Plant Ureases and Related Peptides: Understanding Their Entomotoxic Properties
Abstract
:1. Introduction
2. The Entomotoxic Activity of Plant Ureases
| Insect | Insect stage | Administration route | Effect observed | Reference |
|---|---|---|---|---|
| M. sexta | 2nd instars | Feeding | None | [24] |
| S. americana | 5th instars | Feeding | None | [24] |
| D. melanogaster | Adults | Feeding | None | [24] |
| A. aegypti | Adults | Feeding | None | [24] |
| C. maculatus | Larvae | Feeding | None | [24] |
| R. prolixus | 3rd instars | Feeding | Decreased weight loss after feeding | [24] |
| 80–100% mortality after 72 h | ||||
| 3rd instars | Injection | None | [24] | |
| 4th instars | Feeding | 65–75% mortality after 48 h | [25] | |
| Adults | Feeding | None | [25] | |
| Adults | Injection | None | [25] | |
| D. peruvianus | 3rd instars | Feeding | Reduced body weight gain | [26] |
| Delayed development | ||||
| 90–100% mortality after 10 days | ||||
| Adults | Feeding | None | [26] | |
| N. viridula | 2nd instars | Feeding | 100% lethality after 72 h | [17] |
| Insect | Insect stage | Administration route | Effect observed | Reference |
|---|---|---|---|---|
| R. prolixus | 5th instars | Injection | 96% mortality after 24 h | [31] |
| D. peruvianus | 2nd instars | Feeding | Reduced body weight gain | [27] |
| Delayed development | ||||
| 85% mortality after 20 days | ||||
| Adults | Feeding | None | ||
| O. fasciatus | 3rd instars | Feeding | 90% mortality after 14 days | [32] |
3. The Entomotoxic Peptides
| Insect | Insect stage | Administration route | Effect observed | Reference |
|---|---|---|---|---|
| D. peruvianus | 3rd instars | Feeding | 100% mortality after 11 days | [26] |
| Adults | Feeding | None | [26] | |
| S. frugiperda | 3rd instars | Feeding | Decreased weight gain | [30] |
| 100% mortality after 8 days | ||||
| R. prolixus | 4th instars | Injection | 100% mortality after 48 h | [34] |
| T. infestans | 5th instars | Injection | 100% mortality after 15 h | [34] |
| Adults | Injection | 100% mortality after 20 h | [34] |
4. Urease Processing by Insect Digestive Enzymes
5. Targets of Action
5.1. Malpighian Tubules
5.2. Anterior Midgut
5.3. Other Possible Targets
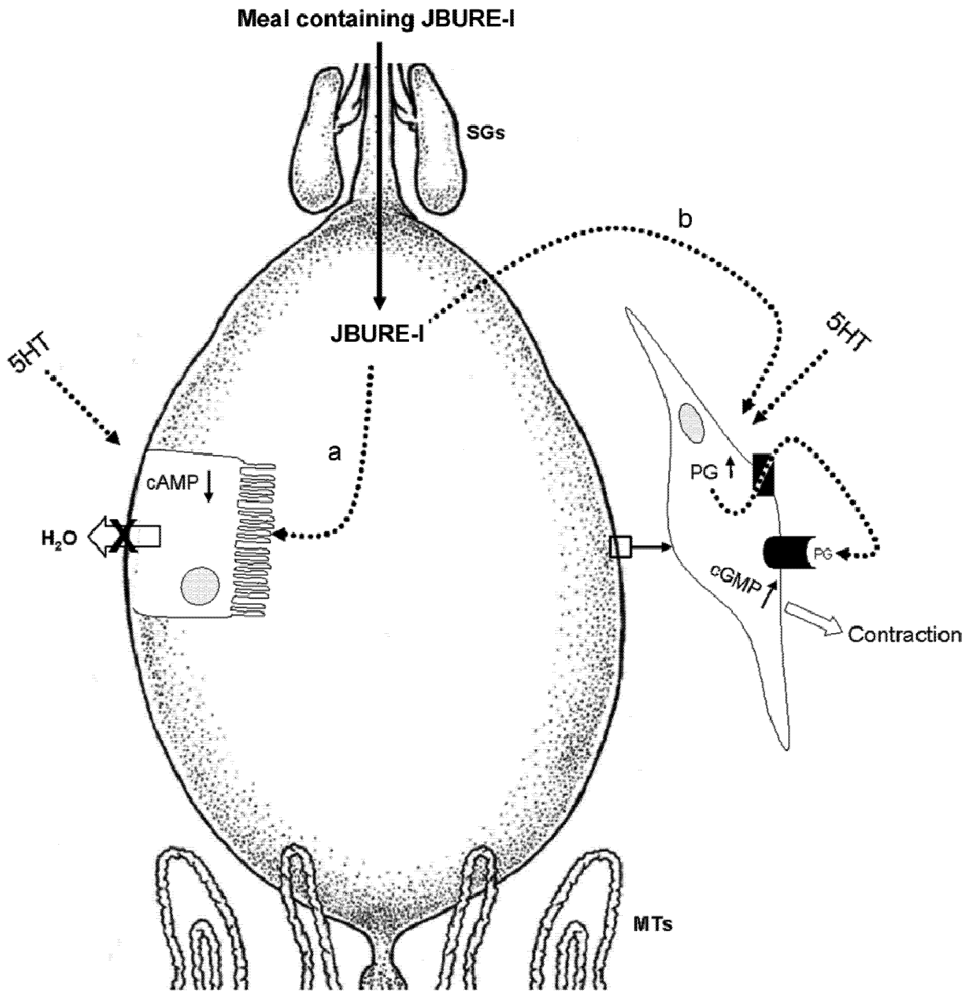
6. Conclusions

Acknowledgements
Conflict of Interest
References
