Hollow Au–Ag Alloy Nanorices and Their Optical Properties
Abstract
:1. Introduction
2. Result and Discussion
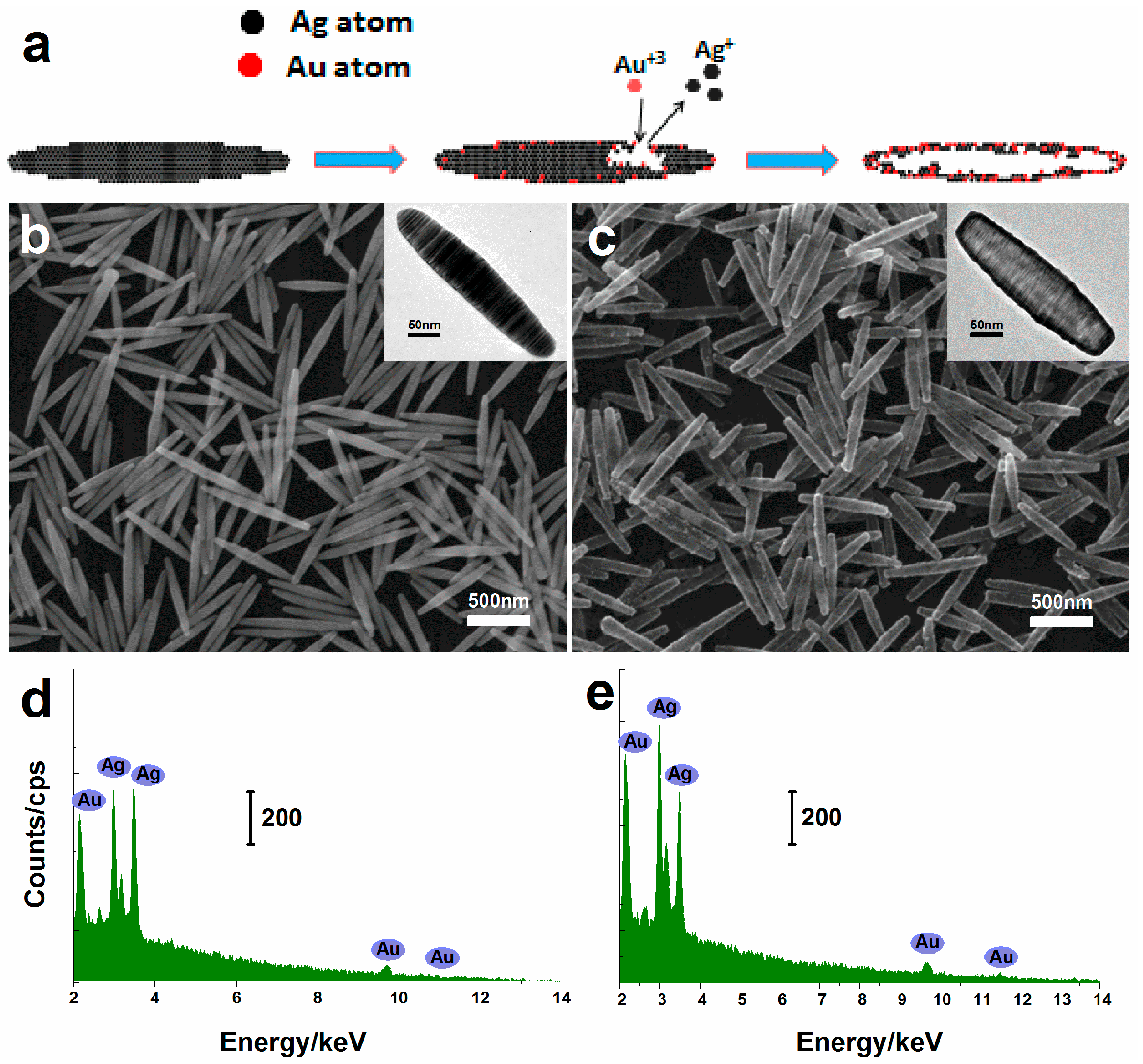
2.1. Characterization of Nanorices
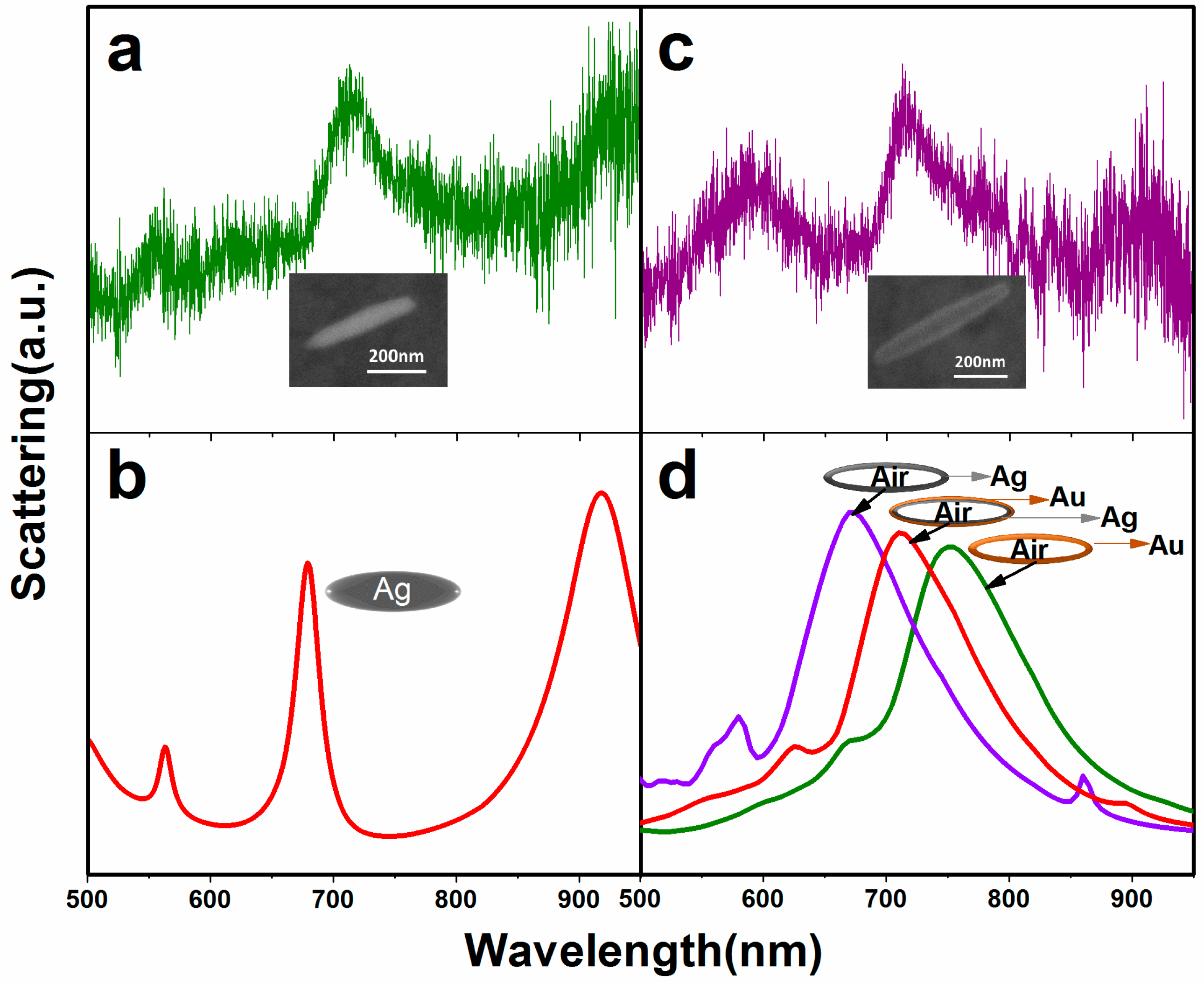
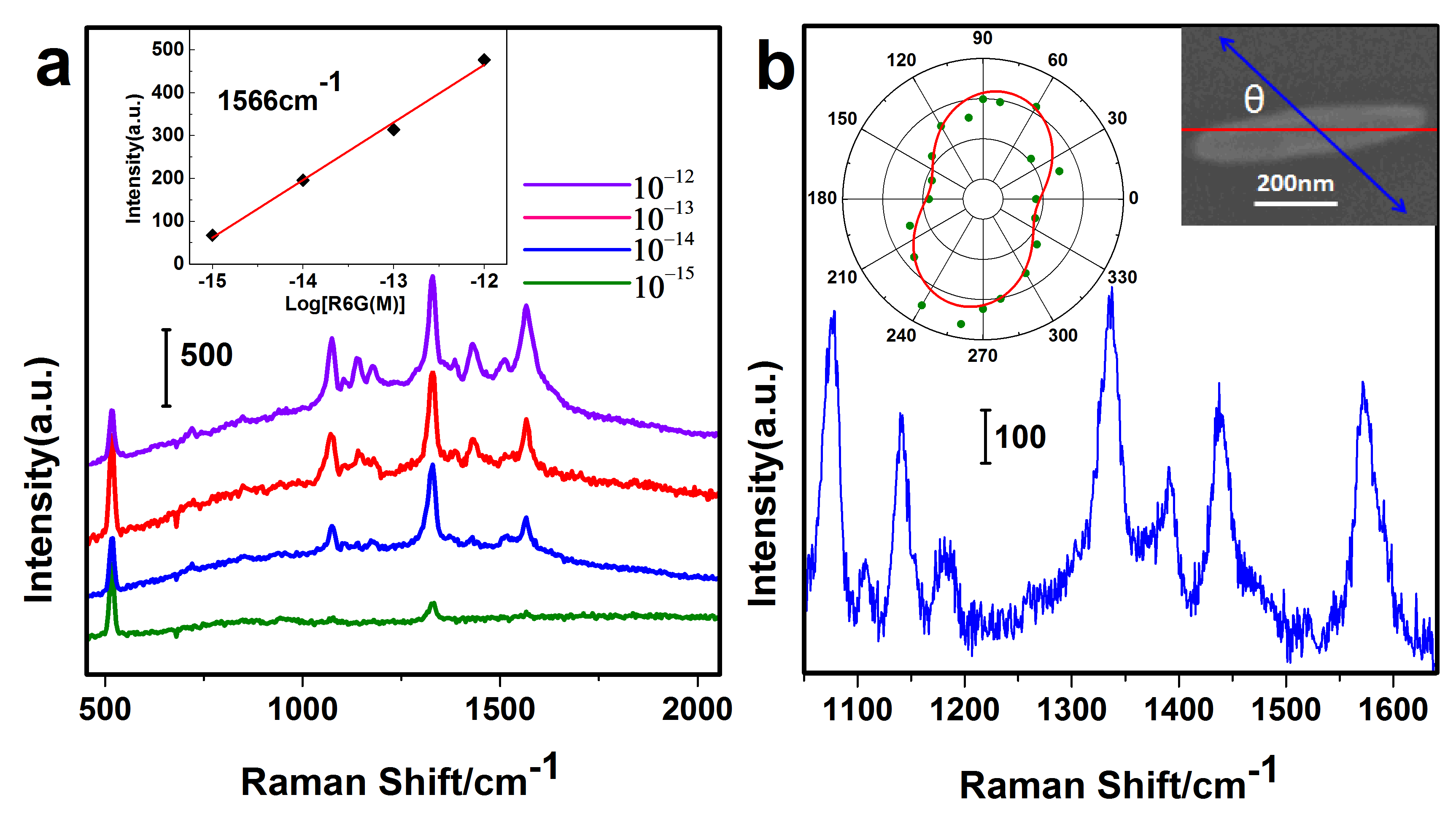
2.2. Optical Properties of Nanorices
2.3. Oxidation Resistance of Nanorices
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Ag Nanorices
3.3. Synthesis of Hollow Au–Ag Alloy Nanorices
3.4. Characterization
3.5. Optical Properties Test
3.6. Oxidation Resistance Analysis
3.7. COMSOL Multiphysics Simulation
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ozbay, E. Plasmonics: Merging photonics and electronics at nanoscale dimensions. Science 2006, 311, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Rycenga, M.; Skrabalak, S.E.; Wiley, B.; Xia, Y. Chemical synthesis of novel plasmonic nanoparticles. Annu. Rev. Phys. Chem. 2009, 60, 167–192. [Google Scholar] [CrossRef] [PubMed]
- Brongersma, M.L.; Shalaev, V.M. Applied physics. The case for plasmonics. Science 2010, 328, 440–441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jin, M.S.; Xia, Y.N. Noble-metal nanocrystals with concave surfaces: Synthesis and applications. Angew. Chem. Int. Ed. 2012, 51, 7656–7673. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Sheng, S.; Wang, R.; Sun, M. Tip-enhanced raman spectroscopy. Anal. Chem. 2016, 88, 9328–9346. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.-Y.; Yi, J.; Li, J.-F.; Ren, B.; Wu, D.-Y.; Panneerselvam, R.; Tian, Z.-Q. Nanostructure-based plasmon-enhanced raman spectroscopy for surface analysis of materials. Nat. Rev. Mater. 2016, 1, 16021. [Google Scholar] [CrossRef]
