Tertiary Denitrification of the Secondary Effluent by Denitrifying Biofilters Packed with Different Sizes of Quartz Sand
Abstract
:1. Introduction
2. Materials and Methods
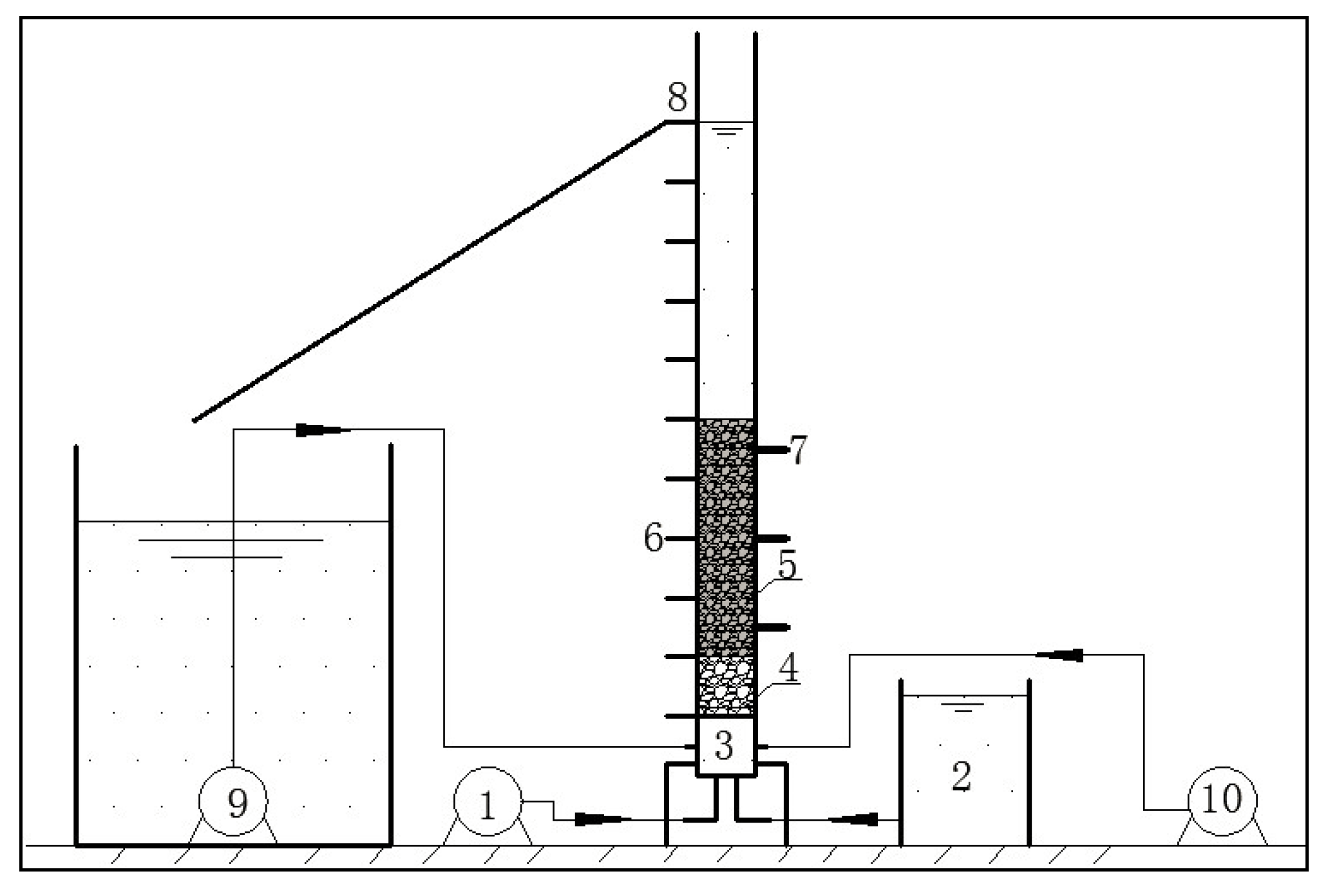
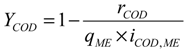
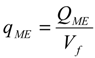
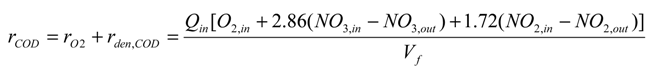
3. Results and Discussion
3.1. Long-Term Performance under Different EBRTs
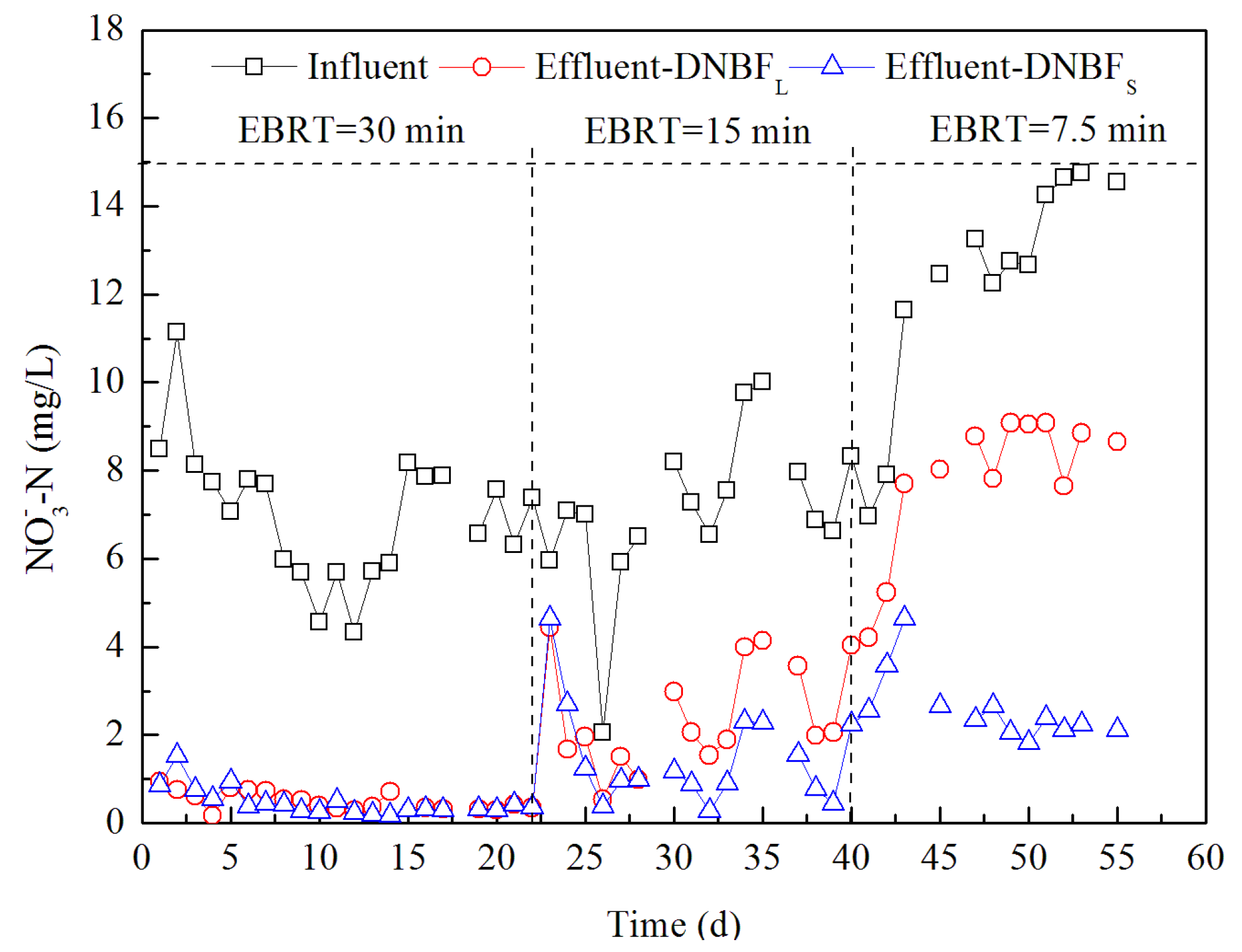
| EBRT | DNBFS | DNBFL | |||||
|---|---|---|---|---|---|---|---|
| 30 min | 15 min | 7.5 min | 30 min | 15 min | 7.5 min | ||
| COD | Influent | 125.02 ± 21.67 | 125.06 ± 9.26 | 120.33 ± 9.87 | 123.22 ± 16.48 | 126.87 ± 8.90 | 115.60 ± 8.82 |
| Effluent | 82.85 ± 19.64 | 77.31 ± 16.85 | 53.44 ± 8.63 | 88.94 ± 18.32 | 83.25 ± 15.82 | 82.20 ± 10.14 | |
| Removal | 33% ± 12% | 38% ± 12% | 55% ± 7% | 29% ± 10% | 34% ± 12% | 28% ± 5% | |
| NO3−-N | Influent | 7.02 ± 1.49 | 7.10 ± 1.74 | 13.5 ± 0.97 | 7.02 ± 1.49 | 7.10 ± 1.74 | 13.5 ± 0.97 |
| Effluent | 0.47 ± 0.31 | 1.27 ± 1.08 | 2.26 ± 0.26 | 0.49 ± 0.21 | 2.45 ± 1.18 | 7.82 ± 1.58 | |
| Removal | 93% ± 3% | 82% ± 8% | 83% ± 2% | 92% ± 2% | 68 ± 8% | 36% ± 5% | |
| NO2−-N | Influent | 0.10 ± 0.12 | 0.07 ± 0.04 | 0.06 ± 0.05 | 0.10 ± 0.12 | 0.07 ± 0.04 | 0.06 ± 0.05 |
| Effluent | 0.12 ± 0.18 | 0.42 ± 0.21 | 0.62 ± 0.16 | 0.22 ± 0.21 | 0.42 ± 0.19 | 0.68 ± 0.11 | |
| NH4+-N | Influent | 2.51 ± 1.78 | 1.85 ± 1.96 | 0.66 ± 0.56 | 2.51 ± 1.78 | 1.85 ± 1.96 | 0.66 ± 0.56 |