- Carlini, C.R.; Guimaraes, J.A. Isolation and characterization of a toxic protein from Canavalia ensiformis (Jackbean) seeds, distinct from Concanavalin-A. Toxicon 1981, 19, 667–675. [Google Scholar] [CrossRef]
- Carlini, C.R.; Gomes, C.; Guimaraes, J.A.; Markus, R.P.; Sato, H.; Trolin, G. Central nervous effects of the convulsant protein Canatoxin. Acta Pharmcol. Toxicol. 1984, 54, 161–166. [Google Scholar]
- Carlini, C.R.; Guimaraes, J.A. Plant and microbial toxic proteins as hemilectins—Emphasis on Canatoxin. Toxicon 1991, 29, 791–806. [Google Scholar]
- Follmer, C.; Barcellos, G.B.S.; Zingali, R.B.; Machado, O.L.T.; Alves, E.W.; Barja-Fidalgo, C.; Guimaraes, J.A.; Carlini, C.R. Canatoxin, a toxic protein from jack beans (Canavalia ensiformis), is a variant form of urease (EC 3.5.1.5): Biological effects of urease independent of its ureolytic activity. Biochem. J. 2011, 360, 217–224. [Google Scholar]
- Dixon, N.E.; Gazzola, C.; Blakeley, R.L.; Zerner, B. Jack-bean urease (EC 3.5.1.5)—Metalloenzyme—Simple biological role for nickel. J. Am. Chem. Soc. 1975, 97, 4131–4133. [Google Scholar] [CrossRef]
- Follmer, C. Insights into the role and structure of plant ureases. Phytochemistry 2008, 69, 18–28. [Google Scholar]
- Sumner, J.B. The isolation and crystallization of the enzyme urease. J. Biol. Chem. 1926, 69, 435–441. [Google Scholar]
- Pires-Alves, M.; Grossi-de-Sa, M.F.; Barcellos, G.B.S.; Carlini, C.R.; Moraes, M.G. Characterization and expression of a novel member (JBURE-II) of the urease gene family from jackbean Canavalia ensiformis (L.) DC. Plant Cell Pysiol. 2003, 44, 139–145. [Google Scholar] [CrossRef]
- Mulinari, F.; Becker-Ritt, A.B.; Demartini, D.R.; Ligabue-Braun, R.; Staniscuaski, F.; Verli, H.; Fragoso, R.R.; Schroeder, E.K.; Carlini, C.R.; Grossi-de-Sa, M.F. Characterization of JBURE-IIb isoform of Canavalia ensiformis (L.) DC urease. Biochim. Biophys. Acta 2011, 1814, 1758–1768. [Google Scholar]
- Holland, M.A.; Griffin, J.D.; Meyer-Bothling, L.E.; Polacco, J.C. Developmental genetics of the soybean urease isozymes. Dev. Genet. 1987, 8, 375–387. [Google Scholar]
- Torisky, R.S.; Polacco, J.C. Soybean roots retain the seed urease isozyme synthesized during embryo development. Plant Physiol. 1990, 94, 681–689. [Google Scholar]
- Goldraij, A.; Beamer, L.J.; Polacco, J.C. Interallelic complementation at the ubiquitous urease coding locus of soybean. Plant Physiol. 2003, 132, 1801–1810. [Google Scholar]