- Huang, Y.Z.; Fang, Y.R.; Sun, M.T. Remote excitation of surface-enhanced raman scattering on single au nanowire with quasi-spherical termini. J. Phys. Chem. C 2011, 115, 3558–3561. [Google Scholar] [CrossRef]
- Zhang, Z.; Fang, Y.; Wang, W.; Chen, L.; Sun, M. Propagating surface plasmon polaritons: Towards applications for remote-excitation surface catalytic reactions. Adv. Sci. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Xu, H. A novel application of plasmonics: Plasmon-driven surface-catalyzed reactions. Small 2012, 8, 2777–2786. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Tian, X.; Huang, Y.; Fang, L.; Fang, Y. Quantitatively analyzing the mechanism of giant circular dichroism in extrinsic plasmonic chiral nanostructures by tracking the interplay of electric and magnetic dipoles. Nanoscale 2016, 8, 3720–3728. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Sun, M. Nanoplasmonic waveguides: Towards applications in integrated nanophotonic circuits. Light Sci. Appl. 2015, 4, e294. [Google Scholar] [CrossRef]
- Pan, D.; Wei, H.; Gao, L.; Xu, H.X. Strong spin-orbit interaction of light in plasmonic nanostructures and nanocircuits. Phys. Rev. Lett. 2016, 117, 166803. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.R.; Jiao, Y.; Xiong, K.L.; Ogier, R.; Yang, Z.J.; Gao, S.W.; Dahlin, A.B.; Kall, M. Plasmon enhanced internal photoemission in antenna-spacer-mirror based Au/TiO2 nanostructures. Nano Lett. 2015, 15, 4059–4065. [Google Scholar] [CrossRef] [PubMed]
- Mubeen, S.; Lee, J.; Singh, N.; Kramer, S.; Stucky, G.D.; Moskovits, M. An autonomous photosynthetic device in which all charge carriers derive from surface plasmons. Nat. Nanotechnol. 2013, 8, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Tan, Y.; Wang, J.; Xu, W.; Yuan, Y.; Cai, W.; Zhu, S.; Zhu, J. 3D self-assembly of aluminium nanoparticles for plasmon-enhanced solar desalination. Nat. Photonics 2016, 10, 393–398. [Google Scholar] [CrossRef]
