Changes in Oscillatory Brain Networks after Lexical Tone Training
Abstract
:1. Introduction
1.1. Lexical Tones
1.2. Oscillatory Brain Activation
1.3. The Current Study
2. Experimental Section
2.1. Participants
2.2. Stimuli and Procedures
2.3. EEG Analysis
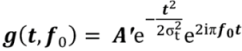
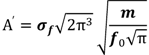
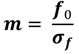
3. Results and Discussion
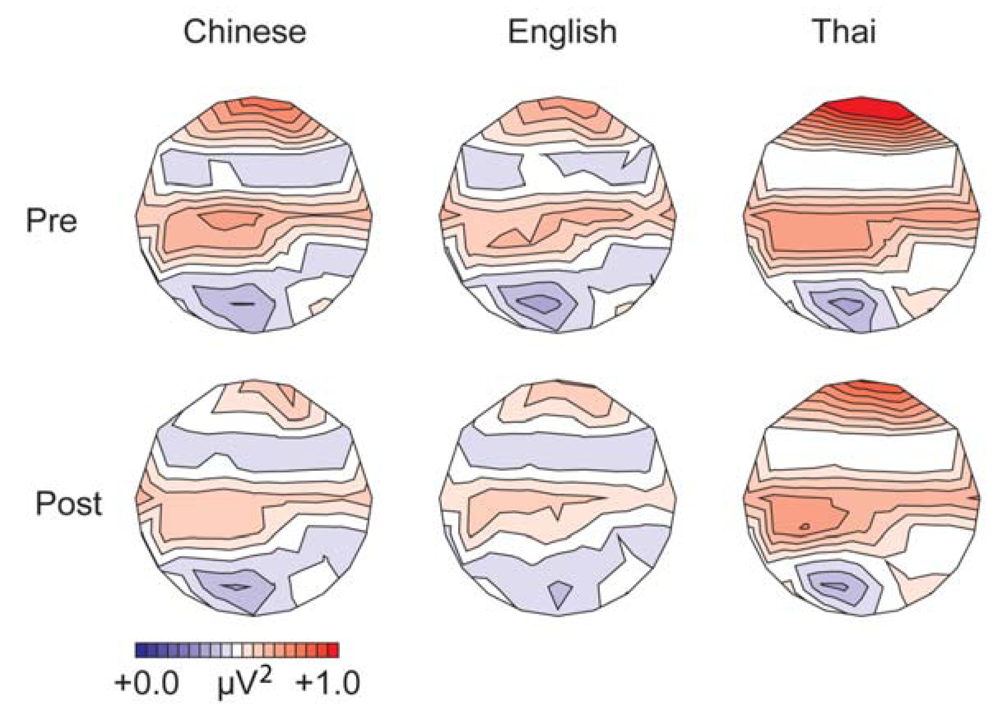
3.1. Differences between the Groups before Training

3.1.1. Gamma Band, Spectral Power (200–400 ms from Stimulus Onset)
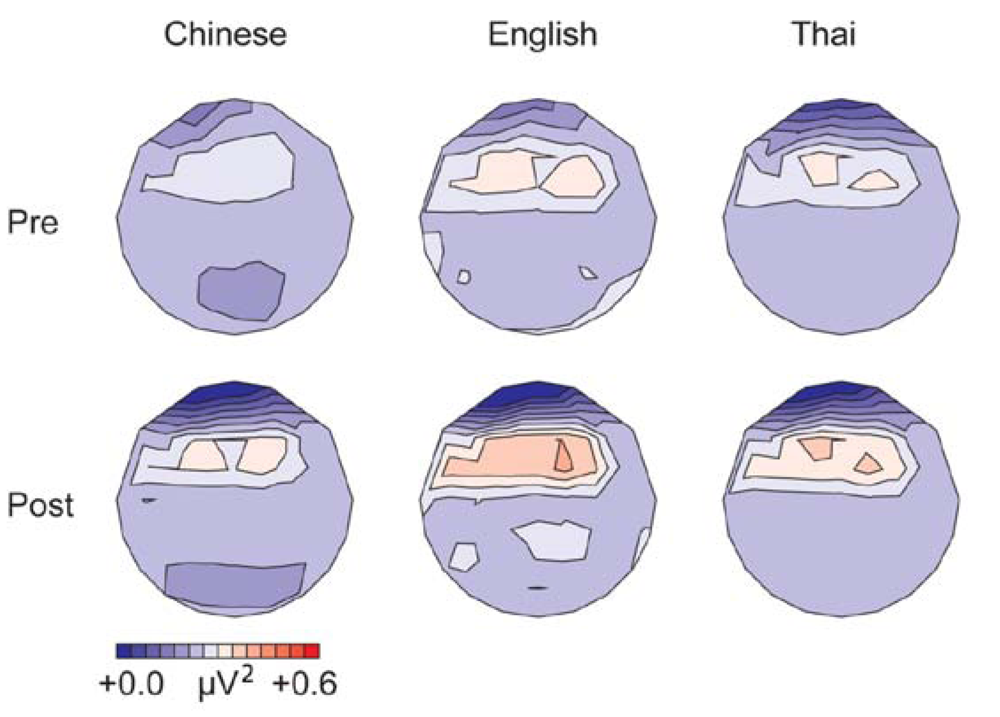
3.1.2. Alpha Band, Spectral Power (100–500 ms from Stimulus Onset)
| Test time | Language group | Low-falling tones | High-rising tones | ||
|---|---|---|---|---|---|
| Standard | Deviant | Standard | Deviant | ||
| Pre training | Chinese | 0.571 (0.021) | 0.526 (0.023) | 0.631 (0.018) | 0.520 (0.019) |
| English | 0.570 (0.021) | 0.477 (0.023) | 0.580 (0.018) | 0.566 (0.019) | |
| Thai | 0.640 (0.020) | 0.574 (0.021) | 0.618 (0.017) | 0.588 (0.018) | |
| Post training | Chinese | 0.567 (0.023) | 0.465 (0.028) | 0.582 (0.027) | 0.507 (0.020) |
| English | 0.527 (0.023) | 0.478 (0.028) | 0.579 (0.027) | 0.504 (0.020) | |
3.1.3. Gamma Band, Phase Synchrony with FCz (200–400 ms from Stimulus Onset)
3.1.4. Alpha Band, Phase Synchrony with Cz (100–500 ms from Stimulus Onset)
3.2. Differences in the Effects of Training between the Native English and the Native Chinese Language Groups

3.2.1. Gamma-Band Power (200–400 ms from Stimulus Onset)
3.2.2. Alpha-Band Power (100–500 ms from Stimulus Onset)
3.2.3. Correlation between Gamma and Alpha-Band Power
3.2.4. Gamma-Band, Phase Synchrony with FCz (200–400 ms from Stimulus Onset)
3.2.5. Alpha-Band, Phase Synchrony with Cz (100-500 ms from Stimulus Onset)
3.2.6. Effects of Training in the Thai Language Group
3.3. Discussion
3.3.1. Overview of Results
| Effect of language group before training (Thai, Chinese, English) | Effect of training (Chinese and English only) | Effect of training (Thai only) | |
|---|---|---|---|
| Gamma-band power | English > Thai > Chinese | English: post > pre; Chinese: post = pre | Post > pre |
| Alpha-band power | Thai > English = Chinese English: difference between deviants and standards larger for low-falling than for high-rising tones | Post training < pre training English: pre-training difference between deviants and standards larger for low-falling than high-rising tones | Post = pre Differences in distribution of differences between standards and deviants before and after training |
| Gamma-band phase synchrony | English = Thai = Chinese | Post > pre English = Chinese | Differences in distribution of differences between standards and deviants before and after training |
| Alpha-band phase synchrony | Thai show symmetric distribution of differences between deviants and standards; English and Chinese show right hemisphere maximum | Post > pre pre: English = Chinese post: English > Chinese | Post > pre Differences in distribution of differences between standards and deviants before and after training |
