Serum Tumor Markers in Pancreatic Cancer—Recent Discoveries
Abstract
:1. Introduction
2. Serum Tumor Markers for Pancreatic Cancer
2.1. Tumor Associated Antigens
2.1.1. Carbohydrate Antigens
2.1.1.1. CA19-9
| Type of marker | Author | Sensitivity | Specificity | Patients tested (n) |
|---|---|---|---|---|
| CA19-9 | Steinberg [13] | 81 | 90 | meta |
| Goonetilleke [14] | 79 | 82 | meta | |
| CA50 | Kobayashi [15] | 84 | 85 | 200 |
| Jiang [19] | 78 | 70 | 129 | |
| CA242 | Banfi [20] | 57 | 93 | 41 |
| Jiang [19] | 82 | 78 | 129 | |
| Ni [21] | 60 | 76 | 68 | |
| CA195 | Banfi [20] | 76 | 85 | 41 |
| Andicoechea [22] | 82 | 73 | 67 | |
| CA125 | Haglund [23] | 45 | 76 | 95 |
| Duraker [24] | 57 | 78 | 123 | |
| PAM4 | Gold [25] | 77 | 95 | 53 |
| TAG-72 | Pasquali [26] | 45 | 95 | 58 |
| CEA | Ni [21] | 45 | 75 | 68 |
| Haglund [23] | 54 | 76 | 95 | |
| Duraker [24] | 39 | 91 | 123 | |
| Zhao [27] | 25 | 86 | 143 | |
| POA | Nishida [28] | 81 | 96 | 21 |
| Zhao [27] | 68 | 88 | 143 | |
| TPA | Panucci [29] | 96 | 67 | 28 |
| Benini [30] | 48 | 80 | 25 | |
| Pasanen [31] | 52 | 85 | 25 | |
| TPS | Banfi [20] | 98 | 22 | 41 |
| Pasanen [32] | 50 | 70 | 26 | |
| Du-PAN 2 | Satake [33] | 48 | 85 | 239 |
| Sawabu [34] | 72 | 94 | 32 | |
| Kawa [35] | 64 | - | 200 | |
| SPan-1 | Kiriyama [36] | 81 | 76 | 64 |
| Chung [37] | 92 | 83 | 67 | |
| Kobayashi [15] | 82 | 85 | 200 | |
| CAM17.1 | Parker [38] | 78 | 76 | 79 |
| Gansauge [39] | 67 | 100 | 91 | |
| TATI | Taccone [40] | 92 | 67 | 36 |
| Pasanen [41] | 41 | 63 | 17 | |
| Aroasio [42] | 63 | - | 52 | |
| Elastase-1 | Zhao [27] | 62 | 67 | 143 |
| GT II | Uemura [43] | 77 | 85 | 13 |
| Tu M2-PK | Ventrucci [44] | 85 | 41 | 60 |
| Cerwenka [45] | 79 | 90 | 38 | |
| Oremek [46] | 71 | 95 | 64 | |
| Mic-1 | Koopmann [47] | 90 | 94 | 50 |
| Koopmann [48] | 71 | 78 | 80 |
2.1.1.2. CA50
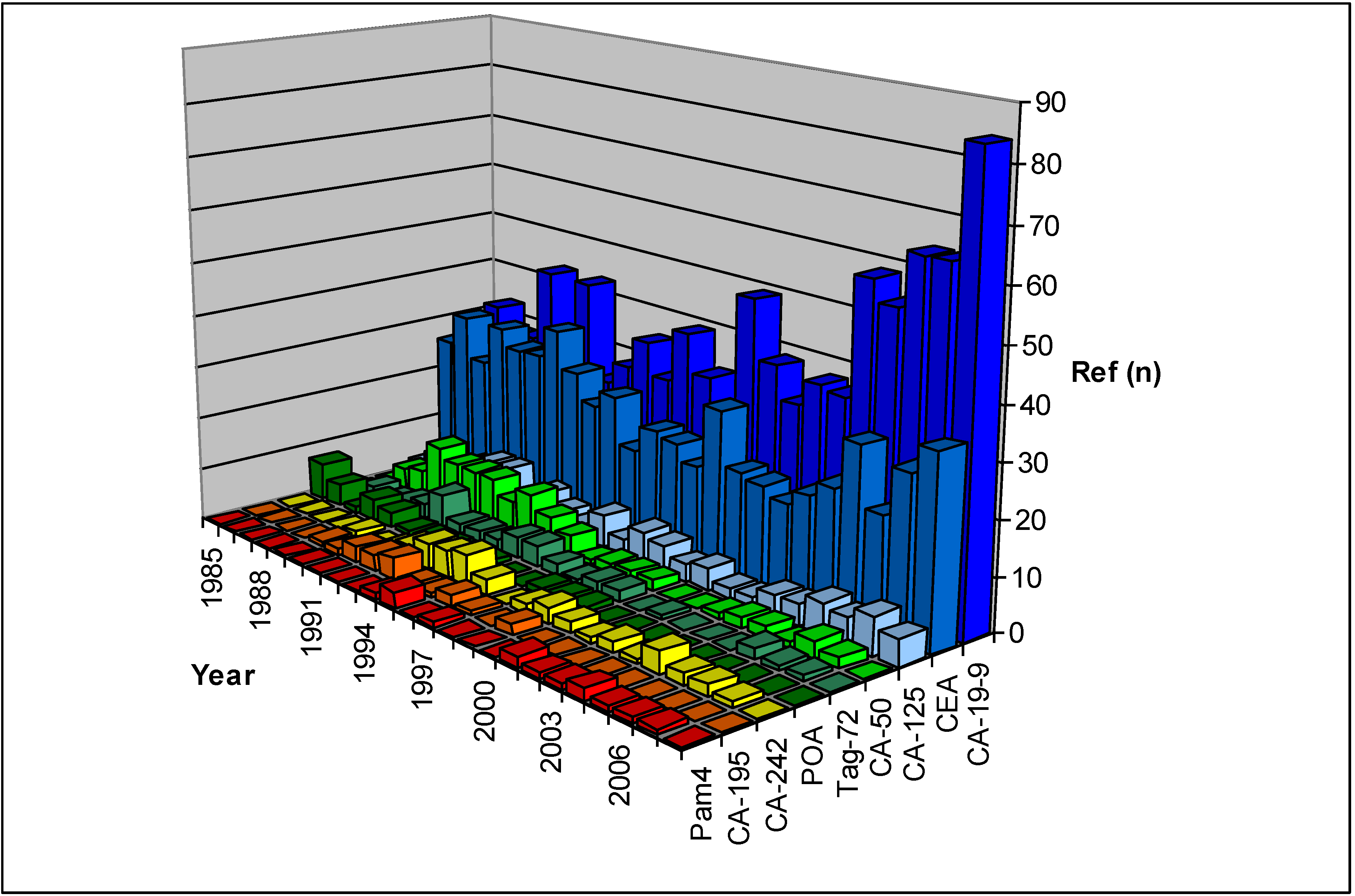
2.1.1.3. CA242
2.1.1.4. CA195
2.1.1.5. CA125
2.1.1.6. Other Carbohydrate Antigens
2.1.2. Glycoproteins
2.1.2.1. CEA
2.1.2.2. POA
2.1.3. Cytokeratins
2.1.3.1. TPA
2.1.3.2. TPS
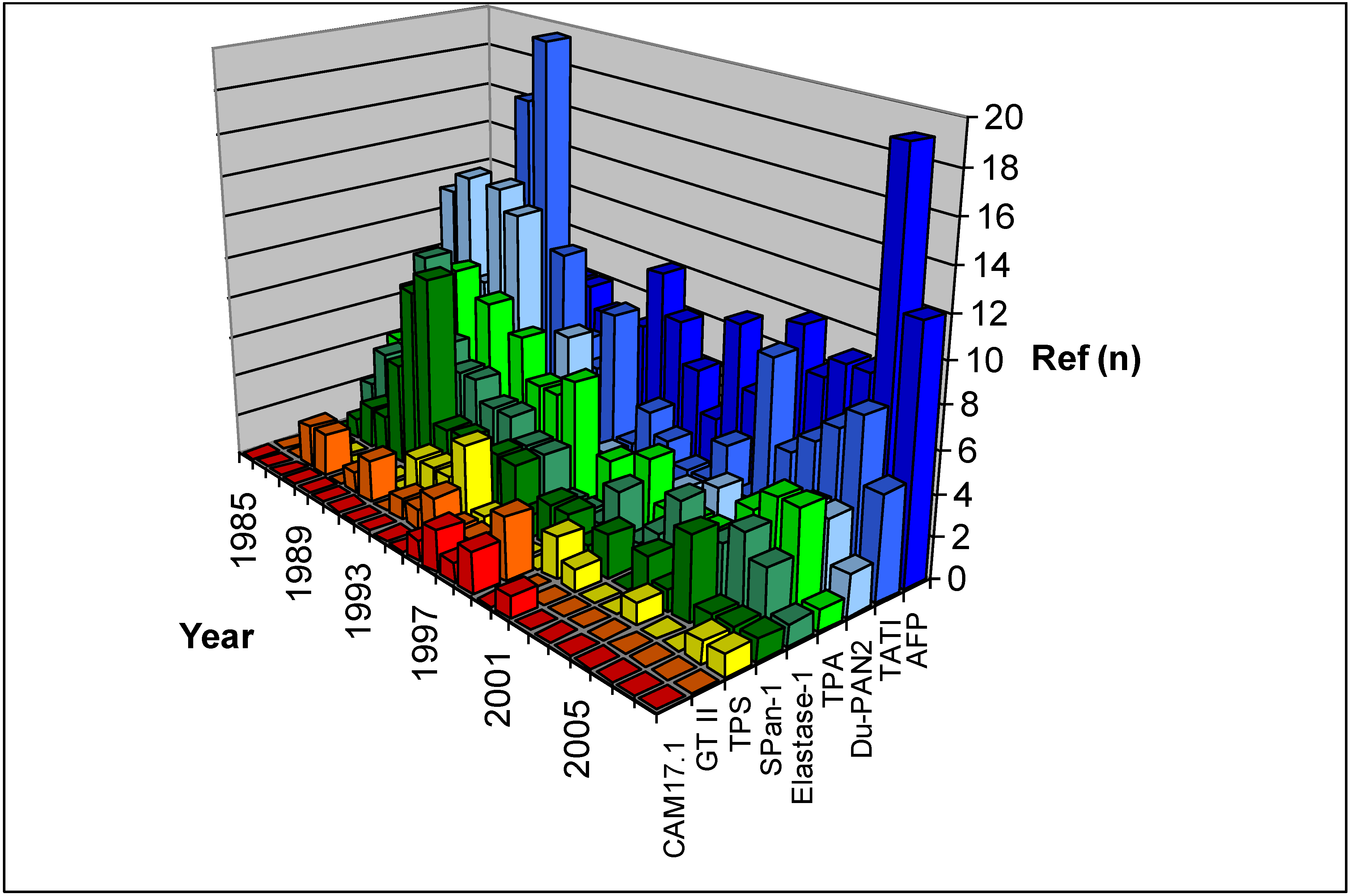
2.1.4. Mucins
2.1.4.1 Du-PAN 2
2.1.4.2 SPan-1
2.1.4.3. CAM17.1
2.2. Enzymatic Proteins
2.2.1. TATI
2.2.2. Tumor M2-Pyruvate Kinase
2.2.3. Elastase-1
2.2.4. Galactosyltransferase Isoenzyme II
2.3. Oncofetal Antigens
2.3.1. AFP
2.4. Other Tumor Markers
2.4.1. EPM-1
2.4.2. OPN
2.4.3. CEACAM1
2.4.4. MIC-1
3. Discussion
4. Conclusions
References
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Murray, T.; Thun, M.J. Cancer statistics, 2008. CA Cancer J. Clin. 2008, 58, 71–96. [Google Scholar] [CrossRef]
- Podolsky, D.K. Serologic markers in the diagnosis and management of pancreatic carcinoma. World J. Surg. 1984, 8, 822–830. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Ching, C.K. Serum diagnostic tests for pancreatic cancer. Baillieres Clin. Gastroenterol. 1990, 4, 833–852. [Google Scholar] [CrossRef]
- Reber, H.A. Pancreatic Cancer: Pathogenesis, Diagnosis, and Treatment; Humana: Totowa, NJ, USA, 1998; pp. 347–353. [Google Scholar]
