Survival and Infectivity of the Insect-Parasitic Nematode Heterorhabditis bacteriophora Poinar in Solutions Containing Four Different Turfgrass Soil Surfactants
Abstract
:1. Introduction
1.1. EPNs used for Biological Control in Turfgrass
1.2. EPN Survival in Turfgrass
1.3. Soil Surfactants
2. Results and Discussion
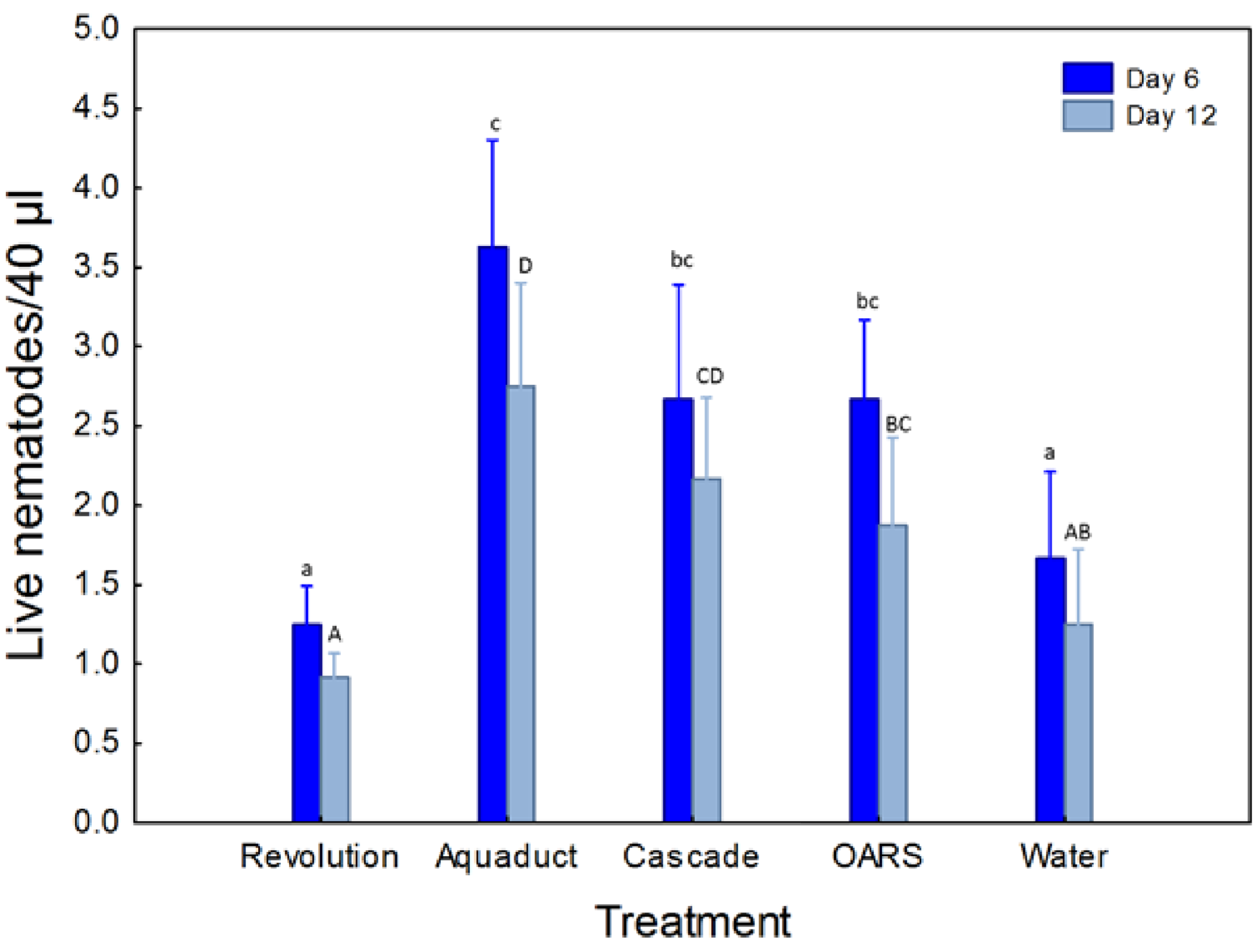
2.1. EPN Survival in Surfactant Solutions
2.2. EPN Infectivity
3. Experimental Section
3.1. Soil Surfactant/Nematode Solutions
3.2. EPN Survival in Surfactant Solutions
3.3. EPN Infectivity
3.4. Statistical Analysis
| Trade Name | Manufacturer | Dilution |
|---|---|---|
| Revolution | Aquatrols Corp. | 1L product/43L H20/km2 |
| Aqueduct | Aquatrols Corp. | 1L product/16L H20/km2 |
| Cascade Plus | Precision Laboratories | 1L product/32L H20/km2 |
| OARS | Aqua-Aid Inc. | 1L product/43L H20/km2 |
4. Conclusions
4.1. Summary and Implications
Acknowledgments
References
- Forst, S.; Dowds, B.; Boemare, N.; Stackebrandt, E. Xenorhabdus and photorhabdus spp.: Bugs that kill bugs. Annu. Rev. Microbiol. 1997, 51, 47–72. [Google Scholar]
- Grewal, P.S.; Ehlers, R.; Shapiro-Ilan, D.I. Nematodes as Biocontrol Agents; CABI: Toronto, ON, USA, 2006. [Google Scholar]
- Lewis, E.E.; Gaugler, R.; Harrison, R. Entomopathogenic nematode host finding: Response to host contact cues by cruise and ambush foragers. Parasitology 1992, 105, 309–315. [Google Scholar] [CrossRef]
- Gaugler, R. Matching Nematode and Insect to Achieve Optimal Field Performance; Polavarapu, S., Ed.; Optimal Use of Insecticidal Nematodes in Pest Management: New Brunswick, NJ, USA, 1999. [Google Scholar]