| Effluent | 1.76 ± 1.96 | 1.45 ± 1.81 | 0.49 ± 0.39 | 1.28 ± 1.79 | 1.39 ± 1.84 | 0.54 ± 0.39 | |
| Removal | 47% ± 27% | 27% ± 19% | 36% ± 11% | 45% ± 29% | 22% ± 15% | 17% ± 12% | |
| Turbidity | Influent | 3.78 ± 0.71 | 4.21 ± 0.93 | 4.38 ± 1.09 | 3.78 ± 0.71 | 4.21 ± 0.93 | 4.38 ± 1.09 |
| Effluent | 1.10 ± 0.11 | 1.63 ± 0.29 | 2.01 ± 0.26 | 1.21 ± 0.25 | 1.71 ± 0.25 | 2.13 ± 0.21 | |
| Removal | 69% ± 6% | 59% ± 9% | 51% ± 11% | 66% ± 8% | 58% ± 7% | 49% ± 9% | |

3.2. Dynamics of Oxidized Nitrogen along the Biofilter Depth
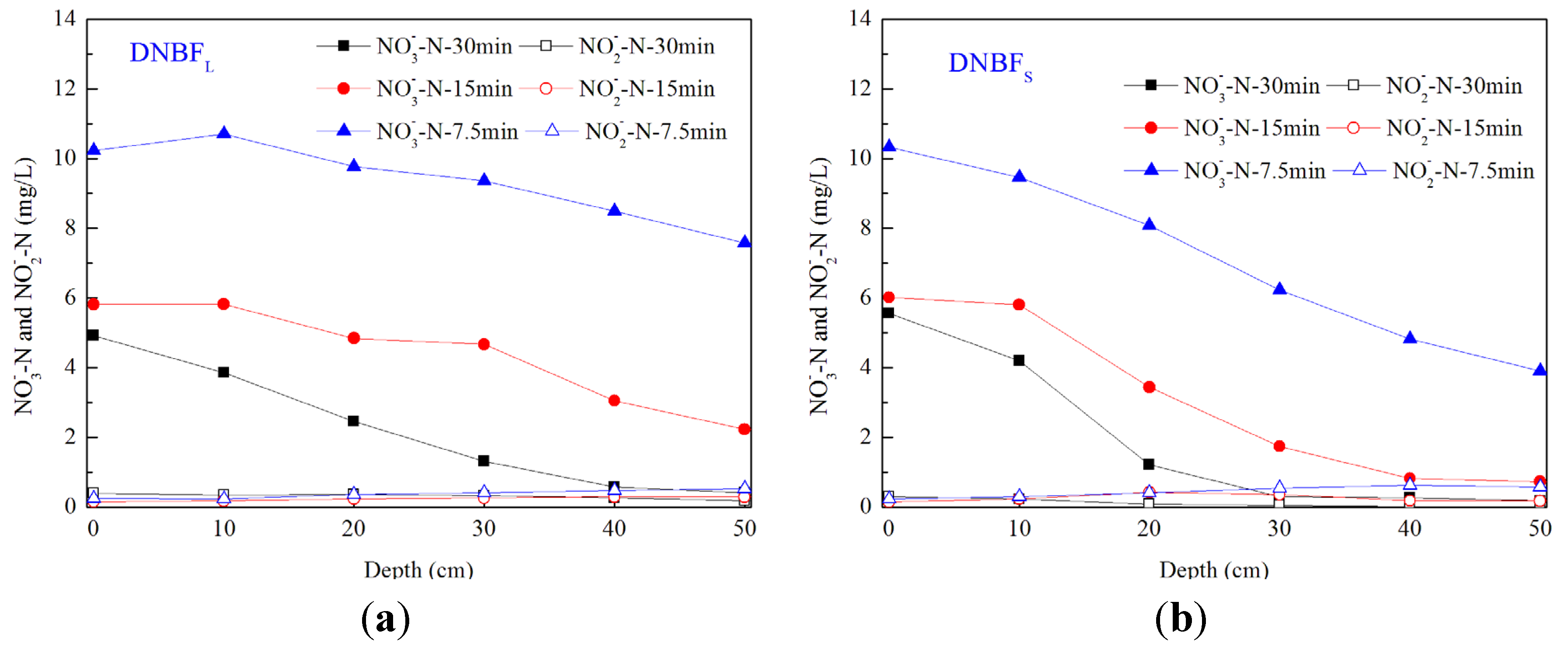
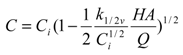

3.3. Performance of Biofilters within a Backwashing Cycle
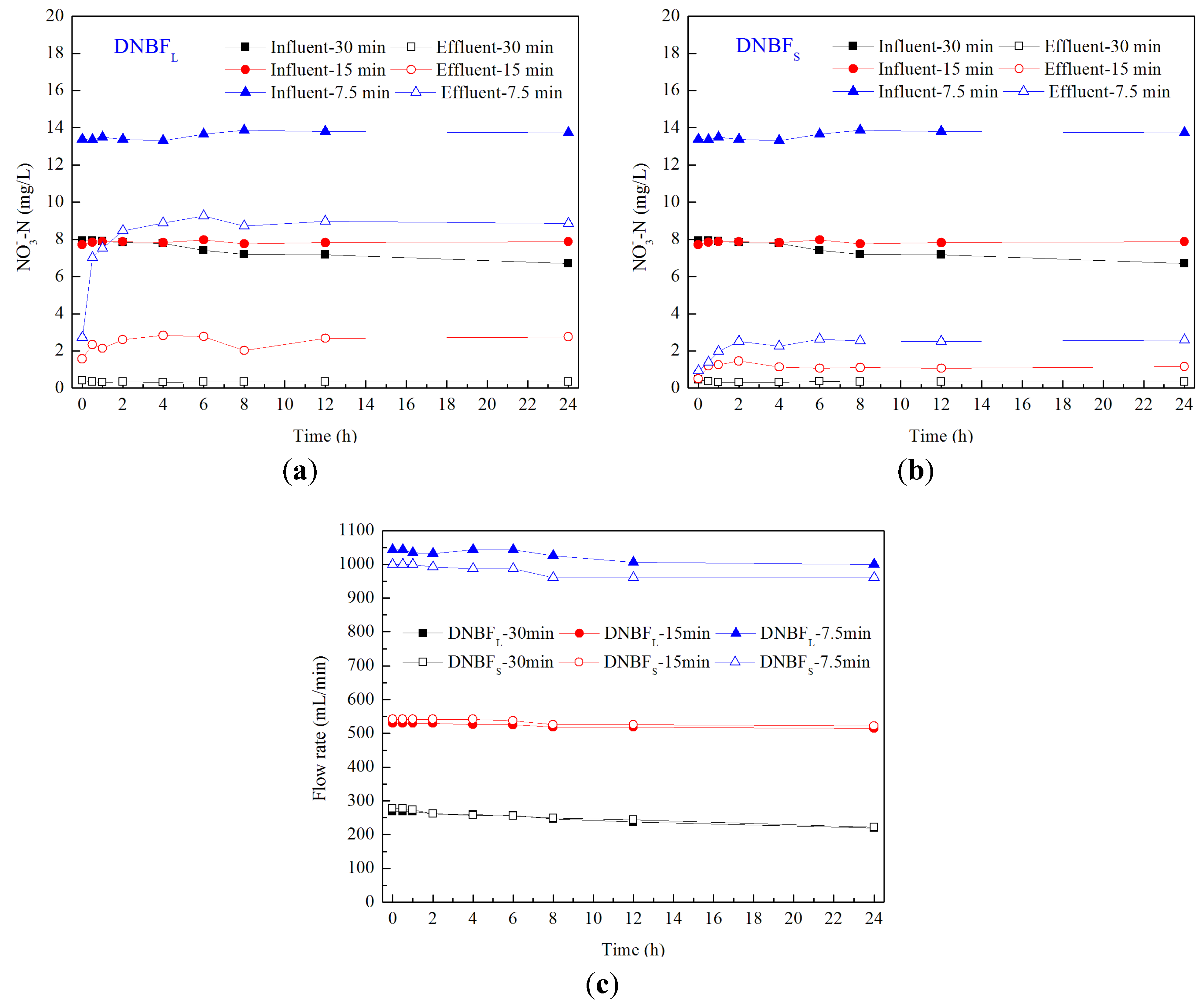
4. Conclusions
Acknowledgment
Author Contributions
Conflicts of Interest
References
- Ryther, J.H.; Dunstan, W.M. Nitrogen, phosphorus, and eutrophication in the coastal marine environment. Science 1971, 171, 1008–1013. [Google Scholar]
- Boltz, J.P.; Morgenroth, E.; Daigger, G.T.; DeBarbadillo, C.; Murthy, S.; Sørensen, K.H.; Stinson, B. Method to identify potential phosphorus rate-limiting conditions in post-denitrification biofilm reactors within systems designed for simultaneous low-level effluent nitrogen and phosphorus concentrations. Water Res. 2012, 46, 6228–6238. [Google Scholar] [CrossRef]
- Park, J.B.K.; Craggs, R.J.; Sukias, J.P.S. Removal of nitrate and phosphorus from hydroponic wastewater using a hybrid denitrification filter (HDF). Bioresour. Technol. 2009, 100, 3175–3179. [Google Scholar] [CrossRef]
