Poly(imide)/Organically-Modified Montmorillonite Nanocomposite as a Potential Membrane for Alkaline Fuel Cells
Abstract
:1. Introduction
2. Results and Discussion
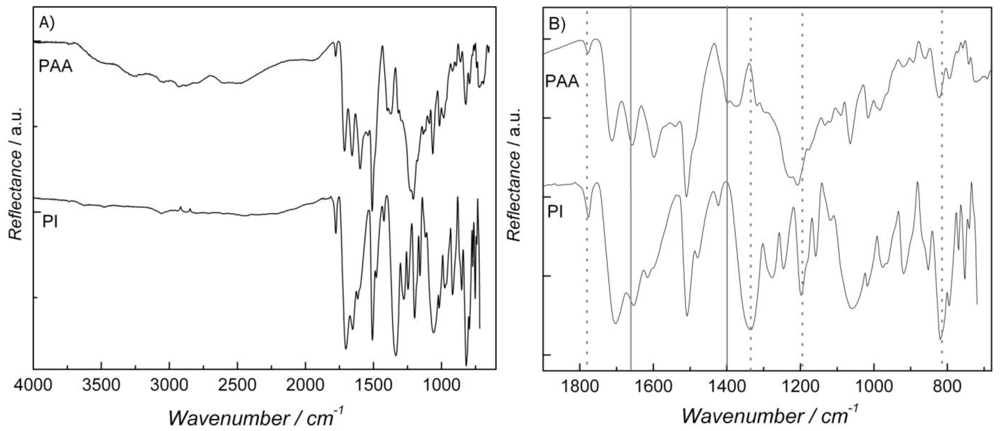
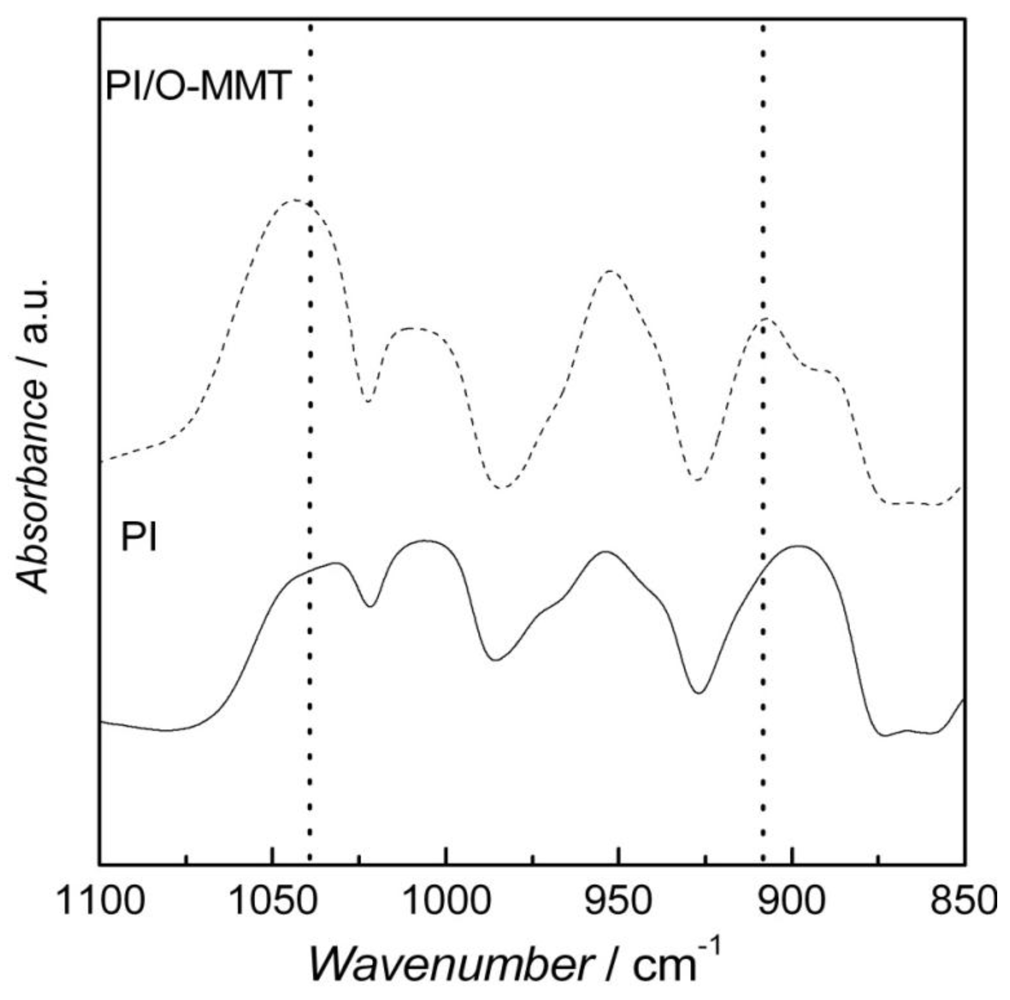
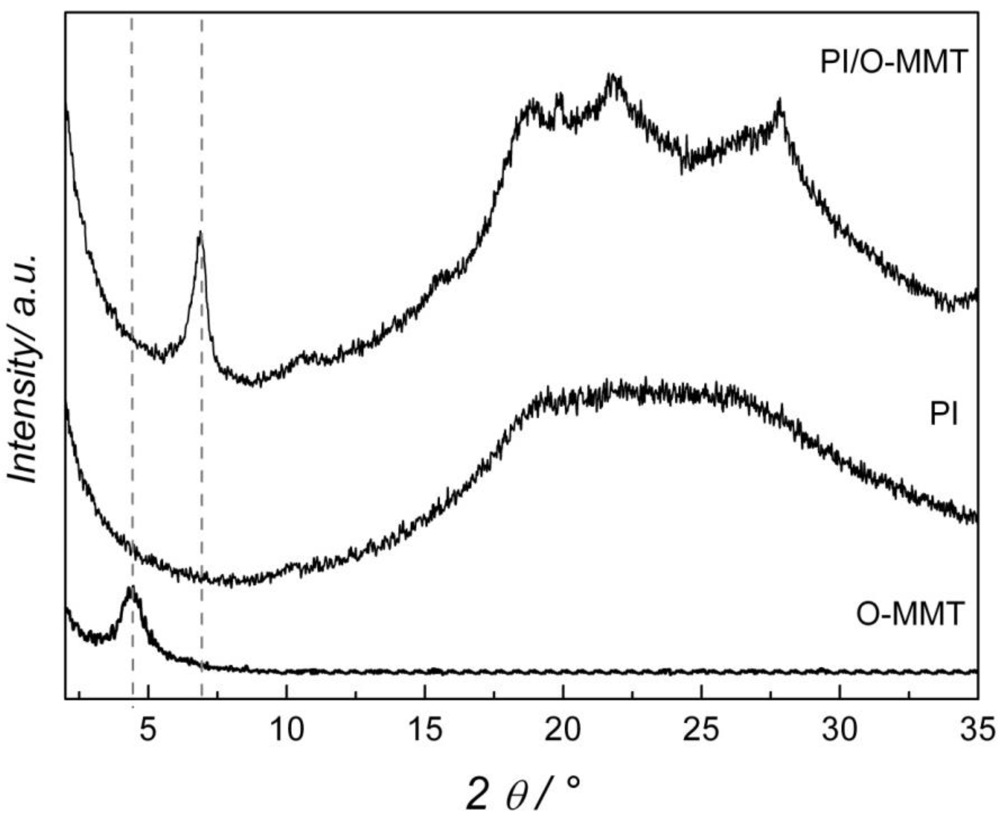

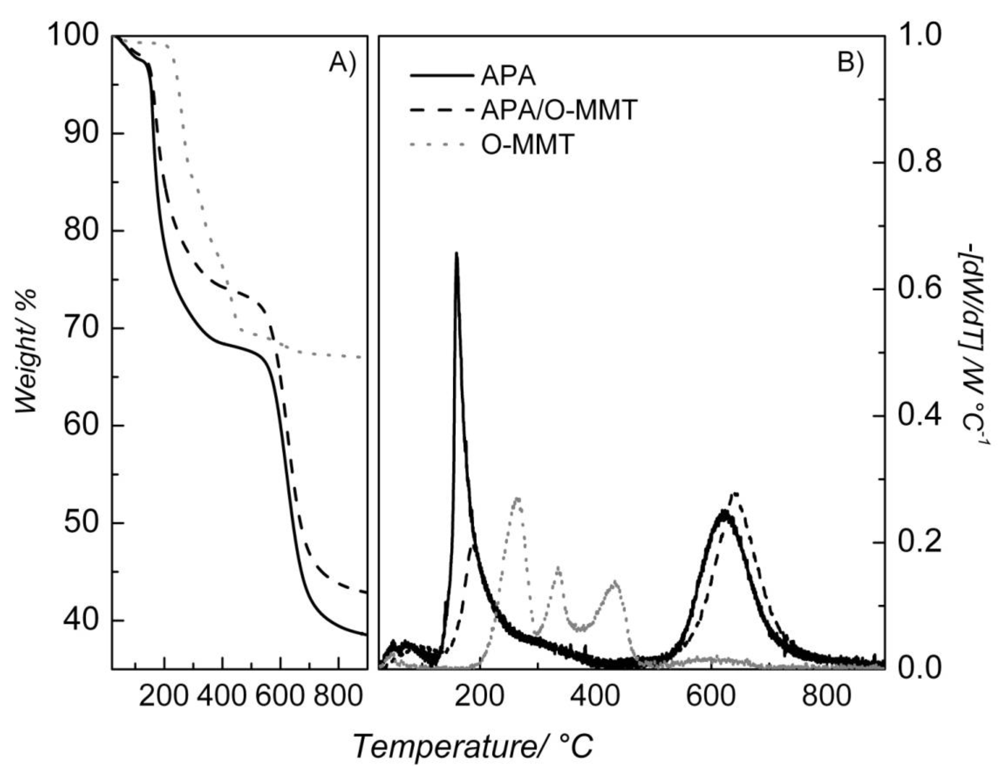
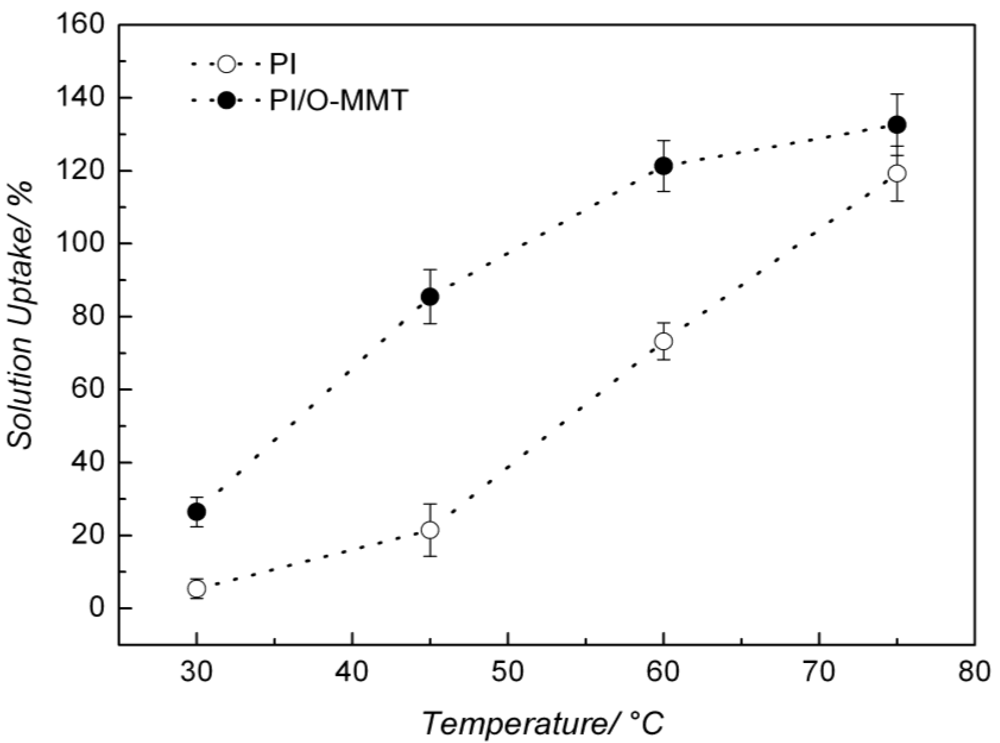
| Temperature (°C) | Solution Uptake (%) | Increment (%) | |
|---|---|---|---|
| PI | PI/O-MMT | ||
| 30 | 5.40 ± 2.70 | 26.46 ± 4.02 | ~390 |
| 45 | 21.49 ± 7.20 | 85.47 ± 7.42 | ~298 |
| 60 | 73.25 ± 5.05 | 121.33 ± 6.97 | ~66 |
| 75 | 119.25 ± 7.58 | 132.63 ± 8.40 | ~11 |
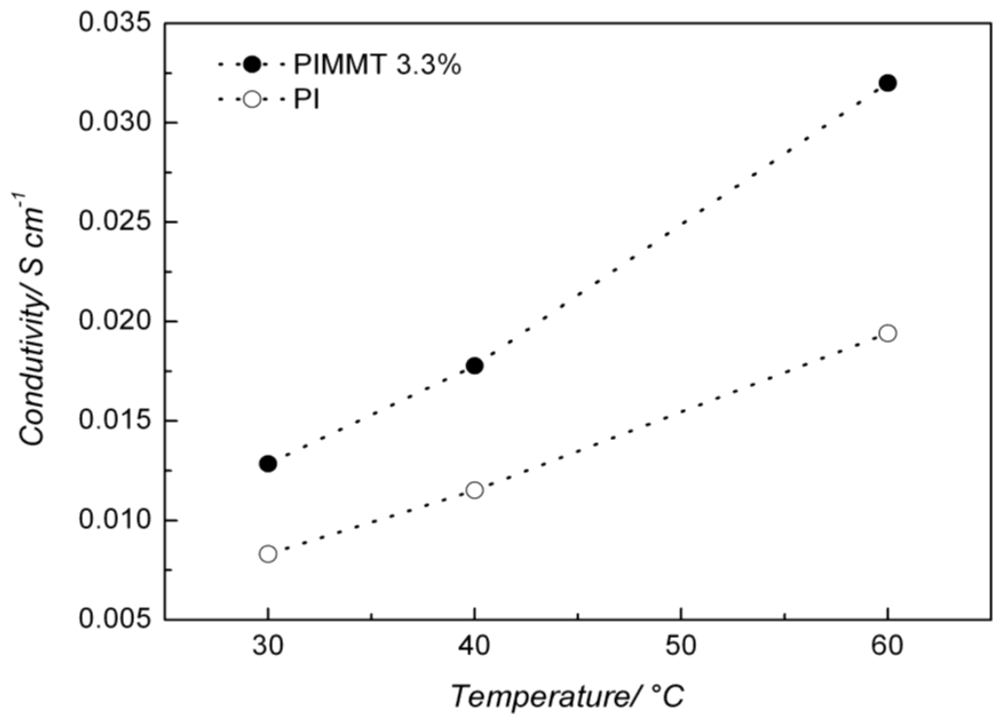
| Temperature (°C) | Conductivity (S cm−1) | Increment (%) | |
|---|---|---|---|
| PI | PI/O-MMT | ||
| 30 | 0.008 | 0.013 | 62 |
| 40 | 0.011 | 0.018 | 63 |
| 60 | 0.019 | 0.032 | 68 |
3. Experimental Section
3.1. Chemicals
3.2. Polymer Synthesis and Modification with O-MMT
3.3. Characterization
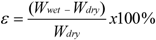
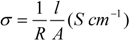
4. Conclusions
Acknowledgments
References
