Simulation and Experimental Characterization of Microscopically Accessible Hydrodynamic Microvortices
Abstract
:1. Introduction
1.1. Single Cell Analysis with Multiple Perspective Imaging
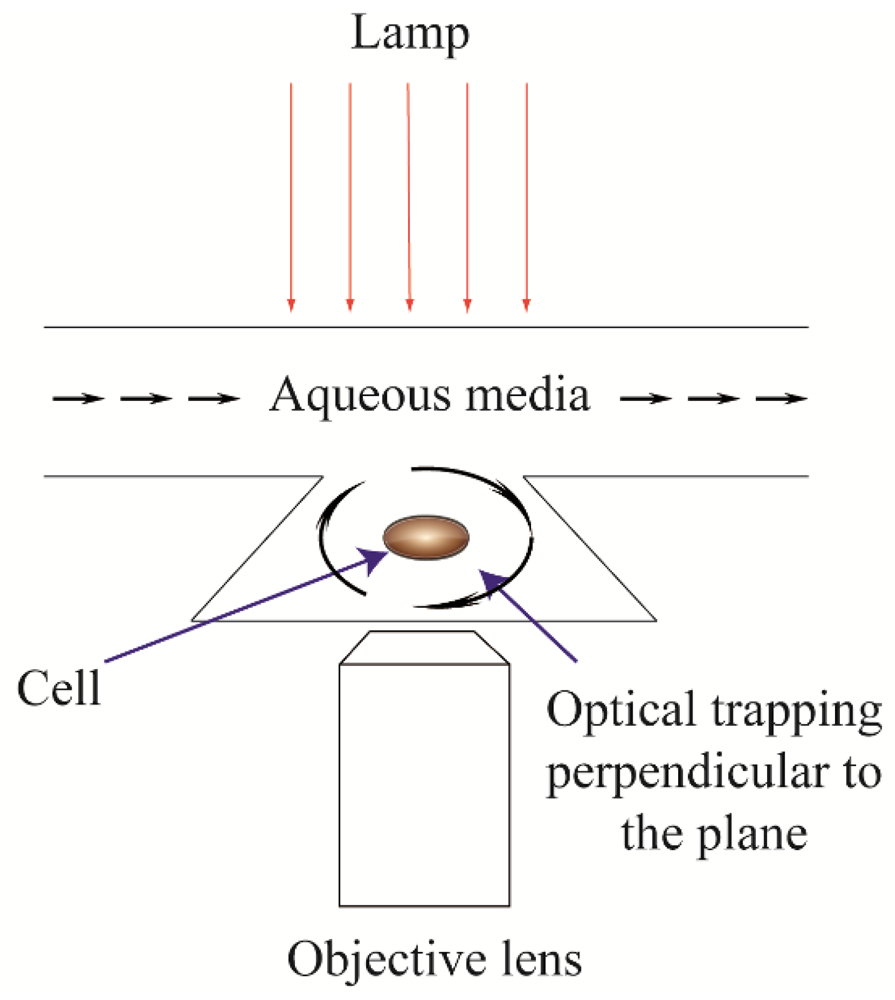
1.2. Existing Techniques for Single Cell Rotation
1.3. Investigations of Flow Profiles in Microvortex Systems
2. Methods
2.1. Computational Modeling
2.2. Microfabrication
2.3. µ-PIV Validation Experiments

3. Results
3.1. CFD Simulation
 ), which represents the rotation of a fluid element in the flow field [28]. Contour plots of vorticity for the three chamber designs are shown in Figure 3 along with contour plots of velocity magnitude and streamlines. The ratio of chamber opening to channel height was 0.8 for all three cases. The geometries of cases (a) and (b) shared lower sides with the same slope, and case (b) had the same chamber height as case (c). The flow direction in the main channel for all three geometries was from left to right, and flow circulation in the side chambers was in a counterclockwise direction. Flow detachment from the channel into the side chamber, forming the microvortices, was apparent in the closed streamline paths.
), which represents the rotation of a fluid element in the flow field [28]. Contour plots of vorticity for the three chamber designs are shown in Figure 3 along with contour plots of velocity magnitude and streamlines. The ratio of chamber opening to channel height was 0.8 for all three cases. The geometries of cases (a) and (b) shared lower sides with the same slope, and case (b) had the same chamber height as case (c). The flow direction in the main channel for all three geometries was from left to right, and flow circulation in the side chambers was in a counterclockwise direction. Flow detachment from the channel into the side chamber, forming the microvortices, was apparent in the closed streamline paths.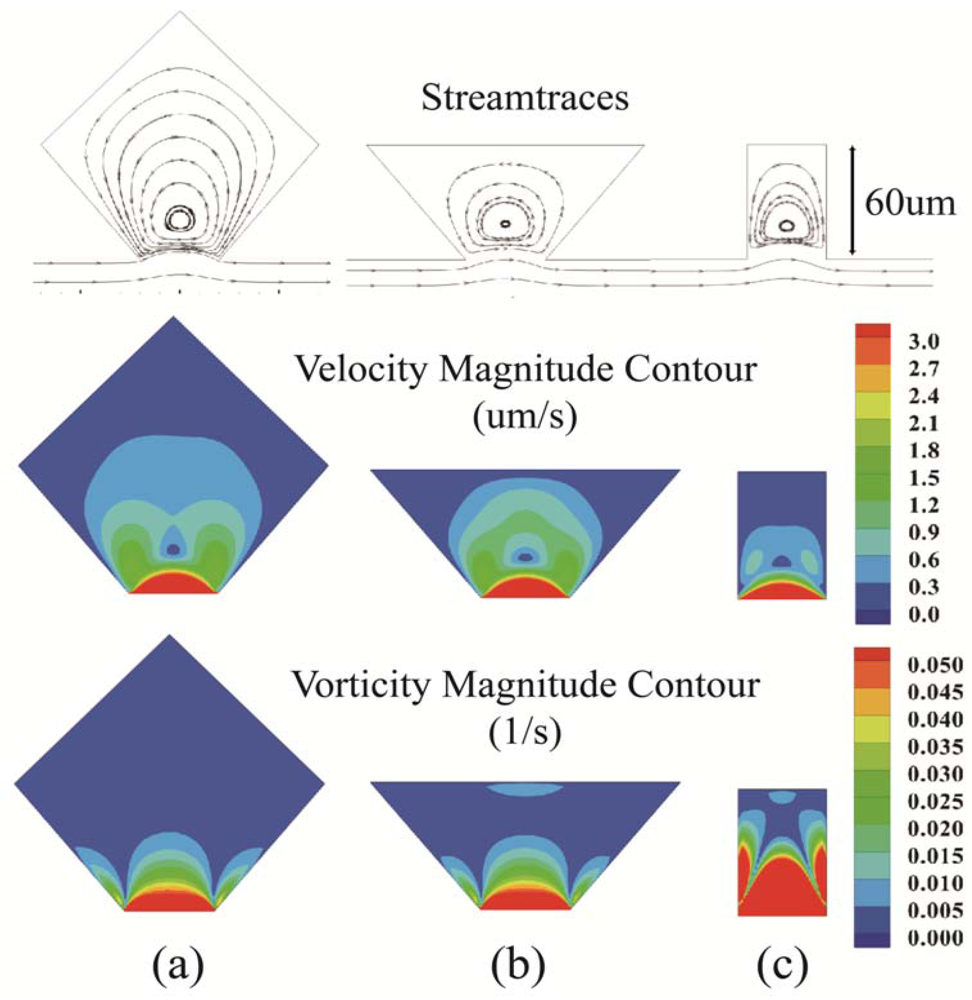
 , the line integral of the tangential velocity component about the boundary of the microvortex, determines the strength of the vortex, and is directly related to the size and shape of the chamber [28]. The value of
, the line integral of the tangential velocity component about the boundary of the microvortex, determines the strength of the vortex, and is directly related to the size and shape of the chamber [28]. The value of  is calculated as:
is calculated as:
3.2. Quantification and Comparison of Flow Profiles in a Trapezoidal Side Chamber


3.3. Rotation of Biological Cells in the Trapezoidal Side Chamber

4. Discussion and Conclusions
Acknowledgments
References
- Avery, S.V. Microbial cell individuality and the underlying sources of heterogeneity. Nat. Rev. Microbiol. 2006, 4, 577–587. [Google Scholar] [CrossRef]
- Suresh, S.; Spatz, J.; Mills, J.P.; Micoulet, A.; Dao, M.; Lim, C.T.; Beil, M.; Seufferlein, T. Connections between single-cell biomechanics and human disease states: Gastrointestinal cancer and malaria. ActaBiomater. 2005, 1, 15–30. [Google Scholar]
- Loeb, L.A. Multiple mutations and cancer. Proc. Natl. Acad. Sci. USA 2003, 100, 776–781. [Google Scholar] [CrossRef]
- Nandakumar, V.; Kelbauskas, L.; Johnson, R.; Meldrum, D. Quantitative characterization of preneoplastic progression using single-cell computed tomography and three-dimensional karyometry. Cytometry 2011, 79A, 25–34. [Google Scholar] [CrossRef]
- Meyer, M.G.; Fauver, M.; Rahn, J.R.; Neumann, T.; Patten, F.W.; Seibel, E.J.; Nelson, A.C. Automated cell analysis in 2D and 3D: A comparative study. Pattern Recogn. 2009, 42, 141–146. [Google Scholar]
- Semwogerere, D.; Weeks, E.R. Confocal microscopy. Biomed. Eng. 2005, 23, 1–10. [Google Scholar]
- Fauver, M.; Seibel, E.; Rahn, J.R.; Meyer, M.; Patten, F.; Neumann, T.; Nelson, A. Three-dimensional imaging of single isolated cell nuclei using optical projection tomography. Opt. Express 2005, 13, 4210–4223. [Google Scholar]