- Olivera-Severo, D.; Wassermann, G.E.; Carlini, C.R. Ureases display biological effects independent of enzymatic activity. Is there a connection to diseases caused by urease-producing bacteria? Braz. J. Med. Biol. Res. 2006, 39, 851–86. [Google Scholar] [CrossRef]
- Wassermann, G.E.; Olivera-Severo, D.; Uberti, A.F.; Carlini, C.R. Helicobacter pylori urease activates blood platelets through a lipoxygenase-mediated pathway. J. Cell. Mol. Med. 2010, 14, 2025–2034. [Google Scholar] [CrossRef]
- Polacco, J.C.; Holland, M.A. Roles of urease in plant cells. Int. Rev. Cytol. 1993, 145, 65–103. [Google Scholar]
- Carlini, C.R.; Polacco, J.C. Toxic properties of urease. Crop Sci. 2008, 48, 1665–1672. [Google Scholar]
- Carlini, C.R.; Grossi-de-Sa, M.F. Plant toxic proteins with insecticidal properties. A review on their potentialities as bioinsecticides. Toxicon 2002, 40, 1515–1539. [Google Scholar]
- Oliveira, A.E.; Gomes, V.M.; Sales, M.P.; Fernandes, K.V.S.; Carlini, C.R.; Xavier-Filho, J. The toxicity of Jack bean [Canavalia ensiformis (L.) DC.] Canatoxin to plant pathogenic fungi. Rev. Bras. Biol. 1999, 59, 59–62. [Google Scholar] [CrossRef]
- Menegassi, A.; Wassermann, G.E.; Olivera-Severo, D.; Becker-Ritt, A.B.; Martinelli, A.H.S.; Feder, V.; Carlini, C.R. Urease from cotton (Gossypium hirsutum) seeds: Isolation, physicochemical characterization, and antifungal properties of the protein. J. Agric. Food Chem. 2008, 56, 4399–4405. [Google Scholar]
- Becker-Ritt, A.B.; Martinelli, A.H.S.; Mitidieri, S.; Feder, V.; Wassermann, G.E.; Santi, L.; Vainstein, M.H.; Oliveira, J.T.A.; Fiuza, L.M.; Pasquali, G.; et al. Antifungal activity of plant and bacterial ureases. Toxicon 2007, 50, 971–983. [Google Scholar]
- Carlini, C.R.; Barcellos, G.B.S.; Baetaneves, A.D.V.; Guimaraes, J.A. Immunoreactivity for Canatoxin and Concanavalin-A among proteins of leguminous seeds. Phytochemistry 1988, 27, 25–30. [Google Scholar]
- Vasconcelos, I.M.; Trentim, A.; Guimaraes, J.A.; Carlini, C.R. Purification and physicochemical characterization of soyatoxin, a novel toxic protein isolated from soybeans (Glycine max). Arch. Biochem. Biophys. 1994, 312, 357–366. [Google Scholar] [CrossRef]
- Barcellos, G.B.S.; Almeida, L.M.; Moreira, R.A.; Cavada, B.S.; Deoliveira, J.T.A.; Carlini, C.R. Canatoxin-cross-reactive, Concanavalin-A-cross-reactive and Canavalin-cross-reactive materials during maturation of Canavalia brasiliensis (Mart) seeds. Planta 1993, 189, 397–402. [Google Scholar] [CrossRef]