- Antolini, E.; Gonzalez, E.R. Alkaline direct alcohol fuel cells. J. Power Sources 2010, 195, 3431–3450. [Google Scholar] [CrossRef]
- Reid, J.H. Process of Generating Electricity. U.S. Patent 736,016,11, 11 August 1903. [Google Scholar]
- McLean, G.F.; Niet, T.; Prince-Richard, S.; Djilali, N. An assessment of alkaline fuel cell technology. Int. J. Hydrog. Energy 2002, 27, 507–526. [Google Scholar] [CrossRef]
- Bartrom, A.M.; Haan, J.L. The direct formate fuel cell with an alkaline anion exchange membrane. J. Power Sources 2012, 214, 68–74. [Google Scholar] [CrossRef]
- Agel, E.; Bouet, J.; Fauvarque, J.F. Characterization and use of anionic membranes for alkaline fuel cells. J. Power Sources 2001, 101, 267–274. [Google Scholar] [CrossRef]
- Liu, G.; Shang, Y.; Xie, X.; Wang, S.; Wang, J.; Wang, Y.; Mao, Z. Synthesis and characterization of anion exchange membranes for alkaline direct methanol fuel cells. Int. J. Hydrog. Energy 2012, 37, 848–853. [Google Scholar]
- Lu, S.; Pan, J.; Huang, A.; Zhuang, L.; Lu, J. Alkaline polymer electrolyte fuel cells completely free from noble metal catalysts. Proc. Natl. Acad. Sci. USA 2008, 105, 20611–20614. [Google Scholar]