- Ringel, J.; Lohr, M. The MUC gene family: their role in diagnosis and early detection of pancreatic cancer. Mol. Cancer 2003, 2, 9. [Google Scholar] [CrossRef]
- Eskelinen, M.; Haglund, U. Developments in serologic detection of human pancreatic adenocarcinoma. Scand. J. Gastroenterol. 1999, 34, 833–844. [Google Scholar] [CrossRef]
- Magnani, J.L.; Steplewski, Z.; Koprowski, H.; Ginsburg, V. Identification of the gastrointestinal and pancreatic cancer-associated antigen detected by monoclonal antibody 19-9 in the sera of patients as a mucin. Cancer Res. 1983, 43, 5489–5492. [Google Scholar]
- Herlyn, M.; Sears, H.F.; Steplewski, Z.; Koprowski, H. Monoclonal antibody detection of a circulating tumor-associated antigen. I. Presence of antigen in sera of patients with colorectal, gastric, and pancreatic carcinoma. J. Clin. Immunol. 1982, 2, 135–140. [Google Scholar] [CrossRef]
- Audisio, R.A.; Veronesi, P.; Maisonneuve, P.; Chiappa, A.; Andreoni, B.; Bombardieri, E.; Geraghty, J.G. Clinical relevance of serological markers in the detection and follow-up of pancreatic adenocarcinoma. Surg. Oncol. 1996, 5, 49–63. [Google Scholar] [CrossRef]
- Aoki, H.; Ohnishi, H.; Hama, K.; Ishijima, T.; Satoh, Y.; Hanatsuka, K.; Ohashi, A.; Wada, S.; Miyata, T.; Kita, H.; Yamamoto, H.; Osawa, H.; Sato, K.; Tamada, K.; Yasuda, H.; Mashima, H.; Sugano, K. Autocrine loop between TGF-beta1 and IL-1beta through Smad3- and ERK-dependent pathways in rat pancreatic stellate cells. Am. J. Physiol. Cell Physiol. 2006, 290, C1100–C1108. [Google Scholar]
- Okusaka, T.; Okada, S.; Ishii, H.; Nose, H.; Nakasuka, H.; Nakayama, H.; Nagahama, H. Clinical response to systemic combined chemotherapy with 5-fluorouracil and cisplatin (FP therapy) in patients with advanced pancreatic cancer. Jpn. J. Clin. Oncol. 1996, 26, 215–220. [Google Scholar] [CrossRef]
- Tanaka, M.; Chari, S.; Adsay, V.; Fernandez-del Castillo, C.; Falconi, M.; Shimizu, M.; Yamaguchi, K.; Yamao, K.; Matsuno, S. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 2006, 6, 17–32. [Google Scholar] [CrossRef]
- Steinberg, W. The clinical utility of the CA 19-9 tumor-associated antigen. Am. J. Gastroenterol. 1990, 85, 350–355. [Google Scholar]
- Goonetilleke, K.S.; Siriwardena, A.K. Systematic review of carbohydrate antigen (CA 19-9) as a biochemical marker in the diagnosis of pancreatic cancer. Eur. J. Surg. Oncol. 2007, 33, 266–270. [Google Scholar] [CrossRef]
- Kobayashi, T.; Kawa, S.; Tokoo, M.; Oguchi, H.; Kiyosawa, K.; Furuta, S.; Kanai, M.; Homma, T. Comparative study of CA-50 (time-resolved fluoroimmunoassay), Span-1, and CA19-9 in the diagnosis of pancreatic cancer. Scand. J. Gastroenterol. 1991, 26, 787–797. [Google Scholar] [CrossRef]
- Tian, F.; Appert, H.E.; Myles, J.; Howard, J.M. Prognostic value of serum CA 19-9 levels in pancreatic adenocarcinoma. Ann. Surg. 1992, 215, 350–355. [Google Scholar] [CrossRef]
- Aoki, K.; Okada, S.; Moriyama, N.; Ishii, H.; Nose, H.; Yoshimori, M.; Kosuge, T.; Ozaki, H.; Wakao, F.; Takayasu, K.; et al. Accuracy of computed tomography in determining pancreatic cancer tumor size. Jpn. J. Clin. Oncol. 1994, 24, 85–87. [Google Scholar]
