Preliminary Study on the Effect of Wastewater Storage in Septic Tank on E. coli Concentration in Summer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and On-site Wastewater Treatment System (OWTS)
2.2. Water Quality Parameters
2.3 Statistical Analysis
3. Results
3.1. Common Water Quality Parameters
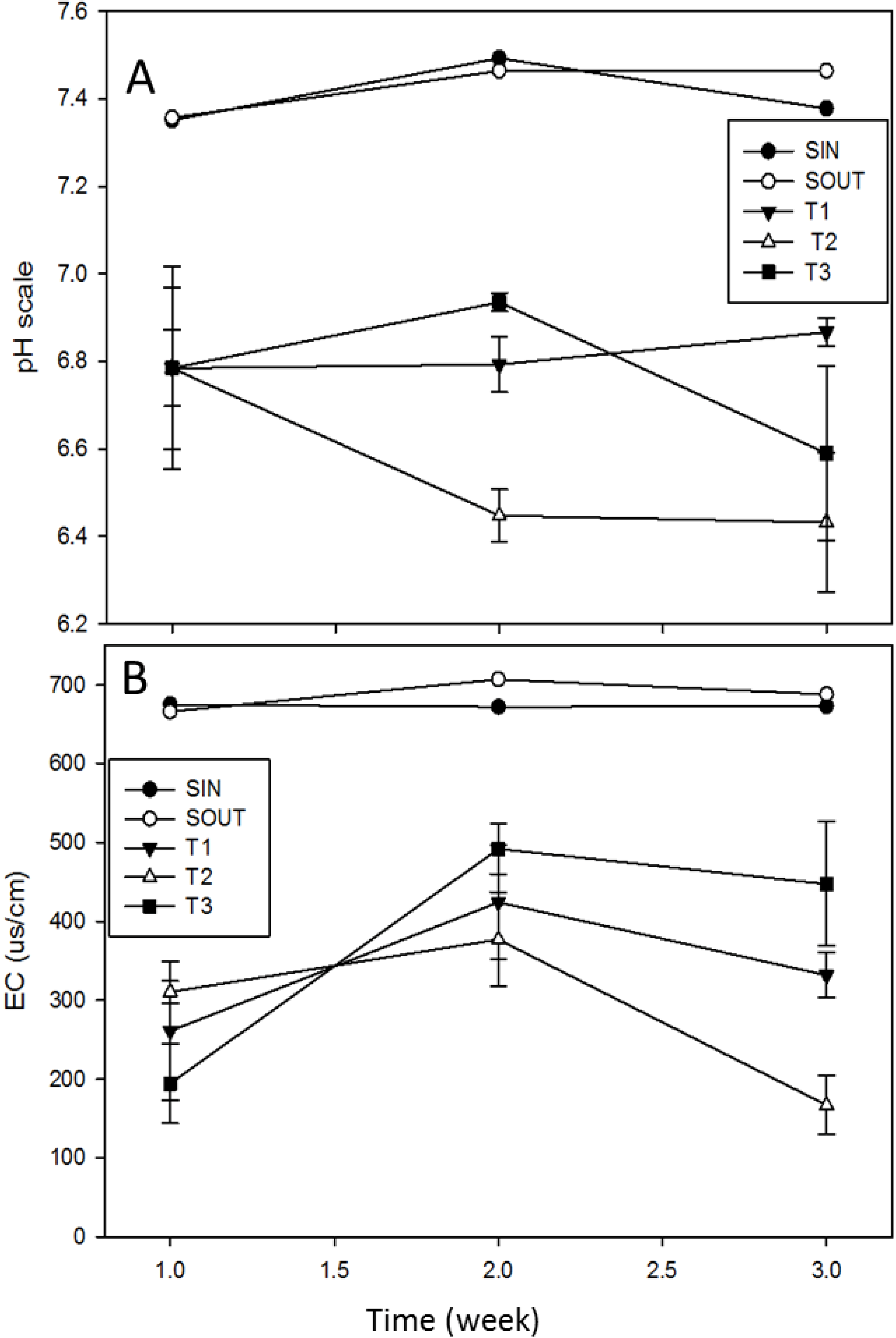
3.2. Fecal Indicator Bacteria

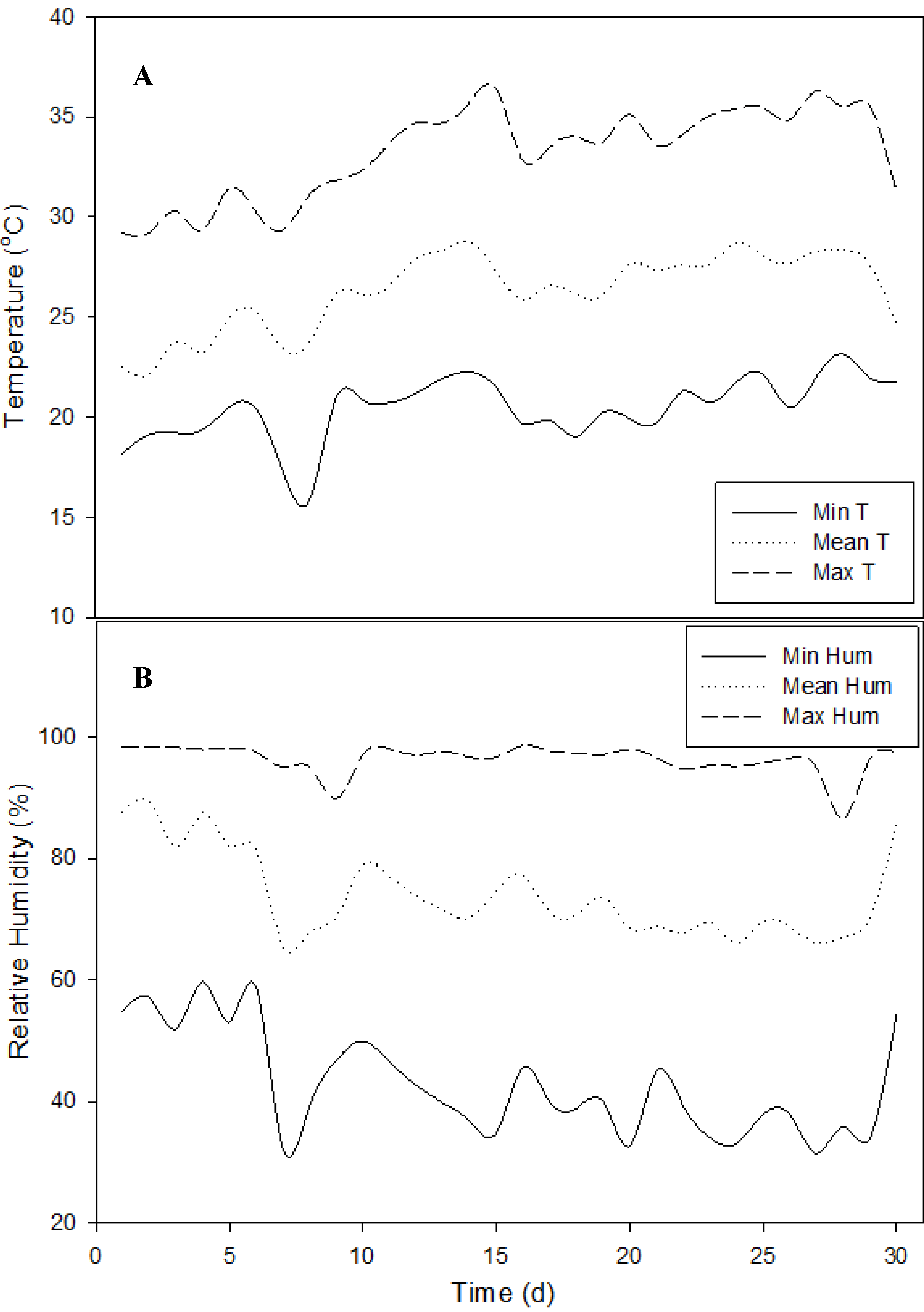
4. Discussions
5. Conclusions
Acknowledgements
References
- U.S. Environmental Protection Agency (USEPA), Onsite Wastewater Treatment Systems Manual; USEPA: Washington, DC, USA, 2002; EPA/625/R-00/008.
