A Preliminary Evaluation of Lyophilized Gelatin Sponges, Enhanced with Platelet-Rich Plasma, Hydroxyapatite and Chitin Whiskers for Bone Regeneration
Abstract
:1. Introduction
| Function | |
|---|---|
| PDGF | Mesenchymal stem cell and progenitor cell recruitment, proliferation, migration and differentiation into osteoblasts |
| TGF-β | Mesenchymal stem cells differentiation, increased production of collagen and mineral matrix |
| IGF-1 | Osteoblast proliferation and differentiation |
| VEGF | Angiogenesis, Endochondral ossification |
| Fibronectin, Vitronectin | Enhance formation of focal adhesions by osteoblasts, osteoblast migration |
2. Results and Discussion
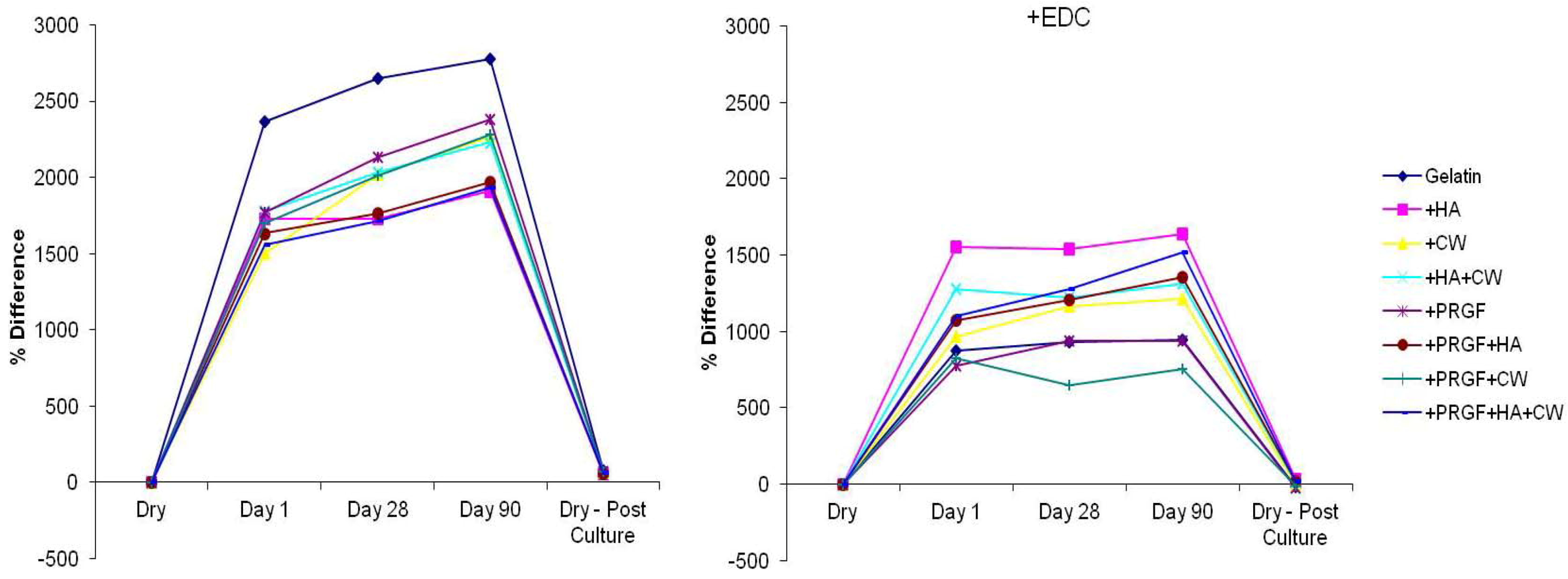
2.1. Mass Loss
| +EDC | ||||
|---|---|---|---|---|
| Pre | Post | Pre | Post | |
| Gelatin | 7.97 ± 0.57 | 14.27 ± 1.51 * | 7.53 ± 1.40 | 6.20 ± 1.71 |
| +HA | 12.17 ± 0.61 | 19.10 ± 0.78 * | 8.17 ± 0.67 | 11.00 ± 1.31 * |
| +CW | 10.67 ± 0.98 | 17.13 ± 9.29 * | 11.23 ± 0.95 | 10.63 ± 1.45 |
| +HA+CW | 11.20 ± 0.70 | 19.13 ± 8.39 * | 7.23 ± 0.29 | 7.93 ± 0.95 |
| +PRGF | 10.83 ± 0.99 | 18.53 ± 1.60 * | 9.40 ± 0.53 | 7.13 ± 0.81 * |
| +PRGF+HA | 11. 07 ± 0.45 | 17.43 ± 0.47 * | 8.13 ± 0.76 | 9.47 ± 0.21 * |
| +PRGF+CW | 9.63 ± 1.01 | 16.10 ± 0.27 * | 8.33 ± 0.50 | 7.73 ± 0.31 |
| +PRGF+HA+CW | 11.33 ± 0.65 | 18.23 ± 0.40 * | 8.10 ± 0.26 | 9.47 ± 0.25 * |
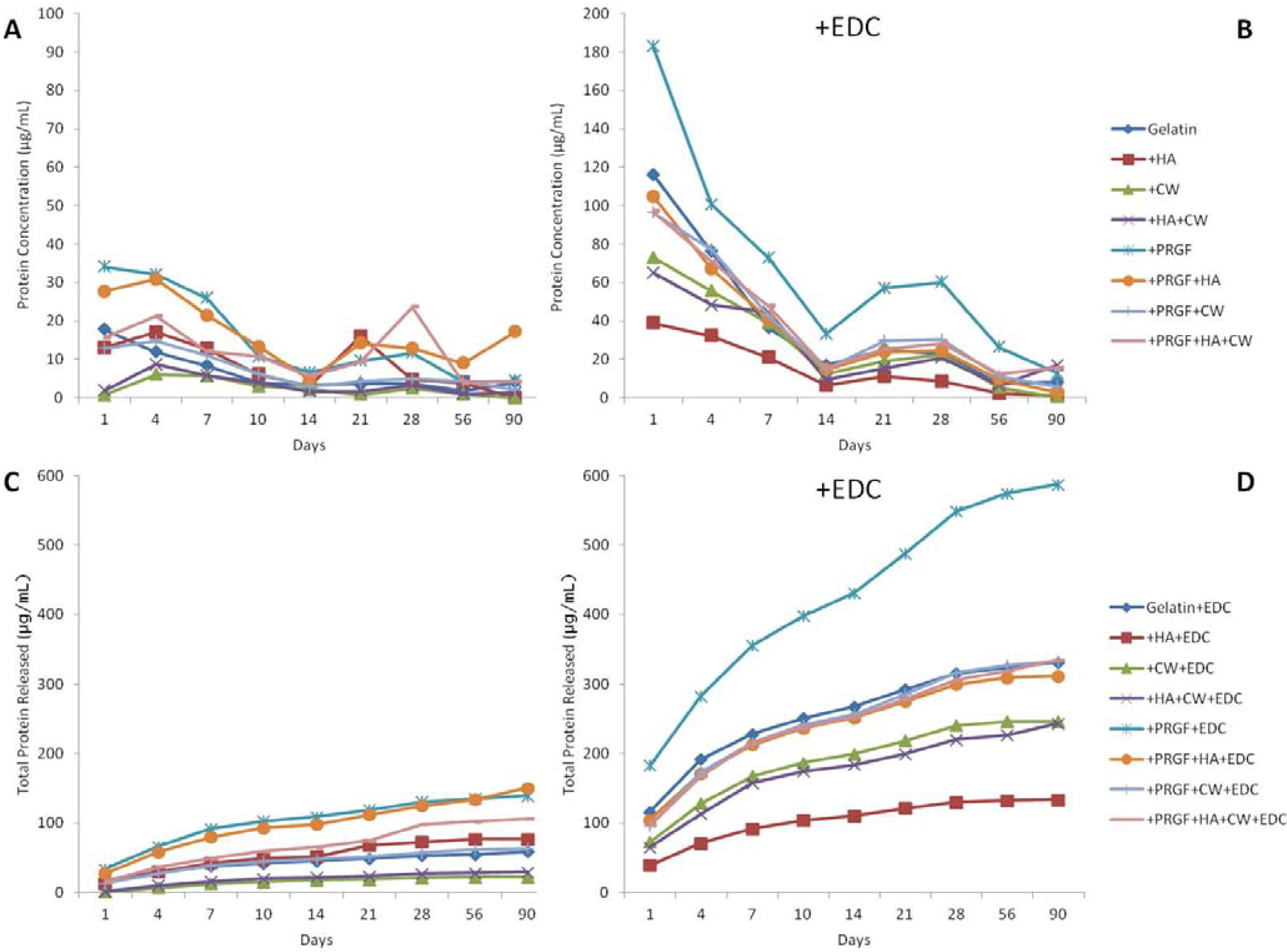
2.2. Protein Release

| % Released | |
|---|---|
| +PRGF+EDC | 27.79 |
| Gelatin+EDC | 19.30 |
| +PRGF+HA+EDC | 19.18 |
| +PRGF+HA+CW+EDC | 16.75 |
| +PRGF+CW+EDC | 15.62 |
| +HA+CW+EDC | 13.94 |
| +CW+EDC | 13.66 |
| +PRGF+HA | 9.29 |
| +HA+EDC | 8.61 |
| +PRGF | 6.59 |
| +PRGF+HA+CW | 5.32 |
| +HA | 4.95 |
| Gelatin | 3.43 |
| +PRGF+CW | 2.98 |
| +HA+CW | 1.68 |
| +CW | 1.23 |
