Integrons in the Intestinal Microbiota as Reservoirs for Transmission of Antibiotic Resistance Genes
Abstract
:1. Global Spread of Antibiotic Resistance Genes
2. Gene Transfer Mechanisms
3. Integrons
3.1. Classification
3.2. Structure
3.3. Evolution
3.4. Mobility
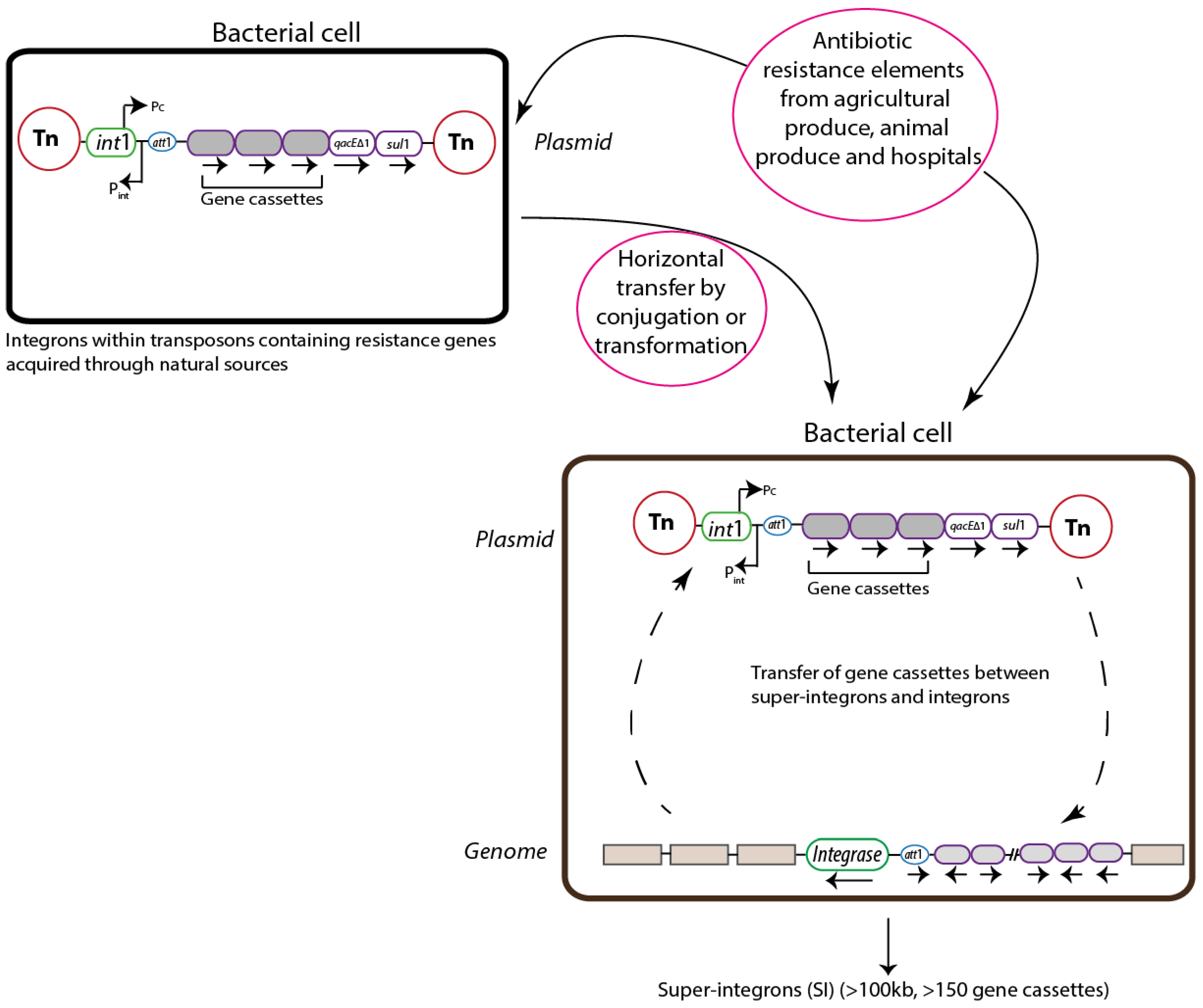
4. Integrons in the Intestinal Microbiota as Reservoirs for AR Genes
| Integron Class | Sampling Source | Integron Source | Resistance to antibiotic | Reference |
|---|---|---|---|---|
| I & II | Human feces | Commensal E.coli | Trimethoprim and aminoglycosides | [64] |
| I | Human feces | Citrobacter freundii | Aminoglycoside | [65] |
| II | Human feces | Commensal E. coli | Trimethoprim, streptomycin & spectinomycin | [1] |
| I | Human feces | Enterobacteriaceae and Pseudomonadaceae | Aminoglycoside, streptomycin and spectinomycin | [63] |
| I | Swine feces | Commensal E coli | Sulfamethoxazole, tetracycline and ampicillin | [66] |
| I | Captive wallabies feces | Metagenome | Spectinomycin, streptomycin and trimethoprim | [67] |
| I | Human feces | Commensal E.coli | Sulfamethoxazole, cefotaxime, gentamycin and ciprofloxacin. | [68] |
| ND* | Poultry and swine feces | Commensal E. coli | Tetracycline, sulfamethoxazole, quinolones and streptomycin | [69] |
| I | Human feces | Commensal E.coli | Streptomycin and Tetracycline | [70] |
| II | Human feces | Commensal E. coli | Streptomycin | [70] |
5. Conclusion
Acknowledgements
Author Contributions
Conflicts of interest
References
- Bailey, J.K.; Pinyon, J.L.; Anantham, S.; Hall, R.M. Commensal Escherichia coli of healthy humans: a reservoir for antibiotic-resistance determinants. J. Med. Microbiol. 2010, 59, 1331–1339. [Google Scholar] [CrossRef]
- Szmolka, A.; Nagy, B. Multidrug resistant commensal Escherichia coli in animals and its impact for public health. FMICB 2013, 4, 258. [Google Scholar]
- Jernberg, C.; Lofmark, S.; Edlund, C.; Jansson, J.K. Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. ISME J. 2007, 1, 56–66. [Google Scholar] [CrossRef]
