Biomarkers for Early Detection of Malignant Mesothelioma: Diagnostic and Therapeutic Application
Abstract
:1. Introduction
2. Biomarkers

2.1. Biomarkers of Exposure
DNA Adducts
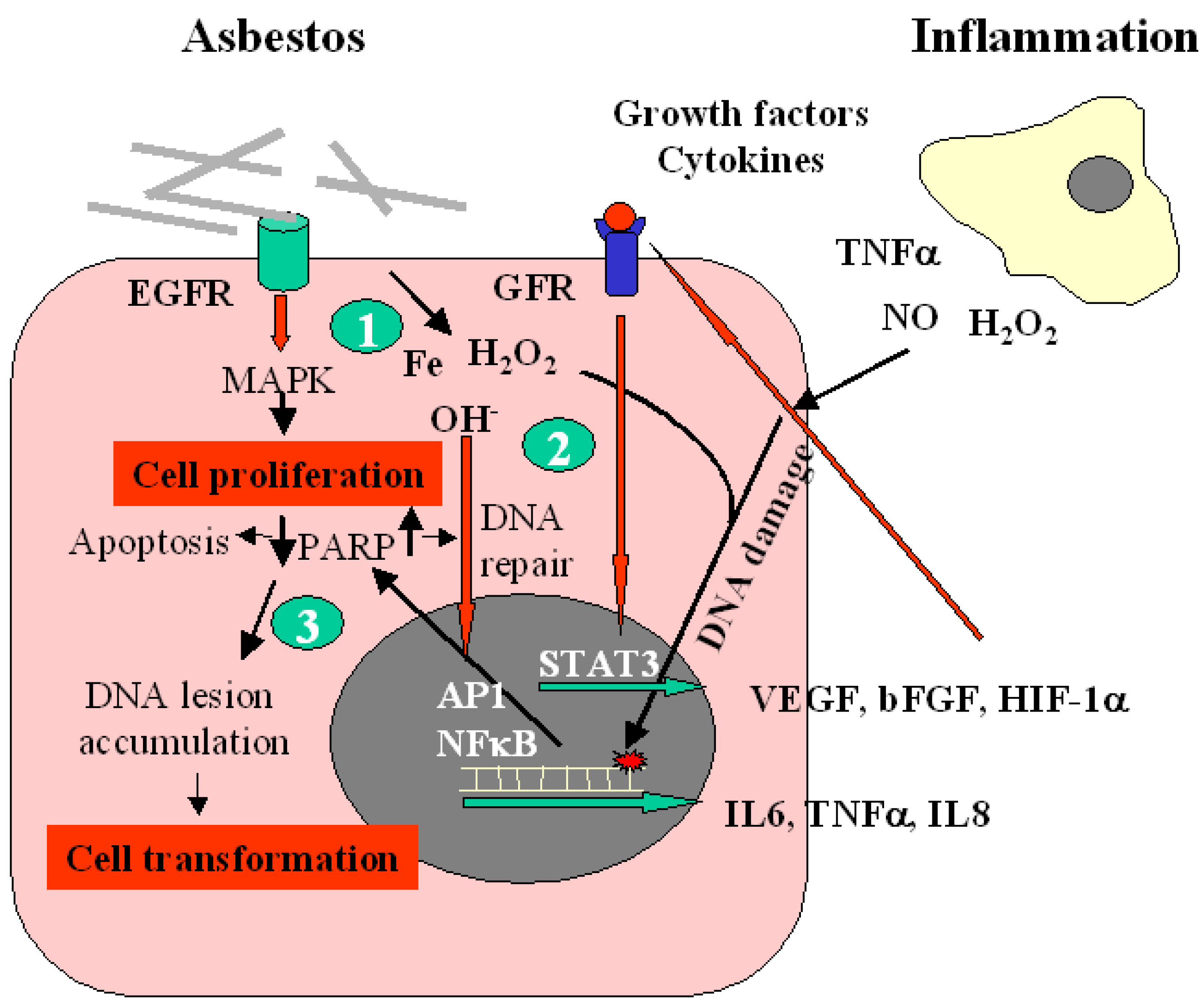
2.2. Biomarkers of Early Effect
2.2.1. DNA Repair Ability
2.2.2. Cytogenetic Assay
2.3. Biomarker for Diagnosis
2.3.1. Conventional Biomarkers
2.3.2. Osteopontin
| Studies | N° | Sample | Biomarker cut-off (ng/mL) | Sensitivity % | Specificity % | AUC |
|---|---|---|---|---|---|---|
| OPN | ||||||
| Pass et al. [45] | 193 | serum | 43.3 | 77.6 | 85.5 | 0.89 (0.83–0.93) |
| Paleari et al. [55] | 94 | plasma | 60.8 | 40.0 | 100.0 | 0.60 (0.47–0.72) |
| Grigoriu et al. [52] | 208 | serum | 68.0 | 95.0 | 50.0 | 0.74 (0.68–0.79) |
| Creany et al. [74] | 107 | serum | 18.0 | 47.0 | 95.0 | 0.76 (0.67–0.85) |
| SMRPs | ||||||
| Grigoriu et al. [52] | 208 | serum | 1.7 | 40.0 | 100.0 | 0.74 (0.68–0.80) |
| Scherpereel et al. [61] | 137 | serum | 1.1 | 71.7 | 69.8 | 0.79 (0.73–0.85) |
| Rodriguez et al. [65] | 362 | serum | 1.1 | 24.0 | 97.2 | 0.75 (0.68–0.83) |
| Amati et al. [24] | 170 | plasma | 1.0 | 90.0 | 78.0 | 0.93 (0.88–0.97) |
| Iwahori et al. [66] | 121 | serum | 93.5 | 59.3 | 86.2 | 0.71 |
| Beyer et al. [67] | 497 | serum | 1.0 | 68.2 | 77.0 | – |
| Cristaudo et al. [68] | 369 | serum | 1.0 | 68.2 | 80.5 | 0.77 (0.71–0.83) |
| Creany et al. [74] | 107 | serum | 1.6 | 73.0 | 95.0 | 0.92 (0.87–0.97) |
| Hollevoet et al. [75] | 507 | serum | 2.0 | – | 64.0 | 0.87 |
| Creaney et al. [77] | 233 | serum | 1.4 | – | 67.0 | 0.77 |
| MPF | ||||||
| Iwahori et al. [65] | 121 | serum | 19.1 | 74.1 | 90.4 | 0.88 |
| Creany et al. [74] | 107 | serum | 1.0 | 34.0 | 95.0 | 0.61 (0.51–0.72) |
| Hollevoet et al. [75] | 507 | serum | 12.4 | – | 68.0 | 0.85 |
2.3.3. Soluble Mesothelin-Related Peptides
2.3.4. Biomarker Combination
| Marker | Ctrl vs. Exp (AUC) | Exp vs. MM (AUC) | Ctrl vs. MM (AUC) |
|---|---|---|---|
| 8OHdG (AU) | 0.775 ± 0.037 | 0.566 ± 0.110 | 0.788 ± 0.090 |
| VEGF (ng/mL) | 0.714 ± 0.062 | 0.705 ± 0.086 | 0.803 ± 0.074 |
| SMRPs (nM) | 0.459 ± 0.042 | 0.927 ± 0.022 | 0.920 ± 0.030 |
| 8OHdG-VEGF-SMRPs | 0.925 ± 0.035 | - | - |
2.3.5. Molecular Biomarkers
2.4. Prognostic Biomarkers
2.5. Target Biomarkers
2.6. Predictive Biomarkers
3. Conclusions

References
- Price, B.; Ware, A. Mesothelioma trends in the United States: an update based on Surveillance, Epidemiology, and End Results Program data for 1973 through 2003. Am. J. Epidemiol. 2004, 159, 107–112. [Google Scholar] [CrossRef]
- Peto, J.; Decarli, A.; La Vecchia, C.; Levi, F.; Negri, E. The European mesothelioma epidemic. Br. J. Cancer 1999, 79, 666–672. [Google Scholar] [CrossRef]
- Bourdès, V.; Boffetta, P.; Pisani, P. Environmental exposure to asbestos and risk of pleural mesothelioma: review and meta-analysis. Eur. J. Epidemiol. 2000, 16, 411–417. [Google Scholar] [CrossRef]
- Hansen, J.; de Klerk, N.H.; Musk, A.W.; Hobbs, M.S.T. Environmental exposure to crocidolite and mesothelioma. Exposure-response relationships. Am. J. Respir. Crit. Care Med. 1998, 157, 69–75. [Google Scholar] [CrossRef]
- Musti, M.; Kettunen, E.; Dragonieri, S.; Lindholm, P.; Cavone, D.; Serio, G.; Knuutila, S. Cytogenetic and molecular genetic changes in malignant mesothelioma. Cancer Genet. Cytogenet. 2006, 170, 9–15. [Google Scholar]
- Shin, H.J.; Shin, D.M.; Tarco, E.; Sneige, N. Detection of numerical aberrations of chromosomes 7 and 9 in cytologic specimens of pleural malignant mesothelioma. Cancer 2003, 99, 233–239. [Google Scholar] [CrossRef]
- Scherpereel, A.; Astoul, P.; Baas, P.; Berghmans, T.; Clayson, H.; de Vuyst, P.; Dienemann, H.; Galateau–Salle, F.; Hennequin, C.; Hillerdal, G.; Le Péchoux, C.; Mutti, L.; Pairon, J.C.; Stahel, R.; van Houtte, P.; van Meerbeeck, J.; Waller, D.; Weder, W. Guidelines of the European Respiratory Society and the European Society of Thoracic Surgeons for the management of malignant pleural mesothelioma. Eur. Respir J. 2010, 35, 479–495. [Google Scholar] [CrossRef]
- Cresoli, G.L.; Gridelli, C.; Santoro, A. Multidisciplinary treatment of malignant pleural mesothelioma. Oncologist 2007, 12, 850–863. [Google Scholar] [CrossRef]
- Scagliotti, G.V.; Selvaggi, G. Advances in diagnosis and treatment of malignant mesothelioma. Oncol. Rev. 2007, 1, 91–102. [Google Scholar] [CrossRef]
- Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001, 69, 29–95.
- Jaurand, M.C. Mechanisms of fiber-induced genotoxicity. Environ. Health Perspect. 1997, 5, 1073–1084. [Google Scholar]
