Incidence of Non-Immunological Defenses of Soil White Grubs on Parasitism Success of Mallophora ruficauda Larva (Diptera: Asilidae)
Abstract
:1. Introduction
2. Experimental Section
2.1. Insects
2.2. Parasitism Patterns
2.2.1. Field Parasitism Pattern
2.2.2. Laboratory Parasitism Pattern
2.3. Parasitism Behavioral Escape
2.4. Statistics
3. Results and Discussion
3.1. Field and Laboratory Parasitism Patterns
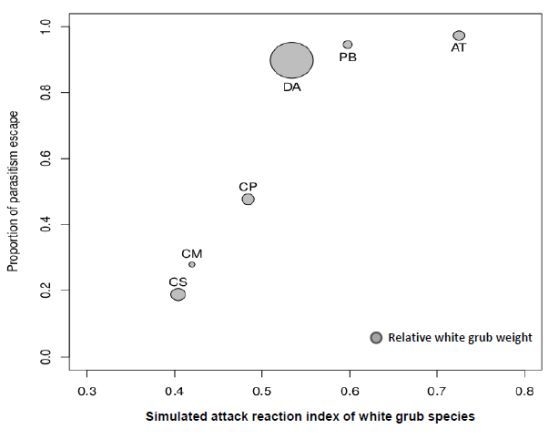
3.2. Magnitude of Non-Immunological Response
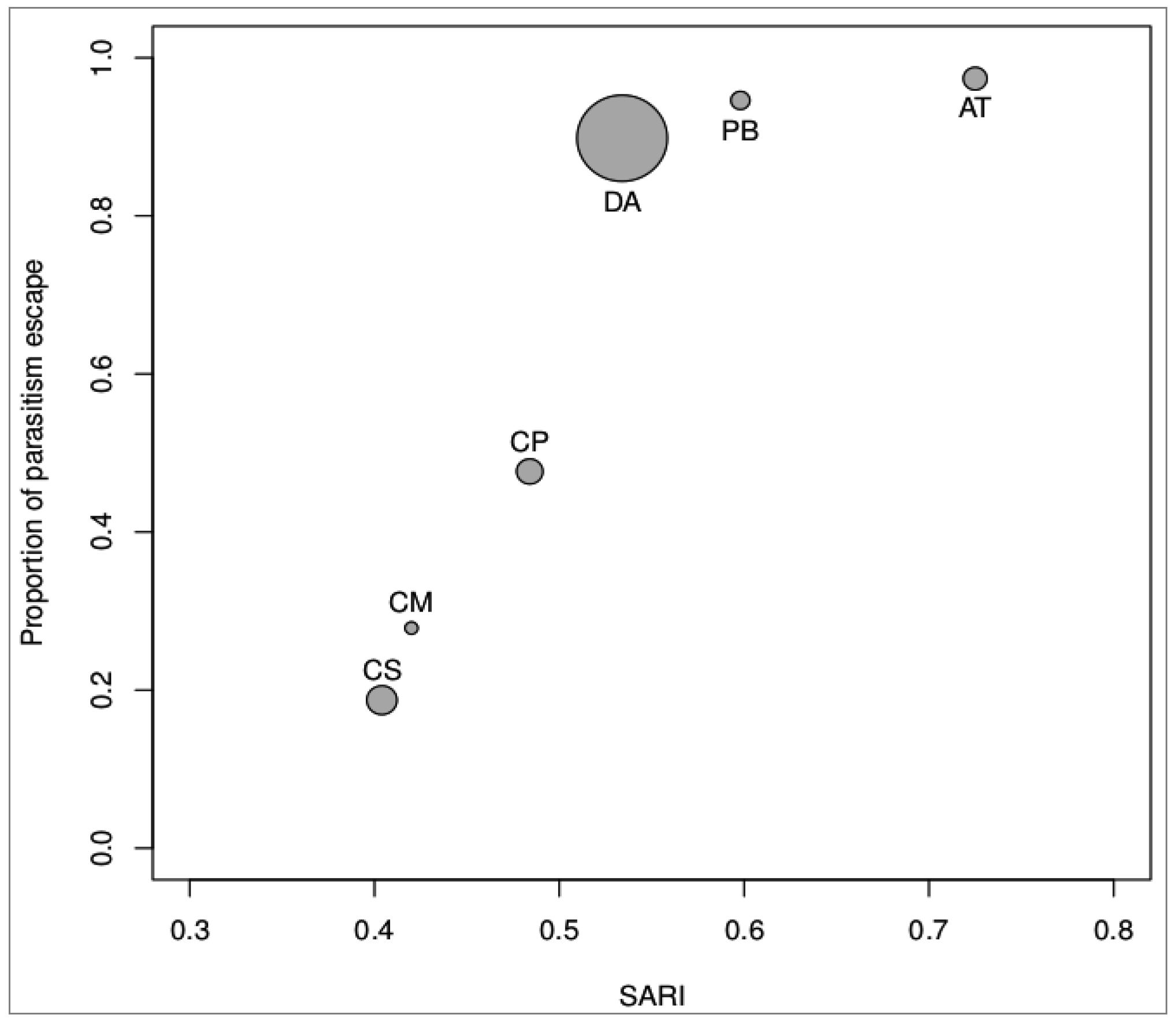
| Species | N Field* | FP (%)* | N Laboratory | LP (%) | 100-LP (%) | Mean Weight (gr) | Mean SARI score |
|---|---|---|---|---|---|---|---|
| CS | 945 (180) | 19.05 | 331 (269) | 81.27 | 18.73 | 0.745 | 0.404 |
| CM | 182 (14) | 7.69 | 79 (57) | 72.15 | 27.85 | 0.320 | 0.420 |
| CP | 81 (3) | 3.70 | 107 (56) | 52.34 | 47.66 | 0.649 | 0.484 |
| AT | 125 (1) | 0.80 | 76 (2) | 2.63 | 97.37 | 0.586 | 0.725 |
| DA | 93 (2) | 2.15 | 59 (6) | 10.17 | 89.83 | 2.225 | 0.534 |
| PB | 1005 (7) | 0.70 | 130 (7) | 5.38 | 94.62 | 0.469 | 0.598 |
| AV | 18 (1) | 5.56 | - | - | - | - | - |
| BS | 2 (0) | 0 | - | - | - | - | - |
| HB | 3 (0) | 0 | - | - | - | - | - |
| Model | s(SARI) | s(mean.weight) | Intercept | Dev. explained | R2 adj | Anova (model 1 vs model 3) | GCV score | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EDF | F | p | EDF | F | p | E | SE | t | p | Dev | F | p | ||||
| Model 1 | 1.878 | 47.013 | 0.020 | 1.082 | 2.084 | 0.292 | 0.627 | 0.032 | 19.74 | <<0.005 | 98% | 0.952 | 0.013 | 2.041 | 0.289 | 0.018 |
| Model 2 | - | - | - | 1.000 | 0.519 | 0.511 | 0.627 | 0.153 | 4.102 | <<0.05 | 11.5% | −0.106 | 0.535 | 70.936 | 0.003 | 0.210 |
| Model 3 | 1.923 | 35.34 | 0.008 | - | - | - | 0.627 | 0.037 | 16.98 | <<0.001 | 96% | 0.935 | - | - | - | 0.016 |
4. Conclusions
Acknowledgments
References
- Hawkins, B.A. Pattern & Process in Host-Parasitoid Interactions; Cambridge University Press: Cambridge, UK, 1994; p. 190. [Google Scholar]
- Vet, L.E.M.; Hemerik, L.; Visser, M.E.; Wäckers, F.E. Flexibility in host-search and patch-use strategies in insect parasitoids. In The Behavioural Ecology of Parasites; Lewis, E.E., Campbell, J.F., Sukhdeo, M.V.K., Eds.; CAB International: New York, NY, USA, 2002; Volume 3, pp. 39–64. [Google Scholar]
- Godfray, H.C.J. Parasitoids: Behavior and Evolutionary Ecology; Princeton University Press: Princeton, NJ, USA, 1994; p. 488. [Google Scholar]
- Gross, P. Insect behavioral and morphological defenses against parasitoids. Annu. Rev. Entomol. 1993, 38, 251–273. [Google Scholar]
- Strand, M.R.; Pech, L.L. Immunological basis for compatibility in parasitoid-host relationships. Annu. Rev. Entomol. 1995, 40, 31–56. [Google Scholar]