3.3.2. Gamma-Band Power
3.3.3. Alpha-Band Power
3.3.4. Gamma and Alpha-Band Phase Synchrony
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Best, C.T.; Tyler, M.D. Nonnative and Second-Language Speech Perception: Commonalities and Complementarities. In Language Experience in Second Language Speech Learning: In Honor of James Emil Flege; Bohn, O.-S., Munro, M., Eds.; John Benjamins: Amsterdam, The Netherlands, 2007; pp. 13–34. [Google Scholar]
- Kuhl, P.K. Early language acquisition: Cracking the speech code. Nat. Rev. Neurosci. 2004, 5, 831–843. [Google Scholar]
- Flege, J.E. Second Language Speech Learning: Theory, Findings, and Problems. In Speech Perception and Linguistic Experience: Issues in Cross-Language Research; Strange, W., Ed.; York Press: Timonium, MD, USA, 1995; pp. 233–277. [Google Scholar]
- Strange, W.; Shafer, V. Speech Perception in Second Language Learners. The Re-Education of Selective Perception. In Phonology and Second Language Acquisition; Hansen Edwards, J.G., Zampini, M.L., Eds.; John Benjamins: Amsterdam, The Netherlands, 2008; pp. 153–191. [Google Scholar]
- Burnham, D.; Mattock, K. The Perception of Tones and Phones. In Language Experience in Second Language Speech Learning: In Honor of James Emil Flege; Bohn, O.-S., Munro, M., Eds.; John Benjamins: Amsterdam, The Netherlands, 2007; pp. 259–280. [Google Scholar]
- Bluhme, H.; Burr, R. An audio-visual display of pitch for teaching Chinese tones. Stud. Linguist. 1971, 22, 51–57. [Google Scholar]
- Kuriloff, C. On the auditory discrimination of tones in Mandarin. Phonetica 1969, 20, 63–69. [Google Scholar]
- Wang, Y.; Spence, M.M.; Jongman, A.; Sereno, J.A. Training American listeners to perceive Mandarin tone. J. Acoust. Soc. Am. 1999, 106, 3649–3658. [Google Scholar]
- Wayland, R.; Guion, S. Training native English and native Chinese speakers to perceive Thai tones. Lang. Learn. 2004, 54, 681–712. [Google Scholar]
- Wayland, R.; Li, B. Effects of two training procedures in cross-language perception of tones. J. Phon. 2008, 36, 250–267. [Google Scholar]
- Krishnan, A.; Xu, Y.; Gandour, J.; Cariani, P. Encoding of pitch in the human brainstem is sensitive to language experience. Brain Res. Cogn. Brain Res. 2005, 25, 161–168. [Google Scholar]
- Kaan, E.; Barkley, C.; Bao, M.; Wayland, R. Thai lexical tone perception in native speakers of Thai, English and Mandarin Chinese: An event-related potentials training study. BMC Neurosci. 2008, 9, 53. [Google Scholar]
- So, C.K.; Best, C.T. Cross-language perception of non-native tonal contrasts: Effects of native phonological and phonetic influences. Lang. Speech 2010, 53, 273–293. [Google Scholar]
- Chandrasekaran, B.; Gandour, J.; Krishnan, A. Neuroplasticity in the processing of pitch dimensions: A multidimensional scaling analysis of the mismatch negativity. Restor. Neurol. Neurosci. 2007, 25, 95–210. [Google Scholar]