- Unfried, K.; Schurkes, C.; Abel, J. Distinct spectrum of mutations induced by crocidolite asbestos: clue for 8-hydroxydeoxyguanosine-dependent mutagenesis in vivo. Cancer Res. 2002, 62, 99–104. [Google Scholar]
- Moriya, M. Single-stranded shuttle phagemid for mutagenesis studies in mammalian cells: 8-oxoguanine in DNA induced targeted G.C → T.A transversions in simian kidney cells. Proc. Natl. Acad. Sci. USA 1993, 90, 1122–1126. [Google Scholar] [CrossRef]
- Halliwell, B. Oxidative stress and cancer: have we moved forward? Biochem. J. 2007, 401, 1–11. [Google Scholar]
- Halliwell, B. Can oxidative DNA damage be used as a biomarker of cancer risk in human? Problems, resolutions and preliminary results from nutritional supplementation studies. Free Radical Res. 1998, 29, 469–486. [Google Scholar] [CrossRef]
- Takahashi, K.; Pan, G.; Kasai, H.; Hanaoka, T.; Feng, Y.; Liu, N.; Zhang, S.; Xu, Z.; Tsuda, T.; Yamato, H.; Higashi, T.; Okubo, T. Relationship between Asbestos Exposures and 8-Hydroxydeoxyguanosine Levels in Leukocytic DNA of Workers at a Chinese Asbestos-material Plant. Int. J. Occup. Environ. Health 1997, 3, 111–119. [Google Scholar]
- Marczynski, B.; Kraus, T.; Rozynek, P.; Raithel, H.J.; Baur, X. Association between 8-hydroxy-2′-deoxyguanosine levels in DNA of workers highly exposed to asbestos and their clinical data, occupational and non-occupational confounding factors, and cancer. Mutat. Res. 2000, 468, 203–212. [Google Scholar] [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, C. 8-hydroxy-2'-deoxyguanosine (8-OHdG): A Critical Biomarker of Oxidative Stress and Carcinogenesis. J. Environ. Sci. Health 2009, 27, 20–139. [Google Scholar]
- Takeuchi, T.; Nakajima, M.; Morimoto, K. Relationship between the intracellular reactive oxygen species and the induction of oxidative DNA damage in human neutrophil-like cells. Carcinogenesis 1996, 17, 1543–1548. [Google Scholar] [CrossRef]
- Sabatini, L.; Barbieri, A.; Tosi, M.; Roda, A.; Violante, F.S. A method for routine quntification of urinary 8-hydroxy-2’-deoxyguanosine based on solid-phase extraction and micro-high-performance liquid chromatography/electrospry ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2005, 19, 147–152. [Google Scholar]
- Collins, A.R.; Dusinská, M.; Horváthová, E.; Munro, E.; Savio, M.; Stĕtina, R. Inter-individual differences in repair of DNA base oxidation, measured in vitro with the comet assay. Mutagenesis 2001, 16, 297–301. [Google Scholar] [CrossRef]
- Tomasetti, M.; Alleva, R.; Borghi, B.; Collins, A.R. In vivo supplementation with coenzyme Q10 enhances the recovery of human lymphocytes from oxidative DNA damage. FASEB J. 2001, 15, 1425–1427. [Google Scholar]
- De Iuliis, G.N.; Thomson, L.K.; Mitchell, L.A.; Finnie, J.M.; Koppers, A.J.; Hedges, A.; Nixon, B.; Aitken, R.J. DNA damage in human spermatozoa is highly correlated with the efficiency of chromatin remodeling and the formation of 8-hydroxy-2'-deoxyguanosine, a marker of oxidative stress. Biol. Reprod. 2009, 81, 517–524. [Google Scholar] [CrossRef]
- Amati, M.; Tomasetti, M.; Scartozzi, M.; Mariotti, L.; Alleva, R.; Pignotti, E.; Borghi, B.; Valentino, M.; Governa, M.; Neuzil, J.; Santarelli, L. Profiling tumor-associated markers for early detection of malignant mesothelioma: an epidemiologic study. Cancer Epidemiol. Biomarkers Prev. 2008, 17, 163–170. [Google Scholar] [CrossRef]
- Amati, M.; Tomasetti, M.; Mariotti, L.; Tarquini, L.M.; Valentino, M.; Santarelli, L. Assessment of biomarkers in asbestos-exposed workers as indicators of cancer risk. Mutat. Res. 2008, 655, 52–58. [Google Scholar] [CrossRef]
- Roushdy-Hammady, I.; Siegel, J.; Emri, S.; Testa, J.R.; Carbone, M. Genetic-susceptibility factor and malignant mesothelioma in the Cappadocian region of Turkey. Lancet 2001, 357, 444–445. [Google Scholar]
- Dusinská, M.; Collins, A.; Kazimírová, A.; Barancoková, M.; Harrington, V.; Volkovová, K.; Staruchová, M.; Horská, A.; Wsólová, L.; Kocan, A.; Petrík, J.; Machata, M.; Ratcliffe, B.; Kyrtopoulos, S. Genotoxic effects of asbestos in humans. Mutat. Res. 2004, 553, 91–102. [Google Scholar] [CrossRef]
- Dusinská, M.; Barancoková, M.; Kazimírová, A.; Harrington, V.; Volkovová, K.; Staruchová, M.; Horská, A.; Wsólová, L.; Collins, A. Does occupational exposure to mineral fibres cause DNA or chromosome damage? Mutat. Res. 2004, 553, 103–110. [Google Scholar]
- Leprat, F.; Alapetite, C.; Rosselli, F.; Ridet, A.; Schlumberger, M.; Sarasin, A.; Suarez, H.G.; Moustacchi, E. Impaired DNA repair as assessed by the "comet" assay in patients with thyroid tumors after a history of radiation therapy: a preliminary study. Int. J. Radiat. Oncol. Biol. Phys. 1998, 40, 1019–1026. [Google Scholar] [CrossRef]