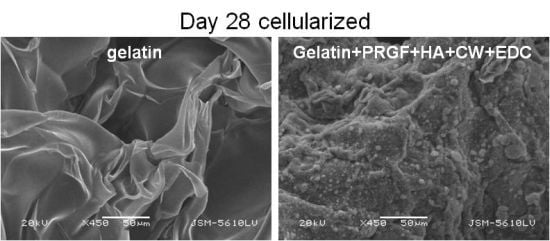
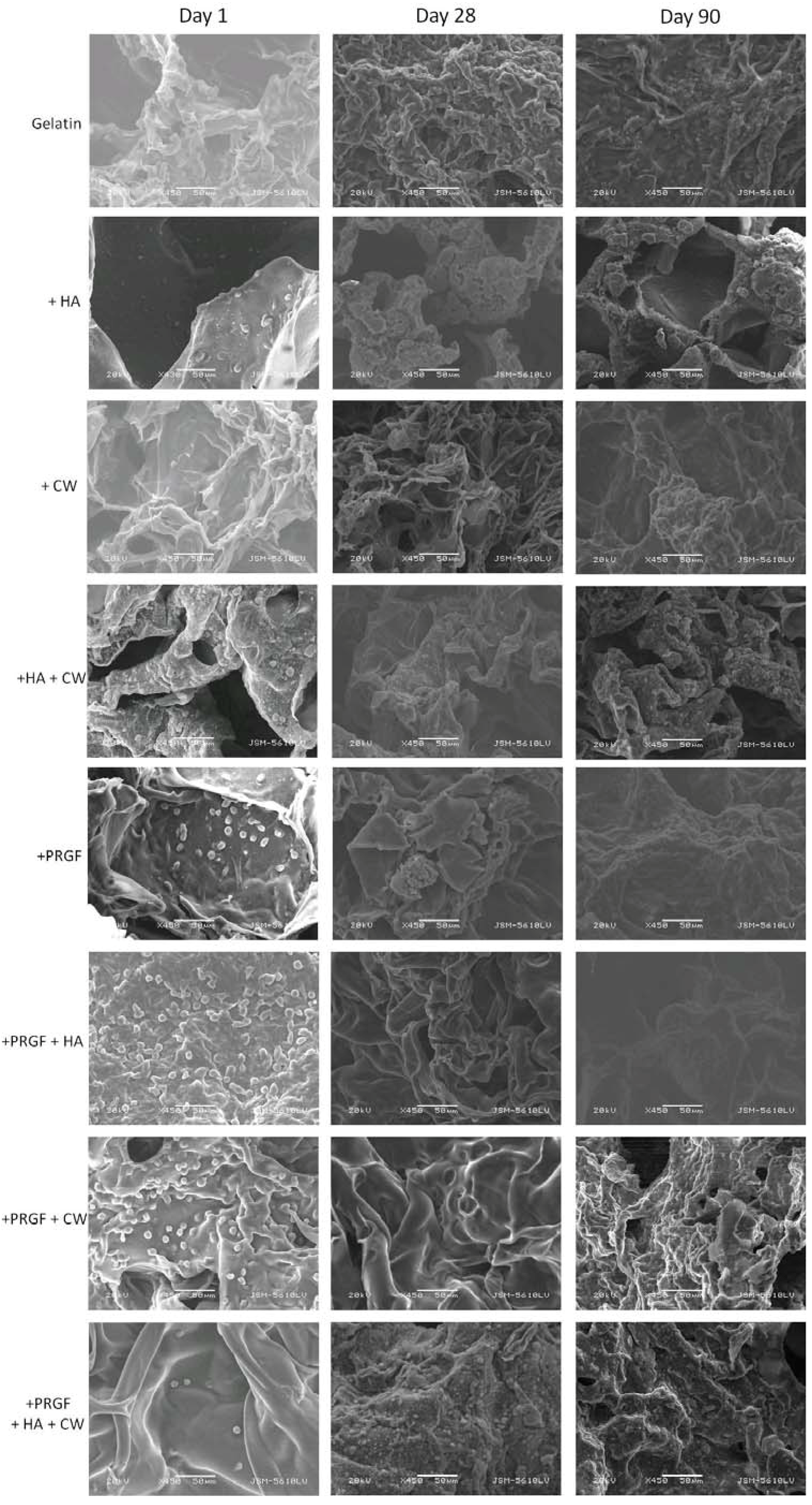
2.3. Cell Attachment, Migration and Matrix Production
2.3.1. Scanning Electron Microscopy
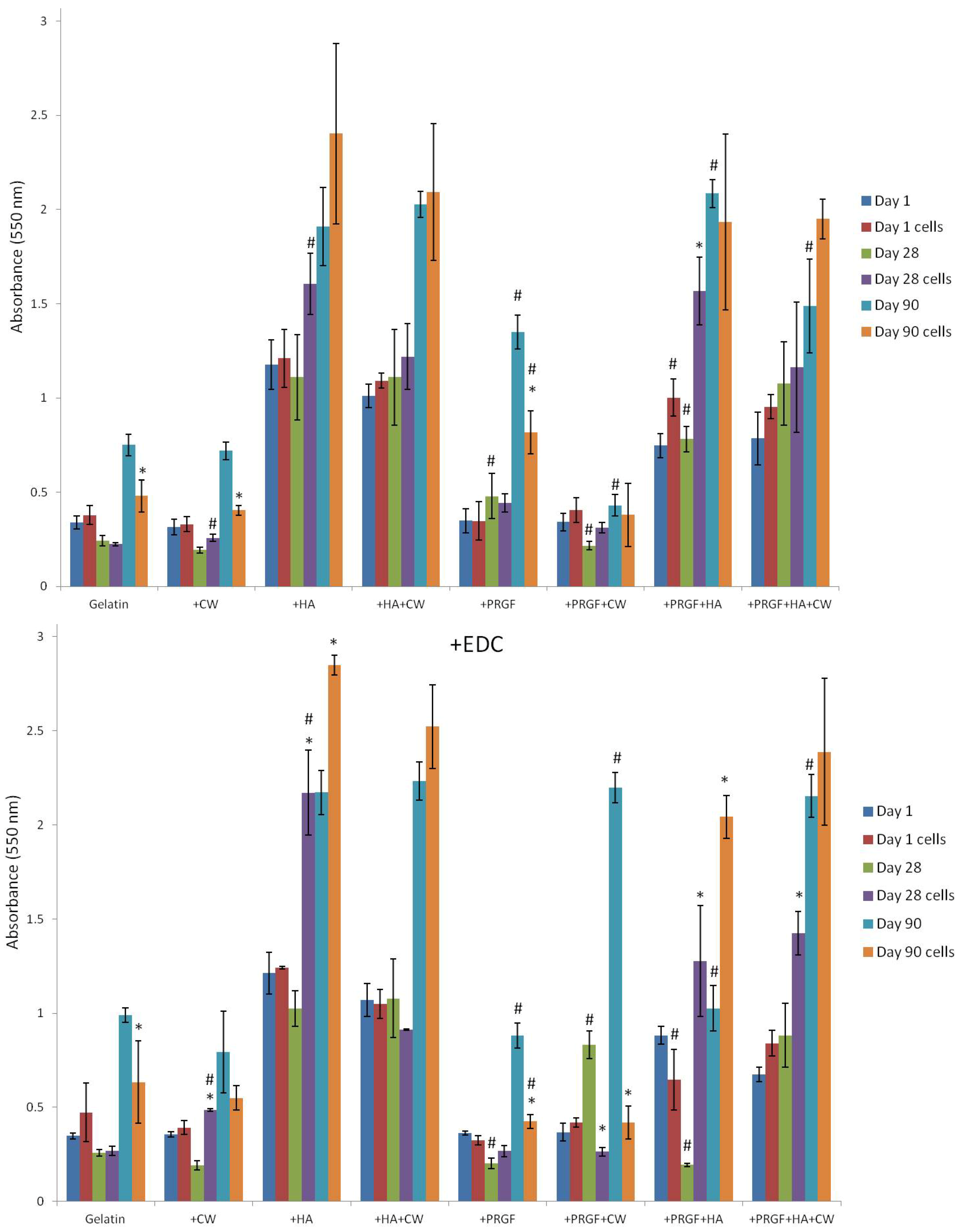
2.3.2. Alizarin Red S Staining

| Post Cross-Linked | +EDC | |
|---|---|---|
| Day 1 cells | +PRGF +HA | |
| Day 28 | ||
| +PRGF +HA | ||
| Day 28 cells | ||
| +HA | ||
| Day 90 | ||
| +PRGF +HA | +PRGF +HA +CW | |
| Day 90 cells | +PRGF |
2.3.3. DAPI Staining


2.4. Uniaxial Compression Testing

3. Experimental Section
3.1. Fabrication of Gelatin Composite Sponges
| Amount (mg/mL) Added to Gelatin Solution | Sonicated | |
|---|---|---|
| +HA | 10 | yes |
| +CW | 10 | yes |
| +HA+CW | 5 (HA) and 5 (CW) | yes |
| +PRGF | 10 | no |
| +PRGF+HA | 5 (PRGF) and 5 (HA) | yes (HA) then PRGF added |
| +PRGF+CW | 5 (PRGF) and 5 (CW) | yes (CW) then PRGF added |
| +PRGF+HA+CW | 3.33 (PRGF), 3.33 (HA), and 3.33 (CW) | yes (HA+CW) then PRGF added |
3.2. Mass Loss
3.3. Protein Release
3.4. Cell Attachment, Migration and Matrix Production
3.4.1. Scanning Electron Microscopy
3.4.2. DAPI Staining
3.4.3. Alizarin Red S Staining
3.5. Uniaxial Compression Testing
3.6. Statistical Analysis
4. Conclusions
References
- Greenbaum, M.A.; Kanat, I.O. Current concepts in bone healing. Review of the literature. J. Am. Podiatr. Med. Assoc. 1993, 83, 123–129. [Google Scholar]
- Logeart-Avramoglou, D.; Anagnostou, F.; Bizios, R.; Petite, H. Engineering bone: Challenges and obstacles. J. Cell. Mol. Med. 2005, 9, 72–84. [Google Scholar] [CrossRef]
- Abarrategi, A.; Moreno-Vicente, C.; Martinez-Vazquez, F.J.; Civantos, A.; Ramos, V.; Sanz-Casado, J.V.; Martinez-Corria, R.; Perera, F.H.; Mulero, F.; Miranda, P.; et al. Biological properties of solid free form designed ceramic scaffolds with BMP-2: in vitro and in vivo evaluation. PLoS One 2012, 7, e34117. [Google Scholar] [CrossRef]
