Heat Generation and Transfer Behaviors of Ti-Coated Carbon Steel Rod Adaptable for Ablation Therapy of Oral Cancer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of the Sample and Tissue-Mimicking Phantom
| Ingredient | Weight (g) | Ratio (%) |
|---|---|---|
| Deionized water | 365.0 | 74.8 |
| Agar | 20.0 | 4.1 |
| Polyethylene powder | 70.0 | 14.4 |
| Sodium chloride (NaCl) | 7.9 | 1.6 |
| Boric acid | 7.9 | 1.6 |
| TX-151 | 16.9 | 3.5 |
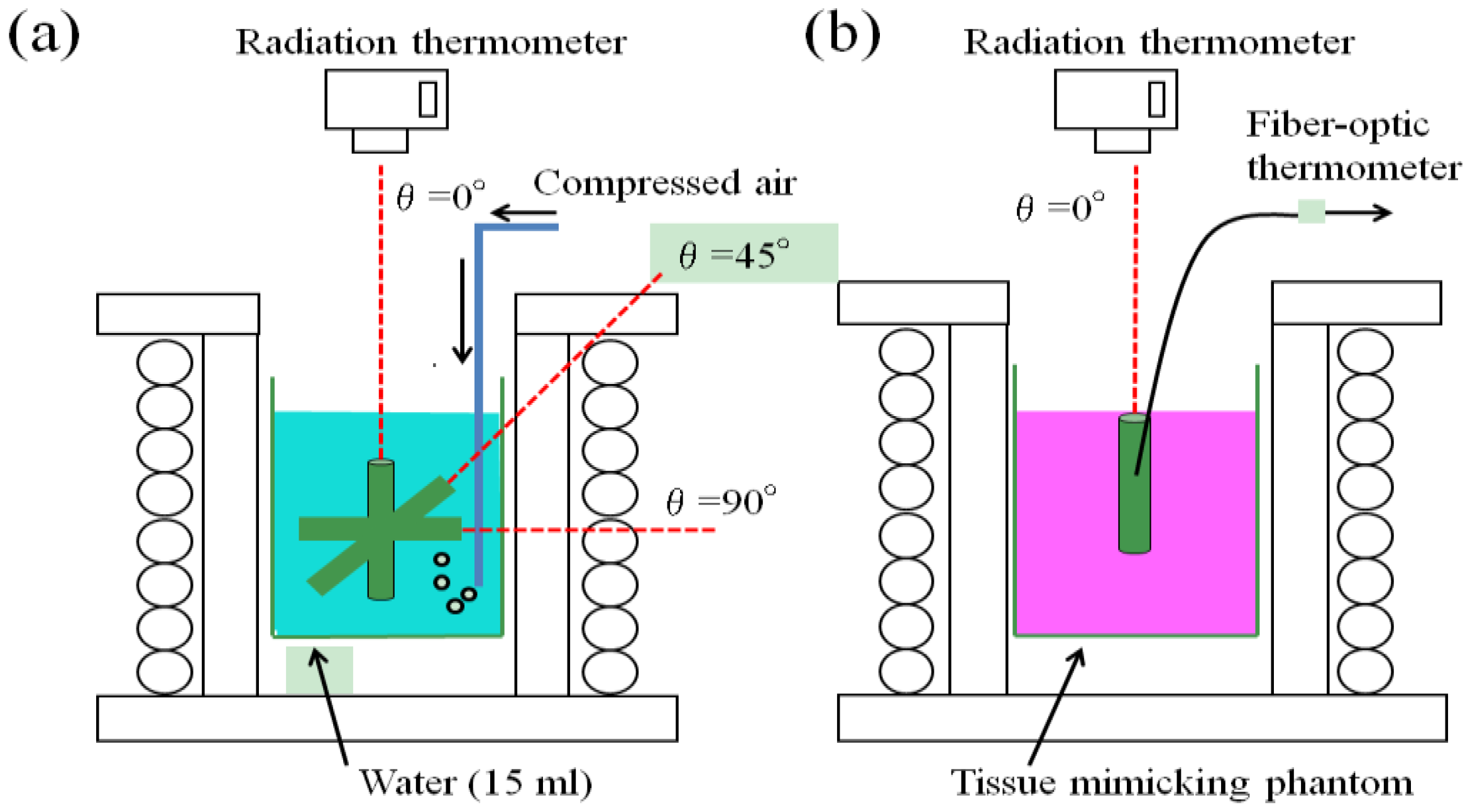
2.2. Experimental Procedures


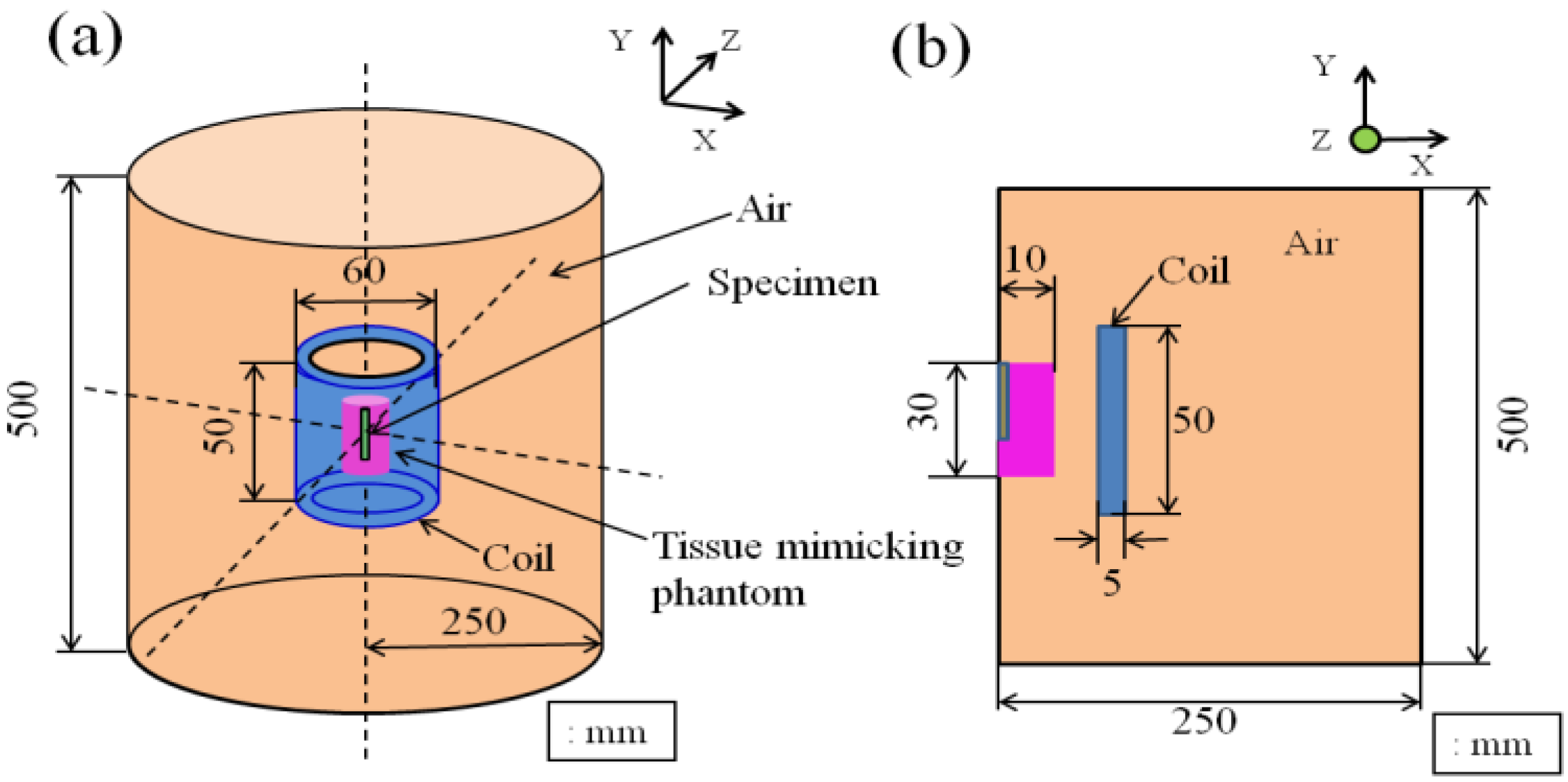
2.3 Computer Simulation of Heat Transfer in the Tissue-Mimicking Phantom
3. Results and Discussion
3.1. Heat Generation Ability of the Ti-Coated Carbon Steel Rod

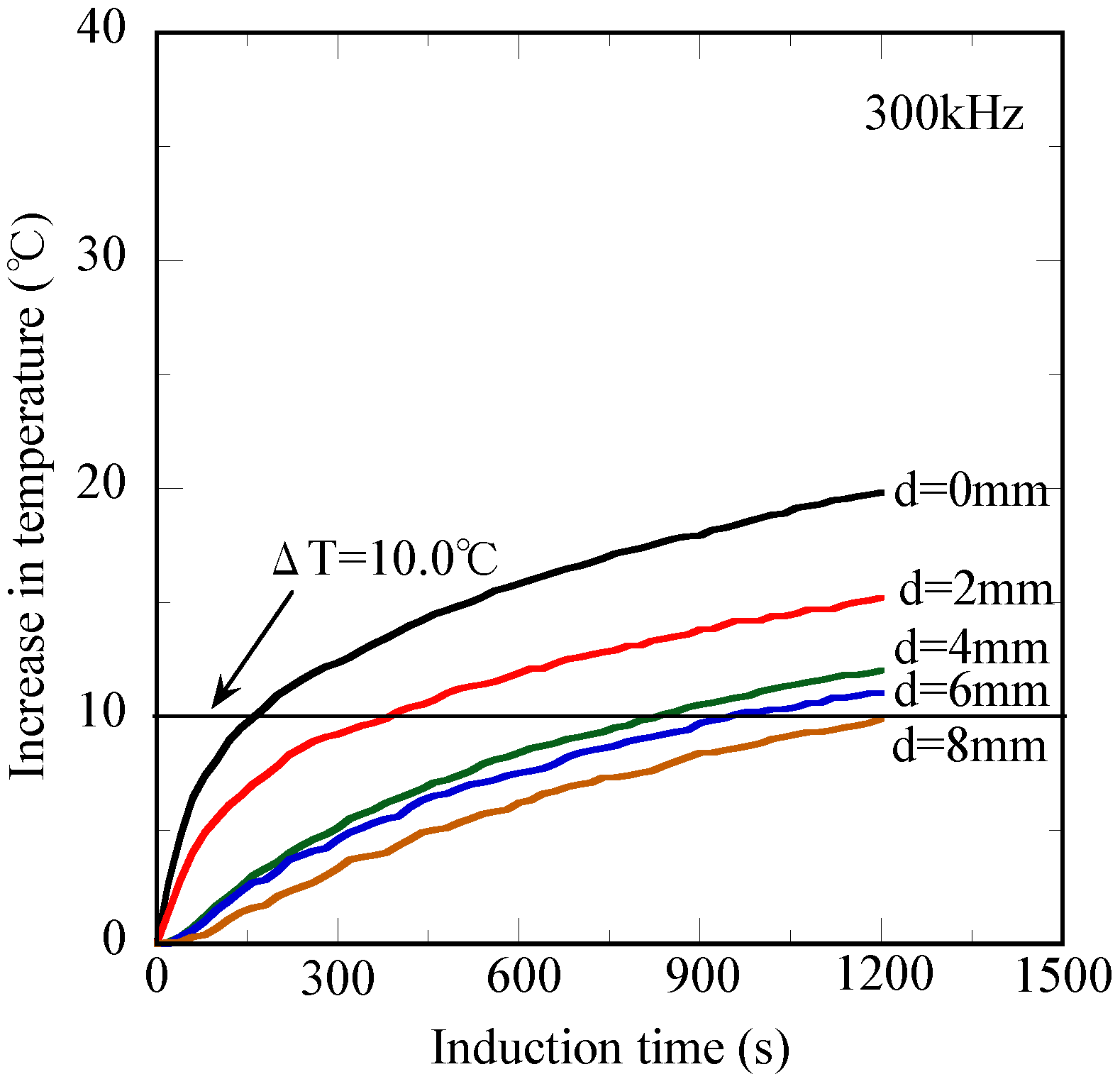
3.2. Heat Transfer Behavior of the Tissue-Mimicking Phantom

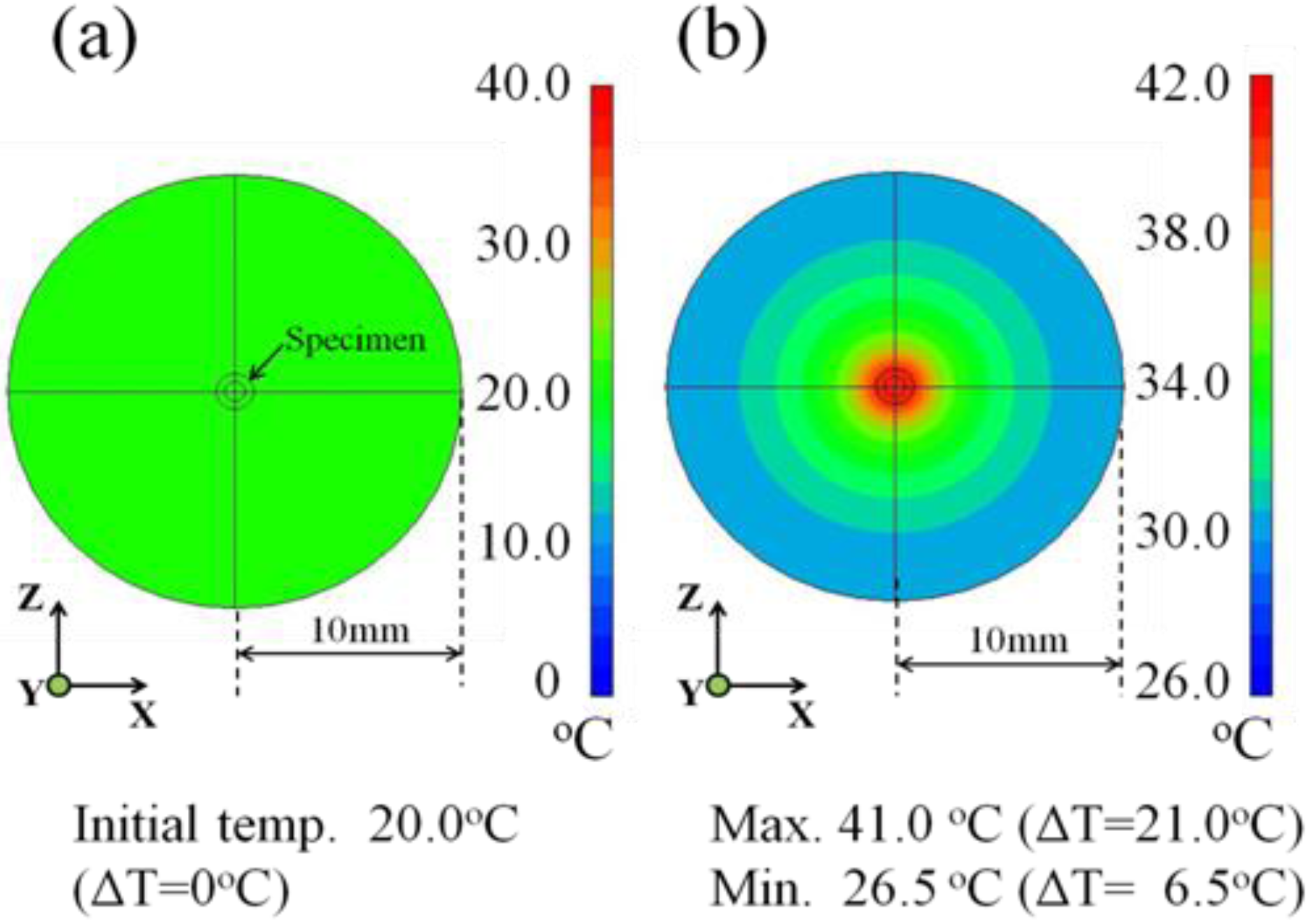
3.3 Heat transfer Simulation Images of the Tissue-Mimicking Phantom

4. Conclusions
Acknowledgment
References