- Neumann, O.; Urban, A.S.; Day, J.; Lal, S.; Nordlander, P.; Halas, N.J. Solar vapor generation enabled by nanoparticles. ACS Nano 2013, 7, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Halas, N.J.; Lal, S.; Chang, W.S.; Link, S.; Nordlander, P. Plasmons in strongly coupled metallic nanostructures. Chem. Rev. 2011, 111, 3913–3961. [Google Scholar] [CrossRef] [PubMed]
- Perrault, S.D.; Chan, W.C. Synthesis and surface modification of highly monodispersed, spherical gold nanoparticles of 50–200 nm. J. Am. Chem. Soc. 2009, 131, 17042–17043. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.G.; Xia, Y.N. Shape-controlled synthesis of gold and silver nanoparticles. Science 2002, 298, 2176–2179. [Google Scholar] [CrossRef] [PubMed]
- Gou, L.F.; Chipara, M.; Zaleski, J.M. Convenient, rapid synthesis of Ag nanowires. Chem. Mater. 2007, 19, 1755–1760. [Google Scholar] [CrossRef]
- Liang, H.Y.; Yang, H.X.; Wang, W.Z.; Li, J.Q.; Xu, H.X. High-yield uniform synthesis and microstructure-determination of rice-shaped silver nanocrystals. J. Am. Chem. Soc. 2009, 131, 6068–6069. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Ji, F.; Xu, Y.; He, L.; Mi, Y.; Bao, F.; Sun, B.; Zhang, X.; Zhang, Q. High-yield seedless synthesis of triangular gold nanoplates through oxidative etching. Nano Lett. 2014, 14, 7201–7206. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhang, Z.; Wang, X. Well-defined metal—Organic-framework hollow nanostructures for catalytic reactions involving gases. Adv. Mater. 2015, 27, 5365. [Google Scholar] [CrossRef] [PubMed]
