Micronutrient Synergy in the Fight against Hepatocellular Carcinoma
Abstract
:1. Introduction
2. Results and Discussion
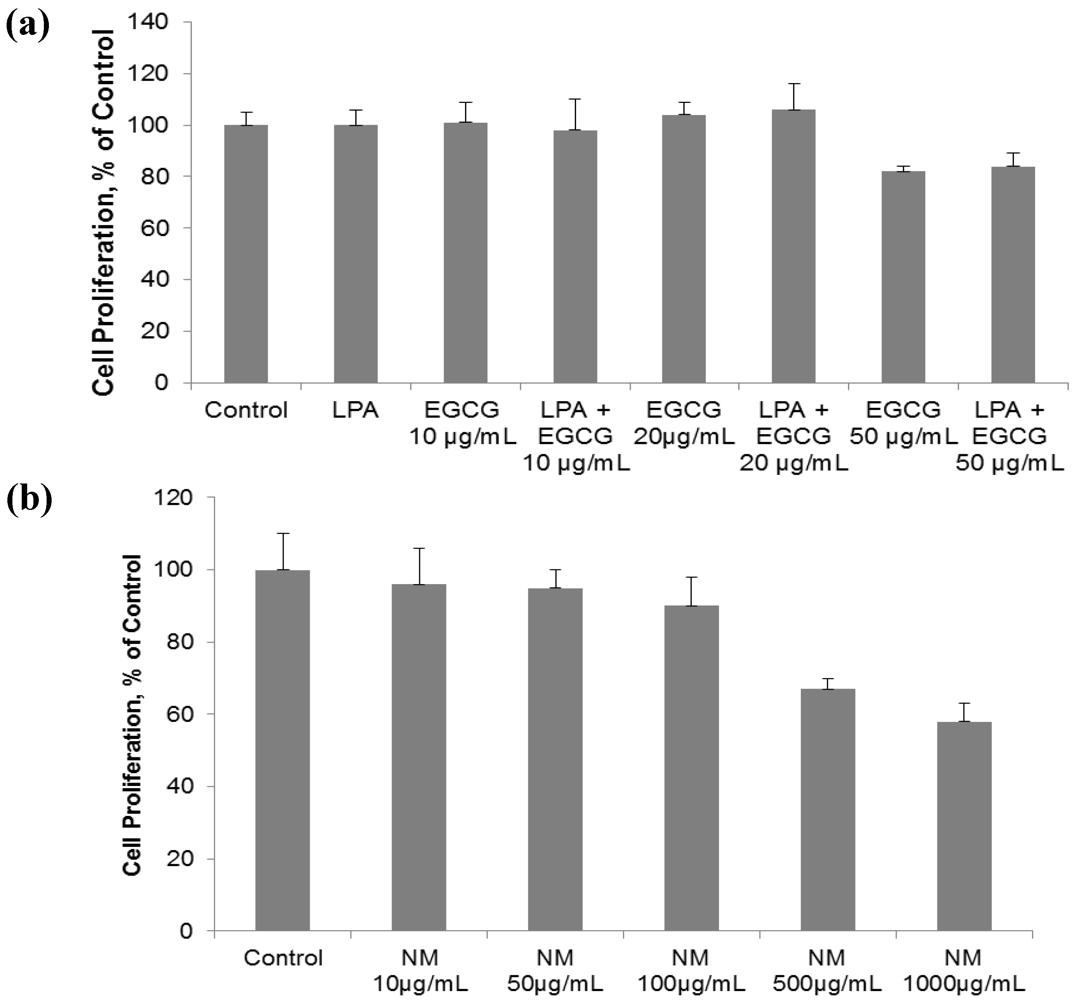
2.1. Comparative Effect of LPA, EGCG and NM on HepG2 Cell Proliferation
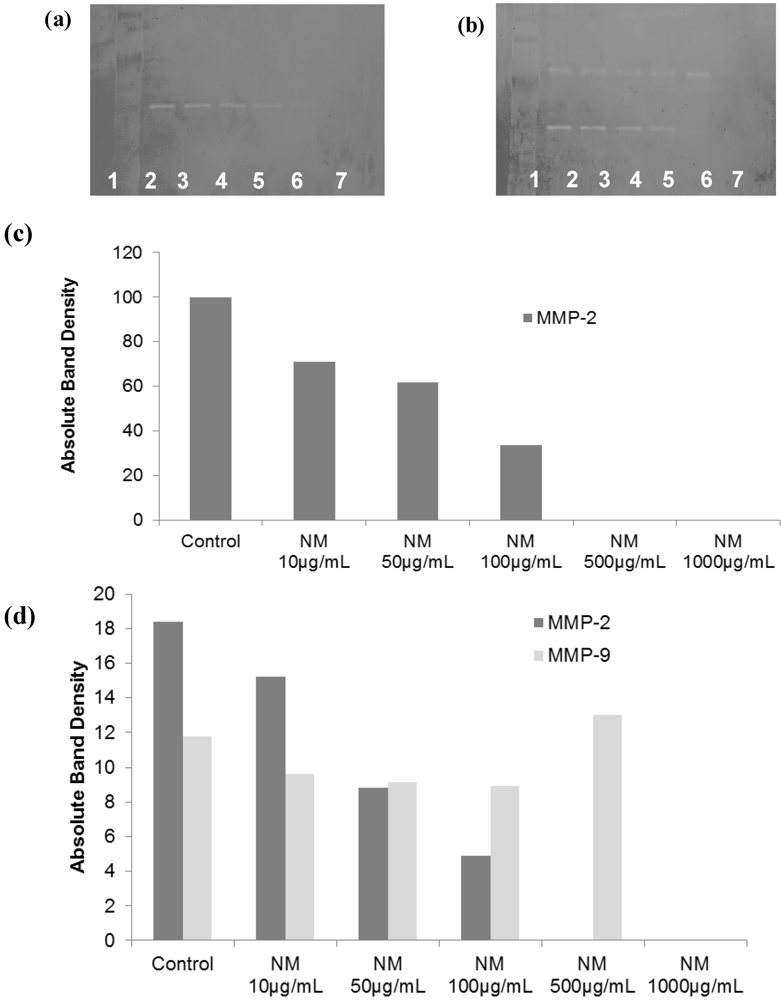
2.2. Comparative Effect of LPA, EGCG and NM on HepG2 MMP Secretion

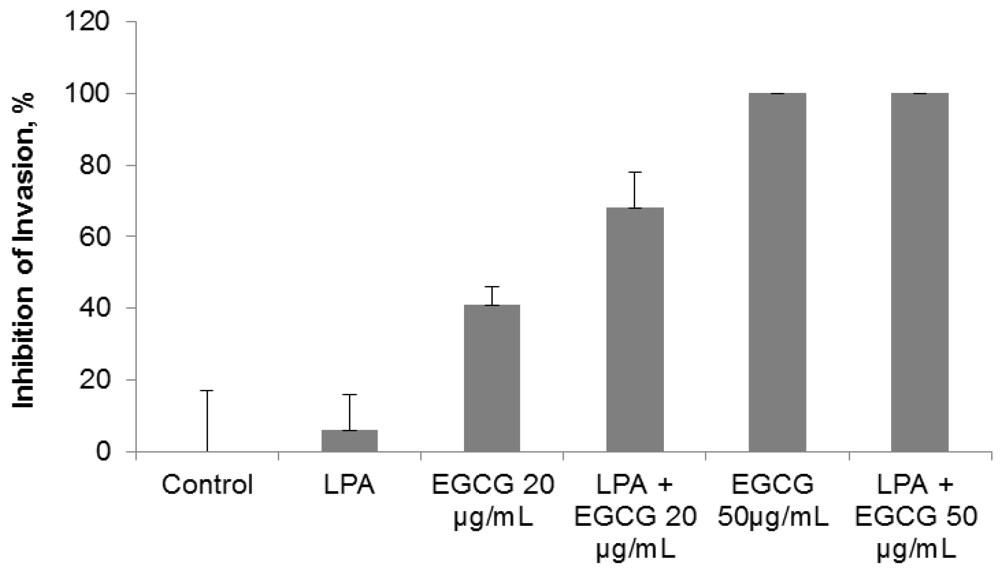
2.3. Comparative Effect of EGCG, LPA and NM on HepG2 Matrigel Invasion
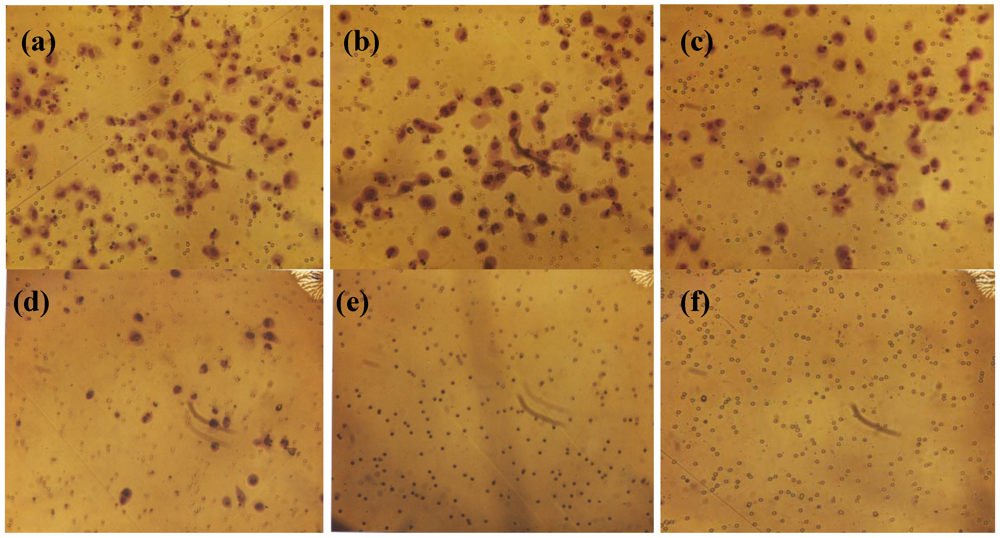


2.4. Effect of NM on HepG2 Apoptosis
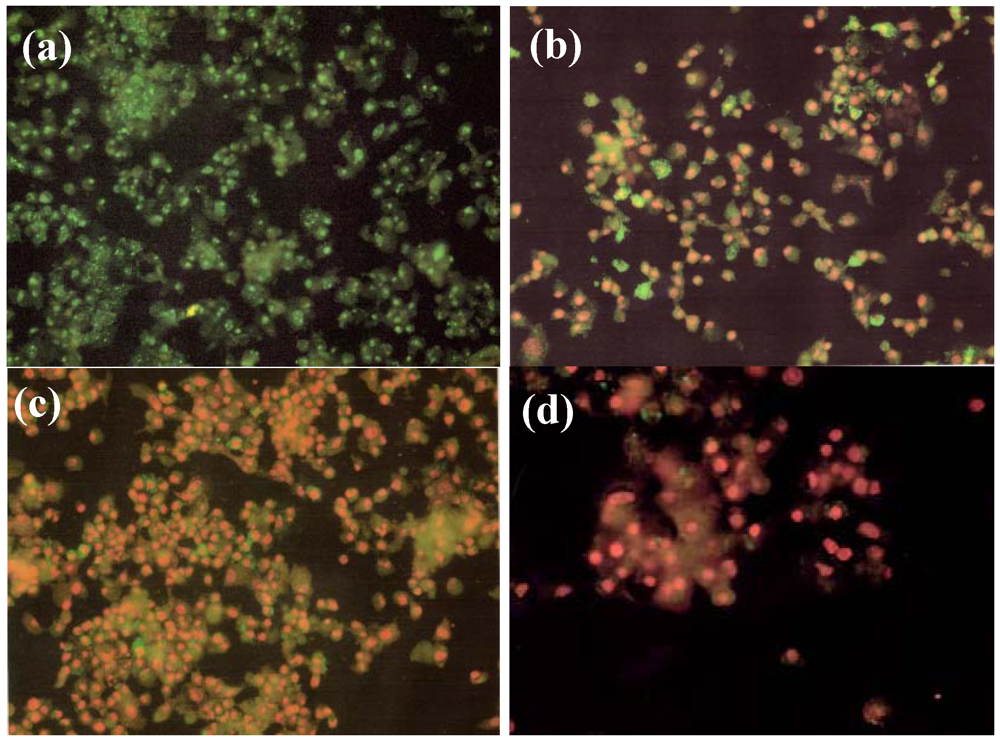
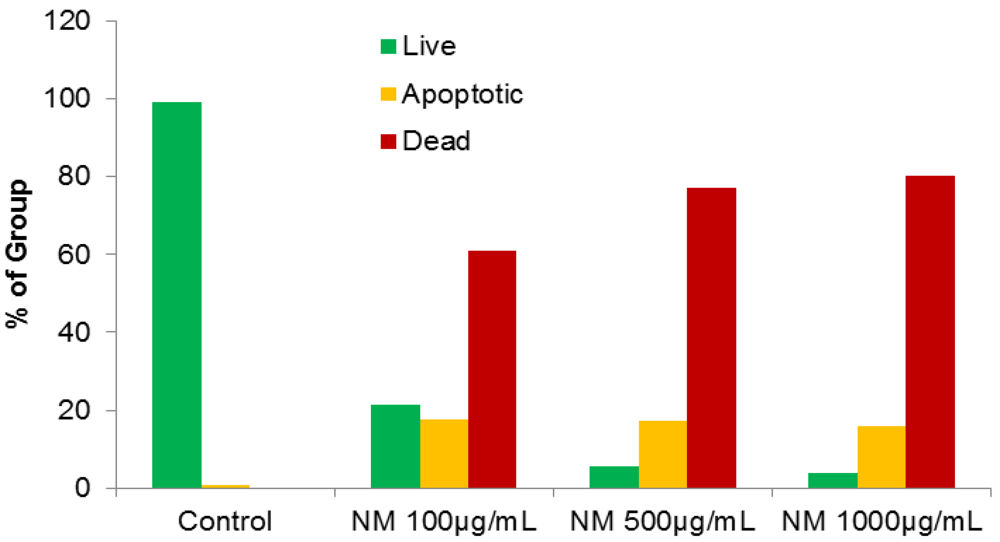
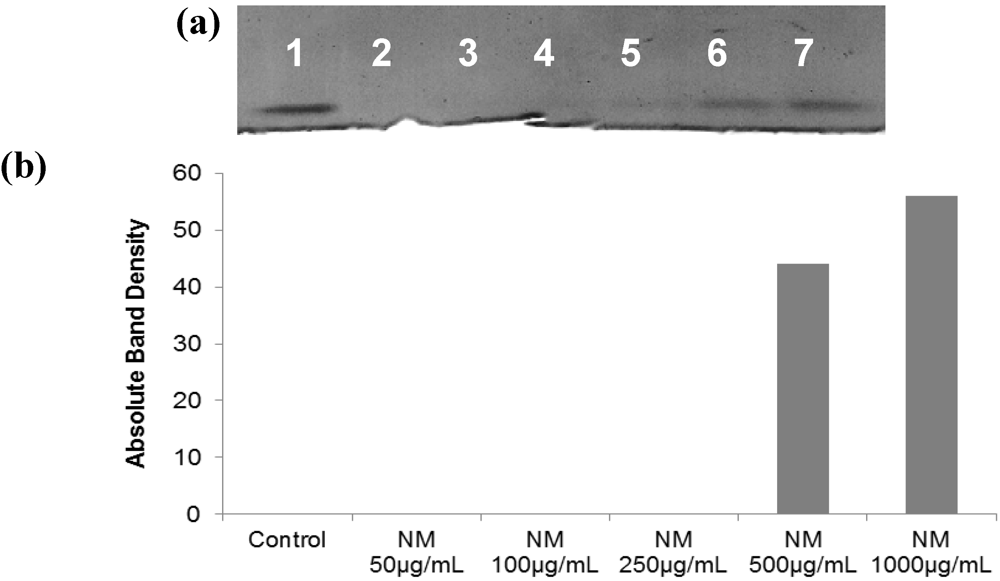
2.5. Effect of NM on u-PA, MMPs and TIMPs Activities in SK-Hep-1 Cells
2.5.1. Effect of NM on u-PA activity in SK-Hep-1 Cells

2.5.2. Effect of NM on SK-Hep-1 Secretion of MMPs
2.5.3. Effect of NM on TIMPs Activity in SK-Hep-1 Cells

3. Experimental Section
3.1. Cancer Cell Lines and Reagents
3.2. Composition of the Nutrient Mixture
3.3. Cell Culture
3.4. MTT Assay
3.5. Fibrin Zymography
3.6. Gelatinase Zymography
3.7. Reverse Zymography
3.8. Scanning of Gelatinase and Fibrin Zymograms
3.9. Matrigel Invasion
3.10. Apoptosis
3.11. Statistical Analysis
4. Conclusions
Acknowledgements
References
- National Cancer Institute. A snapshot of liver and bile duct cancers. Available online: http://www.cancer.gov/aboutnci/servingpeople/snapshots/liver.pdf (accessed on 4 November 2011).