- Smilanich, A.M.; Dyer, L.Q.; Gentry, G.L. The insect immune response and other putative defenses as effective predictors of parasitism. Ecology 2009, 90, 1434–1440. [Google Scholar]
- Parker, B.J.; Barribeau, S.M.; Laughton, A.M.; de Roode, J.C.; Gerardo, N.M. Non-immunological defense in an evolutionary framework. Trends Ecol. Evol. 2011, 26, 242–248. [Google Scholar]
- Blomquist, G.J.; Bagnères, A.G. Insect Hydrocarbons Biology, Biochemistry, and Chemical Ecology; Cambridge University Press: Cambridge, UK, 2010; p. 506. [Google Scholar]
- Fatouros, N.E.; Dicke, M.; Mumm, R.; Meiners, T.; Hilker, M. Foraging behavior of egg parasitoids exploiting chemical information. Behav. Ecol. 2008, 19, 677–689. [Google Scholar]
- Jones, R.L.; Lewis, W.J.; Beroza, M.; Bierl, B.; Sparks, A.N. Host-seeking stimulants (kairomones) for the egg parasite, Trichogramma evanescens. Environ. Entomol. 1973, 2, 593–596. [Google Scholar]
- Morehead, S.A.; Feener, D.H., Jr. Visual and chemical cues used in host location and acceptance by a dipteran parasitoid. J. Insect Behav. 2000, 13, 613–625. [Google Scholar]
- Brodeur, J.; Boivin, G. Functional ecology of immature parasitoids. Annu. Rev. Entomol. 2004, 49, 27–49. [Google Scholar]
- Crespo, J.E.; Castelo, M.K. Insights to the host discrimination and host acceptance behaviour in a parasitoid (Diptera: Asilidae): Implications for fitness. J. Insect Physiol. 2009, 55, 1072–1078. [Google Scholar]
- Mackauer, M. Host discrimination and larval competition in solitary endoparasitoids. In Critical Issues in Biological Control; Mackauer, M., Ehler, L.E., Roland, J., Eds.; Intercept Ltd: Andover, MA, USA, 1990; pp. 41–62. [Google Scholar]
- Royer, L.; Fournet, S.; Brunel, E.; Boivin, G. Intra- and interspecific host discrimination by host-seeking larvae of coleopteran parasitoids. Oecologia 1999, 118, 59–68. [Google Scholar] [CrossRef]
- van Baaren, J.; Boivin, G.; Nénon, J.P. Intra- and interspecific host discrimination in two closely related egg parasitoids. Oecologia 1994, 100, 325–330. [Google Scholar]
- Roitberg, B.D.; Mangel, M. On the evolutionary ecology of marking pheromones. Evol. Ecol. 1988, 2, 289–315. [Google Scholar]
- Crespo, J.E.; Castelo, M.K. The ontogeny of host seeking behavior in a parasitoid dipteran. J. Insect Physiol. 2008, 50, 331–336. [Google Scholar]
- van Baaren, J.; Boivin, G.; Nénon, J.P. Intraspecific hyperparasitism in a primary hymenopteran parasitoid. Behav. Ecol. Sociobiol. 1995, 36, 237–242. [Google Scholar]
- Purrington, F.F.; Uleman, J.S. Brood size of a parasitic wasp: Functional correlation with the mass of a cryptic host. Ann. Entomol. Soc. Am. 1972, 65, 280–281. [Google Scholar]