- Gandour, J. Tone perception in Far Eastern languages. J. Phon. 1983, 11, 49–175. [Google Scholar]
- Gandour, J.; Harshman, R. Cross-language difference in tone perception: A multidimensional scaling investigation. Lang. Speech 1978, 21, 1–33. [Google Scholar]
- Krishnan, A.; Gandour, J.; Bidelman, G.M.; Swaminathan, J. Experience-dependent neural representation of dynamic pitch in the brainstem. Neuroreport 2009, 20, 408–413. [Google Scholar]
- Guion, S.G.; Pederson, E. Investigating the Role of Attention in Phonetic Learning. In Language Experience in Second Language Speech Learning: In Honor of James Emil Flege; Bohn, O.-S., Munro, M., Eds.; John Benjamins: Amsterdam, The Netherlands, 2007; pp. 57–76. [Google Scholar]
- Wayland, R.; Herrera, E.; Kaan, E. Effects of musical experience and training on pitch contour perception. J. Phon. 2010, 38, 654–662. [Google Scholar]
- Wong, P.C.M.; Perrachione, T.K. Learning pitch patterns in lexical identification by native English-speaking adults. Appl. Psycholinguist. 2007, 28, 565–585. [Google Scholar]
- Wang, Y.; Sereno, J.A.; Jongman, A.; Hirsch, J. fMRI evidence for cortical modification during learning of Mandarin lexical tone. J. Cogn. Neurosci. 2003, 15, 1019–1027. [Google Scholar]
- Wong, P.C.M.; Perrachione, T.K.; Parrish, T.B. Neural characteristics of successful and less successful speech and word learning in adults. Hum. Brain Mapp. 2007, 28, 995–1006. [Google Scholar]
- Kaan, E.; Wayland, R.; Bao, M.; Barkley, C. Effects of native language and training on lexical tone perception: An ERP study. Brain Res. 2007, 1148, 113–122. [Google Scholar]
- Menning, H.; Imaizumi, S.; Zwitserlood, P.; Pantev, C. Plasticity of the human auditory cortex induced by discrimination learning of non-native, mora-timed contrasts of the Japanese language. Learn. Mem. 2002, 9, 253–267. [Google Scholar]
- Tremblay, K.; Kraus, N. Auditory training induced asymmetrical changes in cortical neural activity. J. Speech Lang. Hear. Res. 2002, 45, 564–572. [Google Scholar]
- Tremblay, K.; Kraus, N.; McGee, T.; Ponton, C.W.; Otis, B. Central auditory plasticity: Changes in the N1-P2 complex after speech-sound training. Ear Hear. 2001, 22, 79–90. [Google Scholar]
- Zhang, Y.; Kuhl, P.K.; Imada, T.; Iverson, P.; Pruitt, J.; Stevens, E.B.; Kawakatsu, M.; Tohkura, Y.I.; Nemoto, I. Neural signatures of phonetic learning in adulthood: A magnetoencephalography study. Neuroimage 2009, 46, 226–240. [Google Scholar]
- Singer, W.; Gray, C.M. Visual feature integration and the temporal correlation hypothesis. Annu. Rev. Neurosci. 1995, 18, 555–586. [Google Scholar]
- Fries, P.; Reynolds, J.H.; Rorie, A.E.; Desimone, R. Modulation of oscillatory neuronal synchronization by selective visual attention. Science 2001, 291, 1560–1563. [Google Scholar]