- American Cancer Society. Global cancer facts and figures: 2nd edition. Available online: http://www.cancer.org/acs/groups/content/@epidemiologysurveilance/documents/document/acspc-027766.pdf (accessed on 4 November 2011).
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M.J. Cancer statistics, 2009. CA Cancer J. Clin. 2009, 59, 225–249. [Google Scholar]
- Fidler, I.J. Molecular biology of cancer: Invasion and metastasis. In Cancer: Principles and Practice of Oncology, 5th; de Vita, V.T., Hellman, S., Rosenberg, S.A., Eds.; Lippincott-Raven: Philadelphia, PA, USA, 1997; pp. 135–152. [Google Scholar]
- Egeblad, M.; Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2002, 2, 161–174. [Google Scholar]
- Folkman, J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002, 29, 15–18. [Google Scholar]
- Chambers, A.F.; Matrisian, L.M. Changing views on the role of matrix metalloproteinases in metastasis. J. Natl. Cancer Inst. 1997, 89, 1260–1270. [Google Scholar]
- Kleiner, D.L.; Stetler-Stevenson, W.G. Matrix metalloproteinases and metastasis. Cancer Chemother. Pharmacol. 1999, 43, 42S–51S. [Google Scholar]
- Korn, W.M. Moving toward an understanding of the metastatic process in hepatocellular carcinoma. World J. Gastroenterol. 2001, 7, 777–778. [Google Scholar]
