J.; Pugliese-Sivo, C.; Rojas-Triana, A.; Sullivan, L.M.; Narula, S.K. The effects of colony stimulating factors on human monocyte cell function. Int. J. Immunopharmacol. 1995, 17, 385–392. [Google Scholar] [CrossRef]
- Karbassi, A.; Becker, J.M.; Foster, J.S.; Moore, R.N. Enhanced killing of Candida albicans by murine macrophages treated with macrophage colony-stimulating factor: Evidence for augmented expression of mannose receptors. J. Immunol. 1987, 139, 417–421. [Google Scholar] [PubMed]
- Munn, D.H.; Cheung, N.K. Antibody-independent phagocytosis of tumor cells by human monocyte-derived macrophages cultured in recombinant macrophage colony-stimulating factor. Cancer Immunol. Immunother. CII 1995, 41, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Sweet, M.J.; Hume, D.A. CSF-1 as a regulator of macrophage activation and immune responses. Arch. Immunol. Ther. Exp. 2003, 51, 169–177. [Google Scholar]
- Williams, H.; Brenner, S.; Venkatesh, B. Identification and analysis of additional copies of the platelet-derived growth factor receptor and colony stimulating factor 1 receptor genes in fugu. Gene 2002, 295, 255–264. [Google Scholar] [CrossRef]
- Aggad, D.; Stein, C.; Sieger, D.; Mazel, M.; Boudinot, P.; Herbomel, P.; Levraud, J.P.; Lutfalla, G.; Leptin, M. In vivo analysis of IFN-γ1 and IFN-γ2 signaling in zebrafish. J. Immunol. 2010, 185, 6774–6782. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Belosevic, M. Molecular characterization, expression and functional analysis of goldfish (Carassius aurutus L.) interferon gamma. Dev. Comp. Immunol. 2009, 33, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, Y.; Yabu, T.; Araki, K.; Mano, N.; Shiba, H.; Moritomo, T.; Nakanishi, T. Peculiar monomeric interferon gammas, IFNγrel 1 and IFNγrel 2, in ginbuna crucian carp. FEBS J. 2014, 281, 1046–1056. [Google Scholar] [CrossRef] [PubMed]
- Yabu, T.; Toda, H.; Shibasaki, Y.; Araki, K.; Yamashita, M.; Anzai, H.; Mano, N.; Masuhiro, Y.; Hanazawa, S.; Shiba, H.; et al. Antiviral protection mechanisms mediated by ginbuna crucian carp interferon gamma isoforms 1 and 2 through two distinct interferon γ-receptors. J. Biochem. 2011, 150, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Mackaness, G.B. Cellular resistance to infection. J. Exp. Med. 1962, 116, 381–406. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol. 2003, 3, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Robertsen, B. The interferon system of teleost fish. Fish Shellfish Immunol. 2006, 20, 172–191. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, J.J.; Murray, P.J. Cytokine signaling modules in inflammatory responses. Immunity 2008, 28, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Sato, S.; Hemmi, H.; Hoshino, K.; Kaisho, T.; Sanjo, H.; Takeuchi, O.; Sugiyama, M.; Okabe, M.; Takeda, K.; et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 2003, 301, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Van der Vaart, M.; Spaink, H.P.; Meijer, A.H. Pathogen recognition and activation of the innate immune response in zebrafish. Adv. Hematol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Secombes, C.J.; Wang, T.; Bird, S. The interleukins of fish. Dev. Comp. Immunol. 2011, 35, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Secombes, C.J. The cytokine networks of adaptive immunity in fish. Fish Shellfish Immunol. 2013, 35, 1703–1718. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Yoshiura, Y.; Dijkstra, J.M.; Sakai, M.; Ototake, M.; Secombes, C. Identification of an interferon gamma homologue in Fugu, Takifugu rubripes. Fish Shellfish Immunol. 2004, 17, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Carrington, A.; Collet, B.; Dijkstra, J.M.; Yoshiura, Y.; Bols, N.; Secombes, C. Identification and bioactivities of IFN-γ in rainbow trout Oncorhynchus mykiss: The first Th1-type cytokine characterized functionally in fish. J. Immunol. 2005, 175, 2484–2494. [Google Scholar] [CrossRef] [PubMed]
- Igawa, D.; Sakai, M.; Savan, R. An unexpected discovery of two interferon gamma-like genes along with interleukin (IL)-22 and -26 from teleost: IL-22 and -26 genes have been described for the first time outside mammals. Mol. Immunol. 2006, 43, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Milev-Milovanovic, I.; Long, S.; Wilson, M.; Bengten, E.; Miller, N.W.; Chinchar, V.G. Identification and expression analysis of interferon gamma genes in channel catfish. Immunogenetics 2006, 58, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Stolte, E.H.; Savelkoul, H.F.J.; Wiegertjes, G.; Flik, G.; van Kemenade, B.M. Differential expression of two interferon-γ genes in common carp (Cyprinus carpio L.). Dev. Comp. Immunol. 2008, 32, 1467–1481. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Garcia, E.G.; Belosevic, M. Comparison of macrophage antimicrobial responses induced by type II interferons of the goldfish (Carassius Auratus L.). J. Biol. Chem. 2010, 285, 23537–23547. [Google Scholar] [CrossRef] [PubMed]