- Jałoszyński, P.; Kujawski, M.; Czub-Swierczek, M.; Markowska, J.; Szyfter, K. Bleomycin-induced DNA damage and its removal in lymphocytes of breast cancer patients studied by comet assay. Mutat. Res. 1997, 385, 223–233. [Google Scholar] [CrossRef]
- Schmezer, P.; Rajaee-Behbahani, N.; Risch, A.; Thiel, S.; Rittgen, W.; Drings, P.; Dienemann, H.; Kayser, K.W.; Schulz, V.; Bartsch, H. Rapid screening assay for mutagen sensitivity and DNA repair capacity in human peripheral blood lymphocytes. Mutagenesis 2001, 16, 25–30. [Google Scholar] [CrossRef]
- Bonassi, S.; Hagmar, L.; Strömberg, U.; Montagud, A.H.; Tinnerberg, H.; Forni, A.; Heikkilä, P.; Wanders, S.; Wilhardt, P.; Hansteen, I.L.; Knudsen, L.E.; Norppa, H. Chromosomal aberrations in lymphocytes predict human cancer independently of exposure to carcinogens. European Study Group on Cytogenetic Biomarkers and Health. Cancer Res. 2000, 60, 1619–1625. [Google Scholar]
- Rossner, P.; Boffetta, P.; Ceppi, M.; Bonassi, S.; Smerhovsky, Z.; Landa, K.; Juzova, D.; Srám, R.J. Chromosomal aberrations in lymphocytes of healthy subjects and risk of cancer. Environ. Health Perspect. 2005, 113, 517–520. [Google Scholar]
- Trenz, K.; Rothfuss, A.; Schütz, P.; Speit, G. Mutagen sensitivity of peripheral blood from women carrying a BRCA1 or BRCA2 mutation. Mutat. Res. 2002, 500, 89–96. [Google Scholar] [CrossRef]
- Bolognesi, C.; Filiberti, R.; Neri, M.; Perrone, E.; Landini, E.; Canessa, P.A.; Simonassi, C.; Cerrano, P.G.; Mutti, L.; Puntoni, R. High frequency of micronuclei in peripheral blood lymphocytes as index of susceptibility to pleural malignant mesothelioma. Cancer Res. 2002, 62, 5418–5419. [Google Scholar]
- Carbone, M.; Kratzke, R.A.; Testa, J.R. The pathogenesis of mesothelioma. Semin. Oncol. 2002, 29, 2–17. [Google Scholar] [CrossRef]
- Alatas, F.; Alatas, O.; Metintas, M.; Golak, O.; Harmanci, E.; Demir, S. Diagnostic value of CEA, CA 15-3, CA 19-9, CYFRA 21-1, NSE and TSA assay in pleural effutions. Lung Cancer 2001, 31, 9–16. [Google Scholar]
- Paganuzzi, M.; Onetto, M.; Marroni, P.; Filiberti, R.; Tassara, E.; Parodi, S.; Felletti, R. Diagnostic value of CYFRA 21-1 tumor marker and CEA in pleural effusion due to mesothelioma. Chest 2001, 119, 1138–1142. [Google Scholar]
- Grigoriu, B.D.; Grigoriu, C.; Chahine, B.; Gey, T.; Scherpereel, A. Clinical utility of diagnostic markers for malignant pleural mesothelioma. Monaldi Arch. Chest Dis. 2009, 71, 31–38. [Google Scholar]
- van den Heuvel, M.M.; Korse, C.M.; Bonfrer, J.M.; Baas, P. Non-invasive diagnosis of pleural malignancies: the role of tumour markers. Lung Cancer 2008, 59, 350–354. [Google Scholar] [CrossRef]
- Bonfrer, J.M.; Schouwink, J.H.; Korse, C.M.; Baas, P. Cyfra 21-1 and TPA as markers in malignant mesothelioma. Anticancer Res. 1997, 17, 2971–2973. [Google Scholar]
- Salama, G.; Miédougé, M.; Rouzaud, P.; Mauduyt, M.A.; Pujazon, M.C.; Vincent, C.; Carles, P.; Serre, G. Evaluation of pleural CYFRA 21-1 and carcinoembryonic antigen in the diagnosis of malignant pleural effusions. Br. J. Cancer 1998, 77, 472–476. [Google Scholar] [CrossRef]
- Romero, S.; Fernández, C.; Arriero, J.M.; Espasa, A.; Candela, A.; Martín, C.; Sánchez-Payá, J. CEA, CA 15-3 and CYFRA 21-1 in serum and pleural fluid of patients with pleural effusions. Eur. Respir. J. 1996, 9, 17–23. [Google Scholar] [CrossRef]
- Porcel, J.M.; Vives, M.; Esquerda, A.; Salud, A.; Pérez, B.; Rodríguez-Panadero, F. Use of a panel of tumor markers (carcinoembryonic antigen, cancer antigen 125, carbohydrate antigen 15-3, and cytokeratin 19 fragments) in pleural fluid for the differential diagnosis of benign and malignant effusions. Chest 2004, 126, 1757–1763. [Google Scholar] [CrossRef]
- Pettersson, T.; Fröseth, B.; Riska, H.; Klockars, M. Concentration of hyaluronic acid in pleural fluid as a diagnostic aid for malignant mesothelioma. Chest 1988, 94, 1037–1039. [Google Scholar] [CrossRef]
- Atagi, S.; Ogawara, M.; Kawahara, M.; Sakatani, M.; Furuse, K.; Ueda, E.; Yamamoto, S. Utility of hyaluronic acid in pleural fluid for differential diagnosis of pleural effusions: likelihood ratios for malignant mesothelioma. Jpn. J. Clin. Oncol. 1997, 27, 293–297. [Google Scholar] [CrossRef]
- Grigoriu, B.; Chahine, B.; Zerimech, F.; Grégoire, M.; Balduyck, M.; Copin, M.C.; Devos, P.; Lassalle, P.; Scherpereel, A. Serum mesothelin has a higher diagnostic utility than hyaluronic acid in malignant mesothelioma. Clin. Biochem. 2009, 42, 1046–1050. [Google Scholar] [CrossRef] [Green Version]