- Sullivan, Å.; Edlund, C.; Nord, C.E. Effect of antimicrobial agents on the ecological balance of human microflora. Lancet. Infect. Dis. 2001, 1, 101–114. [Google Scholar] [CrossRef]
- Administration, F.D. Combating antibiotic resistance. Available online: http://www.fda.gov/downloads/ForConsumers/ConsumerUpdates/UCM143470.pdf (accessed on 11 November 2011).
- McKenna, M. The Last resort. Available online: http://www.nature.com/news/antibiotic-resistance-the-last-resort-1.13426 (accessed on 1 October 2013).
- Yong, D.; Toleman, M.A.; Giske, C.G.; Cho, H.S.; Sundman, K.; Lee, K.; Walsh, T.R. Characterization of a New Metallo-β-Lactamase Gene, blaNDM-1, and a Novel Erythromycin Esterase Gene Carried on a Unique Genetic Structure in Klebsiella pneumoniae Sequence Type 14 from India. Antimicrob. Agents. Chemother. 2009, 53, 5046–5054. [Google Scholar] [CrossRef]
- Salyers, A.A.; Gupta, A.; Wang, Y. Human intestinal bacteria as reservoirs for antibiotic resistance genes. Trends. Microbiol. 2004, 12, 412–416. [Google Scholar] [CrossRef]
- DiMarzio, M.; Shariat, N.; Kariyawasam, S.; Barrangou, R.; Dudley, E.G. Antibiotic Resistance in Salmonella enterica Serovar Typhimurium Associates with CRISPR Sequence Type. Antimicrob. Agents. Chemother. 2013, 57, 4282–4289. [Google Scholar] [CrossRef]
- Phillips, I.; Casewell, M.; Cox, T.; De Groot, B.; Friis, C.; Jones, R.; Nightingale, C.; Preston, R.; Waddell, J. Does the use of antibiotics in food animals pose a risk to human health? A critical review of published data. J. Antimicrob. Chemother. 2004, 53, 28–52. [Google Scholar]
- Tian, B.; Fadhil, N.H.; Powell, J.E.; Kwong, W.K.; Moran, N.A. Long-Term Exposure to Antibiotics Has Caused Accumulation of Resistance Determinants in the Gut Microbiota of Honeybees. mBio 2012. [Google Scholar] [CrossRef]
- Tavernise, S. FDA restricts Antibiotic Use in lifestock. Available online: http://www.nytimes.com/2013/12/12/health/fda-to-phase-out-use-of-some-antibiotics-in-animals-raised-for-meat.html?pagewanted=all (accessed on 12 December 2013).