- Furnes, C.; Seppola, M.; Robertsen, B. Molecular characterisation and expression analysis of interferon gamma in Atlantic cod (Gadus morhua). Fish Shellfish Immunol. 2009, 26, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.Y.; Hikima, J.; Ohtani, M.; Jang, H.B.; del Castillo, C.S.; Nho, S.W.; Cha, I.S.; Park, S.B.; Aoki, T.; Jung, T.S. Recombinant interferon-γ activates immune responses against Edwardsiella tarda infection in the olive flounder, Paralichthys olivaceus. Fish Shellfish Immunol. 2012, 33, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Skjesol, A.; Hansen, T.; Shi, C.Y.; Thim, H.L.; Jørgensen, J.B. Structural and functional studies of STAT1 from Atlantic salmon (Salmo salar). BMC Immunol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Arts, J.A.J.; Tijhaar, E.J.; Chadzinska, M.; Savelkoul, H.F.J.; van Kemenade, B.M.L. Functional analysis of carp interferon-γ: Evolutionary conservation of classical phagocyte activation. Fish Shellfish Immunol. 2010, 29, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Zhang, S.; Chen, D.; Zhang, A.; Wang, X.; Zhou, H. IFN-γ-activated lymphocytes boost nitric oxide production in grass carp monocytes/macrophages. Fish Shellfish Immunol. 2013, 35, 1635–1641. [Google Scholar] [CrossRef] [PubMed]
- Sieger, D.; Stein, C.; Neifer, D.; van der Sar, A.M.; Leptin, M. The role of gamma interferon in innate immunity in the zebrafish embryo. Dis. Model. Mech. 2009, 2, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Belosevic, M. Molecular characterization of novel interferon gamma receptor 1 isoforms in zebrafish (Danio rerio) and goldfish (Carassius auratus L.). Mol. Immunol. 2009, 46, 3050–3059. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Belosevic, M. Molecular characterization of tumor necrosis factor receptors 1 and 2 of the goldfish (Carassius aurutus L.). Mol. Immunol. 2009, 46, 2190–2199. [Google Scholar] [CrossRef] [PubMed]
- López-Muñoz, A.; Roca, F.J.; Meseguer, J.; Mulero, V. New insights into the evolution of IFNs: Zebrafish group II IFNs induce a rapid and transient expression of IFN-dependent genes and display powerful antiviral activities. J. Immunol. 2009, 182, 3440–3449. [Google Scholar] [CrossRef] [PubMed]
- García-Castillo, J.; Pelegrín, P.; Mulero, V.; Meseguer, J. Molecular cloning and expression analysis of tumor necrosis factor α from a marine fish reveal its constitutive expression and ubiquitous nature. Immunogenetics 2002, 54, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Walsh, J.G.; Belosevic, M. Characterization and functional analysis of goldfish (Carassius auratus L.) tumor necrosis factor-alpha. Dev. Comp. Immunol. 2008, 32, 532–543. [Google Scholar] [CrossRef] [PubMed]
- Hirono, I.; Nam, B.H.; Kurobe, T.; Aoki, T. Molecular cloning, characterization, and expression of TNF cDNA and gene from Japanese flounder Paralychthys olivaceus. J. Immunol. 2000, 165, 4423–4427. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, T.; Harada, H.; Sawada, Y.; Kohchi, C.; Soma, G.I.; Takahashi, Y.; Inagawa, H. Two types of tumor necrosis factor-α in bluefin tuna (Thunnus orientalis) genes: Molecular cloning and expression profile in response to several immunological stimulants. Fish Shellfish Immunol. 2009, 27, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Laing, K.J.; Wang, T.; Zou, J.; Holland, J.; Hong, S.; Bols, N.; Hirono, I.; Aoki, T.; Secombes, C.J. Cloning and expression analysis of rainbow trout Oncorhynchus mykiss tumour necrosis factor-α. Eur. J. Biochem. 2001, 268, 1315–1322. [Google Scholar] [CrossRef] [PubMed]
- Lam, F.W.S.; Wu, S.Y.; Lin, S.J.; Lin, C.C.; Chen, Y.M.; Wang, H.C.; Chen, T.Y.; Lin, H.T.; Lin, J.H.Y. The expression of two novel orange-spotted grouper (Epinephelus coioides) TNF genes in peripheral blood leukocytes, various organs, and fish larvae. Fish Shellfish Immunol. 2011, 30, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, D.S.; Pereira, P.J.B.; Reis, M.I.R.; do Vale, A.; Zou, J.; Silva, M.T.; Secombes, C.J.; dos Santos, N.M.S. Molecular cloning and expression analysis of sea bass (Dicentrarchus labrax L.) tumor necrosis factor-α (TNF-α). Fish Shellfish Immunol. 2007, 23, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Ordás, M.C.; Costa, M.M.; Roca, F.J.; López-Castejón, G.; Mulero, V.; Meseguer, J.; Figueras, A.; Novoa, B. Turbot TNFα gene: Molecular characterization and biological activity of the recombinant protein. Mol. Immunol. 