- Elshahat, A. Correction of craniofacial skeleton contour defects using bioactive glas particles. J. Plast. Reconstr. Surg. 2006, 30, 113–119. [Google Scholar]
- Ni, Z.L.; Liu, H.S.; Qu, Q.Y.; Lu, H.L.; Yan, B.; Zhang, Q.H. Using of titanium mesh for the reconstruction of skull base defect. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 2006, 41, 351–354. [Google Scholar]
- Moioli, E.K.; Clark, P.A.; Xin, X.; Lal, S.; Mao, J.J. Matrices and scaffolds for drug delivery in dental, oral and craniofacial tissue engineering. Adv. Drug Deliv. Rev. 2007, 59, 308–324. [Google Scholar] [CrossRef]
- Ciardelli, G.; Gentile, P.; Chiono, V.; Mattioli-Belmonte, M.; Vozzi, G.; Barbani, N.; Giusti, P. Enzymatically crosslinked porous composite matrices for bone tissue regeneration. J. Biomed. Mater. Res. A 2010, 92, 137–151. [Google Scholar]
- Kane, R.J.; Roeder, R.K. Effects of hydroxyapatite reinforcement on the architecture and mechanical properties of freeze-dried collagen scaffolds. J. Mech. Behav. Biomed. Mater. 2012, 7, 41–49. [Google Scholar] [CrossRef]
- Prosecka, E.; Rampichova, M.; Vojtova, L.; Tvrdik, D.; Melcakova, S.; Juhasova, J.; Plencner, M.; Jakubova, R.; Jancar, J.; Necas, A.; et al. Optimized conditions for mesenchymal stem cells to differentiate into osteoblasts on a collagen/hydroxyapatite matrix. J. Biomed. Mater. Res. A 2011, 99, 307–315. [Google Scholar]
- Wang, G.; Babadagli, M.E.; Uludag, H. Bisphosphonate-derivatized liposomes to control drug release from collagen/hydroxyapatite scaffolds. Mol. Pharm. 2011, 8, 1025–1034. [Google Scholar] [CrossRef]
- Gleeson, J.P.; Plunkett, N.A.; O'Brien, F.J. Addition of hydroxyapatite improves stiffness, Interconnectivity and osteogenic potential of a highly porous collagen-based scaffold for bone tissue regeneration. Eur. Cells Mater. 2010, 20, 218–230. [Google Scholar]