- Davies, J.; Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef]
- Khachatourians, G.G. Agricultural use of antibiotics and the evolution and transfer of antibiotic-resistant bacteria. Can. Med. Assoc. J. 1998, 159, 1129–1136. [Google Scholar]
- Smillie, C.S.; Smith, M.B.; Friedman, J.; Cordero, O.X.; David, L.A.; Alm, E.J. Ecology drives a global network of gene exchange connecting the human microbiome. Nature 2011, 480, 241–244. [Google Scholar] [CrossRef]
- Moore, A.M.; Patel, S.; Forsberg, K.J.; Wang, B.; Bentley, G.; Razia, Y.; Qin, X.; Tarr, P.I.; Dantas, G. Pediatric fecal microbiota harbor diverse and novel antibiotic resistance genes. PLoS One 2013, 8, e78822. [Google Scholar] [CrossRef]
- Ochman, H.; Lawrence, J.G.; Groisman, E.A. Lateral gene transfer and the nature of bacterial innovation. Nature 2000, 405, 299–304. [Google Scholar] [CrossRef]
- Su, J.; Shi, L.; Yang, L.; Xiao, Z.; Li, X.; Yamasaki, S. Analysis of integrons in clinical isolates of Escherichia coli in China during the last six years. FEMS Microbiol. Lett. 2006, 254, 75–80. [Google Scholar] [CrossRef]
- Dobrindt, U.; Hacker, J. Whole genome plasticity in pathogenic bacteria. Curr. Opin. Microbiol. 2001, 4, 550–557. [Google Scholar] [CrossRef]
- Mazel, D. Integrons: Agents of bacterial evolution. Nat. Rev. Microbiol. 2006, 4, 608–620. [Google Scholar] [CrossRef]
- Frost, L.S.; Leplae, R.; Summers, A.O.; Toussaint, A. Mobile genetic elements: the agents of open source evolution. Nat. Rev. Microbiol. 2005, 3, 722–732. [Google Scholar] [CrossRef]
- Mazodier, P.; Davies, J. Gene transfer between distantly related bacteria. Annu. Rev. Genet. 1991, 25, 147–171. [Google Scholar] [CrossRef]
- Baharoglu, Z.; Bikard, D.; Mazel, D. Conjugative DNA Transfer Induces the Bacterial SOS Response and Promotes Antibiotic Resistance Development through Integron Activation. PLoS Genet. 2010, 6, e1001165. [Google Scholar] [CrossRef]
- Brisson-Noel, A.; Arthur, M.; Courvalin, P. Evidence for natural gene transfer from gram-positive cocci to Escherichia coli. J. Bacteriol. 1988, 170, 1739–1745. [Google Scholar]
- Grohmann, E.; Muth, G.; Espinosa, M. Conjugative plasmid transfer in gram-positive bacteria. Microbiol. Mol. Biol. Rev. 2003, 67, 277–301. [Google Scholar] [CrossRef]
- Meng, H.; Zhang, Z.; Chen, M.; Su, Y.; Li, L.; Miyoshi, S.; Yan, H.; Shi, L. Characterization and horizontal transfer of class 1 integrons in Salmonella strains isolated from food products of animal origin. Int. J. Food Microbiol. 2011, 149, 274–277. [Google Scholar] [CrossRef]
- Kruse, H.; Sørum, H. Transfer of multiple drug resistance plasmids between bacteria of diverse origins in natural microenvironments. Appl. Environ. Microbiol. 1994, 60, 4015–4021. [Google Scholar]
- Lester, C.H.; Frimodt-Moller, N.; Hammerum, A.M. Conjugal transfer of aminoglycoside and macrolide resistance between Enterococcus faecium isolates in the intestine of streptomycin-treated mice. FEMS Microbiol. Lett. 2004, 235, 385–391. [Google Scholar] [CrossRef]
- Domingues, S.; da Silva, G.J.; Nielsen, K.M. Integrons: Vehicles and pathways for horizontal dissemination in bacteria. Mob. Genet. Elements. 2012, 2, 211–223. [Google Scholar] [CrossRef]