- Moon, G.D.; Ji, B.J.; Dahl, M.; Jung, H.; Yin, Y. Nitridation and layered assembly of hollow TiO2 shells for electrochemical energy storage. Adv. Funct. Mater. 2014, 24, 848–856. [Google Scholar] [CrossRef]
- Zhang, W.; Hou, X.; Lin, Z.; Yao, L.; Wang, X.; Gao, Y.; Hu, S. Hollow microspheres and nanoparticles MnFe2O4 as superior anode materials for lithium ion batteries. J. Mater. Sci. Mater. Electron. 2015, 26, 9535–9545. [Google Scholar] [CrossRef]
- Young, K.L.; Scott, A.W.; Hao, L.; Mirkin, S.E.; Liu, G.; Mirkin, C.A. Hollow spherical nucleic acids for intracellular gene regulation based upon biocompatible silica shells. Nano Lett. 2012, 12, 3867. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Mayers, B.T.; Xia, Y. Template-engaged replacement reaction: A one-step approach to the large-scale synthesis of metal nanostructures with hollow interiors. Nano Lett. 2002, 2, 481–485. [Google Scholar] [CrossRef]
- Skrabalak, S.E.; Chen, J.; Sun, Y.; Lu, X.; Au, L.; Cobley, C.M.; Xia, Y. Cheminform abstract: Gold nanocages: Synthesis, properties, and applications. Acc. Chem. Res. 2008, 41, 1587. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.G.; Mayers, B.T.; Xia, Y.N. Metal nanostrutures with hollow interiors. Adv. Mater. 2003, 15, 641–646. [Google Scholar] [CrossRef]
- Fang, Z.; Wang, Y.; Liu, C.; Chen, S.; Sang, W.; Wang, C.; Zeng, J. Rational design of metal nanoframes for catalysis and plasmonics. Small 2015, 11, 2593–2605. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, J.; Fu, Z.W.; Qin, D. Galvanic replacement-free deposition of Au on Ag for core-shell nanocubes with enhanced chemical stability and sers activity. J. Am. Chem. Soc. 2014, 136, 8153–8156. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Wang, Y.; Ruditskiy, A.; Xia, Y. 25th anniversary article: Galvanic replacement: A simple and versatile route to hollow nanostructures with tunable and well-controlled properties. Adv. Mater. 