- Carlini, C.R.; Oliveira, A.E.A.; Azambuja, P.; Xavier, J.; Wells, M.A. Biological effects of canatoxin in different insect models: Evidence for a proteolytic activation of the toxin by insect cathepsin-like enzymes. J. Econ. Entomol. 1997, 90, 340–348. [Google Scholar]
- Ferreira-DaSilva, C.T.; Gombarovits, M.E.C.; Masuda, H.; Oliveira, C.M.; Carlini, C.R. Proteolytic activation of canatoxin, a plant toxic protein, by insect cathepsin-like enzymes. Arch. Insect Biochem. Physiol. 2000, 44, 162–171. [Google Scholar]
- Staniscuaski, F.; Ferreira-DaSilva, C.T.; Mulinari, F.; Pires-Alves, M.; Carlini, C.R. Insecticidal effects of canatoxin on the cotton stainer bug Dysdercus peruvianus (Hemiptera : Pyrrhocoridae). Toxicon 2005, 45, 753–760. [Google Scholar] [CrossRef]
- Follmer, C.; Real-Guerra, R.; Wasserman, G.E.; Olivera-Severo, D.; Carlini, C.R. Jackbean, soybean and Bacillus pasteurii ureases—Biological effects unrelated to ureolytic activity. Eur. J. Biochem. 2004, 271, 1357–1363. [Google Scholar] [CrossRef]
- Mobley, H.L.; Garner, R.M.; Bauerfeind, P. Helicobacter pylori nickel-transport gene nixA: Synthesis of catalytically active urease in Escherichia coli independent of growth conditions. Mol. Microbiol. 1995, 16, 97–109. [Google Scholar]
- Sirko, A.; Brodzik, R. Plant ureases: Roles and regulation. Acta Biochem. Pol. 2000, 47, 1189–1195. [Google Scholar]
- Mulinari, F.; Staniscuaski, F.; Bertholdo-Vargas, L.R.; Postal, M.; Oliveira-Neto, O.B.; Riden, D.J.; Grossi-de-Sa, M.F.; Carlini, C.R. Jaburetox-2Ec: An insecticidal peptide derived from an isoform of urease from the plant Canaualia ensiformis. Peptides 2007, 28, 2042–2050. [Google Scholar] [CrossRef]
- Staniscuaski, F.; Brugge, V.T.; Carlini, C.R.; Orchard, I. Jack bean urease alters serotonin-induced effects on Rhodnius prolixus anterior midgut. J. Insect Physiol. 2010, 56, 1078–1086. [Google Scholar] [CrossRef]
- Defferrari, M.S.; Demartini, D.R.; Marcelino, T.B.; Pinto, P.M.; Carlini, C.R. Insecticidal effect of Canavalia ensiformis major urease on nymphs of the milkweed bug Oncopeltus fasciatus and characterization of digestive peptidases. Insect Biochem. Mol. Biol. 2011, 41, 388–399. [Google Scholar]
- Carlini, C.R.; Ferreira-DaSilva, C.T.; Gombarovits, M.E.C. Peptideo Entomotoxico da Canatoxina: Processo de Produção. Instituto Nacional de Propriedade Industrial Patent 0003334–0, 2000.
