In Vitro Antiplasmodial Activity of Phospholipases A2 and a Phospholipase Homologue Isolated from the Venom of the Snake Bothrops asper
Abstract
:1. Introduction
2. Results
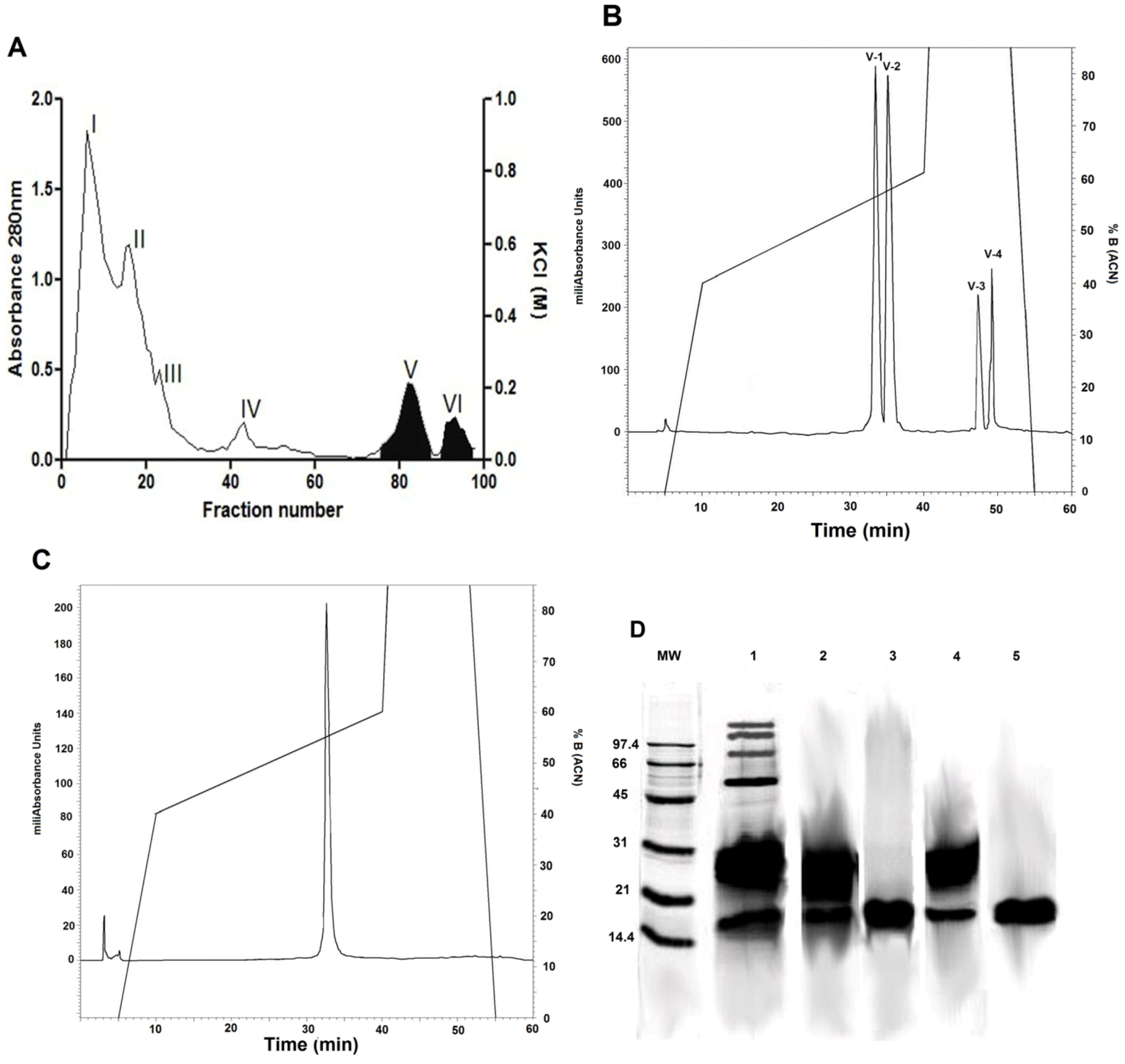
2.1. Isolation of Phospholipase A2 Fractions
2.2. Indirect Hemolytic Activity
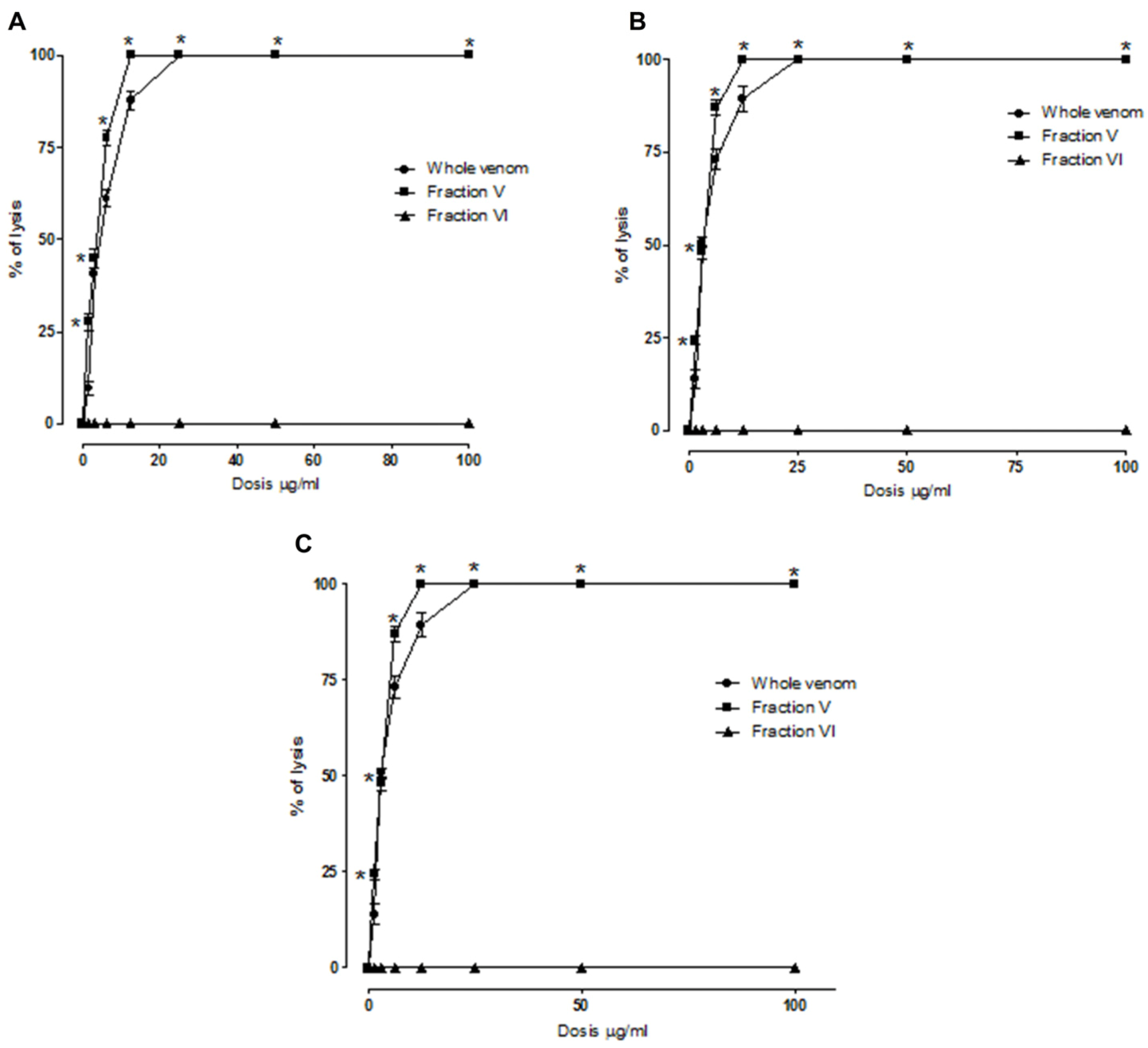
2.3. Antiplasmodial Activity of the Venom, Fractions and Purified PLA2s
| Compound | Antimalarial activity IC50 (µg/mL) | LD50 (µg/kg) | Cytotoxicity CC50 (µg/mL) |
|---|---|---|---|
| B. asper venom | 0.13 ± 0.01 € | 3566 (2561 to 3693) | 38.46 ± 0.95 Ω |
| Fraction V | 1.42 ± 0.56 € | £ > 15000 | 26.98 ± 0.51 Ω |
| Fraction VI | 22.89 ± 1.22 € | £ > 15000 | 67.43 ± 1.03 Ω |
| CQ * | 323.35 ± 6.97 | ND | ND |
2.4. SDS-PAGE
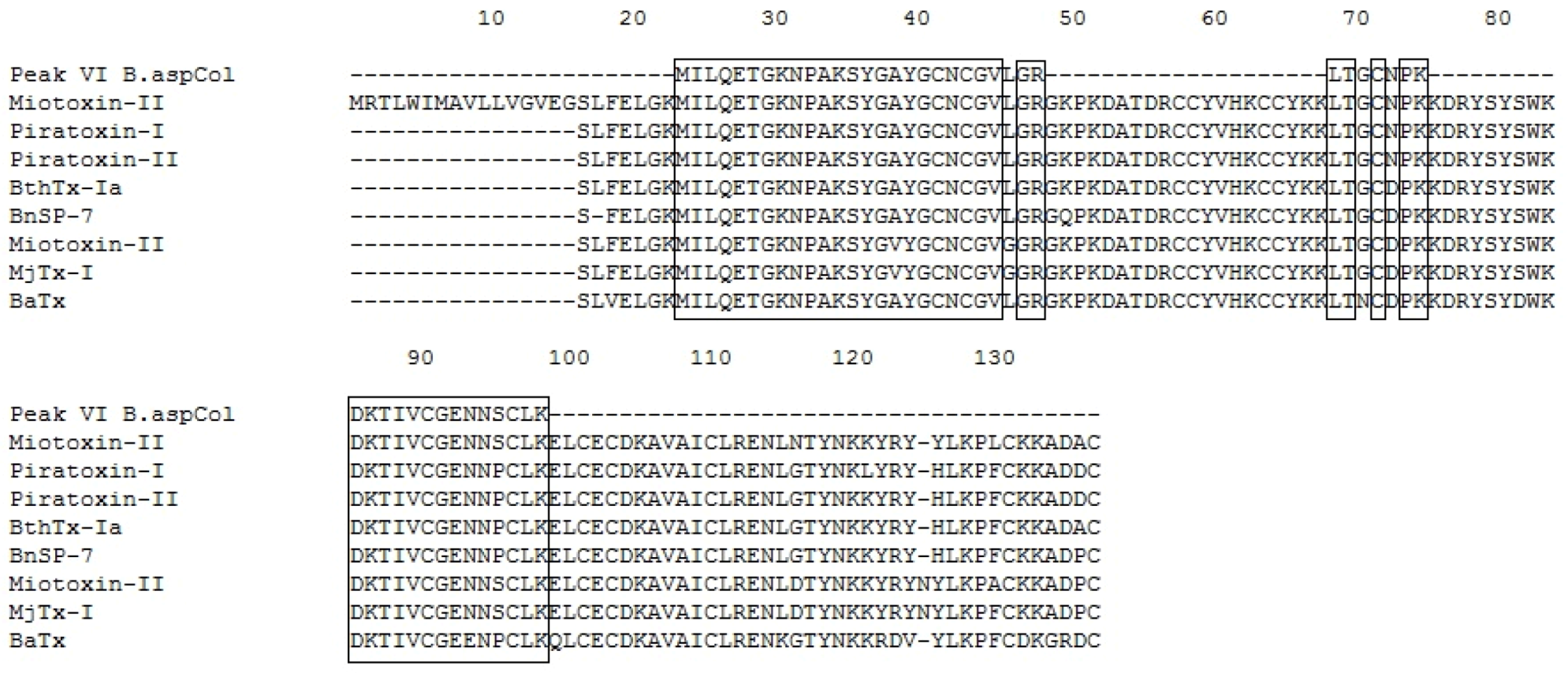
2.5. Mass Spectrometry and Identification of the Protein
| Fraction | MH+ (monoisotopic mass ) | z | MS/MS-derived sequence | Data base ID | Species | Score | Reference | |
|---|---|---|---|---|---|---|---|---|
| Spectrum mill | Mascot | |||||||
| P V-1 | 1944.87 | 3+ | NPVTSYGAYGCNCGVLGR | Q9PVE3.1 | B. asper M1-3-3 | 17.76 | 52 | [35] |