- Pass, H.I.; Lott, D.; Lonardo, F.; Harbut, M.; Liu, Z.; Tang, N.; Carbone, M.; Webb, C.; Wali, A. Asbestos exposure, pleural mesothelioma, and serum osteopontin levels. N. Engl. J. Med. 2005, 353, 1564–1573. [Google Scholar] [CrossRef]
- Cullen, M.R. Serum osteopontin levels--is it time to screen asbestos-exposed workers for pleural mesothelioma? N. Engl. J. Med. 2005, 353, 1617–1618. [Google Scholar] [CrossRef]
- Wai, P.Y.; Kuo, P.C. The role of Osteopontin in tumor metastasis. J. Surg. Res. 2004, 121, 228–241. [Google Scholar] [CrossRef]
- Park, E.K.; Thomas, P.S.; Johnson, A.R.; Yates, D.H. Osteopontin levels in an asbestos-exposed population. Clin. Cancer Res. 2009, 15, 1362–1366. [Google Scholar] [CrossRef]
- Grigoriu, B.D.; Scherpereel, A.; Devos, P.; Chahine, B.; Letourneux, M.; Lebailly, P.; Grégoire, M.; Porte, H.; Copin, M.C.; Lassalle, P. Utility of osteopontin and serum mesothelin in malignant pleural mesothelioma diagnosis and prognosis assessment. Clin. Cancer Res. 2007, 13, 2928–2935. [Google Scholar] [CrossRef]
- Myles, T.; Leung, L.L. Thrombin hydrolysis of human osteopontin is dependent on thrombin anion-binding exosites. J. Biol. Chem. 2008, 283, 17789–17796. [Google Scholar]
- Kazanecki, C.C.; Uzwiak, D.J.; Denhardt, D.T. Control of osteopontin signaling and function by post-translational phosphorylation and protein folding. J. Cell Biochem. 2007, 102, 912–24. [Google Scholar] [CrossRef]
- Paleari, L.; Rotolo, N.; Imperatori, A.; Puzone, R.; Sessa, F.; Franzi, F.; Meacci, E.; Camplese, P.; Cesario, A.; Paganizzi, M. Osteopontin is not a specific marker in malignant pleural mesothelioma. Int. J. Biol. Markers. 2009, 24, 112–117. [Google Scholar]
- Fedarko, N.S.; Jain, A.; Karadag, A.; Van Eman, M.R.; Fisher, L.W. Elevated serum bone sialoprotein and osteopontin in colon, breast, prostate, and lung cancer. Clin. Cancer Res. 2001, 7, 4060–4066. [Google Scholar]
- Ue, T.; Yokozaki, H.; Kitadai, Y.; Yamamoto, S.; Yasui, W.; Ishikawa, T.; Tahara, E. Co-expression of osteopontin and CD44v9 in gastric cancer. Int. J. Cancer. 1998, 79, 127–132. [Google Scholar] [CrossRef]
- Kato, N.; Motoyama, T. Overexpression of osteopontin in clear cell carcinoma of the ovary: close association with HNF-1beta expression. Histopathology 2008, 52, 682–688. [Google Scholar] [CrossRef]
- Zhou, Y.; Dai, D.L.; Martinka, M.; Su, M.; Zhang, Y.; Campos, E.I.; Dorocicz, I.; Tang, L.; Huntsman, D.; Nelson, C.; Ho, V.; Li, G. Osteopontin expression correlates with melanoma invasion. J. Invest. Dermatol. 2005, 124, 1044–1052. [Google Scholar] [CrossRef]
- Robinson, B.W.; Creaney, J.; Lake, R.; Nowak, A.; Musk, A.W.; de Klerk, N.; Winzell, P.; Hellstrom, K.E.; Hellstrom, I. Mesothelin-family proteins and diagnosis of mesothelioma. Lancet 2003, 362, 1612–1616. [Google Scholar] [CrossRef]
- Scherpereel, A.; Grigoriu, B.; Conti, M.; Gey, T.; Grégoire, M.; Copin, M.C.; Devos, P.; Chahine, B.; Porte, H.; Lassalle, P. Soluble mesothelin-related peptides in the diagnosis of malignant pleural mesothelioma. Am. J. Respir. Crit. Care Med. 2006, 173, 1155–1160. [Google Scholar]
- Hartge, P. Designing early detection programs for ovarian cancer. J. Natl. Cancer Inst. 2010, 102, 3–4. [Google Scholar] [CrossRef]
- Johnston, F.M.; Tan, M.C.; Tan, B.R., Jr.; Porembka, M.R.; Brunt, E.M.; Linehan, D.C.; Simon, P.O., Jr.; Plambeck-Suess, S.; Eberlein, T.J.; Hellstrom, K.E.; Hellstrom, I.; Hawkins, W.G.; Goedegebuure, P. Circulating mesothelin protein and cellular antimesothelin immunity in patients with pancreatic cancer. Clin. Cancer Res. 2009, 15, 6511–6518. [Google Scholar] [CrossRef]
- Hassan, R.; Remaley, A.T.; Sampson, M.L.; Zhang, J.; Cox, D.D.; Pingpank, J.; Alexander, R.; Willingham, M.; Pastan, I.; Onda, M. Detection and quantitation of serum mesothelin, a tumor marker for patients with mesothelioma and ovarian cancer. Clin. Cancer Res. 2006, 12, 447–453. [Google Scholar] [CrossRef]
- Rodríguez Portal, J.A.; Rodríguez Becerra, E.; Rodríguez Rodríguez, D.; Alfageme Michavila, I.; Quero Martínez, A.; Diego Roza, C.; León Jiménez, A.; Isidro Montes, I.; Cebollero Rivas, P. Serum levels of soluble mesothelin-related peptides in malignant and nonmalignant asbestos-related pleural disease: relation with past asbestos exposure. Cancer Epidemiol. Biomarkers Prev. 2009, 18, 646–650. [Google Scholar] [CrossRef]