- Sakamoto, Y.; Mafune, K.; Mori, M.; Shiraishi, T.; Imamura, H.; Mori, M.; Takayama, T.; Makuuchi, M. Overexpression of MMP-9 correlates with growth of small hepatocellular carcinoma. Int. J. Oncol. 2000, 17, 237–243. [Google Scholar]
- Jiang, Y.F.; Yang, Z.H.; Hu, J.Q. Recurrence or metastasis of HCC: Predictors, early detection and experimental antiangiogenic therapy. World J. Gastroenterol. 2000, 6, 61–65. [Google Scholar]
- Määta, M.; Soini, Y.; Liakka, A.; Autio-Harmainen, H. Differntial expression of matrix metalloproteinase (MMP)-2 and MMP-9 and membrane type 1-MMP in hepatocellular and pancreatic adenocarcinoma: Implications for tumor progression and clinical prognosis. Clin. Cancer Res. 2000, 6, 2726–2734. [Google Scholar]
- Kashiwagi, A. Matrix metalloproteases in hepatocellular carcinoma. A study in resected specimens from 25 patients. Tokyo Jikekai Med. J. 2001, 118, 33–48. [Google Scholar]
- Qin, L.X.; Tang, Z.Y. The prognostic molecular markers in hepatocellular carcinoma. World J. Gastroenterol. 2002, 8, 385–392. [Google Scholar]
- Guo, R.P.; Zhong, C.; Shi, M.; Zhang, C.Q.; Wei, W.; Zhang, Y.Q.; Li, J.Q. Clinical value of apoptosis and angiogenesis factors in estimating the prognosis of hepatocellular carcinoma. J. Cancer Res. Clin. Oncol. 2006, 132, 547–555. [Google Scholar]
- Rath, M.; Pauling, L. Plasmin-induced proteolysis and the role of apoprotein(a), lysine and synthetic analogs. Orthomolecular Med. 1992, 7, 17–23. [Google Scholar]