2007, 44, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Saeij, J.P.; Stet, R.J.; Groeneveld, A.; van Kemenade, L.B.; van Muiswinkel, W.B.; Wiegertjes, G.F. Molecular and functional characterization of a fish inducible-type nitric oxide synthase. Immunogenetics 2000, 51, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Savan, R.; Kono, T.; Igawa, D.; Sakai, M. A novel tumor necrosis factor (TNF) gene present in tandem with the TNF-α gene on the same chromosome in teleosts. Immunogenetics 2005, 57, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Chen, D.; Wei, H.; Du, L.; Zhao, T.; Wang, X.; Zhou, H. Functional characterization of TNF-α in grass carp head kidney leukocytes: Induction and involvement in the regulation of NF-κB signaling. Fish Shellfish Immunol. 2012, 33, 1123–1132. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Li, R.; Xu, Q.; Secombes, C.J.; Wang, T. Two types of TNF-α exist in teleost fish: Phylogeny, expression, and bioactivity analysis of type-II TNF-α3 in rainbow trout oncorhynchus mykiss. J. Immunol. 2013, 191, 5959–5972. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Chi, M.; Laplace-Builhe, B.; Travnickova, J.; Luz-Crawford, P.; Tejedor, G.; Phan, Q.T.; Duroux-Richard, I.; Levraud, J.P.; Kissa, K.; Lutfalla, G.; et al. Identification of polarized macrophage subsets in zebrafish. eLife 2015. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D.; Ley, K. M1 and M2 macrophages: The chicken and the egg of immunity. J. Innate Immun. 2014, 6, 716–726. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, J.A. Colony-stimulating factors in inflammation and autoimmunity. Nat. Rev. Immunol. 2008, 8, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, J.M. TH2 and Treg candidate genes in elephant shark. Nature 2014, 511, E7–E9. [Google Scholar] [CrossRef] [PubMed]
- Broughton, S.E.; Nero, T.L.; Dhagat, U.; Kan, W.L.; Hercus, T.R.; Tvorogov, D.; Lopez, A.F.; Parker, M.W. The βc receptor family—Structural insights and their functional implications. Cytokine 2015, 74, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D.; Lenz, L.L.; Ley, K. M1/M2 Macrophages: The Arginine Fork in the Road to Health and Disease; Frontiers Media SA: Lausanne, Switzerland, 2015. [Google Scholar]
- Grayfer, L.; Hodgkinson, J.W.; Belosevic, M. Analysis of the antimicrobial responses of primary phagocytes of the goldfish (Carassius auratus L.) against Mycobacterium marinum. Dev. Comp. Immunol. 2011, 35, 1146–1158. [Google Scholar] [CrossRef] [PubMed]
- Briggs, R.T.; Drath, D.B.; Karnovsky, M.L.; Karnovsky, M.J. Localization of NADH oxidase on the surface of human polymorphonuclear leukocytes by a new cytochemical method. J. Cell Biol. 1975, 67, 566–586. [Google Scholar] [CrossRef] [PubMed]
- DeLeo, F.R.; Quinn, M.T. Assembly of the phagocyte NADPH oxidase: Molecular interaction of oxidase proteins. J. Leukoc. Biol. 1996, 60, 677–691. [Google Scholar] [PubMed]
- El-Benna, J.; Dang, P.M.-C.; Gougerot-Pocidalo, M.A. Priming of the neutrophil NADPH oxidase activation: Role of p47phox phosphorylation and NOX2 mobilization to the plasma membrane. Semin. Immunopathol. 2008, 30, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Leto, T.L.; Lomax, K.J.; Volpp, B.D.; Nunoi, H.; Sechler, J.M.; Nauseef, W.M.; Clark, R.A.; Gallin, J.I.; Malech, H.L. Cloning of a 67-kD neutrophil oxidase factor with similarity to a noncatalytic region of p60c-SRC. Science 1990, 248, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Parkos, C.A.; Dinauer, M.C.; Walker, L.E.; Allen, R.A.; Jesaitis, A.J.; Orkin, S.H. Primary structure and unique expression of the 22-kilodalton light chain of human neutrophil cytochrome b. Proc. Natl. Acad. Sci. USA 1988, 85, 3319–3323. [Google Scholar] [CrossRef] [PubMed]
- Parkos, C.A.; Allen, R.A.; Cochrane, C.G.; Jesaitis, A.J. The quaternary structure of the plasma membrane b-type cytochrome of human granulocytes. Biochim. Biophys. Acta 1988, 932, 71–83. [Google Scholar] [CrossRef]
- Parkos, C.A.; Allen, R.A.; Cochrane, C.G.; Jesaitis, A.J. Purified cytochrome b from human granulocyte plasma membrane is comprised of two polypeptides with relative molecular weights of 91,000 and 22,000. J. Clin. Investig. 1987, 80, 732–742. [Google Scholar] [CrossRef] [PubMed]
- Tsunawaki, S.; Mizunari, H.; Nagata, M.; Tatsuzawa, O.; Kuratsuji, T. A novel cytosolic component, p40phox, of respiratory burst oxidase associates with p67phox and is absent in patients with chronic granulomatous disease who lack p67phox. Biochem. Biophys. Res. Commun. 1994, 199, 1378–1387. [Google Scholar] [CrossRef] [PubMed]
- Volpp, B.D.; Nauseef, W.M.; Donelson, J.E.; Moser, D.R.; Clark, R.A. Cloning of the cDNA and functional expression of the 47-kilodalton cytosolic component of human neutrophil respiratory burst oxidase. Proc. Natl. Acad. Sci. USA 1989, 86, 7195–7199. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Belosevic, M. Cytokine regulation of teleost inflammatory responses. In New Advances and Contributions to Fish Biology; Turker, H., Ed.; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Inoue, Y.; Suenaga, Y.; Yoshiura, Y.; Moritomo, T.; Ototake, M.; Nakanishi, T. Molecular cloning and sequencing of Japanese pufferfish (Takifugu rubripes) NADPH oxidase cDNAs. Dev. Comp. Immunol. 2004, 28, 911–925. [Google Scholar] [CrossRef] [PubMed]