- Salt, G. Experimental studies in insect parasitism. III. Host selection. Proc. Roy. Soc. Lond. B Biol. Sci. 1935, 115, 413–435. [Google Scholar] [CrossRef]
- Salt, G. The sense used by Trichogramma to distinguish between parasitized and unparasitized hosts. Proc. Roy. Soc. Lond. B Biol. Sci. 1937, 122, 57–75. [Google Scholar] [CrossRef]
- Takagi, M. The reproductive strategy of the gregarious parasitoid, Pteromalus puparium (Hymenoptera: Pteromalidae) 2. Host size discrimination and regulation of the number and sex ratio of progeny in a single host. Oecologia 1986, 70, 321–325. [Google Scholar] [CrossRef]
- Schimdt, J.M.; Smith, J.J.B. Host volume measurement by the parasitoid wasp Trichogramma minutum: The roles of curvature and surface area. Entomol. Exp. Appl. 1985, 39, 213–221. [Google Scholar]
- Lazzaro, B.P.; Little, T.J. Immunity in a variable world. Phil. Trans. Roy. Soc. Lond. B Biol. Sci. 2009, 364, 15–26. [Google Scholar] [CrossRef]
- Otti, O.; Gantenbein-Ritter, I.; Jacot, A.; Brinkhof, M.G.W. Immune response increases predation risk. Evolution 2012, 66, 732–739. [Google Scholar]
- Feener, D.H., Jr.; Brown, B.V. Diptera as parasitoids. Annu. Rev. Entomol. 1997, 42, 73–97. [Google Scholar]
- Pennacchio, F.; Strand, M.R. Evolution of developmental strategies in parasitic Hymenoptera. Annu. Rev. Entomol. 2006, 51, 233–258. [Google Scholar] [CrossRef]
- Stireman, J.O., III; O’Hara, J.E.; Wood, D.M. Behavior, ecology and evolution of tachinid parasitoids. Annu. Rev. Entomol. 2006, 51, 525–555. [Google Scholar] [CrossRef]
- Castelo, M.K.; Corley, J.C. Oviposition behavior in the robber fly Mallophora ruficauda (Diptera: Asilidae). Ann. Entomol. Soc. Am. 2004, 97, 1050–1054. [Google Scholar] [CrossRef]
- Castelo, M.K.; Ney-Nifle, N.; Corley, J.C.; Bernstein, C. Oviposition height increases parasitism success by the robber fly Mallophora ruficauda (Diptera: Asilidae). Behav. Ecol. Sociobiol. 2006, 61, 231–243. [Google Scholar]
- Copello, A. Biología del moscardón cazador de abejas (Mallophora ruficauda Wied.). Physis 1922, 6, 30–42. [Google Scholar]
- Castelo, M.K. Comportamiento de localización y patrones de explotación de hospedadores (Coleoptera: Scarabaeidae) por el moscardón cazador de abejas Mallophora ruficauda (Diptera: Asilidae) (in Spanish). Ph.D. dissertation, Universidad de Buenos Aires, Buenos Aires, Argentina, December 2003. [Google Scholar]
- Castelo, M.K.; Capurro, A.F. Especificidad y denso-dependencia inversa en parasitoides con oviposición fuera del hospedador: el caso de Mallophora ruficauda (Diptera: Asilidae) en la pampa argentina. Ecología Austral 2000, 10, 89–101. [Google Scholar]
