Pheromonal Communication in the European House Dust Mite, Dermatophagoides pteronyssinus
Abstract
:1. Introduction
2. Experimental
2.1. Cultures
2.2. Chemical Analyses
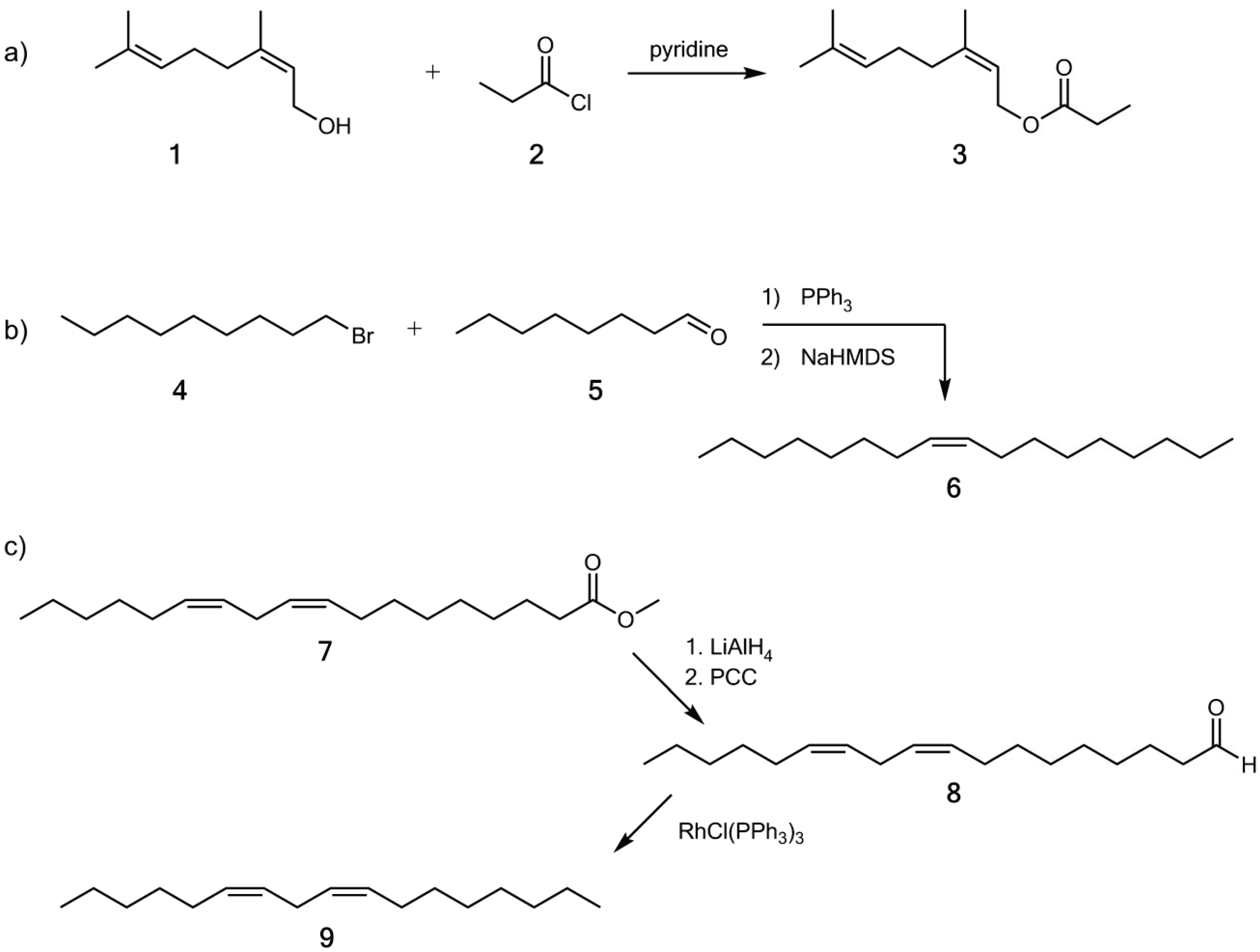
2.3. Synthesis
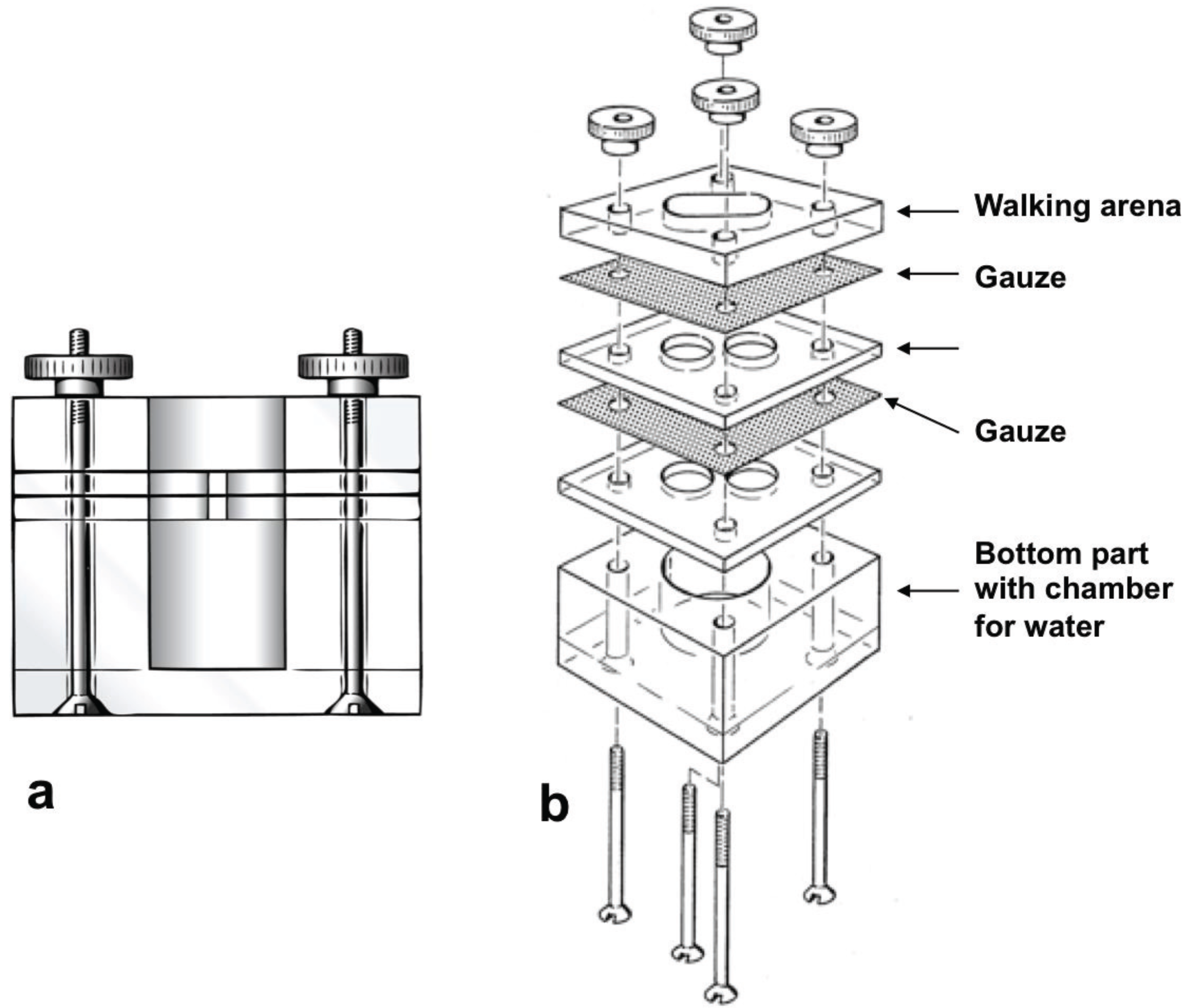
2.4. Reaction to Synthetic Compounds in the Olfactometer
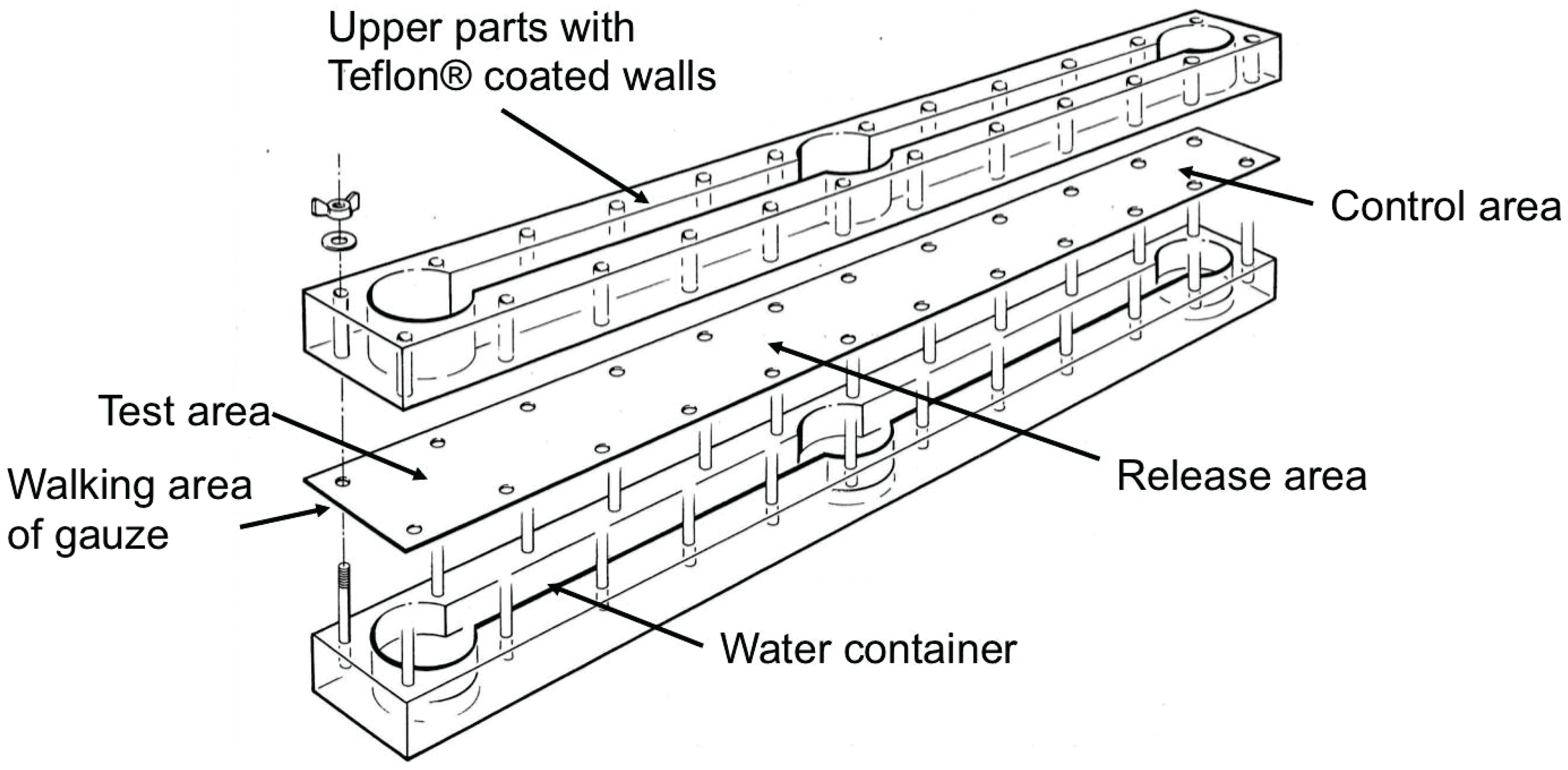
2.5. Use of Neryl Propionate as Bait
3. Results
3.1. Chemical Analyses
3.1.1. Headspace of Mite Cultures
3.1.2. Sex Specific Release of Compounds
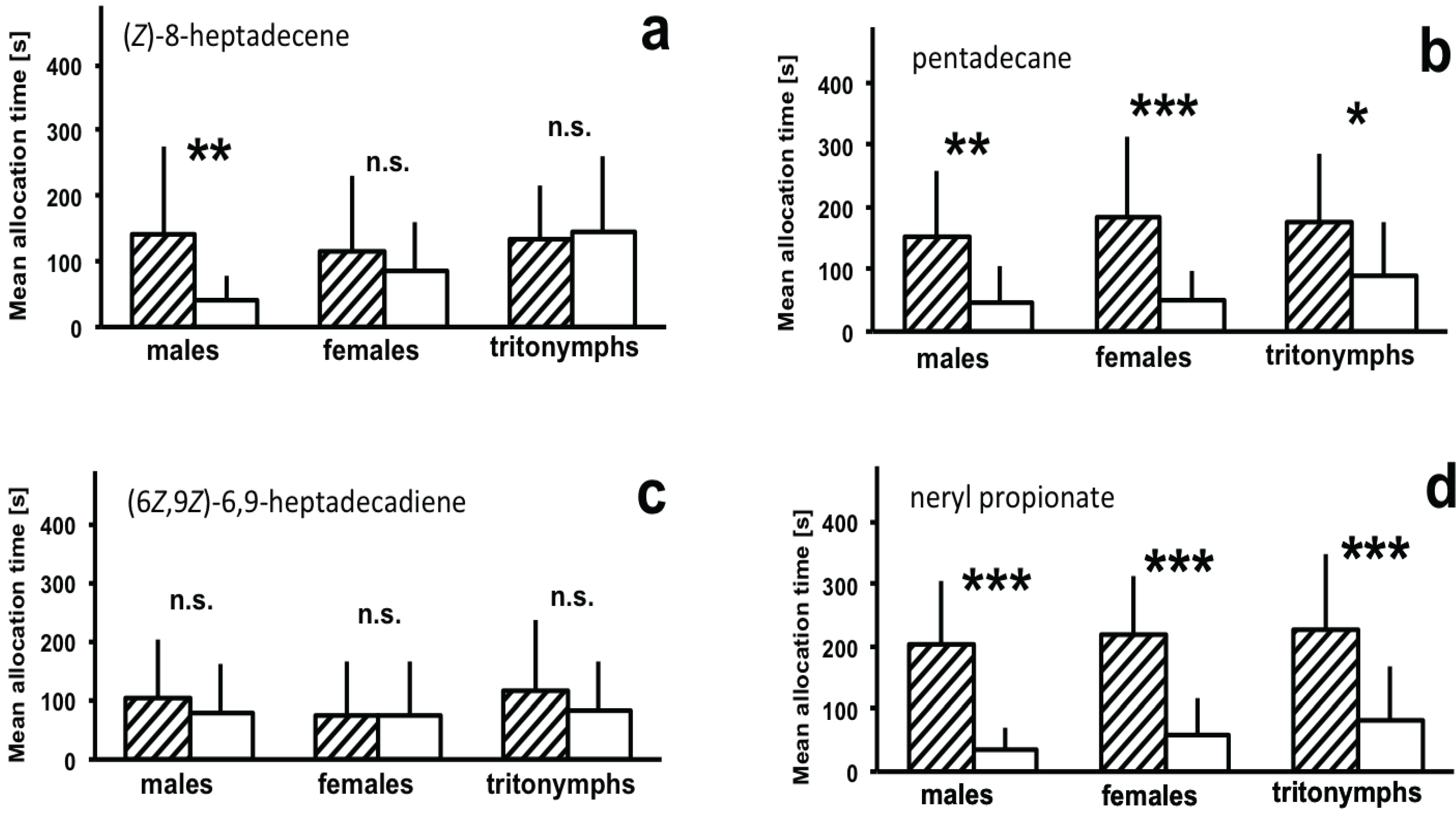
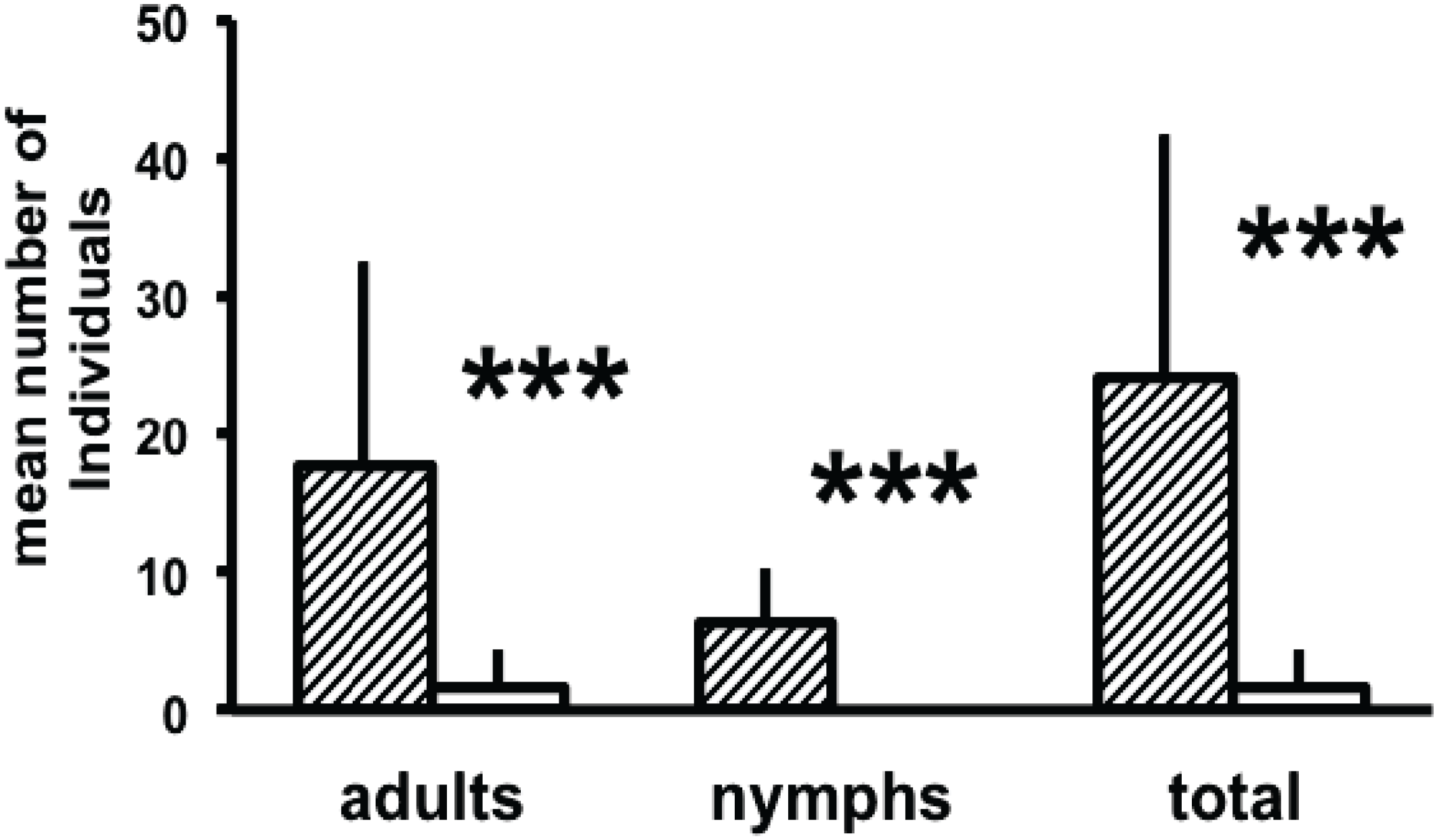
3.2. Attraction to Synthetic Compounds
3.3. Potential Use of Neryl Propionate as Bait for House Dust Mite Control
4. Discussion
| Compound | Demonstration of its presence | Suggested function |
|---|---|---|
| Neral | [10] | unknown |
| Geranial | [10] | unknown |
| Rhizoglyphinyl formate (2-Formyl-3-hydroxybenzyl formate) | [11] | unknown |
| Nerol | this study | unknown |