- Olavarría, V.H.; Gallardo, L.; Figueroa, J.E.; Mulero, V. Lipopolysaccharide primes the respiratory burst of Atlantic salmon SHK-1 cells through protein kinase C-mediated phosphorylation of p47phox. Dev. Comp. Immunol. 2010, 34, 1242–1253. [Google Scholar] [CrossRef] [PubMed]
- Boltaña, S.; Doñate, C.; Goetz, F.W.; MacKenzie, S.; Balasch, J.C. Characterization and expression of NADPH oxidase in LPS-, poly(I:C)- and zymosan-stimulated trout (Oncorhynchus mykiss W.) macrophages. Fish Shellfish Immunol. 2009, 26, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, J.B.; Robertsen, B. Yeast β-glucan stimulates respiratory burst activity of Atlantic salmon (Salmo salar L.) macrophages. Dev. Comp. Immunol. 1995, 19, 43–57. [Google Scholar] [CrossRef]
- Sepulcre, M.P.; López-Castejón, G.; Meseguer, J.; Mulero, V. The activation of gilthead seabream professional phagocytes by different PAMPs underlines the behavioural diversity of the main innate immune cells of bony fish. Mol. Immunol. 2007, 44, 2009–2016. [Google Scholar] [CrossRef] [PubMed]
- Stafford, J.L.; Galvez, F.; Goss, G.G.; Belosevic, M. Induction of nitric oxide and respiratory burst response in activated goldfish macrophages requires potassium channel activity. Dev. Comp. Immunol. 2002, 26, 445–459. [Google Scholar] [CrossRef]
- Ardó, L.; Jeney, Z.; Adams, A.; Jeney, G. Immune responses of resistant and sensitive common carp families following experimental challenge with Aeromonas hydrophila. Fish Shellfish Immunol. 2010, 29, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Hodgkinson, J.W.; Ge, J.Q.; Grayfer, L.; Stafford, J.; Belosevic, M. Analysis of the immune response in infections of the goldfish (Carassius auratus L.) with Mycobacterium marinum. Dev. Comp. Immunol. 2012, 38, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Sharp, G.J.E.; Secombes, C.J. The role of reactive oxygen species in the killing of the bacterial fish pathogen Aeromonas salmonicida by rainbow trout macrophages. Fish Shellfish Immunol. 1993, 3, 119–129. [Google Scholar] [CrossRef]
- Kim, M.S.; Hwang, Y.J.; Yoon, K.J.; Zenke, K.; Nam, Y.K.; Kim, S.K.; Kim, K.H. Molecular cloning of rock bream (Oplegnathus fasciatus) tumor necrosis factor-α and its effect on the respiratory burst activity of phagocytes. Fish Shellfish Immunol. 2009, 27, 618–624. [Google Scholar] [CrossRef] [PubMed]
- MacMicking, J.; Xie, Q.W.; Nathan, C. Nitric Oxide and Macrophage Function. Annu. Rev. Immunol. 1997, 15, 323–350. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C.; Xie, Q.W. Nitric oxide synthases: Roles, tolls, and controls. Cell 1994, 78, 915–918. [Google Scholar] [CrossRef]
- Denicola, A.; Rubbo, H.; Rodriguez, D.; Radi, R. Peroxynitrite-mediated cytotoxicity to trypanosoma cruzi. Arch. Biochem. Biophys. 1993, 304, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Henard, C.A.; Vázquez-Torres, A. Nitric oxide and salmonella pathogenesis. Front. Microbiol. 2011. [Google Scholar] [CrossRef] [PubMed]
- Laing, K.J.; Grabowski, P.S.; Belosevic, M.; Secombes, C.J. A partial sequence for nitric oxide synthase from a goldfish (Carassius auratus) macrophage cell line. Immunol. Cell Biol. 1996, 74, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Laing, K.J.; Hardie, L.J.; Aartsen, W.; Grabowski, P.S.; Secombes, C.J. Expression of an inducible nitric oxide synthase gene in rainbow trout Oncorhynchus mykiss. Dev. Comp. Immunol. 1999, 23, 71–85. [Google Scholar] [CrossRef]
- Vojtech, L.N.; Sanders, G.E.; Conway, C.; Ostland, V.; Hansen, J.D. Host immune response and acute disease in a zebrafish model of francisella pathogenesis. Infect. Immun. 2009, 77, 914–925. [Google Scholar] [CrossRef] [PubMed]
- Losada, A.P.; Bermúdez, R.; Faílde, L.D.; Quiroga, M.I. Quantitative and qualitative evaluation of iNOS expression in turbot (Psetta maxima) infected with Enteromyxum scophthalmi. Fish Shellfish Immunol. 2012, 32, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Joerink, M.; Ribeiro, C.M.S.; Stet, R.J.M.; Hermsen, T.; Savelkoul, H.F.J.; Wiegertjes, G.F. Head kidney-derived macrophages of common carp (Cyprinus carpio L.) show plasticity and functional polarization upon differential stimulation. J. Immunol. 2006, 177, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Ishibe, K.; Yamanishi, T.; Wang, Y.; Osatomi, K.; Hara, K.; Kanai, K.; Yamaguchi, K.; Oda, T. Comparative analysis of the production of nitric oxide (NO) and tumor necrosis factor-alpha (TNF-alpha) from macrophages exposed to high virulent and low virulent strains of Edwardsiella tarda. Fish Shellfish Immunol. 2009, 27, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Stafford, J.L.; Wilson, E.C.; Belosevic, M. Recombinant transferrin induces nitric oxide response in goldfish and murine macrophages. Fish Shellfish Immunol. 2004, 17, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Campos-Pérez, J.J.; Ellis, A.E.; Secombes, C.J. Toxicity of nitric oxide and peroxynitrite to bacterial pathogens of fish. Dis. Aquat. Organ. 2000, 43, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Chettri, J.K.; Raida, M.K.; Kania, P.W.; Buchmann, K. Differential immune response of rainbow trout (Oncorhynchus mykiss) at early developmental stages (larvae and fry) against the bacterial pathogen Yersinia ruckeri. Dev. Comp. Immunol. 2012, 36, 463–474. [Google Scholar] [CrossRef] [PubMed]