- Okusaka, T.; Yamada, T.; Maekawa, M. Serum tumor markers for pancreatic cancer: the dawn of new era? JOP. J. Pancreas 2006, 7, 332–336. [Google Scholar]
- Jiang, X.T.; Tao, H.Q.; Zou, S.C. Detection of serum tumor markers in the diagnosis and treatment of patients with pancreatic cancer. Hepatobiliary Pancreat. Dis. Int. 2004, 3, 464–468. [Google Scholar]
- Banfi, G.; Zerbi, A.; Pastori, S.; Parolini, D.; Di Carlo, V.; Bonini, P. Behavior of tumor markers CA19.9, CA195, CAM43, CA242, and TPS in the diagnosis and follow-up of pancreatic cancer. Clin. Chem. 1993, 39, 420–423. [Google Scholar]
- Ni, X.G.; Bai, X.F.; Mao, Y.L.; Shao, Y.F.; Wu, J.X.; Shan, Y.; Wang, C.F.; Wang, J.; Tian, Y.T.; Liu, Q.; Xu, D.K.; Zhao, P. The clinical value of serum CEA, CA19-9, and CA242 in the diagnosis and prognosis of pancreatic cancer. Eur. J. Surg. Oncol. 2005, 31, 164–169. [Google Scholar] [CrossRef]
- Andicoechea, A.; Vizoso, F.; Alexandre, E.; Martinez, A.; Cruz Diez, M.; Riera, L.; Martinez, E.; Ruibal, A. Comparative study of carbohydrate antigen 195 and carcinoembryonic antigen for the diagnosis of pancreatic carcinoma. World J. Surg. 1999, 23, 227–231; discussion 231–222. [Google Scholar] [CrossRef]
- Haglund, C. Tumour marker antigen CA125 in pancreatic cancer: a comparison with CA19-9 and CEA. Br. J. Cancer 1986, 54, 897–901. [Google Scholar] [CrossRef]
- Duraker, N.; Hot, S.; Polat, Y.; Hobek, A.; Gencler, N.; Urhan, N. CEA, CA 19-9, and CA 125 in the differential diagnosis of benign and malignant pancreatic diseases with or without jaundice. J Surg. Oncol. 2007, 95, 142–147. [Google Scholar] [CrossRef]
- Gold, D.V.; Modrak, D.E.; Ying, Z.; Cardillo, T.M.; Sharkey, R.M.; Goldenberg, D.M. New MUC1 serum immunoassay differentiates pancreatic cancer from pancreatitis. J. Clin. Oncol. 2006, 24, 252–258. [Google Scholar]
- Pasquali, C.; Sperti, C.; D'Andrea, A.A.; Costantino, V.; Filipponi, C.; Pedrazzoli, S. Clinical value of serum TAG-72 as a tumor marker for pancreatic carcinoma. Comparison with CA 19-9. Int. J. Pancreatol. 1994, 15, 171–177. [Google Scholar]
- Zhao, X.Y.; Yu, S.Y.; Da, S.P.; Bai, L.; Guo, X.Z.; Dai, X.J.; Wang, Y.M. A clinical evaluation of serological diagnosis for pancreatic cancer. World J. Gastroenterol. 1998, 4, 147–149. [Google Scholar]
- Nishida, K.; Sugiura, M.; Yoshikawa, T.; Kondo, M. Enzyme immunoassay of pancreatic oncofetal antigen (POA) as a marker of pancreatic cancer. Gut 1985, 26, 450–455. [Google Scholar] [CrossRef]
- Panucci, A.; Fabris, C.; Del Favero, G.; Basso, D.; Marchioro, L.; Piccoli, A.; Burlina, A.; Naccarato, R. Tissue polypeptide antigen (TPA) in pancreatic cancer diagnosis. Br. J. Cancer 1985, 52, 801–803. [Google Scholar] [CrossRef]
- Benini, L.; Cavallini, G.; Zordan, D.; Rizzotti, P.; Rigo, L.; Brocco, G.; Perobelli, L.; Zanchetta, M.; Pederzoli, P.; Scuro, L.A. A clinical evaluation of monoclonal (CA19-9, CA50, CA12-5) and polyclonal (CEA, TPA) antibody-defined antigens for the diagnosis of pancreatic cancer. Pancreas 1988, 3, 61–66. [Google Scholar] [CrossRef]