| Neryl formate | [8], this study | aggregation pheromone [8] |
| Neryl propionate | this study | aggregation pheromone, this study |
| Pentadecane | [10], this study | aggregation pheromone, this study |
| Geranyl formate | [10] | unknown |
| 3-Hydroxybenzene-1,2 -dicarbaldehyde | [5] | unknown |
| Heptadecane | [5] | unknown |
| (Z)-6-Pentadecene | [5] | unknown |
| (6Z,9Z)-6,9-Heptadecadiene | [10], this study | unknown |
| (Z)-8-Heptadecene | [10], this study | female sex pheromone, this study |
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tovey, E.R.; Chapman, M.D.; Platts-Mills, T.A.E. Mite faeces are a major source of house dust allergens. Nature 1981, 289, 592–593. [Google Scholar] [CrossRef]
- World Health Organization. Dust mite allergens and asthma: A worldwide problem. International Workshop Report. Bull. World Health Organ. 1988, 66, 769–780. [Google Scholar]
- Diebschlag, W.; Diebschlag, B. Hausstauballergien-Gesundheitliche und Hygienische Aspekte, 2nd ed.; Herbert Utz Verlag: München, Germany, 2000. [Google Scholar]
- Vallance, G.; McSharry, C.; Wood, S.; Thomson, N.C. House dust mite control measures in the treatment of asthma. Ther. Clin. Risk Manag. 2006, 2, 347–354. [Google Scholar]
- Kuwahara, Y. Chemical ecology of astigmatid mites. In Advances in Insect Chemical Ecology; Cardé, R.T., Millar, J.G., Eds.; Cambridge University Press: Cambridge, UK, 2004; pp. 76–109. [Google Scholar]
- Wyatt, T.D. Pheromones and Animal Behaviour: Chemical Signals and Signatures, 2nd ed.; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Tatami, K.; Mori, N.; Nishida, R.; Kuwahara, Y. 2-Hydroxy-6-methylbenz-aldehyde: The female sex pheromone of the house dust mite Dermatophagoides farinae (Astigmata: Pyroglyphidae). Med. Entomol. Zool. 2001, 52, 279–286. [Google Scholar]
- Skelton, A.C.; Cameron, M.M.; Pickett, J.A.; Birkett, M.A. Identification of neryl formate as the airborne aggregation pheromone for the American house dust mite and the European house dust mite (Acari: Epidermoptidae). J. Med. Entomol. 2010, 47, 798–804. [Google Scholar] [CrossRef]