- Neffe, A.T.; Loebus, A.; Zaupa, A.; Stoetzel, C.; Muller, F.A.; Lendlein, A. Gelatin functionalization with tyrosine derived moieties to increase the interaction with hydroxyapatite fillers. Acta Biomater. 2011, 7, 1693–1701. [Google Scholar] [CrossRef]
- Azami, M.; Samadikuchaksaraei, A.; Poursamar, S.A. Synthesis and characterization of a laminated hydroxyapatite/gelatin nanocomposite scaffold with controlled pore structure for bone tissue engineering. Int. J. Artif. Organs 2010, 33, 86–95. [Google Scholar]
- Kim, H.W.; Knowles, J.C.; Kim, H.E. Porous scaffolds of gelatin-hydroxyapatite nanocomposites obtained by biomimetic approach: Characterization and antibiotic drug release. J. Biomed. Mater. Res. B 2005, 74, 686–698. [Google Scholar]
- Kim, H.W.; Kim, H.E.; Salih, V. Stimulation of osteoblast responses to biomimetic nanocomposites of gelatin-hydroxyapatite for tissue engineering scaffolds. Biomaterials 2005, 26, 5221–5230. [Google Scholar] [CrossRef]
- Woo, K.M.; Seo, J.; Zhang, R.; Ma, P.X. Suppression of apoptosis by enhanced protein adsorption on polymer/hydroxyapatite composite scaffolds. Biomaterials 2007, 28, 2622–2630. [Google Scholar] [CrossRef]
- Rohanizadeh, R.; Swain, M.V.; Mason, R.S. Gelatin sponges (Gelfoam) as a scaffold for osteoblasts. J. Mater. Sci. Mater. Med. 2008, 19, 1173–1182. [Google Scholar] [CrossRef]
- Barbani, N.; Guerra, G.D.; Cristallini, C.; Urciuoli, P.; Avvisati, R.; Sala, A.; Rosellini, E. Hydroxyapatite/gelatin/gellan sponges as nanocomposite scaffolds for bone reconstruction. J. Mater. Sci. Mater. Med. 2012, 23, 51–61. [Google Scholar] [CrossRef]