- Iwahori, K.; Osaki, T.; Serada, S.; Fujimoto, M.; Suzuki, H.; Kishi, Y.; Yokoyama, A.; Hamada, H.; Fujii, Y.; Yamaguchi, K.; Hirashima, T.; Matsui, K.; Tachibana, I.; Nakamura, Y.; Kawase, I.; Naka, T. Megakaryocyte potentiating factor as a tumor marker of malignant pleural mesothelioma: evaluation in comparison with mesothelin. Lung Cancer 2008, 62, 45–54. [Google Scholar] [CrossRef]
- Beyer, H.L.; Geschwindt, R.D.; Glover, C.L.; Tran, L.; Hellstrom, I.; Hellstrom, K.E.; Miller, M.C.; Verch, T.; Allard, W.J.; Pass, H.I.; Sardesai, N.Y. MESOMARK: a potential test for malignant pleural mesothelioma. Clin. Chem. 2007, 53, 666–672. [Google Scholar] [CrossRef]
- Cristaudo, A.; Foddis, R.; Vivaldi, A.; Guglielmi, G.; Dipalma, N.; Filiberti, R.; Neri, M.; Ceppi, M.; Paganuzzi, M.; Ivaldi, G.P.; Mencoboni, M.; Canessa, P.A.; Ambrosino, N.; Chella, A.; Mutti, L.; Puntoni, R. Clinical significance of serum mesothelin in patients with mesothelioma and lung cancer. Clin. Cancer Res. 2007, 13, 5076–5081. [Google Scholar] [CrossRef]
- Park, E.K.; Sandrini, A.; Yates, D.H.; Creaney, J.; Robinson, B.W.; Thomas, P.S.; Johnson, A.R. Soluble mesothelin-related protein in an asbestos-exposed population: the dust diseases board cohort study. Am. J. Respir. Crit. Care Med. 2008, 178, 832–837. [Google Scholar] [CrossRef]
- Amati, M.; Tomasetti, M.; Mariotti, L.; Tarquini, L.M.; Valentino, M.; Santarelli, L. Assessment of biomarkers in asbestos-exposed workers as indicators of cancer risk. Mutat. Res. 2008, 655, 52–58. [Google Scholar] [CrossRef]
- Luo, L.; Shi, H.Z.; Liang, Q.L.; Jiang, J.; Qin, S.M.; Deng, J.M. Diagnostic value of soluble mesothelin-related peptides for malignant mesothelioma: A meta-analysis. Respir. Med. 2010, 104, 149–156. [Google Scholar] [CrossRef]
- Shiomi, K.; Miyamoto, H.; Segawa, T.; Hagiwara, Y.; Ota, A.; Maeda, M.; Takahashi, K.; Masuda, K.; Sakao, Y.; Hino, O. Novel ELISA system for detection of N-ERC/mesothelin in the sera of mesothelioma patients. Cancer Sci. 2006, 97, 928–932. [Google Scholar] [CrossRef]
- Onda, M.; Nagata, S.; Ho, M.; Bera, T.K.; Hassan, R.; Alexander, R.H.; Pastan, I. Megakaryocyte potentiation factor cleaved from mesothelin precursor is a useful tumor marker in the serum of patients with mesothelioma. Clin. Cancer Res. 2006, 12, 4225–4231. [Google Scholar] [CrossRef]
- Creaney, J.; Yeoman, D.; Demelker, Y.; Segal, A.; Musk, A.W.; Skates, S.J.; Robinson, B.W. Comparison of osteopontin, megakaryocyte potentiating factor, and mesothelin proteins as markers in the serum of patients with malignant mesothelioma. J. Thorac. Oncol. 2008, 3, 851–857. [Google Scholar] [CrossRef]
- Hollevoet, K.; Nackaerts, K.; Thimpont, J.; Germonpré, P.; Bosquée, L.; De Vuyst, P.; Legrand, C.; Kellen, E.; Kishi, Y.; Delanghe, J.R.; van Meerbeeck, J.P. Diagnostic Performance of Soluble Mesothelin and Megakaryocyte Potentiating Factor in Mesothelioma. Am. J. Respir. Crit. Care Med. 2010, 181, 620–625. [Google Scholar] [CrossRef]
- Shitrit, D.; Zingerman, B.; Shitrit, A.B.; Shlomi, D.; Kramer, M.R. Diagnostic value of CYFRA 21-1, CEA, CA 19-9, CA 15-3, and CA 125 assays in pleural effusions: analysis of 116 cases and review of the literature. Oncologist 2005, 10, 501–507. [Google Scholar] [CrossRef]
- Creaney, J.; van Bruggen, I.; Hof, M.; Segal, A.; Musk, A.W.; de Klerk, N.; Horick, N.; Skates, S.J.; Robinson, B.W. Combined CA125 and mesothelin levels for the diagnosis of malignant mesothelioma. Chest 2007, 132, 1239–1246. [Google Scholar] [CrossRef]
- Jones, P.A.; Baylin, S.B. The fundamental role of epigenetic events in cancer. Nat. Rev. Genet. 2002, 3, 415–428. [Google Scholar]
- Goto, Y.; Shinjo, K.; Kondo, Y.; Shen, L.; Toyota, M.; Suzuki, H.; Gao, W.; An, B.; Fujii, M.; Murakami, H.; Osada, H.; Taniguchi, T.; Usami, N.; Kondo, M.; Hasegawa, Y.; Shimokata, K.; Matsuo, K.; Hida, T.; Fujimoto, N.; Kishimoto, T.; Issa, J.P.; Sekido, Y. Epigenetic profiles distinguish malignant pleural mesothelioma from lung adenocarcinoma. Cancer Res. 2009, 69, 9073–9082. [Google Scholar] [CrossRef]
- Hussain, S.P.; Harris, C.C. Inflammation and cancer: an ancient link with novel potentials. Int. J. Cancer 2007, 121, 2373–2380. [Google Scholar] [CrossRef]
- Tsou, J.A.; Shen, L.Y.; Siegmund, K.D.; Long, T.I.; Laird, P.W.; Seneviratne, C.K.; Koss, M.N.; Pass, H.I.; Hagen, J.A.; Laird-Offringa, I.A. Distinct DNA methylation profiles in malignant mesothelioma, lung adenocarcinoma, and non-tumor lung. Lung Cancer 2005, 47, 193–204. [Google Scholar] [CrossRef]