- Snyder, J.S.; Large, E.W. Gamma-band activity reflects the metric structure of rhythmic tone sequences. Brain Res. Cogn. Brain Res. 2005, 24, 117–126. [Google Scholar]
- Sokolov, A.; Pavlova, M.; Lutzenberger, W.; Birbaumer, N. Reciprocal modulation of neuromagnetic induced gamma activity by attention in the human visual and auditory cortex. Neuroimage 2004, 22, 521–529. [Google Scholar]
- Tallon-Baudry, C.; Bertrand, O. Oscillatory gamma activity in humans and its role in object representation. Trends Cogn. Sci. 1999, 3, 151–162. [Google Scholar]
- Gruber, O.; Indefrey, P.; Steinmetz, H.; Kleinschmidt, A. Dissociating neural correlates of cognitive components in mental calculation. Cereb. Cortex 2001, 11, 350–359. [Google Scholar]
- Sederberg, P.B.; Kahana, M.J.; Howard, M.W.; Donner, E.J.; Madsen, J.R. Theta and gamma oscillations during encoding predict subsequent recall. J. Neurosci. 2003, 23, 10809–10814. [Google Scholar]
- Lutzenberger, W.; Ripper, B.; Busse, L.; Birbaumer, N.; Kaiser, J. Dynamics of gamma-band activity during an audiospatial working memory task in humans. J. Neurosci. 2002, 22, 5630–5638. [Google Scholar]
- Herrmann, C.S.; Lenz, D.; Junge, S.; Busch, N.A.; Maess, B. Memory-matches evoke human gamma-responses. BMC Neurosci. 2004, 5, 13. [Google Scholar]
- Lutzenberger, W.; Pulvermuller, F.; Birbaumer, N. Words and pseudowords elicit distinct patterns of 30-Hz EEG responses in humans. Neurosci. Lett. 1994, 176, 115–118. [Google Scholar]
- Pantev, C. Evoked and induced gamma-band activity of the human cortex. Brain Topogr. 1995, 7, 321–330. [Google Scholar]
- Lenz, D.; Schadow, J.; Thaerig, S.; Busch, N.A.; Heann, C.S. What’s that sound? Matches with auditory long-term memory induce gamma activity in human EEG. Int. J. Psychophysiol. 2007, 64, 31–38. [Google Scholar]
- Miltner, W.H.R.; Braun, C.; Arnold, M.; Witte, H.; Taub, E. Coherence of gamma-band EEG activity as a basis for associative learning. Nature 1999, 397, 434–436. [Google Scholar]
- Heim, S.; Keil, A. Effects of classical conditioning on identification and cortical processing of speech syllables. Exp. Brain Res. 2006, 175, 411–424. [Google Scholar]
- Hamame, C.M.; Cosmelli, D.; Henriquez, R.; Aboitiz, F. Neural mechanisms of human perceptual learning: electrophysiological evidence for a two-stage process. PLoS One 2011, 6, e19221. [Google Scholar]
- Klimesch, W.; Sauseng, P.; Hanslmayer, S. EEG alpha oscillations: The inhibition-timing hypothesis. Brain Res. Rev. 2007, 53, 63–88. [Google Scholar]
- Palva, S.; Palva, J.M. New vistas for α-frequency band oscillations. Trends Neurosci. 2007, 30, 150–158. [Google Scholar]
- Pfurtscheller, G.; Lopes da Silva, F.H. Functional Meaning of Event-Related Desynchronization (ERD) and Synchronization (ERS). In Event-Related Desynchronizaton. Handbook of Electroencephalography and Clinical Neurophysiology; Pfurtscheller, G., Ed.; Elsevier: Amsterdam, The Netherlands, 1996; pp. 51–65. [Google Scholar]