| 1394.64 | 2+ | TIVCGENNSCLK | AAF66702.1 | B. moojeni Myotoxin II precursor | 14.21 | 87 | [36] | |
| 460.74 | 2+ | MILQETGK | Q9PRT7.1 | B. asper Myotoxin IV | - | 37 | [37] | |
| 434.05 | 2+ | CCYVHK | AAF66702.1 | B. moojeni Myotoxin II precursor | - | 25 | [36] | |
| P V-2 | 1944.87 | 3+ | NPVTSYGAYGCNCGVLGR | Q9PVE3.1 | B. asper M1-3-3 | 12.65 | 68 | [35] |
| 1394,57 | 2+ | TIVCGENNSCLK | 1CLP_B | B. asper Myotoxin II | - | 53 | [38] | |
| 1637.76 | 3+ | DKTIVCGENNSCLK | AAF66702.1 | B. moojeni Myotoxin II precursor | 12.23 | 24 | [36] | |
| 952.78 | 2+ | ELCECDK | AAF66702.1 | B. moojeni Myotoxin II precursor | - | 27 | [36] | |
| 996.80 | 1+ | ENLDTYNK | AAF66702.1 | B. moojeni Myotoxin II precursor | 12.69 | 31 | [36] | |
| 802.36 | 2+ | AVAICLR | Q9PRT7.1 | B. asper Myotoxin IV | - | 36 | [37] | |
| P V-3 | 1944.87 | 3+ | NPVTSYGAYGCNCGVLGR | Q9PVE3.1 | B. asper M1-3-3 | 10.85 | 43 | [35] |
| 1394.64 | 2+ | TIVCGENNSCLK | AAF66702.1 | B. moojeni Myotoxin II precursor | - | 57 | [36] | |
| 1637.74 | 3+ | DKTIVCGENNSCLK | AAF66702.1 | B. moojeni Myotoxin II precursor | 17.52 | 31 | [36] | |