- Tsou, J.A.; Galler, J.S.; Wali, A.; Ye, W.; Siegmund, K.D.; Groshen, S.; Laird, P.W.; Turla, S.; Koss, M.N.; Pass, H.I.; Laird-Offringa, I.A. DNA methylation profile of 28 potential marker loci in malignant mesothelioma. Lung Cancer 2007, 58, 220–230. [Google Scholar] [CrossRef]
- Yang, H.; Bocchetta, M.; Kroczynska, B.; Elmishad, A.G.; Chen, Y.; Liu, Z.; Bubici, C.; Mossman, B.T.; Pass, H.I.; Testa, J.R.; Franzoso, G.; Carbone, M. TNF-alpha inhibits asbestos-induced cytotoxicity via a NF-kappaB-dependent pathway, a possible mechanism for asbestos-induced oncogenesis. Proc. Natl. Acad. Sci. USA 2006, 103, 10397–10402. [Google Scholar] [CrossRef]
- Shapiro, B.; Chakrabarty, M.; Cohn, E.M.; Leon, S.A. Determination of circulating DNA levels in patients with benign or malignant gastrointestinal disease. Cancer 1983, 51, 2116–2120. [Google Scholar] [CrossRef]
- Fischer, J.R.; Ohnmacht, U.; Rieger, N.; Zemaitis, M.; Stoffregen, C.; Kostrzewa, M.; Buchholz, E.; Manegold, C.; Lahm, H. Promoter methylation of RASSF1A, RARbeta and DAPK predict poor prognosis of patients with malignant mesothelioma. Lung Cancer 2006, 54, 109–116. [Google Scholar] [CrossRef]
- Rauhala, H.E.; Jalava, S.E.; Isotalo, J.; Bracken, H.; Lehmusvaara, S.; Tammela, T.L.; Oja, H.; Visakorpi, T. miR-193b is an epigenetically regulated putative tumor suppressor in prostate cancer. Int. J. Cancer 2010. [Google Scholar] [CrossRef]
- Lodygin, D.; Tarasov, V.; Epanchintsev, A.; Berking, C.; Knyazeva, T.; Körner, H.; Knyazev, P.; Diebold, J.; Hermeking, H. Inactivation of miR-34a by aberrant CpG methylation in multiple types of cancer. Cell Cycle 2008, 7, 2591–2600. [Google Scholar] [CrossRef]
- Wijnhoven, B.P.; Michael, M.Z.; Watson, D.I. MicroRNAs and cancer. Br. J. Surg. 2007, 94, 23–30. [Google Scholar] [CrossRef]
- Croce, C.M. Causes and consequences of microRNA dysregulation in cancer. Nat. Rev. Genet. 2009, 10, 704–714. [Google Scholar] [CrossRef]
- Calin, G.A.; Croce, C.M. MicroRNA signatures in human cancers. Nat. Rev. Cancer 2006, 6, 857–866. [Google Scholar] [CrossRef]
- Lu, J.; Getz, G.; Miska, E.A.; Alvarez-Saavedra, E.; Lamb, J.; Peck, D.; Sweet-Cordero, A.; Ebert, B.L.; Mak, R.H.; Ferrando, A.A.; Downing, J.R.; Jacks, T.; Horvitz, H.R.; Golub, T.R. MicroRNA expression profiles classify human cancers. Nature 2005, 435, 834–838. [Google Scholar] [CrossRef]
- Blenkiron, C.; Miska, E.A. miRNAs in cancer: approaches, aetiology, diagnostics and therapy. Hum. Mol. Genet. 2007, 16, R106–R113. [Google Scholar] [CrossRef]
- Guled, M.; Lahti, L.; Lindholm, P.M.; Salmenkivi, K.; Bagwan, I.; Nicholson, A.G.; Knuutila, S. CDKN2A, NF2, and JUN are dysregulated among other genes by miRNAs in malignant mesothelioma -A miRNA microarray analysis. Gene. Chromosome. Canc. 2009, 48, 615–623. [Google Scholar] [CrossRef]
- Busacca, S.; Germano, S.; De Cecco, L.; Rinaldi, M.; Comoglio, F.; Favero, F.; Murer, B.; Mutti, L.; Pierotti, M.; Gaudino, G. MicroRNA Signature of Malignant Mesothelioma with Potential Diagnostic and Prognostic Implications. Am. J. Respir. Cell Mol. Biol. 2010, 42, 312–319. [Google Scholar] [CrossRef]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O'Briant, K.C.; Allen, A.; Lin, D.W.; Urban, N.; Drescher, C.W.; Knudsen, B.S.; Stirewalt, D.L.; Gentleman, R.; Vessella, R.L.; Nelson, P.S.; Martin, D.B.; Tewari, M. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, S.; Marzolf, B.; Troisch, P.; Brightman, A.; Hu, Z.; Hood, L.E.; Galas, D.J. Circulating microRNAs, potential biomarkers for drug-induced liver injury. Proc. Natl. Acad. Sci USA 2009, 106, 4402–4407. [Google Scholar]
- Crispi, S.; Calogero, R.A.; Santini, M.; Mellone, P.; Vincenzi, B.; Citro, G.; Vicidomini, G.; Fasano, S.; Meccariello, R.; Cobellis, G.; Menegozzo, S.; Pierantoni, R.; Facciolo, F.; Baldi, A.; Menegozzo, M. Global gene expression profiling of human pleural mesotheliomas: identification of matrix metalloproteinase 14 (MMP-14) as potential tumour target. PLoS One 2009, 4, e7016. [Google Scholar] [CrossRef]
- Røe, O.D.; Anderssen, E.; Sandeck, H.; Christensen, T.; Larsson, E.; Lundgren, S. Malignant pleural mesothelioma: genome-wide expression patterns reflecting general resistance mechanisms and a proposal of novel targets. Lung Cancer 2010, 67, 57–68. [Google Scholar]
- Gordon, G.J.; Rockwell, G.N.; Godfrey, P.A.; Jensen, R.V.; Glickman, J.N.; Yeap, B.Y.; Richards, W.G.; Sugarbaker, D.J.; Bueno, R. Validation of genomics-based prognostic tests in malignant pleural mesothelioma. Clin. Cancer Res. 2005, 11, 4406–4414. [Google Scholar] [CrossRef]