- Niedzwiecki, A.; Roomi, M.W.; Kalinovsky, T.; Rath, M. Micronutrient synergy—A new tool in effective control of metastasis and other key mechanisms of cancer. Cancer Metastasis Rev. 2010, 29, 529–553. [Google Scholar]
- Roomi, M.W.; Roomi, N.; Ivanov, V.; Kalinovsky, T.; Niedzwiecki, A.; Rath, M. Inhibitory effect of a mixture containing ascorbic acid, lysine, proline and green tea extract on critical parameters in angiogenesis. Oncol. Rep. 2005, 14, 807–815. [Google Scholar]
- Ivanov, V.; Ivanova, S.; Roomi, M.W.; Kalinovsky, T.; Niedzwiecki, A.; Rath, M. Naturally-produced extracellular matrix inhibits growth rate and invasiveness of human osteosarcoma cancer cells. Med. Oncol. 2007, 24, 209–217. [Google Scholar]
- Sun, Z.; Chen, Y.H.; Wang, P.; Zhang, J.; Gurewich, V.; Zhang, P.; Liu, J.N. The blockage of high-affinity lysine binding sites of plasminogen by EACA significantly inhibits prourokinase-induced plasminogen activation. Biochem. Biophys. Acta 2002, 1596, 182–192. [Google Scholar]
- Valcic, S.; Timmermann, B.N.; Alberts, D.S.; Wachter, G.A.; Krutzsch, M.; Wymer, J.; Guillen, J.M. Inhibitory effect of six green tea catechins and caffeine on the growth of four selected human tumor cell lines. Anticancer Drugs 1996, 7, 461–468. [Google Scholar]
- Mukhtar, H.; Ahmed, N. Tea polyphenols: Prevention of cancer and optimizing health. Am. J. Clin. Nutr. 2000, 71, 1698S–1702S. [Google Scholar]