- Neville, B.W.; Day, T.A. Oral cancer and precancerous lesions. CA Cancer J. Clin. 2002, 52, 195–215. [Google Scholar] [CrossRef]
- MacCarthy, D.; Flint, S.R.; Healy, C.; Stassen, L.F. Oral and neck examination for early detection of oral cancer—A practical guide. J. Ir. Dent. Assoc. 2011, 57, 195–199. [Google Scholar]
- Werning, J.W. Oral Cancer: Diagnosis, Management, and Rehabilitation; Thieme Medical Publishers, Inc.: New York, NY, USA, 2007; pp. 38–41. [Google Scholar]
- Sciubba, J.J. Oral cancer: The importance of early diagnosis and treatment. Am. J. Clin. Dermatol. 2001, 2, 239–251. [Google Scholar] [CrossRef]
- Scully, C.; Bagan, J.V. Recent advances in oral oncology 2007: Imaging, treatment and treatment incomes. Oral Oncol. 2008, 44, 211–215. [Google Scholar] [CrossRef]
- Yao, M.; Epstein, J.B.; Modi, B.J.; Pytynia, K.B.; Mundt, A.J.; Feldman, L.E. Current surgical treatment of squamous cell carcinoma of the head and neck. Oral Oncol. 2007, 43, 213–223. [Google Scholar] [CrossRef]