- Castelo, M.K.; Corley, J.C. Spatial density dependent parasitism and specificity in the robber fly Mallophora ruficauda (Diptera: Asilidae). Austral Ecol. 2010, 34, 72–81. [Google Scholar]
- Castelo, M.K.; Lazzari, C.R. Host-seeking behavior in larvae of the robber fly Mallophora ruficauda (Diptera: Asilidae). J. Insect Physiol. 2004, 50, 331–336. [Google Scholar]
- Crespo, J.E.; Castelo, M.K. Life-history traits in a parasitoid dipteran species with free-living and obligate parasitic immature stages. Physiol. Entomol. 2010, 35, 160–167. [Google Scholar]
- Groba, H.F.; Castelo, M.K. Chemical interaction between a dipteran parasitoid larva and its coleopteran host: A case of exploitation of the communication system during the searching behaviour? Bull. Entomol. Res. 2012, 102, 315–323. [Google Scholar]
- Crespo, J.E.; Castelo, M.K. Orientation of M. ruficauda larvae to odors of second instar white grubs. Unpublished work, CONICET, FCEyN: Buenos Aires, Argentina, 2012. [Google Scholar]
- Musso, J.J. Nutritive and ecological requirements of robber flies (Diptera: Brachycera: Asilidae). Entomol. Gen. 1983, 9, 35–50. [Google Scholar]
- Ali, A.D.; Harivandi, M.A. White grubs in lawns. Leaflet - University of California, Cooperative Extension Service 1987, no. 21442. [Google Scholar]
- Cherry, R.H.; Schueneman, T.J. Insect management in sugarcane; Department of Entomology, University of Florida, Cooperative Extension Service, Institute of Food and Agricultural Sciences: Gainesville, FL, USA, 1998; ENY-406 series. [Google Scholar]
- Crutchfield, B.A.; Potter, D.A. Damage relationships of Japanese beetle and southern masked chafer (Coleoptera: Scarabaeidae) grubs in cool-season turfgrasses. J. Econ. Entomol. 1995, 88, 1049–1056. [Google Scholar]
- Potter, D.A. Destructive Turfgrass Insects: Biology, Diagnosis and Control; Ann Arbor Press: Chelsea, MI, USA, 1998; p. 336. [Google Scholar]
- Salvadori, J.R. Manejo de corós em cereais de inverno. EMBRAPA 1997, 3, 1–8. [Google Scholar]
- Smitley, D.R. Incidence of Popillia japonica (Coleoptera: Scarabaeidae) and other scarab larvae in nursery fields. Hortic. Entomol. 1996, 89, 1262–1266. [Google Scholar]
- Alvarado, L. Sistemática y bionomía de los estados inmaduros de coleópteros Scarabaeidae que habitan en el suelo (in Spanish). Ph.D. dissertation, Universidad Nacional de La Plata, La Plata, Argentina, September 1980. [Google Scholar]
- Rakesha, H.S. Studies on arecanut root grub, Leucopholis lepidophora Blanchard and its management by entomopathogenic fungi and plant products. Master thesis; University of Agricultural Sciences: Dharwad, India, July 2007.