- Kohara, H.; Tabata, Y. Enhancement of ectopic osteoid formation following the dual release of bone morphogenetic protein 2 and Wnt1 inducible signaling pathway protein 1 from gelatin sponges. Biomaterials 2011, 32, 5726–5732. [Google Scholar] [CrossRef] [Green Version]
- Zeng, J.B.; He, Y.S.; Li, S.L.; Wang, Y.Z. Chitin whiskers: An overview. Biomacromolecules 2012, 13, 1–11. [Google Scholar] [CrossRef]
- Sriupayo, J.; Supaphol, P.; Blackwell, J.; Rujiravanit, R. Preparation and characterization of α-chitin whisker-reinforced poly(vinyl alcohol) nanocomposite films with or without heat treatment. Polymer 2005, 46, 5637–5644. [Google Scholar] [CrossRef]
- Ji, Y.-L.; Wolfe, P.S.; Rodriguez, I.A.; Bowlin, G.L. Preparation of chitin nanofibril/polycaprolactone nanocomposite from a nonaqueous medium suspension. Carbohydr. Polym. 2012, 87, 2313–2319. [Google Scholar] [CrossRef]
- Junkasem, J.; Rujiravanit, R.; Supaphol, P. Fabrication of α-chitin whisker-reinforced poly(vinyl alcohol) nanocomposite nanofibres by electrospinning. Nanotechnology 2006, 17, 4519–4528. [Google Scholar] [CrossRef]
- Wongpanit, P.; Sanchavanakit, N.; Pavasant, P.; Bunaprasert, T.; Tabata, Y.; Rujiravanit, R. Preparation and characterization of chitin whisker-reinforced silk fibroin nanocomposite sponges. Eur. Polym. J. 2007, 43, 4123–4135. [Google Scholar] [CrossRef]
- Hariraksapitak, P.; Supaphol, P. Preparation and Properties of a-Chitin-Whisker-Reinforced Hyaluronan–Gelatin Nanocomposite Scaffolds. J. Appl. Polym. Sci. 2010, 117, 3406–3418. [Google Scholar]
- Han, T.; Wang, H.; Zhang, Y.Q. Combining platelet-rich plasma and tissue-engineered skin in the treatment of large skin wound. J. Craniofac. Surg. 2012, 23, 439–447. [Google Scholar] [CrossRef]