2013, 25, 6313–6333. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Reyescoronado, A.; Nordlander, P.; Aizpurua, J.; Xu, H. Multipolar plasmon resonances in individual Ag nanorice. ACS Nano 2010, 4, 2649–2654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaffin, E.; O’Connor, R.T.; Barr, J.; Huang, X.; Wang, Y. Dependence of SERS enhancement on the chemical composition and structure of Ag/Au hybrid nanoparticles. J. Chem. Phys. 2016, 145, 054706. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Jia, Y.; Liu, A.; Sun, X.; Su, X.; Zhang, X.; Li, K.; Huang, Y. Effects of substrate and polarization on plasmon driven surface catalysis in nanowire-film hybrid system. Superlattices Microstruct. 2016, 100, 886–891. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Yang, Z.J.; Liu, X.L.; Li, M.; Zhou, L. Multiple plasmon resonances of Au/Ag alloyed hollow nanoshells. Scr. Mater. 2010, 63, 1193–1196. [Google Scholar] [CrossRef]
- Johnson, P.B.; Christy, R.W. Optical constants of noble metals. Phys. Rev. B 1972, 6, 4370. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, K.; Sun, X.; Pan, L.; Liu, T.; Liu, A.; Chen, G.; Huang, Y. Hollow Au–Ag Alloy Nanorices and Their Optical Properties. Nanomaterials 2017, 7, 255. https://doi.org/10.3390/nano7090255
Yu K, Sun X, Pan L, Liu T, Liu A, Chen G, Huang Y. Hollow Au–Ag Alloy Nanorices and Their Optical Properties. Nanomaterials. 2017; 7(9):255. https://doi.org/10.3390/nano7090255
Chicago/Turabian StyleYu, Keke, Xiaonan Sun, Liang Pan, Ting Liu, Anping Liu, Guo Chen, and Yingzhou Huang. 2017. "Hollow Au–Ag Alloy Nanorices and Their Optical Properties" Nanomaterials 7, no. 9: 255. https://doi.org/10.3390/nano7090255