- Couture, G.; Alaaeddine, A.; Boschet, F.; Ameduri, B. Polymeric materials as anion-exchange membranes for alkaline fuel cells. Prog. Polym. Sci. 2011, 36, 1521–1557. [Google Scholar] [CrossRef]
- Sinha Ray, S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Polym. Sci. 2003, 28, 1539–1641. [Google Scholar] [CrossRef]
- Biswas, M.; Ray, S. Recent progress in synthesis and evaluation of polymer-montmorillonite nanocomposites. Adv. Polym. Sci. 2001, 155, 167–221. [Google Scholar] [CrossRef]
- Giannelis, E.P. Polymer-layered silicate nanocomposites: Synthesis, properties and applications. Appl. Organomet. Chem. 1998, 12, 675–680. [Google Scholar]
- Xu, R.; Manias, E.; Snyder, A.J.; Runt, J. New biomedical poly(urethane uera)-layered silicate nanocomposites. Macromolecules 2000, 34, 337–339. [Google Scholar]
- Gilman, J.W.; Jackson, C.L.; Morgan, A.B.; Harris, R.; Manias, E.; Giannelis, E.P.; Wuthenow, M.; Hilton, D.; Phillips, S.H. Flammability properties of polymer—Layered-silicate nanocomposites. Polypropylene and polystyrene nanocomposites. Chem. Mater. 2000, 12, 1866–1873. [Google Scholar]