- Kushida, S.; Kakudo, N.; Suzuki, K.; Kusumoto, K. Effects of Platelet-Rich Plasma on Proliferation and Myofibroblastic Differentiation in Human Dermal Fibroblasts. Ann. Plast. Surg. 2012, Oct 3, 23038148. [Google Scholar]
- Taylor, D.W.; Petrera, M.; Hendry, M.; Theodoropoulos, J.S. A systematic review of the use of platelet-rich plasma in sports medicine as a new treatment for tendon and ligament injuries. Clin. J. Sport Med. 2011, 21, 344–352. [Google Scholar] [CrossRef]
- Xie, X.; Wang, Y.; Zhao, C.; Guo, S.; Liu, S.; Jia, W.; Tuan, R.S.; Zhang, C. Comparative evaluation of MSCs from bone marrow and adipose tissue seeded in PRP-derived scaffold for cartilage regeneration. Biomaterials 2012, 33, 7008–7018. [Google Scholar] [CrossRef]
- Lopez-Lopez, J.; Chimenos-Kustner, E.; Manzanares-Cespedes, C.; Munoz-Sanchez, J.; Castaneda-Vega, P.; Jane-Salas, E.; Alvarez-Lopez, J.M.; Gimeno-Sanding, A. Histomorphological study of the bone regeneration capacity of platelet-rich plasma, bone marrow and tricalcium phosphate: Experimental study on pigs. Med. Oral Patol. Oral 2009, 14, e620–e627. [Google Scholar]
- Gentile, P.; Di Pasquali, C.; Bocchini, I.; Floris, M.; Eleonora, T.; Fiaschetti, V.; Floris, R.; Cervelli, V. Breast Reconstruction With Autologous Fat Graft Mixed With Platelet-Rich Plasma. Surg. Innov. 2012, Sep 10, 22964262. [Google Scholar]
- Jo, C.H.; Kim, J.E.; Yoon, K.S.; Shin, S. Platelet-rich plasma stimulates cell proliferation and enhances matrix gene expression and synthesis in tenocytes from human rotator cuff tendons with degenerative tears. Am. J. Sport. Med. 2012, 40, 1035–1045. [Google Scholar] [CrossRef]
- Messora, M.R.; Nagata, M.J.; Fucini, S.E.; Pola, N.M.; Campos, N.; Oliveira, G.C.; Bosco, A.F.; Garcia, V.G.; Furlaneto, F.A. Effect of platelet-rich plasma on the healing of mandibular defects treated with bone allograft. A radiographic study in dogs. J. Oral Implantol. 2012, Sep 26, 23013307. [Google Scholar]