- Tafalla, C.; Figueras, A.; Novoa, B. Role of nitric oxide on the replication of viral haemorrhagic septicemia virus (VHSV), a fish rhabdovirus. Vet. Immunol. Immunopathol. 1999, 72, 249–256. [Google Scholar] [CrossRef]
- Cambier, C.J.; Takaki, K.K.; Larson, R.P.; Hernandez, R.E.; Tobin, D.M.; Urdahl, K.B.; Cosma, C.L.; Ramakrishnan, L. Mycobacteria manipulate macrophage recruitment through coordinated use of membrane lipids. Nature 2014, 505, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Elks, P.M.; van der Vaart, M.; van Hensbergen, V.; Schutz, E.; Redd, M.J.; Murayama, E.; Spaink, H.P.; Meijer, A.H. Mycobacteria counteract a TLR-mediated nitrosative defense mechanism in a zebrafish infection model. PLoS ONE 2014, 9, e100928. [Google Scholar] [CrossRef] [PubMed]
- Elks, P.M.; Brizee, S.; van der Vaart, M.; Walmsley, S.R.; van Eeden, F.J.; Renshaw, S.A.; Meijer, A.H. Hypoxia inducible factor signaling modulates susceptibility to mycobacterial infection via a nitric oxide dependent mechanism. PLoS Pathog. 2013, 9, e1003789. [Google Scholar] [CrossRef] [PubMed]
- Canton, J.; Khezri, R.; Glogauer, M.; Grinstein, S. Contrasting phagosome pH regulation and maturation in human M1 and M2 macrophages. Mol. Biol. Cell 2014, 25, 3330–3341. [Google Scholar] [CrossRef] [PubMed]
- Delamarre, L.; Pack, M.; Chang, H.; Mellman, I.; Trombetta, E.S. Differential lysosomal proteolysis in antigen-presenting cells determines antigen fate. Science 2005, 307, 1630–1634. [Google Scholar] [CrossRef] [PubMed]
- Cellier, M.F.M. Cell-type specific determinants of NRAMP1 expression in professional phagocytes. Biology 2013, 2, 233–283. [Google Scholar] [CrossRef] [PubMed]
- Aragones, J.; Elorza, A.; Acosta-Iborra, B.; Landazuri, M.O. Myeloid hypoxia-inducible factors in inflammatory diseases. Crit. Rev. Immunol. 2011, 31, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Shay, J.E.S.; Celeste Simon, M. Hypoxia-inducible factors: Crosstalk between inflammation and metabolism. Semin. Cell Dev. Biol. 2012, 23, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.R.; Gros, P. Divalent-metal transport by NRAMP proteins at the interface of host-pathogen interactions. Trends Microbiol. 2001, 9, 397–403. [Google Scholar] [CrossRef]
- Kuhn, D.E.; Baker, B.D.; Lafuse, W.P.; Zwilling, B.S. Differential iron transport into phagosomes isolated from the RAW264.7 macrophage cell lines transfected with NRAMP1Gly169 or NRAMP1Asp169. J. Leukoc. Biol. 1999, 66, 113–119. [Google Scholar] [PubMed]
- Atkinson, P.G.; Barton, C.H. High level expression of NRAMP1G169 in RAW264.7 cell transfectants: Analysis of intracellular iron transport. Immunology 1999, 96, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Jabado, N.; Jankowski, A.; Dougaparsad, S.; Picard, V.; Grinstein, S.; Gros, P. Natural resistance to intracellular infections: Natural resistance-associated macrophage protein 1 (NRAMP1) functions as a pH-dependent manganese transporter at the phagosomal membrane. J. Exp. Med. 2000, 192, 1237–1248. [Google Scholar] [CrossRef] [PubMed]
- Goswami, T.; Bhattacharjee, A.; Babal, P.; Searle, S.; Moore, E.; Li, M.; Blackwell, J.M. Natural-resistance-associated macrophage protein 1 is an H+/bivalent cation antiporter. Biochem. J. 2001, 354, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Saeij, J.P.; Wiegertjes, G.F.; Stet, R.J. Identification and characterization of a fish natural resistance-associated macrophage protein (NRAMP) cDNA. Immunogenetics 1999, 50, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Sibthorpe, D.; Baker, A.M.; Gilmartin, B.J.; Blackwell, J.M.; White, J.K. Comparative analysis of two slc11 (NRAMP) loci in Takifugu rubripes. DNA Cell Biol. 2004, 23, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Waldbieser, G.C.; Rice, C.D.; Elibol, B.; Wolters, W.R.; Hanson, L.A. Isolation and characterization of channel catfish natural resistance associated macrophage protein gene. Dev. Comp. Immunol. 2002, 26, 517–531. [Google Scholar] [CrossRef]
- Cooper, C.A.; Shayeghi, M.; Techau, M.E.; Capdevila, D.M.; MacKenzie, S.; Durrant, C.; Bury, N.R. Analysis of the rainbow trout solute carrier 11 family reveals iron import < or = pH 7.4 and a functional isoform lacking transmembrane domains 11 and 12. FEBS Lett. 2007, 581, 2599–2604. [Google Scholar] [PubMed]