- Ledwell, S.; Fabiyi, M.; Farmer, G. Optimizing denitrification with non-methanol carbon sources in deep-bed denitrification filter technologies. Proc. Water Environ. Fed. 2011, 14, 406–419. [Google Scholar] [CrossRef]
- Sage, M.; Daufin, G.; Gésan-Guiziou, G. Denitrification potential and rates of complex carbon source from dairy effluents in activated sludge system. Water Res. 2006, 40, 2747–2755. [Google Scholar] [CrossRef]
- Moore, R.; Quarmby, J.; Stephenson, T. The effects of media size on the performance of biological aerated filters. Water Res. 2001, 35, 2514–2522. [Google Scholar] [CrossRef] [Green Version]
- Tian, W.; Wen, X.; Yang, A.; Qian, Y. Performance and its influence factors of a Zeolite biofilter for dilute domestic wastewater treatment. Environ. Sci. 2003, 24, 97–101. (In Chinese) [Google Scholar]
- Mendoza-Espinosa, L.; Stephenson, T. A review of biological aerated filters (BAFs) for wastewater treatment. Environ. Eng. Sci. 1999, 16, 201–216. [Google Scholar] [CrossRef]
- Mann, A.T.; Mendoza-Espinosa, L.; Stephenson, T. Performance of floating and sunken media biological aerated filters under unsteady state conditions. Water Res. 1999, 33, 1108–1113. [Google Scholar] [CrossRef]
- Fdz-Polanco, F.; Mendez, E.; Uruena, M.A. Spatial distribution of heterotrophs and nitrifiers in a submerged biofilter for nitrification. Water Res. 2000, 34, 4081–4089. [Google Scholar] [CrossRef]