- Li, N.Y.; Yuan, R.T.; Chen, T.; Chen, L.Q.; Jin, X.M. Effect of platelet-rich plasma and latissimus dorsi muscle flap on osteogenesis and vascularization of tissue-engineered bone in dogs. J. Oral Maxillofac. Surg. 2009, 67, 1850–1858. [Google Scholar] [CrossRef]
- Rai, B.; Oest, M.E.; Dupont, K.M.; Ho, K.H.; Teoh, S.H.; Guldberg, R.E. Combination of platelet-rich plasma with polycaprolactone-tricalcium phosphate scaffolds for segmental bone defect repair. J. Biomed. Mater. Res. A 2007, 81, 888–899. [Google Scholar]
- Huang, S.; Jia, S.; Liu, G.; Fang, D.; Zhang, D. Osteogenic differentiation of muscle satellite cells induced by platelet-rich plasma encapsulated in three-dimensional alginate scaffold. Oral. Surg. Oral Med. O. 2012, Jan 25, 22285139. [Google Scholar]
- Sell, S.A.; Wolfe, P.S.; Ericksen, J.J.; Simpson, D.G.; Bowlin, G.L. Incorporating platelet-rich plasma into electrospun scaffolds for tissue engineering applications. Tissue Eng. Part A 2011, 17, 2723–2737. [Google Scholar] [CrossRef]
- Wolfe, P.S.; Sell, S.A.; Ericksen, J.J.; Simpson, D.G.; Bowlin, G.L. The creation of electrospun nanofibers from platelet rich plasma. J. Tissue Sci. Eng. 2011, 2, 1000107. [Google Scholar]
- Sell, S.A.; Wolfe, P.S.; Spence, A.J.; Rodriguez, I.A.; McCool, J.M.; Petrella, R.L.; Garg, K.; Ericksen, J.J.; Bowlin, G.L. A Preliminary Study on the Potential of Manuka Honey and Platelet-Rich Plasma in Wound Healing. Int. J. Biomater. 2012, 2012, 313781. [Google Scholar]
- Anitua, E.; Orive, G.; Andia, I. Use of PRGF to Accelerate Bone and Soft Tissue Regeneration in Postextraction Sites. Available online: http://www.dentalxp.com/vendors/1/PRGF%20to%20Accelerate%20Bone%20and%20Soft%20Tissue%20Regeneration.pdf (accessed on 12 March 2013).