- Pass, H.I.; Liu, Z.; Wali, A.; Bueno, R.; Land, S.; Lott, D.; Siddiq, F.; Lonardo, F.; Carbone, M.; Draghici, S. Gene expression profiles predict survival and progression of pleural mesothelioma. Clin. Cancer Res. 2004, 10, 849–859. [Google Scholar] [CrossRef]
- Gordon, G.J.; Dong, L.; Yeap, B.Y.; Richards, W.G.; Glickman, J.N.; Edenfield, H.; Mani, M.; Colquitt, R.; Maulik, G.; Van Oss, B.; Sugarbaker, D.J.; Bueno, R. Four-gene expression ratio test for survival in patients undergoing surgery for mesothelioma. J. Natl. Cancer Inst. 2009, 101, 678–686. [Google Scholar] [CrossRef]
- Schouwink, H.; Korse, C.M.; Bonfrer, J.M.; Hart, A.A.; Baas, P. Prognostic value of the serum tumour markers Cyfra 21-1 and tissue polypeptide antigen in malignant mesothelioma. Lung Cancer 1999, 25, 25–32. [Google Scholar]
- Hedman, M.; Arnberg, H.; Wernlund, J.; Riska, H.; Brodin, O. Tissue polypeptide antigen (TPA), hyaluronan and CA 125 as serum markers in malignant mesothelioma. Anticancer Res. 2003, 23, 531–536. [Google Scholar]
- Thylén, A.; Hjerpe, A.; Martensson, G. Hyaluronan content in pleural fluid as a prognostic factor in patients with malignant pleural mesothelioma. Cancer 2001, 92, 1224–1230. [Google Scholar] [CrossRef]
- Filiberti, R.; Marroni, P.; Neri, M.; Ardizzoni, A.; Betta, P.G.; Cafferata, M.A.; Canessa, P.A.; Puntoni, R.; Ivaldi, G.P.; Paganizzi, M. Serum PDGF-AB in pleural mesothelioma. Tumour Biol. 2005, 26, 221–226. [Google Scholar] [CrossRef]
- Aoe, K.; Hiraki, A.; Tanaka, T.; Gemba, K.; Taguchi, K.; Murakami, T.; Sueoka, N.; Kamei, T.; Ueoka, H.; Sugi, K.; Yoshino, T.; Kishimoto, T. Expression of vascular endothelial growth factor in malignant mesothelioma. Anticancer Res. 2006, 26, 4833–4836. [Google Scholar]
- Strizzi, L.; Catalano, A.; Vianale, G.; Orecchia, S.; Casalini, A.; Tassi, G.; Puntoni, R.; Mutti, L.; Procopio, A. Vascular endothelial growth factor is an autocrine growth factor in human malignant mesothelioma. J. Pathol. 2001, 193, 468–475. [Google Scholar] [CrossRef]
- Yarden, Y. EGFR family and its ligand in human cancer. Signalling mechanisms and therapeutic opportunities. Eur. J. Cancer 2001, 37, S3–S8. [Google Scholar] [CrossRef]
- Dazzi, H.; Hasleton, P.S.; Thatcher, N.; Wilkes, S.; Swindell, R.; Chatterjee, A.K. Malignant pleural mesothelioma and epidermal growth factor receptor (EGF-R). Relationship of EGF-R with histology and survival using fixed paraffin embedded tissue and the F4, monoclonal antibody. Br. J. Cancer 1990, 61, 924–926. [Google Scholar] [CrossRef]
- Edwards, J.G.; Swinson, D.E.; Jones, J.L.; Waller, D.A.; O'Byrne, K.J. EGFR expression: associations with outcome and clinicopathological variables in malignant pleural mesothelioma. Lung Cancer 2006, 54, 399–407. [Google Scholar] [CrossRef]
- Pinton, G.; Brunelli, E.; Murer, B.; Puntoni, R.; Puntoni, M.; Fennell, D.A.; Gaudino, G.; Mutti, L.; Moro, L. Estrogen receptor–beta affects the prognosis of human malignant mesothelioma. Cancer Res. 2009, 69, 4598–4604. [Google Scholar] [CrossRef]
- Soini, Y.; Kinnula, V.; Kaarteenaho-Wiik, R.; Kurttila, E.; Linnainmaa, K.; Pääkkö, P. Apoptosis and expression of apoptosis regulating proteins bcl-2, mcl-1, bcl-X, and bax in malignant mesothelioma. Clin. Cancer Res. 1999, 5, 3508–3515. [Google Scholar]
- Kokturk, N.; Firat, P.; Akay, H.; Kadilar, C.; Ozturk, C.; Zorlu, F.; Gungen, Y.; Emri, S. Prognostic significance of Bax and Fas ligand in erionite and asbestos induced Turkish malignant pleural mesothelioma. Lung Cancer 2005, 50, 189–198. [Google Scholar] [CrossRef]
- Gordon, G.J.; Mani, M.; Mukhopadhyay, L.; Dong, L.; Edenfield, H.R.; Glickman, J.N.; Yeap, B.Y.; Sugarbaker, D.J.; Bueno, R. Expression patterns of inhibitor of apoptosis proteins in malignant pleural mesothelioma. J. Pathol. 2007, 211, 447–454. [Google Scholar] [CrossRef]
- Edwards, J.G.; McLaren, J.; Jones, J.L.; Waller, D.A.; O'Byrne, KJ. Matrix metalloproteinases 2 and 9 (gelatinases A and B) expression in malignant mesothelioma and benign pleura. Br. J. Cancer 2003, 88, 1553–1559. [Google Scholar] [CrossRef]
- Edwards, J.G.; Faux, S.P.; Plummer, S.M.; Abrams, K.R.; Walker, R.A.; Waller, D.A.; O'Byrne, K.J. Cyclooxygenase-2 expression is a novel prognostic factor in malignant mesothelioma. Clin. Cancer Res. 2002, 8, 1857–1862. [Google Scholar]
- O'Kane, S.L.; Cawkwell, L.; Campbell, A.; Lind, M.J. Cyclooxygenase-2 expression predicts survival in malignant pleural mesothelioma. Eur. J. Cancer 2005, 41, 1645–1648. [Google Scholar] [CrossRef]
- Zucali, P.A.; Giaccone, G. Biology and management of malignant pleural mesothelioma. Eur. J. Cancer 2006, 42, 2706–2714. [Google Scholar] [CrossRef]