- Pasanen, P.A.; Eskelinen, M.; Partanen, K.; Pikkarainen, P.; Penttila, I. Clinical evaluation of tissue polypeptide antigen (TPA) in the diagnosis of pancreatic carcinoma. Anticancer Res. 1993, 13, 1883–1887. [Google Scholar]
- Pasanen, P.A.; Eskelinen, M.; Partanen, K.; Pikkarainen, P.; Penttila, I.; Alhava, E. Multivariate analysis of six serum tumor markers (CEA, CA 50, CA 242, TPA, TPS, TATI) and conventional laboratory tests in the diagnosis of hepatopancreatobiliary malignancy. Anticancer Res. 1995, 15, 2731–2737. [Google Scholar]
- Satake, K.; Takeuchi, T. Comparison of CA19-9 with other tumor markers in the diagnosis of cancer of the pancreas. Pancreas 1994, 9, 720–724. [Google Scholar] [CrossRef]
- Sawabu, N.; Toya, D.; Takemori, Y.; Hattori, N.; Fukui, M. Measurement of a pancreatic cancer-associated antigen (DU-PAN-2) detected by a monoclonal antibody in sera of patients with digestive cancers. Int. J. Cancer 1986, 37, 693–696. [Google Scholar] [CrossRef]
- Kawa, S.; Oguchi, H.; Kobayashi, T.; Tokoo, M.; Furuta, S.; Kanai, M.; Homma, T. Elevated serum levels of Dupan-2 in pancreatic cancer patients negative for Lewis blood group phenotype. Br. J. Cancer 1991, 64, 899–902. [Google Scholar] [CrossRef]
- Kiriyama, S.; Hayakawa, T.; Kondo, T.; Shibata, T.; Kitagawa, M.; Ono, H.; Sakai, Y. Usefulness of a new tumor marker, Span-1, for the diagnosis of pancreatic cancer. Cancer 1990, 65, 1557–1561. [Google Scholar] [CrossRef]
- Chung, Y.S.; Ho, J.J.; Kim, Y.S.; Tanaka, H.; Nakata, B.; Hiura, A.; Motoyoshi, H.; Satake, K.; Umeyama, K. The detection of human pancreatic cancer-associated antigen in the serum of cancer patients. Cancer 1987, 60, 1636–1643. [Google Scholar] [CrossRef]
- Parker, N.; Makin, C.A.; Ching, C.K.; Eccleston, D.; Taylor, O.M.; Milton, J.D.; Rhodes, J.M. A new enzyme-linked lectin/mucin antibody sandwich assay (CAM 17.1/WGA) assessed in combination with CA 19-9 and peanut lectin binding assay for the diagnosis of pancreatic cancer. Cancer 1992, 70, 1062–1068. [Google Scholar] [CrossRef]
- Gansauge, F.; Gansauge, S.; Parker, N.; Beger, M.I.; Poch, B.; Link, K.H.; Safi, F.; Beger, H.G. CAM 17.1--a new diagnostic marker in pancreatic cancer. Br. J. Cancer 1996, 74, 1997–2002. [Google Scholar]
- Taccone, W.; Mazzon, W.; Belli, M. Evaluation of TATI and other markers in solid tumors. Scand. J. Clin. Lab. Invest. Suppl. 1991, 207, 25–32. [Google Scholar] [CrossRef]
- Pasanen, P.A.; Eskelinen, M.; Partanen, K.; Pikkarainen, P.; Penttila, I.; Alhava, E. Tumour-associated trypsin inhibitor in the diagnosis of pancreatic carcinoma. J. Cancer Res. Clin. Oncol. 1994, 120, 494–497. [Google Scholar] [CrossRef]
- Aroasio, E.; Piantino, P. Tumor-associated trypsin inhibitor in pancreatic diseases. Scand. J. Clin. Lab. Invest. Suppl. 1991, 207, 71–73. [Google Scholar] [CrossRef]
- Uemura, M.; Winant, R.C.; Brandt, A.E. Immunoassay of serum galactosyltransferase isoenzyme II in cancer patients and control subjects. Cancer Res. 1988, 48, 5335–5341. [Google Scholar]