- Domingues, S.; Harms, K.; Fricke, W.F.; Johnsen, P.J.; da Silva, G.J.; Nielsen, K.M. Natural Transformation Facilitates Transfer of Transposons, Integrons and Gene Cassettes between Bacterial Species. PLoS Pathog. 2012, 8, e1002837. [Google Scholar] [CrossRef]
- Davison, J. Genetic exchange between bacteria in the environment. Plasmid 1999, 42, 73–91. [Google Scholar] [CrossRef]
- Kovalevskaya, N.P. Mobile Gene Cassettes and Integrons. Mol. Biol. 2002, 36, 196–201. [Google Scholar] [CrossRef]
- Fluit, A.C.; Schmitz, F.J. Resistance integrons and super-integrons. Clin. Microbiol. Infect. 2004, 10, 272–288. [Google Scholar] [CrossRef]
- Petersen, A.; Guardabassi, L.; Dalsgaard, A.; Olsen, J.E. Class I integrons containing a dhfrI trimethoprim resistance gene cassette in aquatic Acinetobacter spp. FEMS Microbiol. Lett. 2000, 182, 73–76. [Google Scholar] [CrossRef]
- Gallego, L.; Towner, K.J. Carriage of class 1 integrons and antibiotic resistance in clinical isolates of Acinetobacter baumannii from northern Spain. J. Med. Microbiol. 2001, 50, 71–77. [Google Scholar]
- Chang, C.Y.; Chang, L.L.; Chang, Y.H.; Lee, T.M.; Chang, S.F. Characterisation of drug resistance gene cassettes associated with class 1 integrons in clinical isolates of Escherichia coli from Taiwan, ROC. J. Med. Microbiol. 2000, 49, 1097–1102. [Google Scholar]
- Mazel, D.; Dychinco, B.; Webb, V.A.; Davies, J. Antibiotic resistance in the ECOR collection: integrons and identification of a novel aad gene. Antimicrob. Agents Chemother. 2000, 44, 1568–1574. [Google Scholar] [CrossRef]
- Casin, I.; Breuil, J.; Brisabois, A.; Moury, F.; Grimont, F.; Collatz, E. Multidrug-resistant human and animal Salmonella typhimurium isolates in France belong predominantly to a DT104 clone with the chromosome- and integron-encoded beta-lactamase PSE-1. J. Infect. Dis. 1999, 179, 1173–1182. [Google Scholar] [CrossRef]
- Orman, B.E.; Pineiro, S.A.; Arduino, S.; Galas, M.; Melano, R.; Caffer, M.I.; Sordelli, D.O.; Centron, D. Evolution of multiresistance in nontyphoid salmonella serovars from 1984 to 1998 in Argentina. Antimicrob. Agents Chemother. 2002, 46, 3963–3970. [Google Scholar] [CrossRef]
- Gonzalez, G.; Sossa, K.; Bello, H.; Dominguez, M.; Mella, S.; Zemelman, R. Presence of integrons in isolates of different biotypes of Acinetobacter baumannii from Chilean hospitals. FEMS Microbiol. Lett. 1998, 161, 125–128. [Google Scholar] [CrossRef]
- Hochhut, B.; Lotfi, Y.; Mazel, D.; Faruque, S.M.; Woodgate, R.; Waldor, M.K. Molecular analysis of antibiotic resistance gene clusters in vibrio cholerae O139 and O1 SXT constins. Antimicrob. Agents Chemother. 2001, 45, 2991–3000. [Google Scholar] [CrossRef]
- Mazel, D.; Dychinco, B.; Webb, V.A.; Davies, J. A Distinctive Class of Integron in the Vibrio cholerae Genome. Science 1998, 280, 605–608. [Google Scholar] [CrossRef]
- Rowe-Magnus, D.A.; Guerout, A.M.; Ploncard, P.; Dychinco, B.; Davies, J.; Mazel, D. The evolutionary history of chromosomal super-integrons provides an ancestry for multiresistant integrons. Proc. Natl. Acad. Sci. USA 2001, 98, 652–657. [Google Scholar] [CrossRef]
- Clark, C.A.; Purins, L.; Kaewrakon, P.; Focareta, T.; Manning, P.A. The Vibrio cholerae O1 chromosomal integron. Microbiology 2000, 146, 2605–2612. [Google Scholar]