- Tomazetto, G.; Mulinari, F.; Staniscuaski, F.; Settembrini, B.; Carlini, C.R.; Zachia Ayub, M.A. Expression kinetics and plasmid stability of recombinant E. coli encoding urease-derived peptide with bioinsecticide activity. Enzym. Microb. Technol. 2007, 41, 821–827. [Google Scholar] [CrossRef]
- Barros, P.R.; Stassen, H.; Freitas, M.S.; Carlini, C.R.; Nascimento, M.A.C.; Follmer, C. Membrane-disruptive properties of the bioinsecticide Jaburetox-2Ec: Implications to the mechanism of the action of insecticidal peptides derived from ureases. Biochim. Biophys. Acta 2009, 1794, 1848–1854. [Google Scholar]
- Piovesan, A.R.; Staniscuaski, F.; Marco-Salvadori, J.; Real-Guerra, R.; Defferrari, M.S.; Carlini, C.R. Stage-specific gut proteinases of the cotton stainer bug Dysdercus peruvianus: Role in the release of entomotoxic peptides from Canavalia ensiformis urease. Insect Biochem. Mol. Biol. 2008, 38, 1023–1032. [Google Scholar] [CrossRef]
- Defferrari, M.S.; Demartini, D.R.; Marcelino, T.B.; Pinto, P.M.; Carlini, C.R. Insecticidal effect of Canavalia ensiformis major urease on nymphs of the milkweed bug Oncopeltus fasciatus and characterization of digestive peptidases. Insect Biochem. Mol. Biol. 2011, 41, 388–399. [Google Scholar] [CrossRef]
- Carlini, C.R.; Ribeiro, J.M.C.; Guimaraes, J.A. Canatoxin, the neurotoxic protein from Canavalia ensiformis seeds, induces platelet-release reaction and aggregation. Toxicon 1985, 23, 25–25. [Google Scholar]
- Barja-Fidalgo, C.; Guimaraes, J.A.; Carlini, C.R. Canatoxin, a plant protein, induces insulin release from isolated pancreatic islets. Endocrinology 1991, 128, 675–679. [Google Scholar]
- Grassi-Kassisse, D.M.; Ribeiro-DaSilva, G. Canatoxin triggers histamine secretion from rat peritoneal mast cells. Agents Actions 1992, 37, 204–209. [Google Scholar]
- Ghazaleh, F.A.; Araujo, C.F.; Barja-Fidalgo, C.; Carlini, C.R. Canatoxin induces activation on mice peritoneal macrophages. Braz. J. Med. Biol. Res. 1992, 25, 1033–1035. [Google Scholar]
- Ribeiro-DaSilva, G.; Prado, J.F. Increased insulin circulating levels induced by canatoxin in rats. Toxicon 1993, 31, 1131–1136. [Google Scholar]
- Olivera-Severo, D.; Wassermann, G.E.; Carlini, C.R. Bacillus pasteurii urease shares with plant ureases the ability to induce aggregation of blood platelets. Arch. Biochem. Biophys. 2006, 452, 149–155. [Google Scholar] [CrossRef]
- Orchard, I. Serotonin: A coordinator of feeding-related physiological events in the blood-gorging bug, Rhodnius prolixus. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2006, 144, 316–324. [Google Scholar] [CrossRef]
- Staniscuaski, F.; Brugge, V.T.; Carlini, C.R.; Orchard, I. In vitro effect of Canavalia ensiformis urease and the derived peptide Jaburetox-2Ec on Rhodnius prolixus Malpighian tubules. J. Insect Physiol. 2009, 55, 255–263. [Google Scholar] [CrossRef]
- Quinlan, M.C.; Tublitz, N.J.; O’Donnell, M.J. Anti-diuresis in the blood-feeding insect Rhodnius prolixus Stal: The peptide CAP2b and cyclic GMP inhibit Malpighian tubule fluid secretion. J. Exp. Biol. 1997, 200, 2363–2367. [Google Scholar]