- Yang, G.Y.; Liao, J.; Kim, K.; Yurtow, E.J.; Yang, C.S. Inhibition of growth and induction of apoptosis in human cancer cell lines by tea polyphenols. Carcinogenesis 1998, 19, 611–616. [Google Scholar]
- Taniguchi, S.; Fujiki, H.; Kobayashi, H.; Go, H.; Miyado, K.; Sadano, H.; Shimikawa, R. Effect of (-)epigallocatechin gallate, the main constituent of green tea, on lung metastasis with mouse B16 melanoma cell lines. Cancer Lett. 1992, 65, 51–54. [Google Scholar] [CrossRef]
- Hara, Y. Green Tea: Health Benefits and Applications; Marcel Dekker: New York, NY, USA, 2001. [Google Scholar]
- Kawakami, S.; Kageyama, Y.; Fujii, Y.; Kihara, K.; Oshima, H. Inhibitory effects of N-acetylcysteine on invasion and MMP 9 production of T24 human bladder cancer cells. Anticancer Res. 2001, 21, 213–219. [Google Scholar]
- Morini, M.; Cai, T.; Aluigi, M.G.; Noonan, D.M.; Masiello, L.; de Floro, S.; D'Agostinin, F.; Albini, A.; Fassima, G. The role of the thiol N-acetylcysteine in the prevention of tumor invasion and angiogenesis. Int. J. Biol. Markers 1999, 14, 268–271. [Google Scholar]
- Yoon, S.O.; Kim, M.M.; Chung, A.S. Inhibitory effects of selenite on invasion of HT 1080 tumor cells. J. Biol. Chem. 2001, 276, 20085–20092. [Google Scholar]
- Maramag, C.; Menon, M.; Balaji, K.C.; Reddy, P.G.; Laxmanan, S. Effect of vitamin C on prostate cancer cells in vitro: Effect on cell number, viability and DNA synthesis. Prostate 1997, 32, 188–195. [Google Scholar] [CrossRef]