- Guo, X.; Xia, R.; Han, N.; Xu, H. Genetic diversity analyses of class 1 integrons and their associated antimicrobial resistance genes in Enterobacteriaceae strains recovered from aquatic habitats in China. Lett. Appl. Microbiol. 2011, 52, 667–675. [Google Scholar] [CrossRef]
- Leverstein-van Hall, M.A.; Box, A.T.A.; Blok, H.E.M.; Paauw, A.; Fluit, A.C.; Verhoef, J. Evidence of Extensive Interspecies Transfer of Integron-Mediated Antimicrobial Resistance Genes among Multidrug-Resistant Enterobacteriaceae in a Clinical Setting. J. Infect. Dis. 2002, 186, 49–56. [Google Scholar] [CrossRef]
- Hocquet, D.; Llanes, C.; Thouverez, M.; Kulasekara, H.D.; Bertrand, X.; Plesiat, P.; Mazel, D.; Miller, S.I. Evidence for induction of integron-based antibiotic resistance by the SOS response in a clinical setting. PLoS Pathog. 2012, 8, e1002778. [Google Scholar] [CrossRef] [Green Version]
- Rensing, C.; Newby, D.T.; Pepper, I.L. The role of selective pressure and selfish DNA in horizontal gene transfer and soil microbial community adaptation. Soil. Biol. Biochem. 2002, 34, 285–296. [Google Scholar] [CrossRef]
- Gillings, M.R.; Xuejun, D.; Hardwick, S.A.; Holley, M.P.; Stokes, H.W. Gene cassettes encoding resistance to quaternary ammonium compounds: a role in the origin of clinical class 1 integrons? ISME J. 2009, 3, 209–215. [Google Scholar] [CrossRef]
- Toleman, M.A.; Walsh, T.R. Combinatorial events of insertion sequences and ICE in Gram-negative bacteria. FEMS Microbiol. Rev. 2011, 35, 912–935. [Google Scholar] [CrossRef]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Penders, J.; Thijs, C.; Vink, C.; Stelma, F.F.; Snijders, B.; Kummeling, I.; van den Brandt, P.A.; Stobberingh, E.E. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics 2006, 118, 511–521. [Google Scholar] [CrossRef]
- Jimenez, E.; Marin, M.L.; Martin, R.; Odriozola, J.M.; Olivares, M.; Xaus, J.; Fernandez, L.; Rodriguez, J.M. Is meconium from healthy newborns actually sterile? Res. Microbiol. 2008, 159, 187–193. [Google Scholar] [CrossRef]
- Jimenez, E.; Fernandez, L.; Marin, M.L.; Martin, R.; Odriozola, J.M.; Nueno-Palop, C.; Narbad, A.; Olivares, M.; Xaus, J.; Rodriguez, J.M. Isolation of commensal bacteria from umbilical cord blood of healthy neonates born by cesarean section. Curr. Microbiol. 2005, 51, 270–274. [Google Scholar] [CrossRef]
- Satokari, R.; Gronroos, T.; Laitinen, K.; Salminen, S.; Isolauri, E. Bifidobacterium and Lactobacillus DNA in the human placenta. Lett. Appl. Microbiol. 2009, 48, 8–12. [Google Scholar] [CrossRef]
- Palmer, C.; Bik, E.M.; DiGiulio, D.B.; Relman, D.A.; Brown, P.O. Development of the human infant intestinal microbiota. PLoS Biol. 2007, 5, e177. [Google Scholar] [CrossRef]
- Avershina, E.; Storro, O.; Oien, T.; Johnsen, R.; Pope, P.; Rudi, K. Major faecal microbiota shifts in composition and diversity with age in a geographically restricted cohort of mothers and their children. FEMS Microbiol. Ecol. 2013, 87, 280–290. [Google Scholar]
- Chung, H.; Pamp, S.J.; Hill, J.A.; Surana, N.K.; Edelman, S.M.; Troy, E.B.; Reading, N.C.; Villablanca, E.J.; Wang, S.; Mora, J.R.; et al. Gut immune maturation depends on colonization with a host-specific microbiota. Cell 2012, 149, 1578–1593. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Stephen, A.M.; Cummings, J.H. The microbial contribution to human faecal mass. J. Med. Microbiol. 1980, 13, 45–56. [Google Scholar] [CrossRef]