- Jung, D.H.; Cho, S.Y.; Peck, D.H.; Shin, D.R.; Kim, J.S. Preparation and performance of a Nafion®/montmorillonite nanocomposite membrane for direct methanol fuel cell. J. Power Sources 2003, 118, 205–211. [Google Scholar] [CrossRef]
- Chuang, S.W.; Hsu, S.L.C.; Hsu, C.L. Synthesis and properties of fluorine-containing polybenzimidazole/montmorillonite nanocomposite membranes for direct methanol fuel cell applications. J. Power Sources 2007, 168, 172–177. [Google Scholar] [CrossRef]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies: Tables and Charts, 3rd ed; John Wiley & Sons: New York, NY, USA, 2004. [Google Scholar]
- Lan, T.; Kaviratna, P.D.; Pinnavaia, T.J. On the nature of polyimide-clay hybrid composites. Chem. Mater. 1994, 6, 573–575. [Google Scholar] [CrossRef]
- Carroccio, S.; Puglisi, C.; Montaudo, G. New vistas in polymer degradation. Thermal oxidation processes in poly(ether imide). Macromolecules 2005, 38, 6849–6862. [Google Scholar] [CrossRef]
- Liu, P. Polymer modified clay minerals: A review. Appl. Clay Sci. 2007, 38, 64–76. [Google Scholar] [CrossRef]
- Merle, G.; Wessling, M.; Nijmeijer, K. Anion exchange membranes for alkaline fuel cells: A review. J. Membr. Sci. 2011, 377, 1–35. [Google Scholar] [CrossRef]