| 952.78 | 2+ | ELCECDK | AAF66702.1 | B. moojeni Myotoxin II precursor | - | 27 | [36] | |
| 802.36 | 2+ | AVAICLR | Q9PRT7.1 | B. asper Myotoxin IV | - | 32 | [37] | |
| 1533.66 | 2+ | SYGAYGCNCGVLGR | AAF66703.1 | B. neuwiedi pauloensis PLA2 homolog | 17.32 | 63 | [39] | |
| P V-4 | 2064.41 | 2+ | DATDRCCFVHDCCYGK | P20474.2 | B. asper Myotoxin III | 9.51 | 30 | [35] |
| 1728.75 | 2+ | EICECDKAAAVCFR | 1GMZ_A | B. pirajai Piratoxin III | 8.61 | - | [40] | |
| 1506.59 | 2+ | SGVIICCEGTPCEK | P20474.2 | B. asper Myotoxin III | - | 64 | [35] | |
| 862.56 | 2+ | MILEETK | P20474.2 | B. asper Myotoxin III | - | 35 | [35] | |
| 794.57 | 2+ | AAAVCFR | P86974.1 | B. leucurus blD-PLA2 | - | 26 | [41] | |
| 1273.31 | 2+ | YMAYPDLLCK | P20474.2 | B. asper Myotoxin III | - | 42 | [35] | |
| 675.45 | 2+ | YSYSR | P20474.2 | B. asper Myotoxin III | - | 23 | [35] | |
| P VI | 1329.72 | 2+ | MILQETGKNPAK | Q9IAT9.2 | B. neuwiedi pauloensis BnSP-7 | 11.63 | 42 | [39] |
| 1533.66 | 2+ | SYGAYGCNCGVLGR | AAF66703.1 | B. neuwiedi pauloensis PLA2 homolog | 17.92 | 52 | [39] | |