- Ventrucci, M.; Cipolla, A.; Racchini, C.; Casadei, R.; Simoni, P.; Gullo, L. Tumor M2-pyruvate kinase, a new metabolic marker for pancreatic cancer. Dig. Dis. Sci. 2004, 49, 1149–1155. [Google Scholar] [CrossRef]
- Cerwenka, H.; Aigner, R.; Bacher, H.; Werkgartner, G.; el-Shabrawi, A.; Quehenberger, F.; Mischinger, H.J. TUM2-PK (pyruvate kinase type tumor M2), CA19-9 and CEA in patients with benign, malignant and metastasizing pancreatic lesions. Anticancer Res. 1999, 19, 849–851. [Google Scholar]
- Oremek, G.M.; Eigenbrodt, E.; Radle, J.; Zeuzem, S.; Seiffert, U.B. Value of the serum levels of the tumor marker TUM2-PK in pancreatic cancer. Anticancer Res. 1997, 17, 3031–3033. [Google Scholar]
- Koopmann, J.; Rosenzweig, C.N.; Zhang, Z.; Canto, M.I.; Brown, D.A.; Hunter, M.; Yeo, C.; Chan, D.W.; Breit, S.N.; Goggins, M. Serum markers in patients with resectable pancreatic adenocarcinoma: macrophage inhibitory cytokine 1 versus CA19-9. Clin. Cancer Res. 2006, 12, 442–446. [Google Scholar] [CrossRef]
- Koopmann, J.; Buckhaults, P.; Brown, D.A.; Zahurak, M.L.; Sato, N.; Fukushima, N.; Sokoll, L.J.; Chan, D.W.; Yeo, C.J.; Hruban, R.H.; Breit, S.N.; Kinzler, K.W.; Vogelstein, B.; Goggins, M. Serum macrophage inhibitory cytokine 1 as a marker of pancreatic and other periampullary cancers. Clin. Cancer Res. 2004, 10, 2386–2392. [Google Scholar] [CrossRef]
- Palsson, B.; Masson, P.; Andren-Sandberg, A. The influence of cholestasis on CA 50 and CA 242 in pancreatic cancer and benign biliopancreatic diseases. Scand. J. Gastroenterol. 1993, 28, 981–987. [Google Scholar] [CrossRef]
- Johansson, C.; Nilsson, O.; Baeckstrom, D.; Jansson, E.L.; Lindholm, L. Novel epitopes on the CA50-carrying antigen: chemical and immunochemical studies. Tumour Biol. 1991, 12, 159–170. [Google Scholar] [CrossRef]
- Nilsson, O.; Johansson, C.; Glimelius, B.; Persson, B.; Norgaard-Pedersen, B.; Andren-Sandberg, A.; Lindholm, L. Sensitivity and specificity of CA242 in gastro-intestinal cancer. A comparison with CEA, CA50 and CA 19-9. Br. J. Cancer 1992, 65, 215–221. [Google Scholar] [CrossRef]
- Kuus-Reichel, K.; Knott, C.L.; McCormack, R.T.; Guido, M.S.; Beebe, A. Production of IgG monoclonal antibodies to the tumor-associated antigen, CA-195. Hybridoma 1994, 13, 31–36. [Google Scholar] [CrossRef]
- Fukuta, S.; Magnani, J.L.; Gaur, P.K.; Ginsburg, V. Monoclonal antibody CC3C195, which detects cancer-associated antigens in serum, binds to the human Lea blood group antigen and to its sialylated derivative. Arch. Biochem. Biophys. 1987, 255, 214–216. [Google Scholar] [CrossRef]
- Dietel, M.; Arps, H.; Klapdor, R.; Muller-Hagen, S.; Sieck, M.; Hoffmann, L. Antigen detection by the monoclonal antibodies CA 19-9 and CA 125 in normal and tumor tissue and patients' sera. J. Cancer Res. Clin. Oncol. 1986, 111, 257–265. [Google Scholar] [CrossRef]
- Funakoshi, Y.; Suzuki, T. Glycobiology in the cytosol: the bitter side of a sweet world. Biochim. Biophys. Acta 2009, 1790, 81–94. [Google Scholar]