- Chen, S.L.; Zhang, Y.X.; Xu, J.Y.; Meng, L.; Sha, Z.X.; Ren, G.C. Molecular cloning, characterization and expression analysis of natural resistance associated macrophage protein (NRAMP) cDNA from turbot (Scophthalmus maximus). Comp. Biochem. Physiol. B 2007, 147, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.L.; Xu, M.Y.; Ji, X.S.; Yu, G.C. Cloning and characterisation of natural resistance associated macrophage protein (NRAMP) cDNA from red sea bream (Pagrus major). Fish Shellfish Immunol. 2004, 17, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.L.; Wang, Z.J.; Xu, M.Y.; Gui, J.F. Molecular identification and expression analysis of natural resistance associated macrophage protein (NRAMP) cDNA from Japanese flounder (Paralichthys olivaceus). Fish Shellfish Immunol. 2006, 20, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Elibol-Flemming, B.; Waldbieser, G.C.; Wolters, W.R.; Boyle, C.R.; Hanson, L.A. Expression analysis of selected immune-relevant genes in channel catfish during Edwardsiella ictaluri infection. J. Aquat. Anim. Health 2009, 21, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Burge, E.J.; Gauthier, D.T.; Ottinger, C.A.; van Veld, P.A. Mycobacterium-inducible NRAMP in striped bass (Morone saxatilis). Infect. Immun. 2004, 72, 1626–1636. [Google Scholar] [CrossRef] [PubMed]
- Donovan, A.; Brownlie, A.; Zhou, Y.; Shepard, J.; Pratt, S.J.; Moynihan, J.; Paw, B.H.; Drejer, A.; Barut, B.; Zapata, A.; et al. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter. Nature 2000, 403, 776–781. [Google Scholar] [PubMed]
- Abboud, S.; Haile, D.J. A novel mammalian iron-regulated protein involved in intracellular iron metabolism. J. Biol. Chem. 2000, 275, 19906–19912. [Google Scholar] [CrossRef] [PubMed]
- Nairz, M.; Fritsche, G.; Brunner, P.; Talasz, H.; Hantke, K.; Weiss, G. Interferon-gamma limits the availability of iron for intramacrophage Salmonella typhimurium. Eur. J. Immunol. 2008, 38, 1923–1936. [Google Scholar] [CrossRef] [PubMed]
- Nairz, M.; Theurl, I.; Ludwiczek, S.; Theurl, M.; Mair, S.M.; Fritsche, G.; Weiss, G. The co-ordinated regulation of iron homeostasis in murine macrophages limits the availability of iron for intracellular Salmonella typhimurium. Cell. Microbiol. 2007, 9, 2126–2140. [Google Scholar] [CrossRef] [PubMed]
- Zandt, K.E.V.; Sow, F.B.; Florence, W.C.; Zwilling, B.S.; Satoskar, A.R.; Schlesinger, L.S.; Lafuse, W.P. The iron export protein ferroportin 1 is differentially expressed in mouse macrophage populations and is present in the mycobacterial-containing phagosome. J. Leukoc. Biol. 2008, 84, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.E.; Wessling-Resnick, M. Iron metabolism and the innate immune response to infection. Microbes Infect. 2012, 14, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Garrick, M.D.; Singleton, S.T.; Vargas, F.; Kuo, H.-C.; Zhao, L.; Knöpfel, M.; Davidson, T.; Costa, M.; Paradkar, P.; Roth, J.A.; et al. DMT1: Which metals does it transport? Biol. Res. 2006, 39, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.G.; Liu, S.S.; Sun, B.; Wang, X.L.; Wang, N.; Chen, S.L. Iron-metabolic function and potential antibacterial role of Hepcidin and its correlated genes (Ferroportin 1 and Transferrin Receptor) in Turbot (Scophthalmus maximus). Fish Shellfish Immunol. 2013, 34, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.F.; Wang, H.S.; Wang, H.; Zhang, F.; Wang, K.F.; Guo, Q.; Zhang, G.; Cai, S.H.; Du, J. The role of indoleamine 2,3-dioxygenase (IDO) in immune tolerance: Focus on macrophage polarization of THP-1 cells. Cell. Immunol. 2014, 289, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.W.; Feng, G.S. Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J. 1991, 5, 2516–2522. [Google Scholar] [PubMed]
- Grohmann, U.; Bronte, V. Control of immune response by amino acid metabolism. Immunol. Rev. 2010, 236, 243–264. [Google Scholar] [CrossRef] [PubMed]
- Hayaishi, O. Utilization of superoxide anion by indoleamine oxygenase-catalyzed tryptophan and indoleamine oxidation. In Recent Advances in Tryptophan Research; Filippini, G.A., Costa, C.V.L., Bertazzo, A., Eds.; Springer: New York, NY, USA, 1996; pp. 285–289. [Google Scholar]
- Werner, E.R.; Werner-Felmayer, G.; Fuchs, D.; Hausen, A.; Reibnegger, G.; Wachter, H. Parallel induction of tetrahydrobiopterin biosynthesis and indoleamine 2,3-dioxygenase activity in human cells and cell lines by interferon-gamma. Biochem. J. 1989, 262, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Werner-Felmayer, G.; Werner, E.R.; Fuchs, D.; Hausen, A.; Reibnegger, G.; Wachter, H. Induction of indoleamine 2,3-dioxygenase in human cells in vitro. In Kynurenine and Serotonin Pathways; Schwarcz, R., Young, S.N., Brown, R.R., Eds.; Springer: New York, NY, USA, 1991; pp. 505–509. [Google Scholar]