- Gomez, M.A.; Hontoria, E.; Gonzalez-Lopez, J. Effect of dissolved oxygen concentration on nitrate removal from groundwater using a denitrifying submerged filter. J. Hazard. Mater. 2002, 90, 267–278. [Google Scholar] [CrossRef]
- Galvez, J.M.; Gomez, M.A.; Hontoria, E.; Gonzzlez-Lopez, J. Influence of hydraulic loading and air flowrate on urban wastewater nitrogen removal with a submerged fixed-film reactor. J. Hazard. Mater. 2003, 101, 219–229. [Google Scholar] [CrossRef]
- He, S.B.; Xue, G.; Kong, H.N. The performance of BAF using natural zeolite as filter media under conditions of low temperature and ammonium shock load. J. Hazard. Mater. 2007, 143, 291–295. [Google Scholar] [CrossRef]
- Liu, F.; Zhao, C.-C.; Zhao, D.-F.; Liu, G.-H. Tertiary treatment of textile wastewater with combined media biological aerated filter (CMBAF) at different hydraulic loadings and dissolved oxygen concentrations. J. Hazard. Mater. 2008, 160, 161–167. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewater; APHA: Washington, DC, USA, 1995. [Google Scholar]
- Farabegoli, G.; Gavasci, R.; Lombardi, F.; Romani, F. Denitrification in tertiary filtration: Application of an up-flow filter. J. Environ. Sci. Health Part A 2003, 38, 2169–2177. [Google Scholar] [CrossRef]
- Zhou, B.; Cao, J.; Xu, Z. Biofilm accretion and startup of denitrification biofilter. Environ. Sci. Technol. 2009, 22, 5–7. (In Chinese) [Google Scholar]