- Kronenberger, M.B.; Meyers, A.D. Dysphagia following head and neck cancer surgery. Dysphagia 1994, 9, 236–244. [Google Scholar] [CrossRef]
- Davies, A.; Epstein, J. Oral Complications of Oral Cancer and Its Management; Oxford University Press: New York, NY, USA, 2010; pp. 80–82. [Google Scholar]
- Sonis, S.T. Oral mucositis in cancer therapy. J. Support. Oncol. 2004, 2, 3–8. [Google Scholar]
- Vissink, A.; Jansma, J.; Spijkervet, F.K.; Burlage, F.R.; Coppes, R.P. Oral sequelae of head and neck radiotherapy. Crit. Rev. Oral Biol. Med. 2003, 14, 199–212. [Google Scholar] [CrossRef]
- Dreizen, S.; Daly, T.E.; Drane, J.B.; Brown, L.R. Oral complication of cancer radiotherapy. Postgrad. Med. 1977, 61, 85–92. [Google Scholar]
- Burri, R.J.; Lee, N.Y. Concurrent chemotherapy and radiotherapy for head and neck cancer. Expert Rev. Anticancer Ther. 2009, 9, 293–302. [Google Scholar] [CrossRef]
- Molin, Y.; Fayette, J. Current chemotherapies for recurrent/metastatic head and neck cancer. Anticancer Drugs 2011, 22, 621–615. [Google Scholar] [CrossRef]