- Gerba, C.; Smith, J.E., Jr. Sources of pathogenic microorganisms and their fate during land application of wastes. J. Environ. Qual. 2005, 34, 42–48. [Google Scholar]
- Oakley, S.; Greenwood, W.P.; Lee, M. Monitoring Nitrogen and Virus Removal in the Vadose One with Suction Lysimeters. In Proceedings of 10th Northwest On-site Wastewater Treatment Short Course and Equipment Exhibition, Seattle, WA, USA, 20–21 September 1999; University of Washington: Seattle, WA, USA, 1999; pp. 221–232. [Google Scholar]
- Paul, J.H.; McLaughlin, M.R.; Griffin, D.W.; Lipp, E.K.; Stokes, R.; Rose, J.B. Rapid movement of wastewater from onsite disposal systems into surface waters in the lower Florida keys. Estuaries 2000, 23, 662–668. [Google Scholar] [CrossRef]
- Van Cuyk, S.; Siegrist, R.L.; Lowe, K.; Harvey, R.W. Evaluating microbial purification during soil treatment of wastewater with multicomponent tracer and surrogate tests. J. Environ. Qual. 2004, 33, 316–329. [Google Scholar]
- Ahmed, W.; Neller, R.; Katouli, M. Evidence of septic system failure determined by a bacterial biochemical fingerprinting method. J. Appl. Microbiol. 2005, 98, 910–920. [Google Scholar] [CrossRef]
- Keswick, B.H.; Gerba, C.P. Viruses in groundwater. Environ. Sci. Technol. 1980, 14, 1290–1297. [Google Scholar] [CrossRef]
- Arnade, L.J. Seasonal correlation of well contamination and septic tank distance. Ground Water 1999, 37, 920–923. [Google Scholar] [CrossRef]
- Habteselassie, M.Y.; Kirs, M.; Conn, K.E.; Blackwood, A.D.; Kelly, G.; Noble, R.T. Tracking microbial transport through four onsite wastewater treatment systems to receiving waters in eastern north Carolina. J. Appl. Microbiol. 2011, 111, 835–847. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. National Summary of Impaired Waters and TMDL Information. Available online: http://iaspub.epa.gov/waters10/attains_nation_cy.control?p_report_type=T (accessed on 31 May 2013).
- Oakley, S.M.; Gold, A.J.; Oczkowski, A.J. Nitrogen control through decentralized wastewater treatment: Process performance and alternative strategies. Ecol. Engineer. 2010, 36, 1520–1531. [Google Scholar] [CrossRef]
- Moore, B. Innovative and Alternative System Use in Rhode Island. Personal Communication, Rhode Island Department of Environmental Management (RIDEM): Providence, RI, USA, 2008. [Google Scholar]
- Rhode Island Department of Environmental Management (RIDEM). Rules Establishing Minimum Standards Relating to Location, Design, Construction and Maintenance of Onsite Wastewater Treatment Systems. Personal Communication, RIDEM: Providence, RI, USA, 2008.
- Rich, B. Overview of the field test of innovative on-site wastewater treatment systems during the La Pine National Demonstration Project. J. Hydrologic Eng. 2008, 13, 752–760. [Google Scholar] [CrossRef]
- National Sanitation Foundation (NSF) International, Pennsylvania ONLOT Technology Verification Program; NSF International: Ann Arbor, MI, USA, 2009.
- Obropta, C.C.; Berry, D. Onsite Wastewater Treatment Systems: Alternative Technologies; Rutgers University Cooperative Research and Extension: New Brunswick, NJ, USA, 2005. [Google Scholar]
- Humphrey, C.P.; O’Driscoll, M.A.; Zarate, M.A. Evaluation of on-site wastewater system Escherichia coli contributions to shallow groundwater in costal North Carolina. Water Sci. Technol. 2011, 63, 789–795. [Google Scholar] [CrossRef]
- Carroll, S.; Hargreaves, M.; Goonetilleke, A. Source tracking pollution from onsite wastewater treatment systems in surface waters using antibiotic resistance analysis. J. Appl. Microbiol. 2005, 99, 471–482. [Google Scholar] [CrossRef]
- Bradshaw, J.K.; Radcliffe, D.E. Nitrogen Dynamics in a Piedmont Wastewater Treatment System. In Proceedings of the 2011 Georgia Water Resources Conference, Athens, GA, USA, 11–13 April 2011; Carroll, D., Ed.; Georgia Water Resources Association: Athens, GA, USA, 2011; pp. 110–113. [Google Scholar]
- Georgia Department of Human Resources—Division of Public Health (GADHR-DPH). Manual for Onsite Sewage Management Systems. Available online: http://health.state.ga.us/pdfs/environmental/LandUse/Manual/CompleteOnsiteManual.pdf (accessed on 8 March 2013).