| 790.04 | 1+ | LTGCNPK | P86453.1 | B. alternatus BaTx | - | 28 | [42] | |
| 1637.56 | 2+ | DKTIVCGENNSCLK | AAF66702.1 | B. moojeni Myotoxin II precursor | - | 21 | [36] | |
| 1394.57 | 2+ | TIVCGENNSCLK | 1CLP_B | B. asper Myotoxin II | - | 77 | [38] | |




2.6. Cytotoxic Activity

2.7. Acute Toxicity
3. Materials and Methods
3.1. Venom and Reagents
3.2. Venom Fractionation
3.3. Electrophoresis and Molecular Mass Determination
3.4. Protein Iidentification by HPLC-nESI-MS/MS
3.5. Search Database
3.6. BLAST Search of the Identified Peptides
3.7. Acute Toxicity of the Venom and Fractions
3.8. Cytotoxic Activity
3.9. Indirect Hemolysis
3.10. Cultivation of Plasmodium falciparum
3.11. Determination of Percentage of Growth Inhibition of P. falciparum by B. asper PLA2 Fractions
3.12. Statistical Analysis
4. Conclusions
Conflict of Interest
Acknowledgements
References
- World Health Organization. World Malaria Report 2008. Available online: http://malaria.who.int/wmr2008/malaria2008.pdf (accessed on 10 August 2012).
- Breman, J.G.; Alilio, M.S.; Mills, A. Conquering the intolerable burden of malaria: What’s new, what’s needed: A summary. Am. J. