Liquid Core ARROW Waveguides: A Promising Photonic Structure for Integrated Optofluidic Microsensors
Abstract
:1. Introduction
2. Operation Principles and Simulations
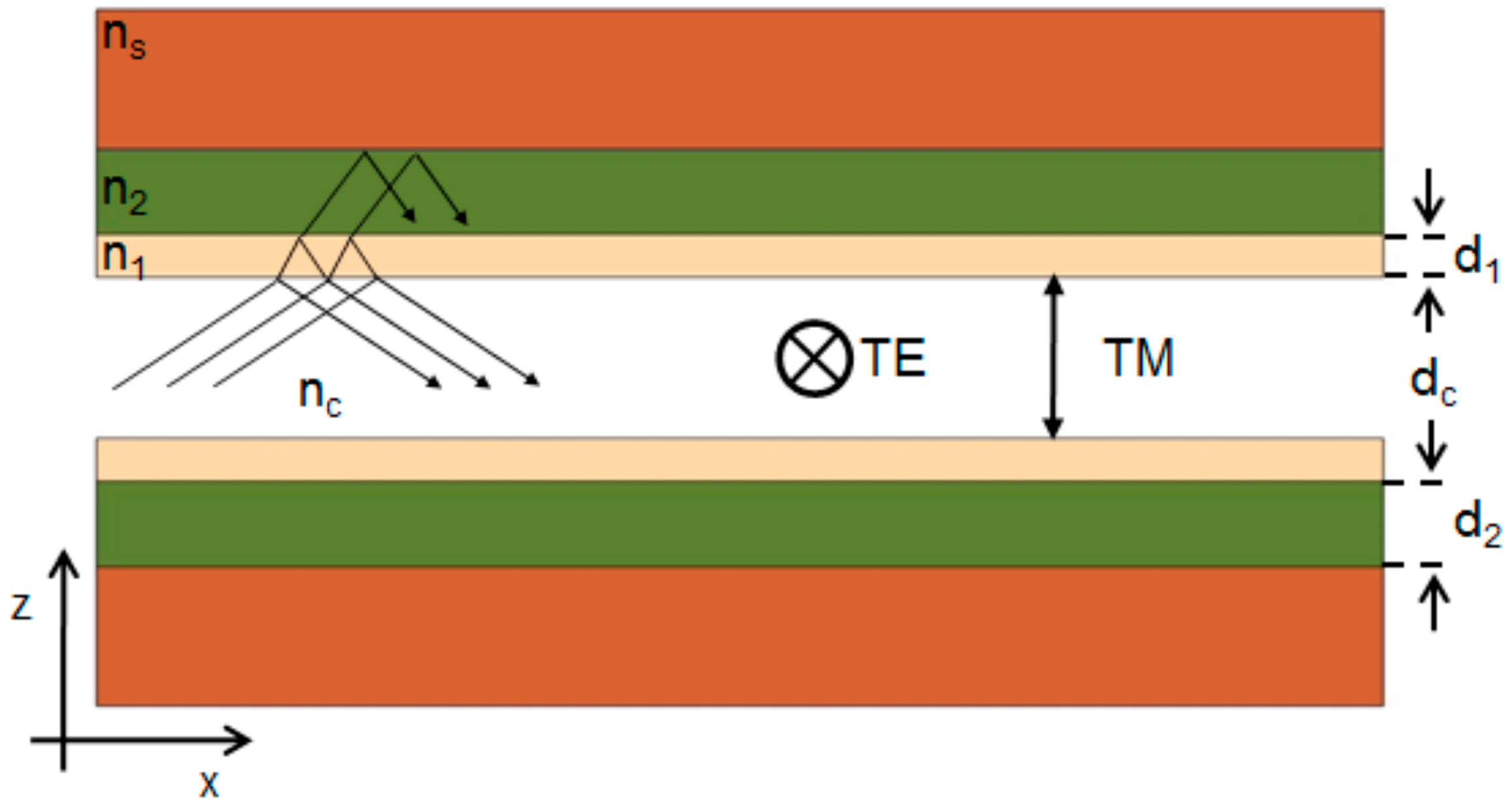
2.1. 1-D Liquid ARROW
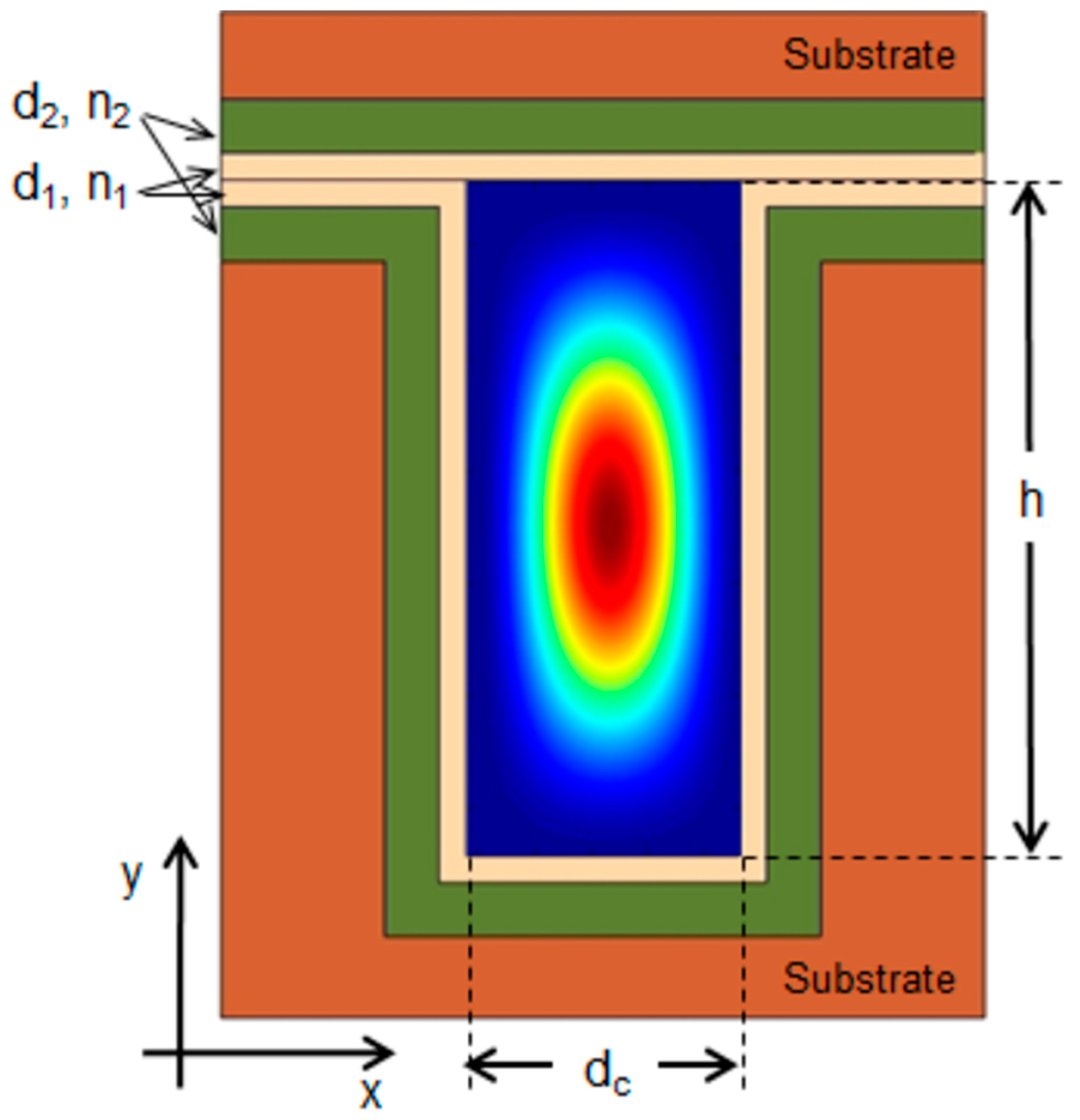
2.2. 2-D Liquid ARROW
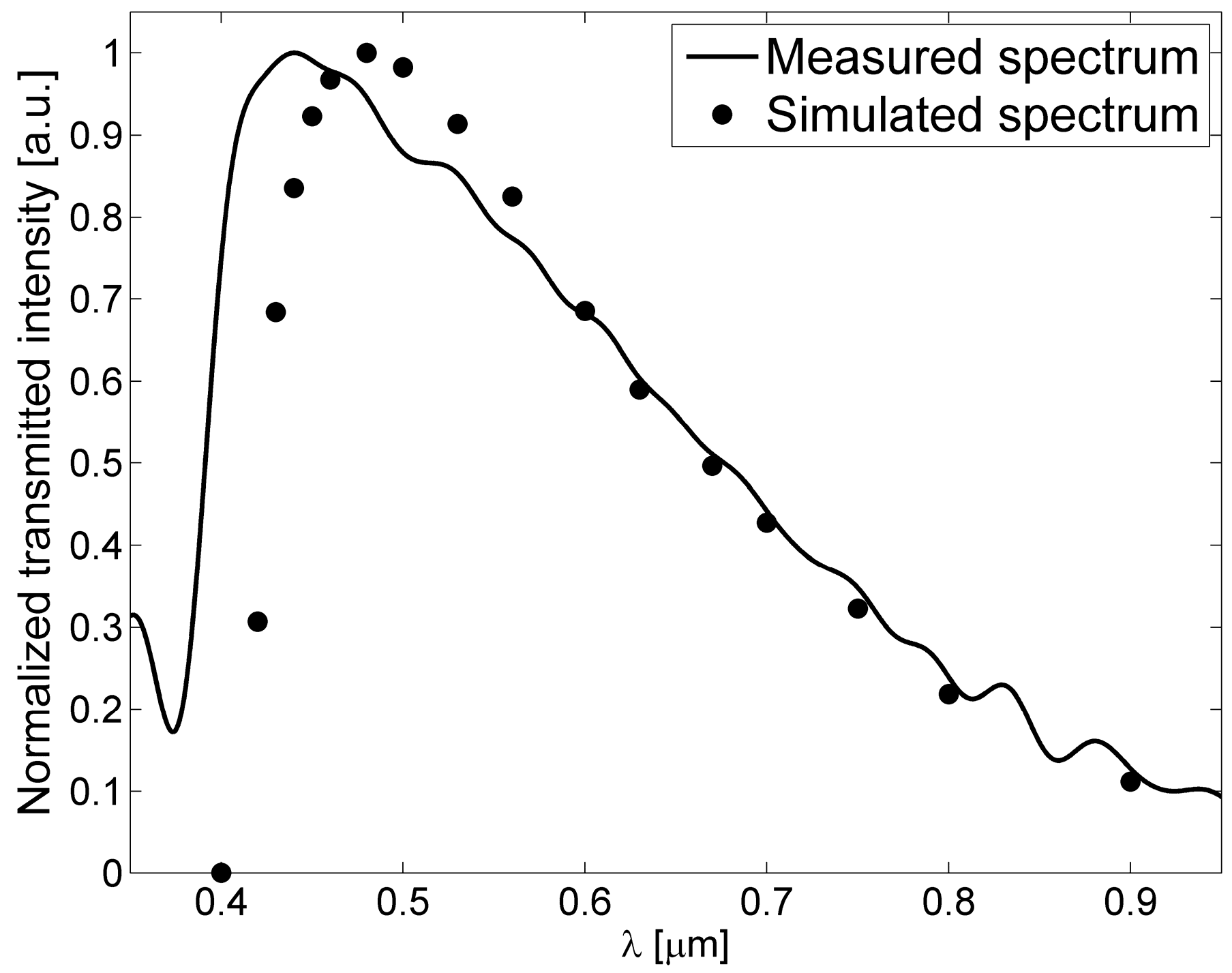
3. Fabrication and Characterization
4. ARROW-Based Devices and Applications
4.1. Liquid ARROW Waveguides as Optofluidic Sensors
4.2. Interferometric Optofluidic Devices for Sensing Applications
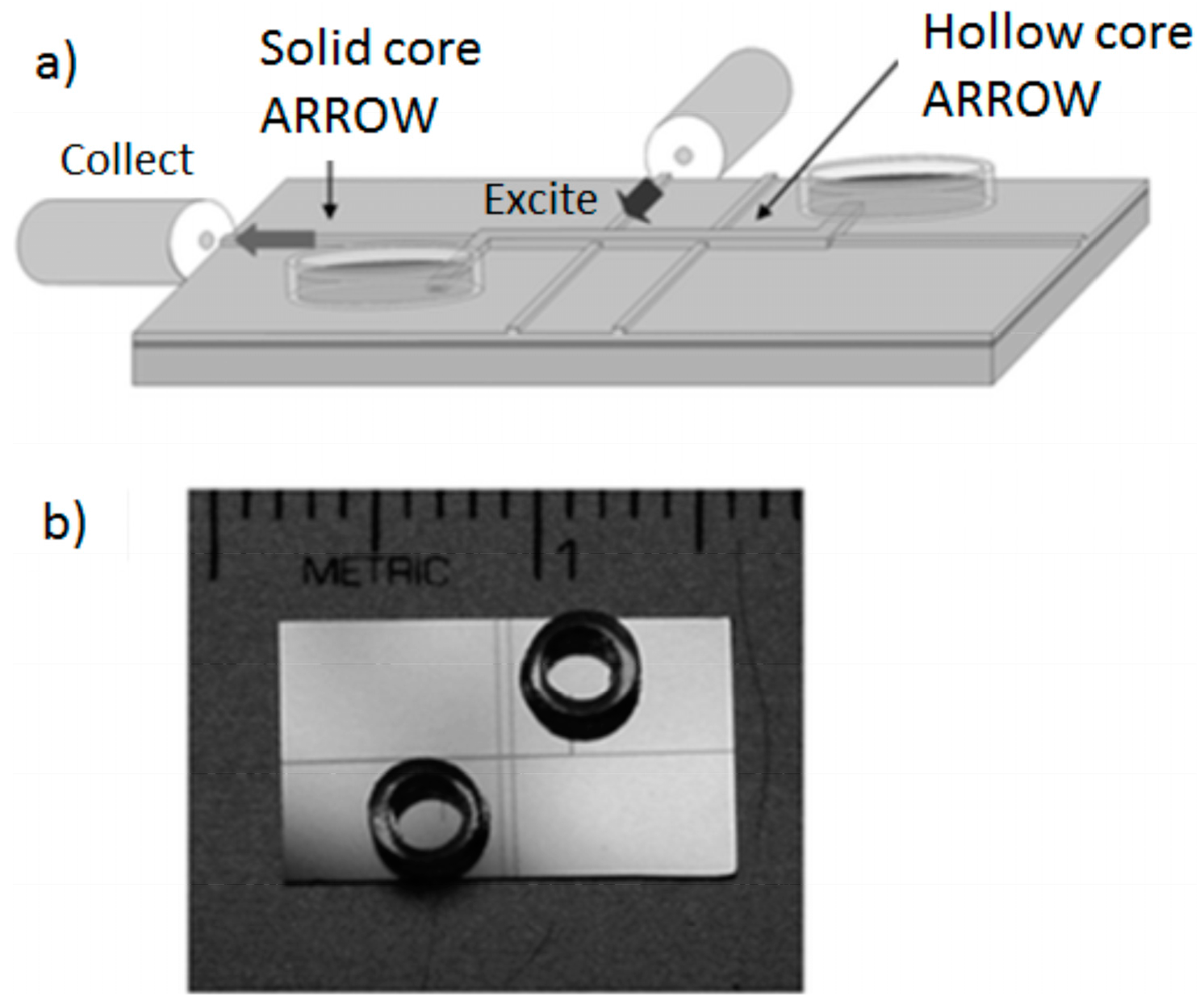
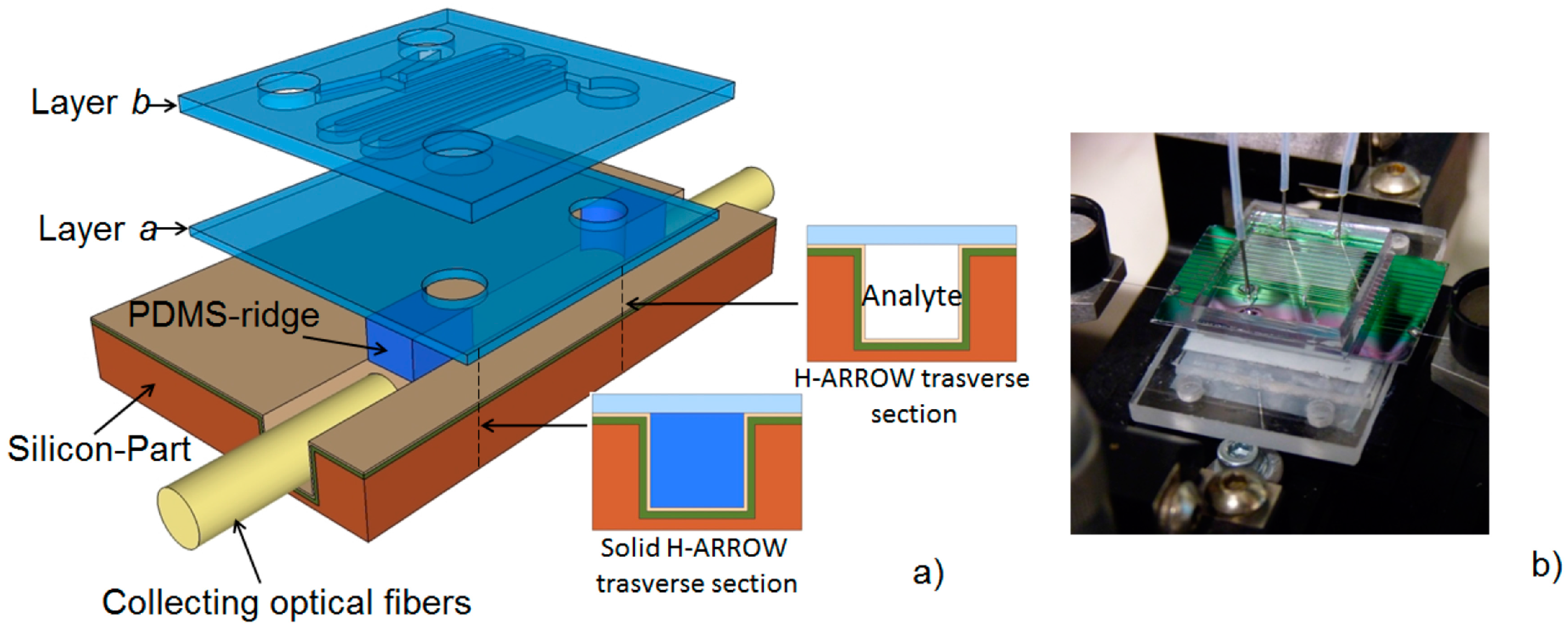
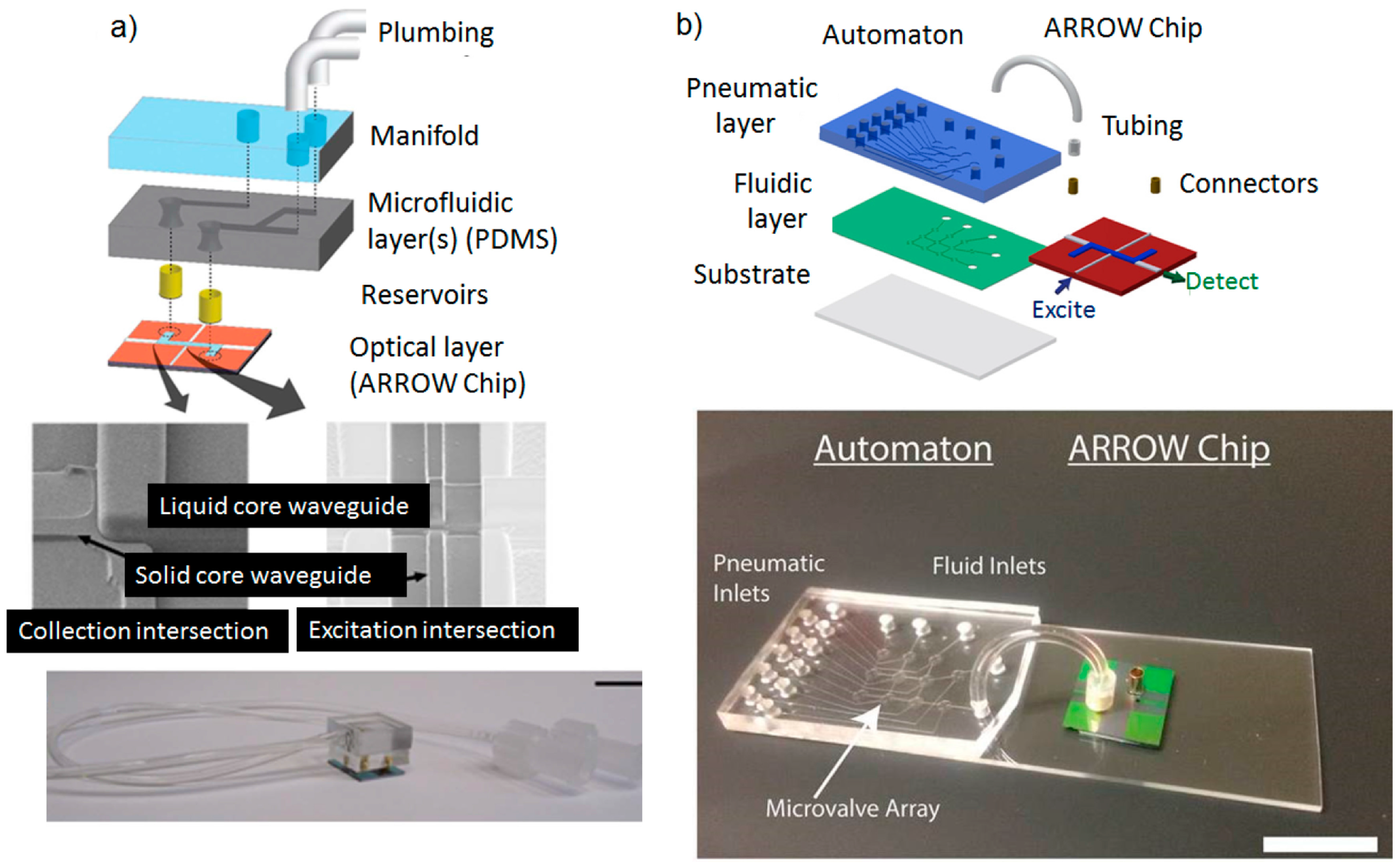
4.3. Planar Integrated ARROW Platform
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Monat, C.; Domachuk, P.; Eggleton, B.J. Integrated optofluidics: A new river of light. Nat. Photon. 2007, 1, 106–114. [Google Scholar] [CrossRef]
- Psaltis, D.; Quake, S.R.; Yang, C. Developing optofluidic technology through the fusion of microfluidics and optics. Nature 2006, 442, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; White, I.M. Optofluidic microsystems for chemical and biological analysis. Nat. Photon. 2011, 5, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Persichetti, G.; Bernini, R. Optofluidic Approaches for Enhanced Microsensor Performances. Sensors 2015, 15, 465–484. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.; Hawkins, A.R. Optofluidic waveguides: I. Concepts and implementations. Microfluid. Nanofluid. 2008, 4, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Bernini, R.; Campopiano, S.; Zeni, L. Silicon micromachined hollow optical waveguides for sensing applications. IEEE J. Sel. Top. Quant. Electron. 2002, 8, 106–110. [Google Scholar] [CrossRef]
- Erickson, D.; Rockwood, T.; Emery, T.; Scherer, A.; Psaltis, D. Nanofluidic tuning of photonic crystal circuits. Opt. Lett. 2006, 31, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Surdo, S.; Merlo, S.; Carpignano, F.; Strambini, L.M.; Trono, C.; Giannetti, A.; Baldini, F.; Barillaro, G. Optofluidic microsystems with integrated vertical one-dimensional photonic crystals for chemical analysis. Lab Chip 2012, 12, 4403–4415. [Google Scholar] [CrossRef] [PubMed]
- Almeida, V.R.; Xu, Q.; Barrios, C.A.; Lipson, M. Guiding and confining light in void nanostructure. Opt. Lett. 2004, 29, 1209–1211. [Google Scholar] [CrossRef] [PubMed]
- Barrios, C.A. Optical Slot-Waveguide Based Biochemical Sensors. Sensors 2009, 9, 4751–4765. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.; Yin, D.; Barber, J.P.; Hawkins, A.R. Hollow-core waveguides and 2-D waveguide arrays for integrated optics of gases and liquids. IEEE J. Sel. Top. Quant. Electron. 2005, 11, 519–527. [Google Scholar] [CrossRef]
- Delonge, T.; Fouckhardt, H. Integrated optical detection cell based on Bragg reflecting waveguides. J. Chromatogr. A 1995, 716, 135–139. [Google Scholar] [CrossRef]
- Baba, T.; Kokubun, Y. Dispersion and radiation loss characteristics of antiresonant reflecting optical waveguides – numerical results and analytical expressions. IEEE J. Quantum. Electron. 1992, 28, 1689–1700. [Google Scholar] [CrossRef]
- Litchinitser, N.M.; Abeeluck, A.K.; Headley, C.; Eggleton, B.J. Antiresonant reflecting photonic crystal optical waveguides. Opt. Lett. 2002, 27, 1592–1594. [Google Scholar] [CrossRef] [PubMed]
- Archambault, J.-L.; Black, R.J.; Lacroix, S.; Bures, J. Loss calculations for antiresonant waveguides. J. Lightwave Technol. 1993, 11, 416–423. [Google Scholar] [CrossRef]
- Testa, G.; Persichetti, G.; Bernini, R. Design and optimization of an optofluidic ring resonator based on liquid-core hybrid ARROWs. IEEE Photon. J. 2014, 6, 2201614. [Google Scholar] [CrossRef]
- Bernini, R.; De Nuccio, E.; Minardo, A.; Zeni, L.; Sarro, P.M. Integrated silicon optical sensors based on hollow core waveguide. Proc. SPIE 2007, 6477, 647714. [Google Scholar]
- Gollub, A.H.; Carvalho, D.O.; Paiva, T.C.; Alayo, M.I. Hollow core ARROW waveguides fabricated with SiOxNy films deposited at low temperatures. Phys. Status Solidi C 2010, 7, 964–967. [Google Scholar]
- Yin, D.; Deamer, D.W.; Schmidt, H.; Barber, J.P.; Hawkins, A.R. Integrated optical waveguides with liquid cores. Appl. Phys. Lett. 2004, 85, 3477. [Google Scholar] [CrossRef]
- Holmes, M.; Keeley, J.; Hurd, K.; Schmidt, H.; Hawkins, A.R. Optimized pirhana etching process for SU8-based MEMS and MOEMS construction. J. Micromech. Microeng. 2010, 20, 115008. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Schmidt, H.; Barber, J.P.; Hawkins, A.R. Integrated ARROW waveguides with hollow cores. Opt. Express 2004, 12, 2710–2715. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Barber, J.P.; Hawkins, A.R.; Schmidt, H. Waveguide loss optimization in hollow-core ARROW waveguides. Opt. Express 2005, 13, 9331–9336. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Barber, J.P.; Hawkins, A.R.; Schmidt, H. Highly efficient fluorescence detection in picoliter volume liquid-core waveguides. Appl. Phys. Lett. 2005, 87, 211111. [Google Scholar] [CrossRef]
- Testa, G.; Huang, Y.; Zeni, L.; Sarro, P.M.; Bernini, R. Liquid core ARROW waveguides by atomic layer deposition. IEEE Photon. Technol. Lett. 2010, 22, 616–618. [Google Scholar] [CrossRef]
- Campopiano, S.; Bernini, R.; Zeni, L.; Sarro, P.M. Microfluidic sensor based on integrated optical hollow waveguides. Opt. Lett. 2004, 29, 1894–1896. [Google Scholar] [CrossRef] [PubMed]
- Barber, J.P.; Lunt, E.J.; George, Z.A.; Yin, D.; Schmidt, H.; Hawkins, A.R. Integrated hollow waveguide with arc shaped core. IEEE Photon. Technol. Lett. 2006, 18, 28–30. [Google Scholar] [CrossRef]
- Yin, D.; Schmidt, H.; Barber, J.P.; Lunt, E.J.; Hawkins, A.R. Optical characterization of arch-shaped ARROW waveguides with liquid cores. Opt. Express 2005, 13, 10564–10570. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Huang, Y.; Zeni, L.; Sarro, P.M.; Bernini, R. Hybrid Silicon-PDMS optofluidic ARROW waveguide. IEEE Photon. Technol. Lett. 2012, 24, 1307–1309. [Google Scholar] [CrossRef]
- Bernini, R.; De Nuccio, E.; Brescia, F.; Minardo, A.; Zeni, L.; Sarro, P.M.; Palumbo, R.; Scarfi, M.R. Development and characterization of an integrated silicon micro flow cytometer. Anal. BioAnal. Chem. 2006, 386, 1267–1272. [Google Scholar] [CrossRef] [PubMed]
- Measor, P.; Seballos, L.; Yin, D.; Barber, J.P.; Hawkins, A.R.; Zhang, J.; Schmidt, H. Integrated ARROW waveguides for molecule specific surface-enhanced Raman sensing. In Proceedings of the 2006 16th Biennial University/Government/Industry Microelectronics Symposium, San Jose, CA, USA, 25–28 June 2006; pp. 181–182.
- Bernini, R.; Testa, G.; Zeni, L.; Sarro, P.M. A 2 × 2 Optofluidic multimode interference coupler. IEEE J. Sel. Top. Quantum Electron. 2009, 15, 1478–1484. [Google Scholar] [CrossRef]
- Testa, G.; Huang, Y.; Sarro, P.M.; Zeni, L.; Bernini, R. High-visibility optofluidic Mach–Zehnder interferometer. Opt. Lett. 2010, 35, 1584–1586. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Collini, C.; Lorenzelli, L.; Bernini, R. Planar silicon-polydimethylsiloxane optofluidic ring resonator sensors. IEEE Photon. Tech. Lett. 2016, 28, 155–158. [Google Scholar] [CrossRef]
- Estevez, M.-C.; Alvarez, M.; Lechuga, L.M. Integrated optical devices for lab-on-a-chip biosensing applications. Laser Photonics Rev. 2012, 6, 463–487. [Google Scholar] [CrossRef] [Green Version]
- Prieto, F.; Sepúlveda, B.; Calle, A.; Llobera, A.; Domínguez, C.; Abad, A.; Montoya, A.; Lechuga, L.M. An integrated optical interferometric nanodevice based on silicon technology for biosensor applications. Nanotechnology 2003, 14, 907–912. [Google Scholar] [CrossRef]
- Nitkowski, A.; Baeumner, A.; Lipson, M. On-chip spectrophotometry for bioanalysis using microring resonators. Biomed. Opt. Express 2011, 2, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Chin, L.K.; Liu, A.Q.; Soh, Y.C.; Lim, C.S.; Lin, C.L. A reconfigurable optofluidic Michelson interferometer using tunable droplet grating. Lab Chip 2010, 10, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Grillet, C.; Domachuk, P.; Ta’eed, V.; Mägi, E.; Bolger, J.A.; Eggleton, B.J.; Rodd, L.E.; Cooper-White, J. Compact tunable microfluidic interferometer. Opt. Express 2004, 12, 5440–5447. [Google Scholar] [CrossRef] [PubMed]
- Domachuk, P.; Grillet, C.; Ta’eed, V.; Mägi, E.; Bolger, J.; Eggleton, B.J.; Rodd, L.E.; Cooper-White, J. Microfluidic interferometer. Appl. Phys. Lett. 2005, 86, 024103. [Google Scholar] [CrossRef]
- Ozcelik, D.; Parks, J.W.; Wall, T.A.; Stott, M.A.; Cai, H.; Parks, J.W.; Hawkins, A.R.; Schmidt, H. Optofluidic wavelength division multiplexing for single-virus detection. Proc. Natl. Acad. Sci. USA 2015, 112, 12933–12937. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Huang, Y.; Sarro, P.M.; Zeni, L.; Bernini, R. Integrated optofluidic ring resonator. Appl. Phys. Lett. 2010, 97, 131110. [Google Scholar] [CrossRef]
- Bernini, R.; Testa, G.; Zeni, L.; Sarro, P.M. Integrated optofluidic Mach–Zehnder interferometer based on liquid core waveguides. Appl. Phys. Lett. 2008, 93, 011106. [Google Scholar] [CrossRef]
- Dumais, P.; Callender, C.L.; Noad, J.P.; Ledderhof, C.J. Integrated optical sensor using a liquid-core waveguide in a Mach–Zehnder interferometer. Opt. Express 2008, 16, 18164–18172. [Google Scholar] [CrossRef] [PubMed]
- Crespi, A.; Gu, Y.; Ngamsom, B.; Hoekstra, H.J.W.M.; Dongre, C.; Pollnau, M.; Ramponi, R.; van den Vlekkert, H.H.; Watts, P.; Cerullo, G.; et al. Three-dimensional Mach–Zehnder interferometer in a microfluidic chip for spatially-resolved label-free detection. Lab Chip 2010, 10, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Lapsley, M.I.; Chiang, I.-K.; Zheng, Y.B.; Ding, X.; Mao, X.; Huang, T.J. A single-layer, planar, optofluidic Mach–Zehnder interferometer for label-free detection. Lab Chip 2011, 11, 1795–1800. [Google Scholar] [CrossRef] [PubMed]
- Kozma, P.; Kehl, F.; Ehrentreich-Förster, E.; Stamm, C.; Bier, F.F. Integrated planar optical waveguide interferometer biosensors: A comparative review. Biosens. Bioelectron. 2014, 58, 287–307. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.; Deutsch, B.; Ignatovich, F.; Dykes, C.; Novotny, L. Nano-optofluidic Detection of Single Viruses and Nanoparticles. ACS Nano 2010, 4, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Bolaños Quiñones, V.A.; Ding, F.; Kiravittaya, S.; Mei, Y.; Schmidt, O.G. Rolled-up optical microcavities with subwavelength wall thicknesses for enhanced liquid sensing applications. ACS Nano 2010, 4, 3123–3130. [Google Scholar] [CrossRef] [PubMed]
- Berneschi, S.; Farnesi, D.; Cosi, F.; Conti, G.N.; Pelli, S.; Righini, G.C.; Soria, S. High Q silica microbubble resonators fabricated by arc discharge. Opt. Lett. 2011, 36, 3521–3523. [Google Scholar] [CrossRef] [PubMed]
- White, I.M.; Oveys, H.; Fan, X. Liquid-core optical ring-resonator sensors. Opt. Lett. 2006, 31, 1319–1321. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Fan, X. Characterization of sensing capability of optofluidic ring resonator biosensors. Appl. Phys. Lett. 2010, 97, 011105. [Google Scholar] [CrossRef]
- Carlborg, C.F.; Gylfason, K.B.; Kaźmierczak, A.; Dortu, F.; Bañuls Polo, M.J.; Maquieira Catala, A.; Kresbach, G.M.; Sohlström, H.; Moh, T.; Vivien, L.; et al. A packaged optical slot-waveguide ring resonator sensor array for multiplex label-free assays in labs-on-chips. Lab Chip 2010, 10, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Dale, P.S.; Caldwell, C.W.; Fan, X. Rapid and Label-Free Detection of Breast Cancer Biomarker CA15-3 in Clinical Human Serum Samples with Optofluidic Ring Resonator Sensors. Anal. Chem. 2009, 81, 9858–9865. [Google Scholar] [CrossRef] [PubMed]
- Fei, P.; Chen, Z.; Men, Y.; Li, A.; Shen, Y.; Huang, Y. A compact optofluidic cytometer with integrated liquid-core/PDMS-cladding waveguides. Lab Chip 2012, 12, 3700–3706. [Google Scholar] [CrossRef] [PubMed]
- Fleger, M.; Neyer, A. PDMS microfluidic chip with integrated waveguides for optical detection. Microelectron. Eng. 2006, 83, 1291–1293. [Google Scholar] [CrossRef]