- Klein, M.G. Efficacy against soil-inhabiting insect pests. In Entomopathogenic Nematodes in Biological Control; Gaugler, R., Kaya, H.K., Eds.; CRC Press Inc: Boca Raton, FL, USA, 1990; pp. 195–214. [Google Scholar]
- Georgis, R.; Kaya, H.K.; Gaugler, R. Effect of steinernematid and heterorhabditid nematodes rhabditida steinernematidae and heterorhabitidae on nontarget arthropods. Environ. Entomol. 1991, 20, 814–822. [Google Scholar]
- Grewal, P.; Georgis, R. Entomopathogenic nematodes. Meth. Biotechnol. 1999, 5, 271–299. [Google Scholar]
- Norton, D.C. Ecology of Plant-parasitic Nematodes; Wiley: Hoboken, NJ, USA, 1978. [Google Scholar]
- Koppenhofer, A.M.; Kaya, H.K.; Taormino, S.P. Infectivity of entomopathogenic nematodes (rhabditida, steinernematidae) at different soil depths and moistures. J. Invertebr. Pathol. 1995, 65, 193–199. [Google Scholar] [CrossRef]
- Kung, S.P.; Gaugler, R.; Kaya, H.K. Effects of soil-temperature, moisture, and relative-humidity on entomopathogenic nematode persistence. J. Invertebr. Pathol. 1991, 57, 242–249. [Google Scholar] [CrossRef]
- Kaya, H.K.; Gaugler, R. Entomopathogenic nematodes. Annu. Rev. Entomol. 1993, 38, 181–206. [Google Scholar] [CrossRef]
- Smits, P.H. Post-application persistence of entomopathogenic nematodes. Biocontrol Sci. Technol. 1996, 6, 379–387. [Google Scholar] [CrossRef]
- Kostka, S.J. Amelioration of water repellency in highly managed soils and the enhancement of turfgrass performance through the systematic application of surfactants. J. Hydrol. 2000, 231, 359–368. [Google Scholar] [CrossRef]
- Moore, D.; Kostka, S.J.; Franklin, M.K.; Lennert, L.L.; Moore, R.A. The Evolution of Soil Wetting Agents for Managing Water Repellency in Soils. In Proceedings of ASA-CSSA-SSSA 2006 International Meetings, Indianapolis, IN, USA, 12–16 November 2006.