- Cooke, J.P.; Dzau, V.J. Nitric oxide synthase: Role in the genesis of vascular disease. Annu. Rev. Med. 1997, 48, 489–509. [Google Scholar]
- Roomi, M.W.; Kalinovsky, T.; Roomi, N.W.; Monterrey, J.; Rath, M.; Niedzwiecki, A. A nutrient mixture suppresses hepatic metastasis in athymic nude mice injected with murine B16FO melanoma cells. BioFactors 2009, 33, 181–189. [Google Scholar]
- Roomi, M.W.; Roomi, N.W.; Kalinovsky, T.; Niedzwiecki, A.; Rath, M. In vivo and in vitro effect of a nutrient mixture on human hepatocarcinoma cell line SK-Hep-1. Exp. Oncol. 2010, 32, 84–91. [Google Scholar]
- Nelson, A.R.; Fingleton, B.; Rothenberg, M.L.; Matrisian, L.M. Matrix metalloproteinases: Biologic activity and clinical implications. J. Clin. Oncol. 2000, 18, 1135–1149. [Google Scholar]
- Zheng, Q.; Tang, Z.Y.; Xue, Q.; Shi, D.R.; Song, H.Y.; Tang, H.B. Invasion and metastasis of hepatocellular carcinoma in relation to urokinase-type plasminogen activator, its receptor and inhibitor. J. Cancer Res. Clin. Oncol. 2000, 126, 641–646. [Google Scholar]
- Yurchenko, P.D.; Schitny, J.C. Molecular architecture of basement membranes. FASEB J. 1990, 4, 1577–1590. [Google Scholar]
- Barsky, S.H.; Siegel, G.P.; Jannotta, F.; Liotta, L.A. Loss of basement membrane components by invasive tumors but not by their benign counterparts. Lab. Invest. 1983, 49, 140–147. [Google Scholar]