- Mogensen, K.B.; El-Ali, J.; Wolff, A.; Kutter, J.P. Integration of polymer waveguides for optical detection in microfabricated chemical analysis systems. Appl. Opt. 2003, 42, 4072–4079. [Google Scholar] [CrossRef] [PubMed]
- Friis, P.; Hoppe, K.; Leistiko, O.; Mogensen, K.B.; Hübner, J.; Kutter, J.P. Monolithic integration of microfluidic channels and optical waveguides in silica on silicon. Appl. Opt. 2001, 40, 6246–6251. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yan, H.; Wang, Z.; Zou, Y.; Yang, C.-J.; Chakravarty, S.; Subbaraman, H.; Tang, N.; Xu, X.; Fan, D.L.; et al. Ultralow-loss waveguide crossings for the integration of microfluidics and optical waveguide sensors. Proc. SPIE 2015, 9320, 932012. [Google Scholar]
- Yin, D.; Deamer, D.W.; Schmidt, H.; Barber, J.P.; Hawkins, A.R. Single-molecule detection sensitivity using planar integrated optics on a chip. Opt. Lett. 2006, 31, 2136–2138. [Google Scholar] [CrossRef] [PubMed]
- Measor, P.; Seballos, L.; Yin, D.; Zhang, J.Z.; Lunt, E.J.; Hawkins, A.R.; Schmidt, H. On-chip surface-enhanced Raman scattering detection using integrated liquid-core waveguides. Appl. Phys. Lett. 2007, 90, 211107. [Google Scholar] [CrossRef]
- Li, Q.-L.; Li, B.-W.; Wang, Y.-Q. Surface-enhanced Raman scattering microfluidic sensor. RSC Adv. 2013, 3, 13015–13026. [Google Scholar] [CrossRef]
- Wang, M.; Jing, N.; Chou, I.-H.; Cote, G.L.; Kameoka, J. An optofluidic device for surface enhanced Raman spectroscopy. Lab Chip 2007, 7, 630–632. [Google Scholar] [CrossRef] [PubMed]
- Lunt, E.J.; Phillips, B.S.; Jones, C.J.; Hawkins, A.R.; Measor, P.; Kuehn, S.; Schmidt, H. Hollow waveguide optimization for fluorescence based detection. Proc. SPIE 2008, 6883, 68830H. [Google Scholar]
- Lunt, E.J.; Wu, B.; Keeley, J.M.; Measor, P.; Schmidt, H.; Hawkins, A.R. Hollow ARROW Waveguides on Self-Aligned Pedestals for Improved Geometry and Transmission. IEEE Photonics Technol. Lett. 2010, 22, 1147–1149. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Jenkins, M.H.; Measor, P.; Leake, K.; Liu, S.; Schmidt, H.; Hawkins, A.R. Hollow waveguides with low intrinsic photoluminescence fabricated with Ta2O5 and SiO2 film. Appl. Phys. Lett. 2011, 98, 091104. [Google Scholar] [CrossRef] [PubMed]
- Phillips, B.S.; Jenkins, M.H.; Liu, S.; Schmidt, H.; Hawkins, A.R. Selective Thin-Film deposition for optofluidic platforms with optimized transmission. IEEE Photon. Technol. Lett. 2011, 23, 721–723. [Google Scholar] [CrossRef]
- Zhao, Y.; Leake, K.D.; Measor, P.; Jenkins, M.H.; Keeley, J.; Schmidt, H.; Hawkins, A.R. Optimization of interface transmission between integrated solid core and optofluidic waveguides. IEEE Photon. Technol. Lett. 2012, 24, 46–48. [Google Scholar] [CrossRef]
- Yin, D.; Lunt, E.J.; Rudenko, M.I.; Deamer, D.W.; Hawkins, A.R.; Schmidt, H. Planar optofluidic chip for single particle detection, manipulation, and analysis. Lab Chip 2007, 7, 1171–1175. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Eberle, M.M.; Lunt, E.J.; Liu, S.; Leake, K.; Rudenko, M.I.; Hawkins, A.R.; Schmidt, H. Dual-color fluorescence cross-correlation spectroscopy on a planar optofluidic chip. Lab Chip 2011, 11, 1502–1506. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, M.I.; Kühn, S.; Lunt, E.J.; Deamer, D.W.; Hawkins, A.R.; Schmidt, H. Ultrasensitive Qβ phage analysis using fluorescence correlation spectroscopy on an optofluidic chip. Biosens. Bioelectron. 2009, 24, 3258–3263. [Google Scholar] [CrossRef] [PubMed]
- Bao, N.; Wang, J.; Lu, C. Recent advances in electric analysis of cells in microfluidic systems. Anal. Bioanal. Chem. 2008, 391, 933–942. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Kang, M.; Jung, J.H. Integrated micro-optofluidic platform for real-time detection of airborne microorganisms. Sci. Rep. 2015, 5, 15983. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.S.; Walter, T.M.; Chang, W.J.; Lim, K.S.; Yang, L.; Lee, S.W.; Aronson, A.; Bashir, R. Electrical detection of germination of viable model Bacillus anthracis spores in microfluidic biochips. Lab Chip 2007, 7, 603–610. [Google Scholar]
- Mukundan, H.; Anderson, A.S.; Grace, W.K.; Grace, K.M.; Hartman, N.; Martinez, J.S.; Swanson, B.I. Waveguide-Based Biosensors for Pathogen Detection. Sensors 2009, 9, 5783–5809. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.S.; Grace, W.K.; Grace, K.M.; Hartman, N.; Swanson, B.I. Pathogen detection using single mode planar optical waveguides. J. Mater. Chem. 2005, 15, 4639–4647. [Google Scholar] [CrossRef]
- Donaldson, K.A.; Kramer, M.F.; Lim, D.V. A rapid detection method for Vaccinia virus, the surrogate for smallpox virus. Biosens. Bioelectron. 2004, 20, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Patolsky, F.; Zheng, G.; Hayden, O.; Lakadamyali, M.; Zhuang, X.; Lieber, C.M. Electrical detection of single viruses. Proc. Natl. Acad. Sci. USA 2004, 101, 14017–14022. [Google Scholar] [CrossRef] [PubMed]
- Ilic, B.; Yang, Y.; Craighead, H.G. Virus detection using nanoelectromechanical devices. Appl. Phys. Lett. 2004, 85, 2604. [Google Scholar] [CrossRef]
- Shuo, L.; Zhao, Y.; Parks, J.W.; Deamer, D.W.; Hawkins, A.R.; Schmidt, H. Correlated electrical and optical analysis of single nanoparticles and biomolecules on a nanopore-gated optofluidic chip. Nano Lett. 2014, 14, 4816–4820. [Google Scholar]