- Yoshida, R.; Imanishi, J.; Oku, T.; Kishida, T.; Hayaishi, O. Induction of pulmonary indoleamine 2,3-dioxygenase by interferon. Proc. Natl. Acad. Sci. USA 1981, 78, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, H.J.; Takubo, M.; Takahashi, A.; Hasegawa, T.; Noma, H.; Suzuki, T. Evolution of vertebrate indoleamine 2,3-dioxygenases. J. Mol. Evol. 2007, 65, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000Prime Rep. 2014. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Martinez, F.O. Alternative activation of macrophages: Mechanism and functions. Immunity 2010, 32, 593–604. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, M.R.; Gongora, M.M.; Grimmond, S.M.; Perkins, A.C. A global role for zebrafish KLF4 in embryonic erythropoiesis. Mech. Dev. 2007, 124, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Martinez, F.O.; Helming, L.; Milde, R.; Varin, A.; Melgert, B.N.; Draijer, C.; Thomas, B.; Fabbri, M.; Crawshaw, A.; Ho, L.P.; et al. Genetic programs expressed in resting and IL-4 alternatively activated mouse and human macrophages: Similarities and differences. Blood 2013, 121, e57–e69. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.B.; Langenau, D.M.; Goetz, F.W. Cloning and characterization of prostaglandin endoperoxide synthase-1 and -2 from the brook trout ovary. Mol. Cell. Endocrinol. 2000, 160, 89–97. [Google Scholar] [CrossRef]
- Skjesol, A.; Liebe, T.; Iliev, D.B.; Thomassen, E.I.S.; Tollersrud, L.G.; Sobhkhez, M.; Lindenskov Joensen, L.; Secombes, C.J.; Jørgensen, J.B. Functional conservation of suppressors of cytokine signaling proteins between teleosts and mammals: Atlantic salmon SOCS1 binds to JAK/STAT family members and suppresses type I and II IFN signaling. Dev. Comp. Immunol. 2014, 45, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Varela, M.; Diaz-Rosales, P.; Pereiro, P.; Forn-Cuní, G.; Costa, M.M.; Dios, S.; Romero, A.; Figueras, A.; Novoa, B. Interferon-induced genes of the expanded IFIT family show conserved antiviral activities in non-mammalian species. PLoS ONE 2014, 9, e100015. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, M.; Hayashi, N.; Hashimoto, K.; Nakanishi, T.; Dijkstra, J.M. Comprehensive clarification of two paralogous interleukin 4/13 loci in teleost fish. Immunogenetics 2008, 60, 383–397. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Secombes, C.J. The evolution of IL-4 and IL-13 and their receptor subunits. Cytokine 2015, 75, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Huang, W.; Costa, M.M.; Martin, S.A.M.; Secombes, C.J. Two copies of the genes encoding the subunits of putative interleukin (IL)-4/IL-13 receptors, IL-4Rα, IL-13Rα1 and IL-13Rα2, have been identified in rainbow trout (Oncorhynchus mykiss) and have complex patterns of expression and modulation. Immunogenetics 2011, 63, 235–253. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Pan, P.; Fang, W.; Shao, J.; Xiang, L. Essential role of IL-4 and IL-4Rα interaction in adaptive immunity of zebrafish: Insight into the origin of Th2-like regulatory mechanism in ancient vertebrates. J. Immunol. 2012, 188, 5571–5584. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.-L.; Xiang, L.-X.; Shao, J.-Z. Identification and characterization of a novel immunoglobulin Z isotype in zebrafish: Implications for a distinct B cell receptor in lower vertebrates. Mol. Immunol. 2010, 47, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.F.; Xiang, L.X.; Wang, Q.L.; Dong, W.R.; Gong, Y.F.; Shao, J.Z. The DC-SIGN of zebrafish: Insights into the existence of a CD209 homologue in a lower vertebrate and its involvement in adaptive immunity. J. Immunol. 2009, 183, 7398–7410. [Google Scholar] [CrossRef] [PubMed]
- Barksdale, A.R.; Bernard, A.C.; Maley, M.E.; Gellin, G.L.; Kearney, P.A.; Boulanger, B.R.; Tsuei, B.J.; Ochoa, J.B. Regulation of arginase expression by T-helper II cytokines and isoproterenol. Surgery 2004, 135, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Green, S.J.; Crawford, R.M.; Hockmeyer, J.T.; Meltzer, M.S.; Nacy, C.A. Leishmania major amastigotes initiate the l-arginine-dependent killing mechanism in IFN-γ-stimulated macrophages by induction of tumor necrosis factor-alpha. J. Immunol. 1990, 145, 4290–4297. [Google Scholar] [PubMed]
- Iniesta, V.; Gómez-Nieto, L.C.; Molano, I.; Mohedano, A.; Carcelén, J.; Mirón, C.; Alonso, C.; Corraliza, I. Arginase I induction in macrophages, triggered by Th2-type cytokines, supports the growth of intracellular Leishmania parasites. Parasite Immunol. 2002, 24, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Liew, F.Y.; Li, Y.; Moss, D.; Parkinson, C.; Rogers, M.V.; Moncada, S. Resistance to Leishmania major infection correlates with the induction of nitric oxide synthase in murine macrophages. Eur. J. Immunol. 1991, 21, 3009–3014. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.D. Macrophage arginine metabolism to ornithine/urea or nitric oxide/citrulline: A life or death issue. Crit. Rev. Immunol. 2001, 21, 399–425. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.M. Recent advances in arginine metabolism: Roles and regulation of the arginases. Br. J. Pharmacol. 2009, 157, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Munder, M.; Eichmann, K.; Morán, J.M.; Centeno, F.; Soler, G.; Modolell, M. Th1/Th2-regulated expression of arginase isoforms in murine macrophages and dendritic cells. J. Immunol. 1999, 163, 3771–3777. [Google Scholar] [PubMed]