- Miao, Q.; Rahn, J.R.; Tourovskaia, A.; Meyer, M.G.; Neumann, T.; Nelson, A.C.; Seibel, E.J. Dual-modal three-dimensional imaging of single cells with isometric high resolution using an optical projection tomography microscope. J. Biomed. Opt. 2009. [Google Scholar]
- Arai, F.; Kawaji, A.; Luangjarmekom, P.; Fukuda, T.; Itoigawa, K. Three-dimensional Bio-micromanipulation under the microscope. In Proceedings of the 2001 IEEE International Conference on Robotics and Automation, Seoul, Korea, 21–26 May 2001; 1, pp. 604–609.
- Lin, C.M.; Lai, Y.S.; Liu, H.P.; Chen, C.Y.; Wo, A.M. Trapping of bioparticles via microvortices in a microfluidic device for bioassay applications. Anal. Chem. 2008, 80, 8937–8945. [Google Scholar] [CrossRef]
- Lidstrom, M.E.; Meldrum, D.R. Life-on-a-chip. Nat. Rev. Microbiol. 2003, 1, 158–164. [Google Scholar] [CrossRef]
- Eriksson, E.; Enger, J.; Nordlander, B.; Erjavec, N.; Ramser, K.; Goksor, M.; Hohmann, S.; Nystrom, T.; Hanstorp, D. A microfluidic system in combination with optical tweezers for analyzing rapid and reversible cytological alterations in single cells upon environmental changes. Lab Chip 2007, 7, 71–76. [Google Scholar] [CrossRef]
- Brehm-Stecher, B.F.; Johnson, E.A. Single-cell microbiology: Tools, technologies, and applications. Microbiol. Mol. Biol. Rev. 2004, 68, 538–559. [Google Scholar] [CrossRef]
- Shelby, J.P.; Lim, D.S.W.; Kuo, J.S.; Chiu, D.T. High radial acceleration in microvortices. Nature 2003. [Google Scholar]
- Shelby, J.P.; Chiu, D.T. Controlled rotation of biological micro- and nano-particles in microvortices. Lab Chip 2004, 4, 168–170. [Google Scholar] [CrossRef]
- Ashkin, A.; Dziedzic, J.M.; Yamane, T. Optical trapping and manipulation of single cells using infrared-laser beams. Nature 1987, 330, 769–771. [Google Scholar]
- Neuman, K.C.; Block, S.M. Optical trapping. Rev. Sci. Instrum. 2004, 75, 2787–2809. [Google Scholar] [CrossRef]
- Liu, K.; Pitchimani, R.; Dang, D.; Bayer, K.; Harrington, T.; Pappas, D. Cell culture chip using low-shear mass transport. Langmuir 2008, 24, 5955–5960. [Google Scholar] [CrossRef]
- Shelby, J.P.; Mutch, S.A.; Chiu, D.T. Direct manipulation and observation of the rotational motion of single optically trapped microparticles and biological cells in microvortices. Anal. Chem. 2004, 76, 2492–2497. [Google Scholar] [CrossRef]
- Ha, J.B.; Bahk, Y.K.; Yoon, S.H.; Lee, J.H.; Jeong, E.H.; Yoon, S.Y.; Arakawa, T.; Ko, J.S.; Shin, B.S.; Kim, K.C.; Boo, J.S.; Shoji, S.; Go, J.S. Microfludic centrifuge of nano particles using rotating flow in a microchamber. In Proceedings of the Solid-State SensorsActuators and Microsystems ConferenceTRANSDUCERS 2007, Lyon, France, 10–14 June 2007; pp. 927–930.