- Lee, C.H.; Park, H.B.; Lee, Y.M.; Lee, R.D. Importance of proton conductivity measurement in polymer electrolyte membrane for fuel cell application. Ind. Eng. Chem. Res. 2005, 44, 7617–7626. [Google Scholar] [CrossRef]
- Tricoli, V.; Carretta, N.; Bartolozzi, M. A comparative investigation of proton and methanol transport in fluorinated ionomeric membranes. J. Electrochem. Soc. 2000, 147, 1286–1290. [Google Scholar] [CrossRef]
- Litina, K.; Miriouni, A.; Gournis, D.; Karakassides, M.A.; Georgiou, N.; Klontzas, E.; Ntoukas, E.; Avgeropoulos, A. Nanocomposites of polystyrene-b-polyisoprene copolymer with layered silicates and carbon nanotubes. Eur. Polym. J. 2006, 42, 2098–2107. [Google Scholar] [CrossRef]
- Paganin, V.A.; Oliveira, C.L.F.; Ticianelli, E.A.; Springer, T.E.; Gonzalez, E.R. Modelistic interpretation of the impedance response of a polymer electrolyte fuel cell. Electrochim. Acta 1998, 43, 3761–3766. [Google Scholar]
- Springer, T.E.; Zawodzinski, T.A.; Wilson, M.S.; Gottesfeld, S. Characterization of polymer electrolyte fuel cells using AC impedance spectroscopy. J. Electrochem. Soc. 1996, 143, 587–599. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Battirola, L.C.; Gasparotto, L.H.S.; Rodrigues-Filho, U.P.; Tremiliosi-Filho, G. Poly(imide)/Organically-Modified Montmorillonite Nanocomposite as a Potential Membrane for Alkaline Fuel Cells. Membranes 2012, 2, 430-439. https://doi.org/10.3390/membranes2030430
Battirola LC, Gasparotto LHS, Rodrigues-Filho UP, Tremiliosi-Filho G. Poly(imide)/Organically-Modified Montmorillonite Nanocomposite as a Potential Membrane for Alkaline Fuel Cells. Membranes. 2012; 2(3):430-439. https://doi.org/10.3390/membranes2030430
Chicago/Turabian StyleBattirola, Liliane C., Luiz H. S. Gasparotto, Ubirajara P. Rodrigues-Filho, and Germano Tremiliosi-Filho. 2012. "Poly(imide)/Organically-Modified Montmorillonite Nanocomposite as a Potential Membrane for Alkaline Fuel Cells" Membranes 2, no. 3: 430-439. https://doi.org/10.3390/membranes2030430
APA StyleBattirola, L. C., Gasparotto, L. H. S., Rodrigues-Filho, U. P., & Tremiliosi-Filho, G. (2012). Poly(imide)/Organically-Modified Montmorillonite Nanocomposite as a Potential Membrane for Alkaline Fuel Cells. Membranes, 2(3), 430-439. https://doi.org/10.3390/membranes2030430