- Chaveli Lopez, B.; Gavalda Esteve, C.; Sarrion Perez, M.G. Dental treatment consideration in the chemotherapy patient. J. Clin. Exp. Dent. 2011, 3, e31–e42. [Google Scholar]
- Lionel, D.; Christophe, L.; Marc, A.; Jean-Luc, C. Oral mucositis induced by anticancer treatments: Physiopathology and treatments. Ther. Clin. Risk Manag. 2006, 2, 159–168. [Google Scholar] [CrossRef]
- Dąbrowski, T. Hair loss as a consequence of cancer chemotherapy—Physical methods of prevention. A review of the literature. Contemp. Oncol. 2011, 15, 95–101. [Google Scholar]
- Naohara, T.; Aono, H.; Maehara, T.; Hirazawa, H.; Matsutomo, S.; Watanabe, Y. Development of Ti-coated ferromagnetic needle adaptable for ablation cancer therapy by high-frequency induction heating. J. Funct. Biomater. 2012, 3, 163–172. [Google Scholar]
- Furuya, K.; Hamada, L.; Ito, K.; Kasai, H. A new muscle-equivalent phantom for SAR estimation. IEICE Trans. Commun. 1995, E78, 871–873. [Google Scholar]
- Onishi, T.; Ishido, R.; Takimoto, T.; Saito, K.; Uebayashi, S.; Ito, K. Biological tissue-equivalent agar-based solid phantom and SAR estimation using the thermographic method in the range of 3–6 GHz. IEICE Trans. Commun. 2005, E88-B, 3733–3741. [Google Scholar] [CrossRef]
- Naohara, T.; Aono, H.; Hirazawa, H.; Maehara, T.; Watanabe, Y.; Matsutomo, S. Heat generation ability in AC magnetic field of needle-type Ti-coated mild steel for ablation cancer therapy. Int. J. Comput. Math. Electr. Electron. Eng. 2011, 30, 1582–1588. [Google Scholar] [CrossRef]
- Cullity, B.D.; Graham, C.D. Introduction to Magnetic Materials; Willey IEEE Press: Hoboken, NJ, USA, 2008; pp. 234–237. [Google Scholar]
- Yamada, K.; Oda, T.; Hashimoto, S.; Enomoto, T.; Ohkohchi, N.; Ikeda, H.; Yanagihara, H.; Kishimoto, M.; Kita, E.; Tasaki, A.; Satake, M.; Ikehata, Y.; Nagae, H.; Nagano, I.; Takagi, T.; Kanamori, T. Minimally required heat doses for various tumor sizes in induction heating cancer therapy determined by computer simulation using experimental data. Int. J. Hyperthermia 2010, 26, 465–474. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Naohara, T.; Aono, H.; Maehara, T.; Hirazawa, H.; Matsutomo, S.; Watanabe, Y. Heat Generation and Transfer Behaviors of Ti-Coated Carbon Steel Rod Adaptable for Ablation Therapy of Oral Cancer. J. Funct. Biomater. 2013, 4, 27-37. https://doi.org/10.3390/jfb4010027
Naohara T, Aono H, Maehara T, Hirazawa H, Matsutomo S, Watanabe Y. Heat Generation and Transfer Behaviors of Ti-Coated Carbon Steel Rod Adaptable for Ablation Therapy of Oral Cancer. Journal of Functional Biomaterials. 2013; 4(1):27-37. https://doi.org/10.3390/jfb4010027
Chicago/Turabian StyleNaohara, Takashi, Hiromichi Aono, Tsunehiro Maehara, Hideyuki Hirazawa, Shinya Matsutomo, and Yuji Watanabe. 2013. "Heat Generation and Transfer Behaviors of Ti-Coated Carbon Steel Rod Adaptable for Ablation Therapy of Oral Cancer" Journal of Functional Biomaterials 4, no. 1: 27-37. https://doi.org/10.3390/jfb4010027