- American Public Health Association (APHA)American Water Works Association (AWWA)Water Pollution Control Research (WPCR) Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 1999.
- Hurley, M.A.; Roscoe, M.E. Automated statistical analysis of microbial enumeration by dilution series. J. Appl. Bacteriol. 1983, 55, 159–164. [Google Scholar] [CrossRef]
- Georgia Weather Net Home Page. Available online: http://www.georgiaweather.net/ (accessed on 10 April 2013).
- Byappanahalli, M.; Fujioka, R. Indigenous soil bacteria and low moisture may limit but allow faecal bacteria to multiply and become minor population in tropical soils. Water Sci. Technol. 2004, 50, 27–32. [Google Scholar]
- Fujioka, R.S. Monitoring coastal marine waters for spore-forming bacteria of faecal and soil origin to determine point from non-point source pollution. Water Sci. Technol. 2001, 44, 181–188. [Google Scholar]
- Ishii, S.; Ksoll, W.B.; Hicks, R.E.; Sadowsky, M.J. Presence and growth of naturalized Escherichia coli in temperate soils from Lake Superior watersheds. Appl. Environ. Microbiol. 2006, 72, 612–621. [Google Scholar] [CrossRef]
- Ottoson, J.; Stenstrom, T.A. Faecal contamination of greywater and associated microbial risks. Water Research 2003, 37, 645–655. [Google Scholar] [CrossRef]
- Leclerc, H.; Mossel, D.A.A.; Edberg, S.C.; Struijk, C.B. Advances in the bacteriology of the coliform group: Their suitability as markers of microbial water safety. Annu. Rev. Microbiol. 2001, 55, 201–234. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA), Ambient Water Quality Criteria for Bacteria; United States Environmental Protection Agency: Washington, DC, USA, 1986.
- Meschke, J.S.; Sobsey, M.D. Comparative adsorption of Norwalk virus, Poliovirus 1 and F + RNA coliphage MS2 to soils suspended in treated wastewater. Water Sci. Technol. 1998, 38, 187–189. [Google Scholar]
- Sobsey, M.D.; Dean, C.H.; Knuckles, M.E.; Wagner, R.A. Interactions and survival of enteric viruses in soil materials. Appl. Environ. Microbiol. 1980, 40, 92–101. [Google Scholar]
- Scandura, J.E.; Sobsey, M.D. Viral and bacterial contamination of groundwater from on-site sewage treatment systems. Water Sci. Technol. 1997, 35, 141–146. [Google Scholar] [CrossRef]
- Trueba, F.J.; Woldringh, C.L. Changes in cell diameter during the division cycle of Escherichia coli. J. Bacteriol. 1980, 142, 869–878. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Appling, D.; Habteselassie, M.Y.; Radcliffe, D.; Bradshaw, J.K. Preliminary Study on the Effect of Wastewater Storage in Septic Tank on E. coli Concentration in Summer. Water 2013, 5, 1141-1151. https://doi.org/10.3390/w5031141
Appling D, Habteselassie MY, Radcliffe D, Bradshaw JK. Preliminary Study on the Effect of Wastewater Storage in Septic Tank on E. coli Concentration in Summer. Water. 2013; 5(3):1141-1151. https://doi.org/10.3390/w5031141
Chicago/Turabian StyleAppling, Dominique, Mussie Y. Habteselassie, David Radcliffe, and James K. Bradshaw. 2013. "Preliminary Study on the Effect of Wastewater Storage in Septic Tank on E. coli Concentration in Summer" Water 5, no. 3: 1141-1151. https://doi.org/10.3390/w5031141
APA StyleAppling, D., Habteselassie, M. Y., Radcliffe, D., & Bradshaw, J. K. (2013). Preliminary Study on the Effect of Wastewater Storage in Septic Tank on E. coli Concentration in Summer. Water, 5(3), 1141-1151. https://doi.org/10.3390/w5031141