- Wightman, J.A. Influence of low temperature on pupation induction in Costelytra zealandica (Coleoptera: Scarabaeidae). New Zeal. J. Zool. 1974, 1, 503–507. [Google Scholar]
- Grassé, P.P. Insectes supérieurs et Hémiptérodes. In Traité de Zoologie. Anatomie, systématique, biologie (in French); Masson & Cie: Paris, France, 1951; pp. 771–975. [Google Scholar]
- Villani, M.G.; Allee, L.L.; Díaz, A.; Robbins, P.S. Adaptative strategies of edaphic arthropods. Annu. Rev. Entomol. 1999, 44, 233–256. [Google Scholar]
- Gaugler, R.; Wang, Y.; Campbell, J.F. Aggressive and evasive behaviors in Popillia japonica (Coleoptera: Scarabaeidae) larvae: Defenses against entomopathogenic nematode attack. J. Invertebr. Pathol. 1994, 64, 193–199. [Google Scholar] [CrossRef]
- Villani, M.G.; Nyrop, J.P. Age-dependent movement patterns of Japanese beetle and European chafer (Coleoptera: Scarabaeidae) grubs in soil-turfgrass microcosms. Environ. Entomol. 1991, 20, 241–251. [Google Scholar]
- Wood, S.N. Thin plate regression splines. J. Roy. Stat. Soc. B Stat. Meth. 2003, 65, 95–114. [Google Scholar]
- Crawley, M.J. The R Book; J Wiley & Sons Ltd: Chichester, UK, 2007; p. 951. [Google Scholar]
- Wood, S.N. Generalized Additive Models: An Introduction with R; Taylor & Francis Group: Boca Raton, FL, USA, 2006; p. 385. [Google Scholar]
- Crespo, J.E. CONICET, FCEyN, UBA (Buenos Aires, Argentina). Parasitism escape of white grubs. Unpublished work, 2012. [Google Scholar]
- Brodeur, J.; Geervliet, J.B.F.; Vet, L.E.M. The role of host species, age and defensive behaviour on ovipositional decisions in a solitary specialist and gregarious generalist parasitoid (Cotesia species). Entomol. Exp. Appl. 1996, 81, 125–132. [Google Scholar]
- Firlej, A.; Lucas, E.; Coderre, D.; Boivin, G. Impact of host behavioral defenses on parasitization efficacy of a larval and adult parasitoid. Biocontrol 2010, 55, 339–348. [Google Scholar] [CrossRef]
- Potting, R.P.J.; Vermeulen, N.E.; Conlong, D.E. Active defense of herbivorous hosts against parasitism: Adult parasitoid mortality risk involved in attacking a concealed stemboring host. Entomol. Exp. Appl. 1999, 91, 143–148. [Google Scholar]
- Klemperer, H.G. The repair of larval cells and other larval activities in Geotrupes spiniger Marsham and other species (Coleoptera, Scarabaeidae). Ecol. Entomol. 1978, 3, 119–131. [Google Scholar] [CrossRef]
- Mallampalli, N.; Barbosa, P.; Weinges, K. Effects of condensed tannins and catalpol on growth and development of Compsilura concinnata (Diptera: Tachinidae) reared in gypsy moth (Lepidoptera: Lymantriidae). J. Entomol. Sci. 1996, 31, 289–300. [Google Scholar]
- Gentry, G.L.; Dyer, D.L. On the conditional nature of neotropical caterpillar defenses against their natural enemies. Ecology 2002, 83, 3108–3119. [Google Scholar] [CrossRef]
- Rogers, M.E.; Cole, T.; Ramaswamy, S.; Potter, D.A. Behavioral changes in Japanese beetle and masked chafer grubs following parasitism by tiphiid wasps. Environ. Entomol. 2003, 32, 618–625. [Google Scholar]
- Rogers, M.E.; Potter, D.A. Kairomones from scarabaeid grubs and their frass as cues in below-ground host location by the parasitoids Tiphia vernalis and Tiphia pygidialis. Entomol. Exp. Appl. 2002, 102, 307–314. [Google Scholar]