- Kuwahara, Y.; Matsumoto, K.; Wada, Y. Pheromone study on acarid mites IV. Citral: Composition and function as an alarm pheromone and its secretory gland in four species of acarid mites. Jpn. J. Sanit. Zool. 1980, 31, 73–80. [Google Scholar]
- Kuwahara, Y.; Leal, W.S.; Suzuki, T. Pheromone study on astigmatid mites: XXVI. Comparison of volatile components between Dermatophagoides farinae and D. pteronyssinus (Astigmata, Pyroglyphidae). Jpn. J. Sanit. Zool. 1990, 41, 23–28. [Google Scholar]
- Sato, M.; Kuwahara, Y.; Matsumama, S.; Suzuki, T. 2-Formyl-3-hydroxy-benzylformate (Rhizoglyphinyl Formate), a novel Salicylaldehyde analog from the house dust mite Dermatophagoides pteronyssinus (Astigmata, Pyroglyphidae). Biosci. Biotechnol. Biochem. 1993, 57, 1299–1301. [Google Scholar] [CrossRef]
- Raspotnig, G.; Krisper, K.; Schuster, R. Duftstoffproduktion bei Hornmilben (Acari, Oribatida). Entomol. Austriaca 2004, 10, 11–18. [Google Scholar]
- Shimano, S.; Sakata, T.; Mizutani, Y.; Kuahara, Y.; Aoki, J.-I. Geranial: The alarm pheromone in the nymphal stage of the oribatid mite, Nothrus palustris. J. Chem. Ecol. 2002, 28, 1831–1837. [Google Scholar] [CrossRef]
- Raspotnig, G.; Krisper, G.; Schuster, R. Ontogenetic changes in the chemistry and morphology of oil glands in Hermannia convexa (Acari: Oribatida). Exp. Appl. Acarol. 2005, 35, 47–58. [Google Scholar] [CrossRef]
- Takada, W.; Sakata, T.; Shimano, S.; Enami, Y.; Mori, N.; Nishida, R.; Kuwahara, Y. Scheloribatid mites as the source of pumiliotoxins in dendrobatid frogs. J. Chem. Ecol. 2005, 31, 2403–2415. [Google Scholar] [CrossRef]
- Hughes, A.M. The mites of stored food and houses. In Technical Bulletin, Ministry of Agriculture, Fisheries and Food; Her Majesty’s Stationary Office: London, UK, 1976; pp. 1–400. [Google Scholar]