- Shelby, J.P.; Chiu, D.T. Mapping fast flows over micrometer-length scales using flow-tagging velocimetry and single-molecule detection. Anal. Chem. 2003, 75, 1387–1392. [Google Scholar] [CrossRef]
- Santiago, J.G.; Wereley, S.T.; Meinhart, C.D.; Beebe, D.J.; Adrian, R.J. A particle image velocimetry system for microfluidics. Exp. Fluids 1998, 25, 316–319. [Google Scholar] [CrossRef]
- Raffel, M.; Willert, C.E.; Wereley, S.T.; Kompenhaus, J. Particle Image Velocimetry a Practical Guide, 2nd ed; Springer: Heidelberg, Germany, 2007. [Google Scholar]
- Qin, J.J.; Yeo, L.Y.; Friend, J.R. MicroPIV and micromixing study of corona wind induced microcentrifugation flows in a cylindrical cavity. Microfluid. Nanofluid. 2009, 8, 231–241. [Google Scholar]
- Ota, H.; Yamamoto, R.; Deguchi, K.; Tanaka, Y.; Kazoe, Y.; Sato, Y.; Miki, N. Three-dimensional spheroid-forming lab-on-a-chip using micro-rotational flow. Sens. Actuat. B Chem. 2010, 147, 359–365. [Google Scholar] [CrossRef]
- Yang, J.-T.; Huang, K.-J.; Lin, Y.-C. Geometric effects on fluid mixing in passive grooved micromixers. Lab Chip 2005, 5, 1140–1147. [Google Scholar] [CrossRef]
- Park, J.-S.; Song, S.-H.; Jung, H.-I. Continuous focusing of microparticles using inertial lift force and vorticity via multi-orifice microfluidic channels. Lab Chip 2009, 9, 939–948. [Google Scholar] [CrossRef]
- Fox, R.W.; Pritchard, P.J.; McDonald, A.T. Introduction to Fluid Mechanics, 7th ed; Wiley: Hoboken, NJ, USA, 2008; p. 181. [Google Scholar]
- Matheron, G. Principles of geostatistics. Econ. Geol. 1963, 58, 1246–1266. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, W.; Frakes, D.H.; Babiker, H.; Chao, S.-h.; Youngbull, C.; Johnson, R.H.; Meldrum, D.R. Simulation and Experimental Characterization of Microscopically Accessible Hydrodynamic Microvortices. Micromachines 2012, 3, 529-541. https://doi.org/10.3390/mi3020529
Zhang W, Frakes DH, Babiker H, Chao S-h, Youngbull C, Johnson RH, Meldrum DR. Simulation and Experimental Characterization of Microscopically Accessible Hydrodynamic Microvortices. Micromachines. 2012; 3(2):529-541. https://doi.org/10.3390/mi3020529
Chicago/Turabian StyleZhang, Wenjie, David H. Frakes, Haithem Babiker, Shih-hui Chao, Cody Youngbull, Roger H. Johnson, and Deirdre R. Meldrum. 2012. "Simulation and Experimental Characterization of Microscopically Accessible Hydrodynamic Microvortices" Micromachines 3, no. 2: 529-541. https://doi.org/10.3390/mi3020529
APA StyleZhang, W., Frakes, D. H., Babiker, H., Chao, S.-h., Youngbull, C., Johnson, R. H., & Meldrum, D. R. (2012). Simulation and Experimental Characterization of Microscopically Accessible Hydrodynamic Microvortices. Micromachines, 3(2), 529-541. https://doi.org/10.3390/mi3020529