- Hammarstrom, S.; Shively, J.E.; Paxton, R.J.; Beatty, B.G.; Larsson, A.; Ghosh, R.; Bormer, O.; Buchegger, F.; Mach, J.P.; Burtin, P.; et al. Antigenic sites in carcinoembryonic antigen. Cancer Res. 1989, 49, 4852–4858. [Google Scholar]
- Friess, H.; Buchler, M.; Auerbach, B.; Weber, A.; Malfertheiner, P.; Hammer, K.; Madry, N.; Greiner, S.; Bosslet, K.; Beger, H.G. CA 494--a new tumor marker for the diagnosis of pancreatic cancer. Int. J. Cancer 1993, 53, 759–763. [Google Scholar] [CrossRef]
- Guadagni, F.; Roselli, M.; Cosimelli, M.; Ferroni, P.; Spila, A.; Cavaliere, F.; Casaldi, V.; Wappner, G.; Abbolito, M.R.; Greiner, J.W.; et al. CA 72-4 serum marker--a new tool in the management of carcinoma patients. Cancer Invest. 1995, 13, 227–238. [Google Scholar] [CrossRef]
- Basso, D.; Fabris, C.; Panucci, A.; Del Favero, G.; Angonese, C.; Plebani, M.; Petrin, P.; Burlina, A.; Naccarato, R. Tissue polypeptide antigen, galactosyltransferase isoenzyme II and pancreatic oncofetal antigen serum determination: role in pancreatic cancer diagnosis. Int. J. Pancreatol 1988, 3 (Suppl. 1), S95–S100. [Google Scholar]
- Franke, W.W.; Schmid, E.; Osborn, M.; Weber, K. Intermediate-sized filaments of human endothelial cells. J. Cell Biol. 1979, 81, 570–580. [Google Scholar] [CrossRef]
- Barak, V.; Goike, H.; Panaretakis, K.W.; Einarsson, R. Clinical utility of cytokeratins as tumor markers. Clin. Biochem. 2004, 37, 529–540. [Google Scholar] [CrossRef]
- Bjorklund, B.; Bjorklund, V. Specificity and basis of the tissue polypeptide antigen. Cancer Detect. Prev. 1983, 6, 41–50. [Google Scholar]
- Metzgar, R.S.; Gaillard, M.T.; Levine, S.J.; Tuck, F.L.; Bossen, E.H.; Borowitz, M.J. Antigens of human pancreatic adenocarcinoma cells defined by murine monoclonal antibodies. Cancer Res. 1982, 42, 601–608. [Google Scholar]
- Takasaki, H.; Uchida, E.; Tempero, M.A.; Burnett, D.A.; Metzgar, R.S.; Pour, P.M. Correlative study on expression of CA 19-9 and DU-PAN-2 in tumor tissue and in serum of pancreatic cancer patients. Cancer Res. 1988, 48, 1435–1438. [Google Scholar]
- Frena, A. SPan-1 and exocrine pancreatic carcinoma. The clinical role of a new tumor marker. Int. J. Biol. Markers 2001, 16, 189–197. [Google Scholar]
- Ventrucci, M.; Pezzilli, R.; Gullo, L.; Plate, L.; Sprovieri, G.; Barbara, L. Role of serum pancreatic enzyme assays in diagnosis of pancreatic disease. Dig. Dis. Sci. 1989, 34, 39–45. [Google Scholar] [CrossRef]
- Masson, P.; Palsson, B.; Andren-Sandberg, A. Evaluation of CEA, CA 19-9, CA-50, CA-195, and TATI with special reference to pancreatic disorders. Int. J. Pancreatol. 1991, 8, 333–344. [Google Scholar]
- Tatsuta, M.; Yamamura, H.; Noguchi, S.; Ichii, M.; Iishi, H.; Okuda, S. Values of serum carcinoembryonic antigen and elastase 1 in diagnosis of pancreatic carcinoma. Gut 1984, 25, 1347–1351. [Google Scholar] [CrossRef]
- Prall, F.; Nollau, P.; Neumaier, M.; Haubeck, H.D.; Drzeniek, Z.; Helmchen, U.; Loning, T.; Wagener, C. CD66a (BGP), an adhesion molecule of the carcinoembryonic antigen family, is expressed in epithelium, endothelium, and myeloid cells in a wide range of normal human tissues. J. Histochem. Cytochem. 1996, 44, 35–41. [Google Scholar] [CrossRef]