- Paluzzi, J.P.; Orchard, I. Distribution, activity and evidence for the release of an anti-diuretic peptide in the kissing bug Rhodnius prolixus. J. Exp. Biol. 2006, 209, 907–915. [Google Scholar] [CrossRef]
- Barja-Fidalgo, C.; Guimaraes, J.A.; Carlini, C.R. Lipoxygenase-mediated secretory effect of canatoxin the toxic protein from Canavalia ensiformis seeds. Toxicon 1991, 29, 453–459. [Google Scholar] [CrossRef]
- Norregaard, R.; Jensen, B.L.; Topcu, S.O.; Diget, M.; Schweer, H.; Knepper, M.A.; Nielsen, S.; Frokiaer, J. COX-2 activity transiently contributes to increased water and NaCl excretion in the polyuric phase after release of ureteral obstruction. Am. J. Physiol. Renal Physiol. 2007, 292, F1322–F1333. [Google Scholar] [CrossRef] [Green Version]
- Echevarria, M.; Ramirez-Lorca, R.; Hernandez, C.S.; Gutierrez, A.; Mendez-Ferrer, S.; Gonzalez, E.; Toledo-Aral, J.J.; Ilundain, A.A.; Whittembury, G. Identification of a new water channel (Rp-MIP) in the Malpighian tubules of the insect Rhodnius prolixus. Pflugers Arch. Eur. J. Physiol. 2001, 442, 27–34. [Google Scholar] [CrossRef]
- Martini, S.V.; Goldenberg, R.C.; Fortes, F.S.; Campos-de-Carvalho, A.C.; Falkenstein, D.; Morales, M.M. Rhodnius prolixus Malpighian tubule’s aquaporin expression is modulated by 5-hydroxytryptamine. Arch. Insect. Biochem. Physiol. 2004, 57, 133–141. [Google Scholar]
- Paluzzi, J.P.; Young, P.; Defferrari, M.S.; Orchard, I.; Carlini, C.R.; O’Donnell, M.J. Investigation of the potential involvement of eicosanoid metabolites in anti-diuretic hormone signaling in Rhodnius prolixus. Peptides 2011, in press. [Google Scholar]
- Hirayama, C.; Sugimura, M.; Saito, H.; Nakamura, M. Host plant urease in the hemolymph of the silkworm, Bombyx mori. J. Insect Physiol. 2000, 46, 1415–1421. [Google Scholar] [CrossRef]
- Kurahashi, H.; Atiwetin, P.; Nagaoka, S.; Miyata, S.; Kitajima, S.; Sugimura, Y. Absorption of mulberry root urease to the hemolymph of the silkworm. Bombyx mori. J. Insect Physiol. 2005, 51, 1055–1061. [Google Scholar] [CrossRef]
- Sugimura, M.; Hirayama, C.; Nakamura, M. Selective transport of the mulberry leaf urease from the midgut into the larval hemolymph of the silkworm, Bombyx mori. J. Insect Physiol. 2001, 47, 1133–1138. [Google Scholar] [CrossRef]
- Staniscuaski, F.; Carlini, C.R. Universidade Federal do Rio Grande do Sul: Porto Alegre, Brazil, 2007; Unpublished work.
- Mykles, D.L.; Adams, M.E.; Gade, G.; Lange, A.B.; Marco, H.G.; Orchard, I. Neuropeptide action in insects and crustaceans. Physiol. Biochem. Zool. 2010, 83, 836–846. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stanisçuaski, F.; Carlini, C.R. Plant Ureases and Related Peptides: Understanding Their Entomotoxic Properties. Toxins 2012, 4, 55-67. https://doi.org/10.3390/toxins4020055
Stanisçuaski F, Carlini CR. Plant Ureases and Related Peptides: Understanding Their Entomotoxic Properties. Toxins. 2012; 4(2):55-67. https://doi.org/10.3390/toxins4020055
Chicago/Turabian StyleStanisçuaski, Fernanda, and Célia R. Carlini. 2012. "Plant Ureases and Related Peptides: Understanding Their Entomotoxic Properties" Toxins 4, no. 2: 55-67. https://doi.org/10.3390/toxins4020055