- Liotta, L.A.; Tryggvason, K.; Garbisa, A.; Hart, I.; Foltz, C.M.; Shafie, S. Metastatic potential correlates with enzymatic degradation of basement membrane collagen. Nature 1980, 284, 67–68. [Google Scholar]
- Stetler-Stevenson, W.G. The role of matrix metalloproteinases in tumor invasion, metastasis and angiogenesis. Surg. Oncol. Clin. N. Am. 2001, 10, 383–392. [Google Scholar]
- Stetler-Stevenson, W.G. Type IV collagenases in tumor invasion and metastasis. Cancer Metastasis Rev. 1990, 9, 289–303. [Google Scholar]
- Dano, K.; Andreasen, P.A.; Grondahl-Hansen, J.; Kristensen, P.; Nielsen, L.S.; Skriver, L. Plasminogen activators, tissue degradation and cancer. Adv. Cancer Res. 1985, 44, 139–266. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Roomi, M.W.; Roomi, N.W.; Kalinovsky, T.; Niedzwiecki, A.; Rath, M. Micronutrient Synergy in the Fight against Hepatocellular Carcinoma. Cancers 2012, 4, 323-339. https://doi.org/10.3390/cancers4020323
Roomi MW, Roomi NW, Kalinovsky T, Niedzwiecki A, Rath M. Micronutrient Synergy in the Fight against Hepatocellular Carcinoma. Cancers. 2012; 4(2):323-339. https://doi.org/10.3390/cancers4020323
Chicago/Turabian StyleRoomi, M. Waheed, Nusrath W. Roomi, Tatiana Kalinovsky, Aleksandra Niedzwiecki, and Matthias Rath. 2012. "Micronutrient Synergy in the Fight against Hepatocellular Carcinoma" Cancers 4, no. 2: 323-339. https://doi.org/10.3390/cancers4020323