- Simeone, D.M.; Ji, B.; Banerjee, M.; Arumugam, T.; Li, D.; Anderson, M.A.; Bamberger, A.M.; Greenson, J.; Brand, R.E.; Ramachandran, V.; Logsdon, C.D. CEACAM1, a novel serum biomarker for pancreatic cancer. Pancreas 2007, 34, 436–443. [Google Scholar] [CrossRef]
- Roulston, J.E. Novel tumour markers: a diagnostic role in pancreatic cancer? Br. J. Cancer 1994, 70, 389–390. [Google Scholar] [CrossRef]
- Chan, M.H.; Shing, M.M.; Poon, T.C.; Johnson, P.J.; Lam, C.W. Alpha-fetoprotein variants in a case of pancreatoblastoma. Ann. Clin. Biochem. 2000, 37 (Pt 5), 681–685. [Google Scholar]
- Kohda, E.; Iseki, M.; Ikawa, H.; Endoh, M.; Yokoyama, J.; Mukai, M.; Hata, J.; Yamazaki, H.; Miyauchi, J.; Saeki, M. Pancreatoblastoma. Three original cases and review of the literature. Acta Radiol. 2000, 41, 334–337. [Google Scholar] [CrossRef]
- Dippold, W.G.; Bernhard, H.; Klingel, R.; Dienes, H.P.; Kron, G.; Schneider, B.; Knuth, A.; Meyer zum Buschenfelde, K.H. A common epithelial cell surface antigen (EPM-1) on gastrointestinal tumors and in human sera. Cancer Res. 1987, 47, 3873–3879. [Google Scholar]
- Koopmann, J.; Fedarko, N.S.; Jain, A.; Maitra, A.; Iacobuzio-Donahue, C.; Rahman, A.; Hruban, R.H.; Yeo, C.J.; Goggins, M. Evaluation of osteopontin as biomarker for pancreatic adenocarcinoma. Cancer Epidemiol. Biomarkers Prev. 2004, 13, 487–491. [Google Scholar]
- Bootcov, M.R.; Bauskin, A.R.; Valenzuela, S.M.; Moore, A.G.; Bansal, M.; He, X.Y.; Zhang, H.P.; Donnellan, M.; Mahler, S.; Pryor, K.; Walsh, B.J.; Nicholson, R.C.; Fairlie, W.D.; Por, S.B.; Robbins, J.M.; Breit, S.N. MIC-1, a novel macrophage inhibitory cytokine, is a divergent member of the TGF-beta superfamily. Proc. Natl. Acad. Sci. USA 1997, 94, 11514–11519. [Google Scholar] [CrossRef]
- Satake, K.; Chung, Y.S.; Umeyama, K.; Takeuchi, T.; Kim, Y.S. The possibility of diagnosing small pancreatic cancer (less than 4.0 cm) by measuring various serum tumor markers. A retrospective study. Cancer 1991, 68, 149–152. [Google Scholar] [CrossRef]
- Grote, T.; Logsdon, C.D. Progress on molecular markers of pancreatic cancer. Curr. Opin. Gastroenterol. 2007, 23, 508–514. [Google Scholar] [CrossRef]
- Pilarsky, C.; Ammerpohl, O.; Sipos, B.; Dahl, E.; Hartmann, A.; Wellmann, A.; Braunschweig, T.; Lohr, M.; Jesnowski, R.; Friess, H.; Wente, M.N.; Kristiansen, G.; Jahnke, B.; Denz, A.; Ruckert, F.; Schackert, H.K.; Kloppel, G.; Kalthoff, H.; Saeger, H.D.; Grutzmann, R. Activation of Wnt signalling in stroma from pancreatic cancer identified by gene expression profiling. J. Cell. Mol. Med. 2008, 12, 2823–2835. [Google Scholar] [CrossRef]
- Pilarsky, C.; Wenzig, M.; Specht, T.; Saeger, H.D.; Grutzmann, R. Identification and validation of commonly overexpressed genes in solid tumors by comparison of microarray data. Neoplasia 2004, 6, 744–750. [Google Scholar] [CrossRef]