- Freilich, S.; Kreimer, A.; Meilijson, I.; Gophna, U.; Sharan, R.; Ruppin, E. The large-scale organization of the bacterial network of ecological co-occurrence interactions. Nucleic. Acids. Res. 2010, 38, 3857–3868. [Google Scholar] [CrossRef]
- Lévesque, C.; Piché, L.; Larose, C.; Roy, P.H. PCR mapping of integrons reveals several novel combinations of resistance genes. Antimicrob. Agents Chemother. 1995, 39, 185–191. [Google Scholar] [CrossRef]
- Lee, J.C.; Kang, H.Y.; Oh, J.Y.; Jeong, J.H.; Kim, J.; Seol, S.Y.; Cho, D.T.; Lee, Y.C. Antimicrobial Resistance and Integrons Found in Commensal Escherichia coli Isolates from Healthy Humans. J. Bacteriol. Virol. 2006, 36, 133–139. [Google Scholar] [CrossRef]
- Norskov-Lauritsen, N.; Sandvang, D.; Hedegaard, J.; Fussing, V.; Mortensen, K.K.; Sperling-Petersen, H.U.; Schonheyder, H.C. Clonal origin of aminoglycoside-resistant Citrobacter freundii isolates in a Danish county. J. Med. Microbiol. 2001, 50, 636–641. [Google Scholar]
- Phongpaichit, S.; Liamthong, S.; Mathew, A.G.; Chethanond, U. Prevalence of class 1 integrons in commensal Escherichia coli from pigs and pig farmers in Thailand. J. Food Prot. 2007, 70, 292–299. [Google Scholar]
- Power, M.L.; Emery, S.; Gillings, M.R. Into the Wild: Dissemination of Antibiotic Resistance Determinants via a Species Recovery Program. PLoS ONE 2013, 8, e63017. [Google Scholar] [CrossRef]
- Sepp, E.; Stsepetova, J.; Loivukene, K.; Truusalu, K.; Koljalg, S.; Naaber, P.; Mikelsaar, M. The occurrence of antimicrobial resistance and class 1 integrons among commensal Escherichia coli isolates from infants and elderly persons. Ann. Clin. Microbiol. Antimicrob. 2009, 8, 34. [Google Scholar] [CrossRef]
- Koo, H.J.; Woo, G.J. Characterization of antimicrobial resistance of Escherichia coli recovered from foods of animal and fish origin in Korea. J. Food Prot. 2012, 75, 966–972. [Google Scholar] [CrossRef]
- Skurnik, D.; Le Menac'h, A.; Zurakowski, D.; Mazel, D.; Courvalin, P.; Denamur, E.; Andremont, A.; Ruimy, R. Integron-Associated Antibiotic Resistance and Phylogenetic Grouping of Escherichia coli Isolates from Healthy Subjects Free of Recent Antibiotic Exposure. Antimicrob. Agents Chemother. 2005, 49, 3062–3065. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ravi, A.; Avershina, E.; Ludvigsen, J.; L'Abée-Lund, T.M.; Rudi, K. Integrons in the Intestinal Microbiota as Reservoirs for Transmission of Antibiotic Resistance Genes. Pathogens 2014, 3, 238-248. https://doi.org/10.3390/pathogens3020238
Ravi A, Avershina E, Ludvigsen J, L'Abée-Lund TM, Rudi K. Integrons in the Intestinal Microbiota as Reservoirs for Transmission of Antibiotic Resistance Genes. Pathogens. 2014; 3(2):238-248. https://doi.org/10.3390/pathogens3020238
Chicago/Turabian StyleRavi, Anuradha, Ekaterina Avershina, Jane Ludvigsen, Trine M. L'Abée-Lund, and Knut Rudi. 2014. "Integrons in the Intestinal Microbiota as Reservoirs for Transmission of Antibiotic Resistance Genes" Pathogens 3, no. 2: 238-248. https://doi.org/10.3390/pathogens3020238