- Shuo, L.; Wall, T.A.; Ozcelik, D.; Parks, J.W.; Hawkins, A.R.; Schmidt, H. Electro-optical detection of single λ-DNA. Chem. Comm. 2015, 51, 2084–2087. [Google Scholar]
- Gilboa, T.; Meller, A. Optical sensing and analyte manipulation in solid-state nanopores. Analyst 2015, 140, 4733–4747. [Google Scholar] [CrossRef] [PubMed]
- Kayani, A.A.; Khoshmanesh, K.; Ward, S.A.; Mitchell, A.; Kalantar-zadeh, K. Optofluidics incorporating actively controlled micro- and nano-particles. Biomicrofluidics 2012, 6, 031501. [Google Scholar] [CrossRef] [PubMed]
- Kühn, S.; Measor, P.; Lunt, E.J.; Phillips, B.S.; Deamer, D.W.; Hawkins, A.R.; Schmidt, H. Loss-based optical trap for on-chip particle analysis. Lab Chip 2009, 9, 2212–2216. [Google Scholar] [CrossRef] [PubMed]
- Kühn, S.; Lunt, E.J.; Phillips, B.S.; Hawkins, A.R.; Schmidt, H. Optofluidic particle concentration by a long-range dual-beam trap. Opt. Lett. 2009, 34, 2306–2308. [Google Scholar] [CrossRef] [PubMed]
- Kühn, S.; Phillips, B.S.; Lunt, E.J.; Hawkins, A.R.; Schmidt, H. Ultralow power trapping and fluorescence detection of single particles on an optofluidic chip. Lab Chip 2010, 10, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Leake, K.D.; Phillips, B.S.; Yuzvinsky, T.D.; Hawkins, A.R.; Schmidt, H. Optical particle sorting on an optofluidic chip. Opt. Express 2013, 21, 32605–32610. [Google Scholar] [CrossRef] [PubMed]
- Schuhladen, S.; Banerjee, K.; Stürmer, M.; Müller, P.; Wallrabe, U.; Zappe, H. Variable optofluidic slit aperture. Light Sci. Appl. 2016, 5, e16005. [Google Scholar] [CrossRef]
- Seow, Y.C.; Lim, S.P.; Lee, H.P. Optofluidic variable-focus lenses for light manipulation. Lab Chip 2012, 12, 3810–3815. [Google Scholar] [CrossRef] [PubMed]
- Müller, P.; Kopp, D.; Llobera, A.; Zappe, H. Optofluidic router based on tunable liquid-liquid mirrors. Lab Chip 2014, 14, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Llobera, A.; Demming, S.; Joensson, H.N.; Vila-Planas, J.; Andersson-Svahn, H.; Büttgenbach, S. Monolithic PDMS passband filters for fluorescence detection. Lab Chip. 2010, 10, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Dandin, M.; Abshire, P.; Smela, E. Optical filtering technologies for integrated fluorescence sensors. Lab Chip 2007, 7, 955–977. [Google Scholar] [CrossRef] [PubMed]
- Phillips, B.S.; Measor, P.; Zhao, Y.; Schmidt, H.; Hawkins, A.R. Optofluidic notch filter integration by lift-off of thin films. Opt. Express 2010, 18, 4790–4795. [Google Scholar] [CrossRef] [PubMed]
- Measor, P.; Phillips, B.S.; Chen, A.; Hawkins, A.R.; Schmidt, H. Tailorable integrated optofluidic filters for biomolecular detection. Lab Chip 2011, 11, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.M.; Gitlin, I.; Stroock, A.D.; Whitesides, G.M. Components for integrated poly(dimethylsiloxane) microfluidic systems. Electrophoresis 2002, 23, 3461–3473. [Google Scholar] [CrossRef]
- Testa, G.; Persichetti, G.; Sarro, P.M.; Bernini, R. A hybrid silicon-PDMS optofluidic platform for sensing applications. Biomed. Opt. Express 2014, 5, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Taitt, C.R.; Anderson, G.P.; Ligler, F.S. Evanescent wave fluorescence biosensors. Biosens. Bioelectron. 2005, 20, 2470–2487. [Google Scholar] [CrossRef] [PubMed]
- Parks, J.W.; Cai, H.; Zempoaltecatl, L.; Yuzvinsky, T.D.; Leake, K.; Hawkins, A.R.; Schmidt, H. Hybrid optofluidic integration. Lab Chip 2013, 13, 4118–4123. [Google Scholar] [CrossRef] [PubMed]
- Parks, J.W.; Olson, M.A.; Kim, J.; Ozcelik, D.; Cai, H.; Carrion, R.; Patterson, J.L.; Mathies, R.A.; Hawkins, A.R.; Schmidt, H. Integration of programmable microfluidics and on-chip fluorescence detection for biosensing applications. Biomicrofluidics 2014, 8, 054111. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Parks, J.W.; Wall, T.A.; Stott, M.A.; Stambaugh, A.; Alfson, K.; Griffiths, A.; Mathies, R.A.; Carrion, R.; Patterson, J.L.; et al. Optofluidic analysis system for amplification-free, direct detection of Ebola infection. Sci. Rep. 2015, 5, 14494. [Google Scholar] [CrossRef] [PubMed]
- Trombley, A.R.; Wachter, L.; Garrison, J.; Buckley-Beason, V.-A.; Jahrling, J.; Hensley, L.E.; Schoepp, R.J.; Norwood, D.A.; Goba, A.; Fair, J.N.; et al. Comprehensive panel of real-time TaqMan™ polymerase chain reaction assays for detection and absolute quantification of Filoviruses, Arenaviruses, and New World Hantaviruses. Am. J. Trop. Med. Hyg. 2010, 82, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Daaboul, G.G.; Lopez, C.A.; Chinnala, J.; Goldberg, B.B.; Connor, J.H.; Ünlü, M.S. Digital sensing and sizing of vesicular stomatitis virus pseudotypes in complex media: A model for Ebola and Marburg detection. ACS Nano 2014, 8, 6047–6055. [Google Scholar] [CrossRef] [PubMed]



© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Testa, G.; Persichetti, G.; Bernini, R. Liquid Core ARROW Waveguides: A Promising Photonic Structure for Integrated Optofluidic Microsensors. Micromachines 2016, 7, 47. https://doi.org/10.3390/mi7030047
Testa G, Persichetti G, Bernini R. Liquid Core ARROW Waveguides: A Promising Photonic Structure for Integrated Optofluidic Microsensors. Micromachines. 2016; 7(3):47. https://doi.org/10.3390/mi7030047
Chicago/Turabian StyleTesta, Genni, Gianluca Persichetti, and Romeo Bernini. 2016. "Liquid Core ARROW Waveguides: A Promising Photonic Structure for Integrated Optofluidic Microsensors" Micromachines 7, no. 3: 47. https://doi.org/10.3390/mi7030047