- Jahan, T.; Gu, L.; Wang, X.; Kratzke, R.; Green, M.; Vokes, E.; Kindler, H. Vatalanib in patients with previously untreated advanced malignant mesothelioma (MM): Preliminary analysis of a phase II study by the Cancer and Leukemia Group B (CALGB 30107). Lung Cancer 2005, 49, S221. [Google Scholar]
- Baas, P.; Boogerd, W.; Dalesio, O.; Haringhuizen, A.; Custers, F.; van Zandwijk, N. Thalidomide in patients with malignant pleural mesothelioma. Lung Cancer 2005, 48, 291–296. [Google Scholar] [CrossRef]
- Kindler, H.L.; Vogelzang, N.J.; Chien, K. SU5416 in malignant mesothelioma: A University of Chicago phase II consortium study. J. Clin. Oncol. 2001, 2, 1359. [Google Scholar]
- Jänne, P.A.; Wang, X.F.; Krug, L.M.; Hodgson, L.; Vokes, E.E.; Kindler, H.L. Phase II trial of sorafenib (BAY 43–9006) in malignant mesothelioma: CALGB 30307. Lung Cancer 2006, 54, S51. [Google Scholar]
- Huang, S.M.; Harari, PM. Epidermal growth factor receptor inhibition in cancer therapy: biology, rationale and preliminary clinical results. Invest. New Drugs 1999, 17, 259–269. [Google Scholar] [CrossRef]
- Govindan, R.; Kratzke, R.A.; Herndon, J.E., II; Niehans, G.A.; Vollmer, R.; Watson, D.; Green, M.R.; Kindler, H.L. Cancer and Leukemia Group B (CALGB 30101). Gefitinib in patients with malignant mesothelioma: a phase II study by the Cancer and Leukemia Group B. Clin. Cancer Res 2005, 11, 2300–2304. [Google Scholar] [CrossRef]
- Garland, L.L.; Rankin, C.; Gandara, D.R.; Rivkin, S.E.; Scott, K.M.; Nagle, R.B.; Klein-Szanto, A.J.; Testa, J.R.; Altomare, D.A.; Borden, E.C. Phase II study of erlotinib in patients with malignant pleural mesothelioma: a Southwest Oncology Group Study. J. Clin. Oncol. 2007, 25, 2406–2413. [Google Scholar] [CrossRef]
- Kelly, W.K.; O'Connor, O.A.; Krug, L.M.; Chiao, J.H.; Heaney, M.; Curley, T.; MacGregore-Cortelli, B.; Tong, W.; Secrist, J.P.; Schwartz, L.; Richardson, S.; Chu, E.; Olgac, S.; Marks, P.A.; Scher, H.; Richon, V.M. Phase I study of an oral histone deacetylase inhibitor, suberoylanilide hydroxamic acid, in patients with advanced cancer. J. Clin. Oncol. 2005, 23, 3923–3931. [Google Scholar] [CrossRef]
- Park, D.J.; Lenz, H.J. The role of proteasome inhibitors in solid tumors. Ann. Med. 2004, 36, 296–303. [Google Scholar] [CrossRef]
- Zangemeister-Wittke, U.; Hopkins-Donaldson, S. Apoptosis regulation and drug resistance in malignant pleural mesothelioma. Lung Cancer 2005, 49, 105–108. [Google Scholar] [CrossRef]
- Gordon, G.J.; Mani, M.; Mukhopadhyay, L.; Dong, L.; Yeap, B.Y.; Sugarbaker, D.J.; Bueno, R. Inhibitor of apoptosis proteins are regulated by tumour necrosis factor-alpha in malignant pleural mesothelioma. J. Pathol. 2007, 211, 439–446. [Google Scholar] [CrossRef]
- Smythe, W.R.; Mohuiddin, I.; Ozveran, M.; Cao, X.X. Antisense therapy for malignant mesothelioma with oligonucleotides targeting the bcl-xl gene product. J. Thorac. Cardiovasc. Surg. 2002, 123, 1191–1198. [Google Scholar] [CrossRef]
- Altomare, D.A.; You, H.; Xiao, G.H.; Ramos-Nino, M.E.; Skele, K.L.; De Rienzo, A.; Jhanwar, S.C.; Mossman, B.T.; Kane, A.B.; Testa, J.R. Human and mouse mesotheliomas exhibit elevated AKT/PKB activity, which can be targeted pharmacologically to inhibit tumor cell growth. Oncogene 2005, 24, 6080–6089. [Google Scholar] [CrossRef]
- Hassan, R.; Remaley, A.T.; Sampson, M.L.; Zhang, J.; Cox, D.D.; Pingpank, J.; Alexander, R.; Willingham, M.; Pastan, I.; Onda, M. Detection and quantitation of serum mesothelin, a tumor marker for patients with mesothelioma and ovarian cancer. Clin. Cancer Res. 2006, 12, 447–453. [Google Scholar] [CrossRef]
- Grigoriu, B.D.; Chahine, B.; Vachani, A.; Gey, T.; Conti, M.; Sterman, D.H.; Marchandise, G.; Porte, H.; Albelda, S.M.; Scherpereel, A. Kinetics of soluble mesothelin in patients with malignant pleural mesothelioma during treatment. Am. J. Respir. Crit. Care Med. 2009, 179, 950–954. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tomasetti, M.; Santarelli, L. Biomarkers for Early Detection of Malignant Mesothelioma: Diagnostic and Therapeutic Application. Cancers 2010, 2, 523-548. https://doi.org/10.3390/cancers2020523
Tomasetti M, Santarelli L. Biomarkers for Early Detection of Malignant Mesothelioma: Diagnostic and Therapeutic Application. Cancers. 2010; 2(2):523-548. https://doi.org/10.3390/cancers2020523
Chicago/Turabian StyleTomasetti, Marco, and Lory Santarelli. 2010. "Biomarkers for Early Detection of Malignant Mesothelioma: Diagnostic and Therapeutic Application" Cancers 2, no. 2: 523-548. https://doi.org/10.3390/cancers2020523