- Koch, G.; Siegrist, H. Denitrification with methanol in tertiary filtration at wastewater treatment plant Zürich-Werdhoölzli. Water Sci. Technol. 1997, 36, 165–172. [Google Scholar] [CrossRef]
- Aesoy, A.; Odegaard, H.; Bach, K.; Pujol, R.; Hamon, M. Denitrification in a packed bed biofilm reactor (BIOFOR)—Experiments with different carbon sources. Water Res. 1998, 32, 1463–1470. [Google Scholar] [CrossRef]
- Holloway, R.; Zhao, H.; Rinne, T.; Thesing, G.; Parker, J.; Beals, M. The impact of temperature and loading on meeting stringent nitrogen requirements in a two-stage BAF a comparison of pilot and full-scale performance. Proc. Water Environ. Fed. 2008, 16, 3586–3601. [Google Scholar]
- DeBarbadillo, C.; Miller, P.; Ledwell, S. A comparison of operating issues and dosing requirements for alternative carbon sources in denitrification filters. Proc. Water Environ. Fed. 2008, 15, 6603–6617. [Google Scholar] [CrossRef]
- Purtschert, I.; Siegrist, H.; Gujer, W. Enhanced denitrification with methanol at WWTP Zürich-Werdhölzli. Water Sci. Technol. 1996, 33, 117–126. [Google Scholar] [CrossRef]
- Gomez, M.A.; Gonzalez-Lopez, J.; Hontoria-Garcıa, E. Influence of carbon source on nitrate removal of contaminated groundwater in a denitrify submerged filter. J. Hazard. Mater. 2000, 80, 69–80. [Google Scholar] [CrossRef]
- Foglar, L.; Briski, F. Wastewater denitrification process—The influence of methanol and kinetic analysis. Process Biochem. 2003, 39, 95–103. [Google Scholar] [CrossRef]