- Grutzmann, R.; Foerder, M.; Alldinger, I.; Staub, E.; Brummendorf, T.; Ropcke, S.; Li, X.; Kristiansen, G.; Jesnowski, R.; Sipos, B.; Lohr, M.; Luttges, J.; Ockert, D.; Kloppel, G.; Saeger, H.D.; Pilarsky, C. Gene expression profiles of microdissected pancreatic ductal adenocarcinoma. Virchows Arch. 2003, 443, 508–517. [Google Scholar] [CrossRef]
- Guo, J.; Wang, W.; Liao, P.; Lou, W.; Ji, Y.; Zhang, C.; Wu, J.; Zhang, S. Identification of serum biomarkers for pancreatic adenocarcinoma by proteomic analysis. Cancer Sci. 2009, 100, 2292–2301. [Google Scholar] [CrossRef]
- Chen, R.; Pan, S.; Brentnall, T.A.; Aebersold, R. Proteomic profiling of pancreatic cancer for biomarker discovery. Mol. Cell. Proteomics 2005, 4, 523–533. [Google Scholar] [CrossRef]
- Harsha, H.C.; Kandasamy, K.; Ranganathan, P.; Rani, S.; Ramabadran, S.; Gollapudi, S.; Balakrishnan, L.; Dwivedi, S.B.; Telikicherla, D.; Selvan, L.D.; Goel, R.; Mathivanan, S.; Marimuthu, A.; Kashyap, M.; Vizza, R.F.; Mayer, R.J.; Decaprio, J.A.; Srivastava, S.; Hanash, S.M.; Hruban, R.H.; Pandey, A. A compendium of potential biomarkers of pancreatic cancer. PLoS Med. 2009, 6, e1000046. [Google Scholar] [CrossRef]
- Ruckert, F.; Hennig, M.; Petraki, C.D.; Wehrum, D.; Distler, M.; Denz, A.; Schroder, M.; Dawelbait, G.; Kalthoff, H.; Saeger, H.D.; Diamandis, E.P.; Pilarsky, C.; Grutzmann, R. Co-expression of KLK6 and KLK10 as prognostic factors for survival in pancreatic ductal adenocarcinoma. Br. J. Cancer 2008, 99, 1484–1492. [Google Scholar] [CrossRef]
- Olive, K.P.; Jacobetz, M.A.; Davidson, C.J.; Gopinathan, A.; McIntyre, D.; Honess, D.; Madhu, B.; Goldgraben, M.A.; Caldwell, M.E.; Allard, D.; Frese, K.K.; Denicola, G.; Feig, C.; Combs, C.; Winter, S.P.; Ireland-Zecchini, H.; Reichelt, S.; Howat, W.J.; Chang, A.; Dhara, M.; Wang, L.; Ruckert, F.; Grutzmann, R.; Pilarsky, C.; Izeradjene, K.; Hingorani, S.R.; Huang, P.; Davies, S.E.; Plunkett, W.; Egorin, M.; Hruban, R.H.; Whitebread, N.; McGovern, K.; Adams, J.; Iacobuzio-Donahue, C.; Griffiths, J.; Tuveson, D.A. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 2009, 324, 1457–1461. [Google Scholar] [CrossRef]
- Conrad, K.; Bartsch, H.; Canzler, U.; Pilarsky, C.; Grutzmann, R.; Bachmann, M. Search for and identification of novel tumor-associated autoantigens. Methods Mol. Biol. 576, 213–230.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rückert, F.; Pilarsky, C.; Grützmann, R. Serum Tumor Markers in Pancreatic Cancer—Recent Discoveries. Cancers 2010, 2, 1107-1124. https://doi.org/10.3390/cancers2021107
Rückert F, Pilarsky C, Grützmann R. Serum Tumor Markers in Pancreatic Cancer—Recent Discoveries. Cancers. 2010; 2(2):1107-1124. https://doi.org/10.3390/cancers2021107
Chicago/Turabian StyleRückert, Felix, Christian Pilarsky, and Robert Grützmann. 2010. "Serum Tumor Markers in Pancreatic Cancer—Recent Discoveries" Cancers 2, no. 2: 1107-1124. https://doi.org/10.3390/cancers2021107
APA StyleRückert, F., Pilarsky, C., & Grützmann, R. (2010). Serum Tumor Markers in Pancreatic Cancer—Recent Discoveries. Cancers, 2(2), 1107-1124. https://doi.org/10.3390/cancers2021107