- Schulz, S.; Fuhlendorff, J.; Steidle, J.L.M.; Collatz, J.; Franz, T.C. Identification and biosynthesis of an aggregation pheromone of the storage mite, Chortoglyphus arcuatus. Chembiochem 2004, 5, 1500–1507. [Google Scholar] [CrossRef]
- Pempo, D.; Cintrat, J.-C.; Parrain, J.-L.; Santelli, M. Synthesis of [3H2]-(11S,17R)-11,17-Dimethylhentriacontane: A useful tool for the study of the Internalisation of communication pheromones of ant Camponotus vagus. Tetrahedron 2000, 56, 5493–5497. [Google Scholar] [CrossRef]
- Shimizu, N.; Mori, N.; Kuwahara, Y. Identification of the new hydrocarbon (Z,Z)-1,6,9-heptadecatriene as the secretory component of Caloglyphus polyphyllae (Astigmata: Acaridae). Biosci. Biotechnol. Biochem. 1999, 63, 1478–1480. [Google Scholar] [CrossRef]
- Jain, S.C.; Dussourd, D.E.; Conner, W.E.; Eisner, T.; Guerrero, A.; Meinwald, J. Polyene pheromone components from an arctiid moth (Utetheisa ornatrix): Characterization and synthesis. J. Org. Chem. 1983, 48, 2266–2270. [Google Scholar] [CrossRef]
- Corey, E.J.; Suggs, W.J. Pyridinium chlorochromate. An efficient reagent for oxidation of primary and secondary alcohols to carbonyl compounds. Tetrahedron Lett. 1975, 16, 2647–2650. [Google Scholar] [CrossRef]
- Young, J.F.; Osborn, J.A.; Jardine, F.H.; Wilkinson, G. Hydride intermediates in homogeneous hydrogenation reactions of olefins and acetylenes using rhodium catalysts. Chem. Commun. (London) 1965, 7, 131–132. [Google Scholar]
- Tsuji, J.; Ohno, K. Organic syntheses by means of noble metal compounds XXI. Decarbonylation of aldehydes using rhodium complex. Tetrahedron Lett. 1965, 44, 3969–3971. [Google Scholar] [CrossRef]
- Hutzinger, M.W.; Oehlschlager, A.C. Stereospecific reduction and cross-coupling of γ-monosubstituted allylic chlorides using coordinatively unsaturated palladium catalysts. J. Org. Chem. 1991, 56, 2918–2920. [Google Scholar] [CrossRef]
- The Observer. Noldus: Wageningen, The Netherlands, 2003.