- Jimenez, B.; Buitron, G. Comparison between three secondary effluents in tertiary high rate filtration. Environ. Technol. 1996, 17, 987–995. [Google Scholar] [CrossRef]
- Kennedy, T.J.; Hernandez, E.A.; Morse, A.N.; Anderson, T.A. Hydraulic loading rate effect on removal rates in a BioSand filter: A pilot study of three conditions. Water Air Soil Poll. 2012, 223, 4527–4537. [Google Scholar] [CrossRef]
- Harremoes, P. The significance of pore diffusion to filter denitrification. J. Water Poll. Control Fed. 1976, 48, 377–388. [Google Scholar]
- Hanning, K.F.; Harremoes, P.; Nielsen, M. Evaluating and modelling the kinetics in a full scale submerged denitrification filter. Water Sci. Technol. 1995, 32, 115–123. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wei, N.; Shi, Y.; Wu, G.; Hu, H.; Wu, Y.; Wen, H. Tertiary Denitrification of the Secondary Effluent by Denitrifying Biofilters Packed with Different Sizes of Quartz Sand. Water 2014, 6, 1300-1311. https://doi.org/10.3390/w6051300
Wei N, Shi Y, Wu G, Hu H, Wu Y, Wen H. Tertiary Denitrification of the Secondary Effluent by Denitrifying Biofilters Packed with Different Sizes of Quartz Sand. Water. 2014; 6(5):1300-1311. https://doi.org/10.3390/w6051300
Chicago/Turabian StyleWei, Nan, Yunhong Shi, Guangxue Wu, Hongying Hu, Yihui Wu, and Hui Wen. 2014. "Tertiary Denitrification of the Secondary Effluent by Denitrifying Biofilters Packed with Different Sizes of Quartz Sand" Water 6, no. 5: 1300-1311. https://doi.org/10.3390/w6051300