- Walker, A.M.; Hoy, M.A. Responses of Lipolexis oregmae (Hymenoptera: Aphidiidae) to different instars of Toxoptera citricida (Homoptera: Aphididae). J. Econ. Entomol. 2003, 96, 1685–1692. [Google Scholar]
- Hance, T.; van Baaren, J.; Vernon, P.; Boivin, G. Impact of temperature extremes on parasitoids in a climate change perspective. Annu. Rev. Entomol. 2007, 52, 107–126. [Google Scholar] [CrossRef]
- Thomas, M.B.; Blanford, S. Thermal biology in insect-parasite interactions. Trends Ecol. Evol. 2003, 18, 344–350. [Google Scholar]
- Fellowes, M.D.E.; Kraaijeveld, A.R.; Godfray, H.C.J. Cross-resistance following artificial selection for increased defense against parasitoids in Drosophila melanogaster. Evolution 1999, 53, 966–972. [Google Scholar]
- Hegazi, E.; Khafagi, W. Developmental interaction between suboptimal instars of Spodoptera littoralis (Lepidoptera: Noctuidae) and its parasitoid Microplitis rufiventris (Hymenoptera: Braconidae). Arch. Insect Biochem. Physiol. 2005, 60, 172–184. [Google Scholar] [CrossRef]
- Blumberg, D. Seasonal variations in the encapsulation of eggs of the encyrtid parasitoid Metaphycus stanleyi by the pyriform scale, Protopulvinaria pyriformis. Entomol. Exp. Appl. 1991, 58, 231–237. [Google Scholar] [CrossRef]
- Castelo, M.K. Attachment preference of larvae of M. ruficauda on the tegument of white grubs. Unpublished work, CONICET, FCEyN, UBA: Buenos Aires, Argentina, 2012. [Google Scholar]
- Rao, A.; Henderson, R.E.; Vinson, S.B. The probable significance of tracheal tufts in the 8th abdominal segment of Heliothis virescens (F.) on the development of its parasitoid, Toxoneuron nigriceps (Viereck). J. Insect Physiol. 2009, 55, 769–773. [Google Scholar] [CrossRef]
- Hassell, M.P. The Spatial and Temporal Dynamics of Host-Parasitoid Interactions; Oxford University Press: Oxford, UK, 2000; p. 199. [Google Scholar]
- Comins, H.N.; Hassell, M.P.; May, R.M. The spatial dynamics of host-parasitoids systems. J. Anim. Ecol. 1992, 61, 735–748. [Google Scholar]
- Godfray, H.C.J.; Hassell, M.P.; Holt, R.D. The population dynamic consequences of phenological asynchrony between parasitoids and their hosts. J. Anim. Ecol. 1994, 63, 1–10. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Castelo, M.K.; Crespo, J.E. Incidence of Non-Immunological Defenses of Soil White Grubs on Parasitism Success of Mallophora ruficauda Larva (Diptera: Asilidae). Insects 2012, 3, 692-708. https://doi.org/10.3390/insects3030692
Castelo MK, Crespo JE. Incidence of Non-Immunological Defenses of Soil White Grubs on Parasitism Success of Mallophora ruficauda Larva (Diptera: Asilidae). Insects. 2012; 3(3):692-708. https://doi.org/10.3390/insects3030692
Chicago/Turabian StyleCastelo, Marcela K., and José E. Crespo. 2012. "Incidence of Non-Immunological Defenses of Soil White Grubs on Parasitism Success of Mallophora ruficauda Larva (Diptera: Asilidae)" Insects 3, no. 3: 692-708. https://doi.org/10.3390/insects3030692
APA StyleCastelo, M. K., & Crespo, J. E. (2012). Incidence of Non-Immunological Defenses of Soil White Grubs on Parasitism Success of Mallophora ruficauda Larva (Diptera: Asilidae). Insects, 3(3), 692-708. https://doi.org/10.3390/insects3030692