- Lang, R.; Patel, D.; Morris, J.J.; Rutschman, R.L.; Murray, P.J. Shaping gene expression in activated and resting primary macrophages by IL-10. J. Immunol. 2002, 169, 2253–2263. [Google Scholar] [CrossRef] [PubMed]
- Wright, P.A.; Campbell, A.; Morgan, R.L.; Rosenberger, A.G.; Murray, B.W. Dogmas and controversies in the handling of nitrogenous wastes: Expression of arginase Type I and II genes in rainbow trout: Influence of fasting on liver enzyme activity and mRNA levels in juveniles. J. Exp. Biol. 2004, 207, 2033–2042. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.I.; Zoghi, B.; Liao, J.C.; Kuo, L. The involvement of tyrosine kinases, cyclic AMP/protein kinase A, and p38 mitogen-activated protein kinase in IL-13-mediated arginase I induction in macrophages: Its implications in IL-13-inhibited nitric oxide production. J. Immunol. 2000, 165, 2134–2141. [Google Scholar] [CrossRef] [PubMed]
- Forlenza, M.; Fink, I.R.; Raes, G.; Wiegertjes, G.F. Heterogeneity of macrophage activation in fish. Dev. Comp. Immunol. 2011, 35, 1246–1255. [Google Scholar] [CrossRef] [PubMed]
- Wiegertjes, G.F.; Wentzel, A.S.; Spaink, H.P.; Elks, P.M.; Fink, I.R. Polarization of immune responses in fish: The “macrophages first” point of view. Mol. Immunol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gensel, J.C.; Zhang, B. Macrophage activation and its role in repair and pathology after spinal cord injury. Brain Res. 2015, 1619, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ehrchen, J.; Steinmüller, L.; Barczyk, K.; Tenbrock, K.; Nacken, W.; Eisenacher, M.; Nordhues, U.; Sorg, C.; Sunderkötter, C.; Roth, J. Glucocorticoids induce differentiation of a specifically activated, anti-inflammatory subtype of human monocytes. Blood 2007, 109, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- Van de Garde, M.D.B.; Martinez, F.O.; Melgert, B.N.; Hylkema, M.N.; Jonkers, R.E.; Hamann, J. Chronic exposure to glucocorticoids shapes gene expression and modulates innate and adaptive activation pathways in macrophages with distinct changes in leukocyte attraction. J. Immunol. 2014, 192, 1196–1208. [Google Scholar] [CrossRef] [PubMed]
- Maule, A.G.; Tripp, R.A.; Kaattari, S.L.; Schreck, C.B. Stress alters immune function and disease resistance in chinook salmon (Oncorhynchus tshawytscha). J. Endocrinol. 1989, 120, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Montero, D.; Marrero, M.; Izquierdo, M.S.; Robaina, L.; Vergara, J.M.; Tort, L. Effect of vitamin E and C dietary supplementation on some immune parameters of gilthead seabream (Sparus aurata) juveniles subjected to crowding stress. Aquaculture 1999, 171, 269–278. [Google Scholar] [CrossRef]
- Pickering, A.D.; Pottinger, T.G. Cortisol can increase the susceptibility of brown trout, Salmo trutta L., to disease without reducing the white blood cell count. J. Fish Biol. 1985, 27, 611–619. [Google Scholar] [CrossRef]
- Wang, R.; Belosevic, M. The in vitro effects of estradiol and cortisol on the function of a long-term goldfish macrophage cell line. Dev. Comp. Immunol. 1995, 19, 327–336. [Google Scholar] [CrossRef]
- Ferrante, C.J.; Leibovich, S.J. Regulation of macrophage polarization and wound healing. Adv. Wound Care 2012, 1, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, L.; Belosevic, M. Identification and molecular characterization of the interleukin-10 receptor 1 of the zebrafish (Danio rerio) and the goldfish (Carassius auratus L.). Dev. Comp. Immunol. 2012, 36, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Wang, S.; Qin, L.; Wang, X.; Zhou, H. Molecular characterization, 3D modeling of grass carp interleukin-10 receptor 1 (IL10R1). Engineering 2013, 5, 214–219. [Google Scholar] [CrossRef]
- Monte, M.M.; Wang, T.; Collet, B.; Zou, J.; Secombes, C.J. Molecular characterisation of four class 2 cytokine receptor family members in rainbow trout, Oncorhynchus mykiss. Dev. Comp. Immunol. 2015, 48, 43–54. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodgkinson, J.W.; Grayfer, L.; Belosevic, M. Biology of Bony Fish Macrophages. Biology 2015, 4, 881-906. https://doi.org/10.3390/biology4040881
Hodgkinson JW, Grayfer L, Belosevic M. Biology of Bony Fish Macrophages. Biology. 2015; 4(4):881-906. https://doi.org/10.3390/biology4040881
Chicago/Turabian StyleHodgkinson, Jordan W., Leon Grayfer, and Miodrag Belosevic. 2015. "Biology of Bony Fish Macrophages" Biology 4, no. 4: 881-906. https://doi.org/10.3390/biology4040881