- Neubauer, A.C.; Grabner, R.H.; Freudenthaler, H.; Beckmann, J.; Guthke, J. Intelligence and individual differences in becoming neurally efficient. Acta Psychol. (Amst.) 2004, 116, 55–74. [Google Scholar]
- Gruber, T.; Muller, M.M. Effects of picture repetition on induced gamma band responses, evoked potentials, and phase synchrony in the human EEG. Brain Res. Cogn. Brain Res. 2002, 13, 377–392. [Google Scholar]
- Reiterer, S.; Pereda, E.; Bhattacharya, J. Measuring second language proficiency with EEG synchronization: How functional cortical networks and hemispheric involvement differ as a function of proficiency level in second language speakers. Second Lang. 2009, 25, 77–106. [Google Scholar]
- Reiterer, S.; Hemmelmann, C.; Rappelsberger, P.; Berger, M.L. Decreased EEG coherence between prefrontal electrodes: A correlate of high language proficiency? Exp. Brain Res. 2005, 163, 109–113. [Google Scholar]
- Chandrasekaran, B.; Krishnan, A.; Gandour, J. Relative influence of musical and linguistic experience on early cortical processing of pith contours. Brain Lang. 2009, 108, 1–9. [Google Scholar]
- Gottfried, T.L. Music and Language Learning: Effect of Musical Training on Learning L2 Speech Contrasts. In Language Experience in Second Language Speech Learning: In Honor of James Emil Flege; Bohn, O.-S., Munro, M., Eds.; John Benjamins: Amsterdam, The Netherlands, 2007; pp. 221–237. [Google Scholar]
- Wong, P.C.M.; Skoe, E.; Russo, N.; Dees, T.; Kraus, N. Musical experience shapes human brainstem encoding of linguistic pitch pattern. Nat. Neurosci. 2007, 10, 420–422. [Google Scholar]
- Bertrand, O.; Bohorquez, J.; Pernier, J. Time-frequency digital filtering based on an invertible wavelet transform: An application to evoked potentials. IEEE Trans. Biomed. Eng. 1994, 41, 77–88. [Google Scholar]
- Lachaux, J.P.; Rodriguez, E.; Martinerie, J.; Varela, F.J. Measuring phase synchrony in brain signals. Hum. Brain Mapp. 1999, 8, 194–208. [Google Scholar]
- Rodriguez, E.; George, N.; Lachaux, J.P.; Martinerie, J.; Renault, B.; Varela, F.J. Perception’s shadow: Long-distance synchronization of human brain activity. Nature 1999, 397, 430–433. [Google Scholar]
- Heim, S.; Thomas Friedman, J.; Keil, A.; Benasich, A.A. Reduced sensory oscillatory activity during rapid auditory processing as a correlate of language-learning impairment. J. Neurolinguist. 2011, 24, 538–555. [Google Scholar]
- Weisz, N.; Keil, A.; Wienbruch, C.; Hoffmeister, S.; Elbert, T. One set of sounds, two tonotopic maps: Exploring auditory cortex with amplitude-modulated tones. Clin. Neurophysiol. 2004, 115, 1249–1258. [Google Scholar]
- Sanders, L.D.; Neville, H.J. An ERP study of continuous speech processing. I. Segmentation, semantics, and syntax in native speakers. Brain Res. Cogn. Brain Res. 2003, 15, 228–240. [Google Scholar]
- Gross, J.; Kujala, J.; Hamalainen, M.; Timmermann, L.; Schnitzler, A.; Salmelin, R. Dynamic imaging of coherent sources: Studying neural interactions in the human brain. Proc. Natl. Acad. Sci. USA 2001, 98, 694–699. [Google Scholar]