- Mulligan, C.N.; Yong, R.N.; Gibbs, B.F. Surfactant-enhanced remediation of contaminated soil: A review. Eng. Geol. 2001, 60, 371–380. [Google Scholar] [CrossRef]
- Leinauer, B.; Karcher, D.; Barrick, T.; Ikemura, Y.; Hubble, H.; Makk, J. Water repellency varies with depth and season in sandy rootzones treated with ten wetting agents. Available online: http://www.plantmanagementnetwork.org/pub/ats/research/2007/repel/ (accessed on 27 October 2012).
- Lacey, L.A.; Neven, L.G.; Headrick, H.L.; Fritts, R. Factors affecting entomopathogenic nematodes (steinernematidae) for control of overwintering codling moth (lepidoptera: Tortricidae) in fruit bins. J. Econ. Entomol. 2005, 98, 1863–1869. [Google Scholar] [CrossRef]
- Lacey, L.A.; Arthurs, S.P.; Unruh, T.R.; Headrick, H.; Fritts, R. Entomopathogenic nematodes for control of codling moth (lepidoptera : Tortricidae) in apple and pear orchards: Effect of nematode species and seasonal temperatures, adjuvants, application equipment, and post-application irrigation. Biol. Control 2006, 37, 214–223. [Google Scholar] [CrossRef]
- Shapiro-Ilan, D.I.; Cottrell, T.E.; Brown, I.; Gardner, W.A.; Hubbard, R.K.; Wood, B.W. Effect of soil moisture and a surfactant on entomopathogenic nematode suppression of the pecan weevil, curculio caryae. J. Nematol. 2006, 38, 474–482. [Google Scholar]
- Alumai, A.; Grewal, P.S. Tank-mix compatibility of the entomopathogenic nematodes, heterorhabditis bacteriophora and steinernema carpocapsae, with selected chemical pesticides used in turfgrass. Biocontrol Sci. Technol. 2004, 14, 725–730. [Google Scholar] [CrossRef]
- Garcia-Del-Pino, F.; Morton, A. Synergistic effect of the herbicides glyphosate and mcpa on survival of entomopathogenic nematodes. Biocontrol Sci. Technol. 2010, 20, 483–488. [Google Scholar] [CrossRef]
- Gaugler, R.; Kaya, H.K. Entomopathogenic Nematodes in Biological Control; CRC Press Inc: Boca Raton, FL, USA, 1990. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hoctor, T.L.; Gibb, T.J.; Bigelow, C.A.; Richmond, D.S. Survival and Infectivity of the Insect-Parasitic Nematode Heterorhabditis bacteriophora Poinar in Solutions Containing Four Different Turfgrass Soil Surfactants. Insects 2013, 4, 1-8. https://doi.org/10.3390/insects4010001
Hoctor TL, Gibb TJ, Bigelow CA, Richmond DS. Survival and Infectivity of the Insect-Parasitic Nematode Heterorhabditis bacteriophora Poinar in Solutions Containing Four Different Turfgrass Soil Surfactants. Insects. 2013; 4(1):1-8. https://doi.org/10.3390/insects4010001
Chicago/Turabian StyleHoctor, Terri L., Timothy J. Gibb, Cale A. Bigelow, and Douglas S. Richmond. 2013. "Survival and Infectivity of the Insect-Parasitic Nematode Heterorhabditis bacteriophora Poinar in Solutions Containing Four Different Turfgrass Soil Surfactants" Insects 4, no. 1: 1-8. https://doi.org/10.3390/insects4010001