- Statistica. Statsoft Inc.: Tulsa, OK, USA, 1995.
- Heethoff, M.; Raspotnig, G. Investigating chemical communication in oribatid and astigmatid mites in bioassays—Pitfalls and suggestions. Soil Org. 2012, 84, 409–421. [Google Scholar]
- Wertheim, B.; van Baalen, E.J.; Dicke, M.; Vet, L.E.M. Pheromone-mediated aggregation in nonsocial arthropods: An evolutionary ecological perspective. Annu. Rev. Entomol. 2005, 50, 321–346. [Google Scholar] [CrossRef]
- Reka, S.A.; Suto, C.; Yamaguchi, M. Evidence of aggregation pheromone in the feces of house dust mite Dermatophagoides farinae. Jpn. J. Sanit. Zool. 1992, 43, 399–341. [Google Scholar]
- Van Bronswijk, J.E.M.H. House Dust Biology for Allergists, Acarologist and Mycologists; NIB Publishers: Zeist, The Netherlands, 1981. [Google Scholar]
- Arlian, L.G. Water balance and humidity requirements of house dust mites. Exp. Appl. Acarol. 1992, 16, 15–35. [Google Scholar] [CrossRef]
- Glass, E.V.; Yoder, J.A.; Needham, G.R. Clustering reduces water loss by adult American house dust mites Dermatophagoides farinae (Acari: Pyroglyphidae). Exp. Appl. Acarol. 1998, 22, 31–37. [Google Scholar] [CrossRef]
- Peschke, K. Male aggression, female mimicry and female choice in the rove beetle, Aleochara curtula (Coleoptera, Staphylinida). Ethology 1987, 75, 265–284. [Google Scholar] [CrossRef]
- Steiner, S.; Steidle, J.L.M.; Ruther, J. Female sex pheromone in immature insect males—a case of pre-emergence chemical mimicry? Behav. Ecol. Sociobiol. 2005, 58, 111–120. [Google Scholar] [CrossRef]
- Howse, P.; Stevens, J.M.; Jones, O. Insect Pheromones and Their Use in Pest Management; Chapman & Hall: London, UK, 1997. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Steidle, J.L.M.; Barcari, E.; Hradecky, M.; Trefz, S.; Tolasch, T.; Gantert, C.; Schulz, S. Pheromonal Communication in the European House Dust Mite, Dermatophagoides pteronyssinus. Insects 2014, 5, 639-650. https://doi.org/10.3390/insects5030639
Steidle JLM, Barcari E, Hradecky M, Trefz S, Tolasch T, Gantert C, Schulz S. Pheromonal Communication in the European House Dust Mite, Dermatophagoides pteronyssinus. Insects. 2014; 5(3):639-650. https://doi.org/10.3390/insects5030639
Chicago/Turabian StyleSteidle, Johannes L.M., Elena Barcari, Marc Hradecky, Simone Trefz, Till Tolasch, Cornelia Gantert, and Stefan Schulz. 2014. "Pheromonal Communication in the European House Dust Mite, Dermatophagoides pteronyssinus" Insects 5, no. 3: 639-650. https://doi.org/10.3390/insects5030639