- Keil, A.; Costa, V.; Smith, J.C.; Sabatinelli, D.; McGinnis, E.M.; Bradley, M.M.; Lang, P.J. Tagging cortical networks in emotion: A topographical analysis. Hum. Brain Mapp. 2012, 33, 2920–2931. [Google Scholar]
- Greenblatt, R.E.; Pflieger, M.E.; Ossadtchi, A.E. Connectivity measures applied to human brain electrophysiological data. . J. Neurosci. Methods 2012, 207, 1–16. [Google Scholar]
- Greenhouse, S.W.; Geisser, S. On methods in the analysis of profile data. Psychometrika 1959, 24, 95–112. [Google Scholar]
- Sutoh, T.; Yabe, H.; Sato, Y.; Hiruma, T.; Kaneko, S. Event-related desynchronization during an auditory oddball task. Clin. Neurophysiol. 2000, 111, 858–862. [Google Scholar]
- Höller, Y.; Bergmann, J.; Kronbichler, M.; Crone, J.S.; Schmid, E.V.; Golaszewski, S.; Ladurner, G. Preserved oscillatory response but lack of mismatch negativity in patients with disorders of consciousness. Clin. Neurophysiol. 2011, 122, 1744–1754. [Google Scholar]
- Kaiser, J.; Lutzenberger, W. Human gamma-band activity: A window to cognitive processing. Neuroreport 2005, 16, 207–211. [Google Scholar]
- Pfurtscheller, G.; Stancák, A.J.; Neuper, C. Event-related synchronization (ERS) in the alpha band—An electrophysiological correlate of cortical idling: A review. Int. J. Psychophysiol. 1996, 24, 39–46. [Google Scholar]
- Foxe, J.J.; Simpson, G.V.; Ahlfors, S.P. Parieto-occipital ~10 Hz activity reflects anticipatory state of visual attention mechanisms. Neuroreport 1998, 9, 3929–3033. [Google Scholar]
- Fuentemilla, L.; Marco-Pallarésa, J.; Münte, T.F.; Grau, C. Theta EEG oscillatory activity and auditory change detection. Brain Res. 2008, 1220, 93–101. [Google Scholar]
- Hsiao, F.-J.; Wu, Z.-A.; Ho, L.-T.; Lin, Y.-Y. Theta oscillation during auditory change detection: An MEG study. Biol. Psychol. 2009, 81, 58–66. [Google Scholar]
- Ko, D.; Kwon, S.; Lee, G.-T.; Im, C.; Kim, K.; Jung, K.-Y. Theta oscillation related to the auditory discrimination process in Mismatch Negativity: Oddball versus control paradigm. J. Clin. Neurol. 2012, 8, 35–42. [Google Scholar]
- Hauk, O.; Keil, A.; Elbert, T.; Muller, M.M. Comparison of data transformation procedures to enhance topographical accuracy in time-series analysis of the human EEG. J. Neurosci. Methods 2002, 113, 111–122. [Google Scholar]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kaan, E.; Wayland, R.; Keil, A. Changes in Oscillatory Brain Networks after Lexical Tone Training. Brain Sci. 2013, 3, 757-780. https://doi.org/10.3390/brainsci3020757
Kaan E, Wayland R, Keil A. Changes in Oscillatory Brain Networks after Lexical Tone Training. Brain Sciences. 2013; 3(2):757-780. https://doi.org/10.3390/brainsci3020757
Chicago/Turabian StyleKaan, Edith, Ratree Wayland, and Andreas Keil. 2013. "Changes in Oscillatory Brain Networks after Lexical Tone Training" Brain Sciences 3, no. 2: 757-780. https://doi.org/10.3390/brainsci3020757