- Lu, H.H.; Vo, J.M.; Chin, H.S.; Lin, J.; Cozin, M.; Tsay, R.; Eisig, S.; Landesberg, R. Controlled delivery of platelet-rich plasma-derived growth factors for bone formation. J. Biomed. Mater. Res. A 2008, 86, 1128–1136. [Google Scholar]
- Arvidson, K.; Abdallah, B.M.; Applegate, L.A.; Baldini, N.; Cenni, E.; Gomez-Barrena, E.; Granchi, D.; Kassem, M.; Konttinen, Y.T.; Mustafa, K.; et al. Bone regeneration and stem cells. J. Cell. Mol. Med. 2011, 15, 718–746. [Google Scholar] [CrossRef]
- Anitua, E.; Orive, G.; Andia, I. The Therapeutic Potential of PRGF in Dentistry and Oral Implantology. In Implant and Regenerative Therapy in Dentistry: A Guide to Decision Making; John Wiley & Sons: Hoboken, NJ, USA, 2009; pp. 113–121. [Google Scholar]
- Sell, S.; Barnes, C.; Smith, M.; McClure, M.; Madurantakam, P.; Grant, J.; McManus, M.; Bowlin, G. Extracellular matrix regenerated: Tissue engineering via electrospun biomimetic nanofibers. Polym. Int. 2007, 56, 1349–1360. [Google Scholar] [CrossRef]
- Barnes, C.P.; Pemble, C.W.; Brand, D.D.; Simpson, D.G.; Bowlin, G.L. Cross-linking electrospun type II collagen tissue engineering scaffolds with carbodiimide in ethanol. Tissue Eng. 2007, 13, 1593–1605. [Google Scholar] [CrossRef]
- Sell, S.A.; Francis, M.P.; Garg, K.; McClure, M.J.; Simpson, D.G.; Bowlin, G.L. Cross-linking methods of electrospun fibrinogen scaffolds for tissue engineering applications. Biomed. Mater. 2008, 3, 045001. [Google Scholar] [CrossRef]
- Olde Damink, L.H.; Dijkstra, P.J.; van Luyn, M.J.; van Wachem, P.B.; Nieuwenhuis, P.; Feijen, J. Cross-linking of dermal sheep collagen using a water-soluble carbodiimide. Biomaterials 1996, 17, 765–773. [Google Scholar] [CrossRef]
- McClure, M.; Sell, S.; Barnes, C.; Bowen, W.; Bowlin, G. Cross-linking electrospun polydioxanone-soluble elastin blends: Material characterization. J. Eng. Fiber. Fabr. 2008, 3, 1–10. [Google Scholar]
- Rodriguez, I.A.; Madurantakam, P.A.; McCool, J.M.; Sell, S.A.; Yang, H.; Moon, P.C.; Bowlin, G.L. Mineralization potential of electrospun PDO-hydroxyapatite-fibrinogen blended scaffolds. Int. J. Biomater 2012, 2012, 159484. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rodriguez, I.A.; Sell, S.A.; McCool, J.M.; Saxena, G.; Spence, A.J.; Bowlin, G.L. A Preliminary Evaluation of Lyophilized Gelatin Sponges, Enhanced with Platelet-Rich Plasma, Hydroxyapatite and Chitin Whiskers for Bone Regeneration. Cells 2013, 2, 244-265. https://doi.org/10.3390/cells2020244
Rodriguez IA, Sell SA, McCool JM, Saxena G, Spence AJ, Bowlin GL. A Preliminary Evaluation of Lyophilized Gelatin Sponges, Enhanced with Platelet-Rich Plasma, Hydroxyapatite and Chitin Whiskers for Bone Regeneration. Cells. 2013; 2(2):244-265. https://doi.org/10.3390/cells2020244
Chicago/Turabian StyleRodriguez, Isaac A., Scott A. Sell, Jennifer M. McCool, Gunjan Saxena, Andrew J. Spence, and Gary L. Bowlin. 2013. "A Preliminary Evaluation of Lyophilized Gelatin Sponges, Enhanced with Platelet-Rich Plasma, Hydroxyapatite and Chitin Whiskers for Bone Regeneration" Cells 2, no. 2: 244-265. https://doi.org/10.3390/cells2020244
APA StyleRodriguez, I. A., Sell, S. A., McCool, J. M., Saxena, G., Spence, A. J., & Bowlin, G. L. (2013). A Preliminary Evaluation of Lyophilized Gelatin Sponges, Enhanced with Platelet-Rich Plasma, Hydroxyapatite and Chitin Whiskers for Bone Regeneration. Cells, 2(2), 244-265. https://doi.org/10.3390/cells2020244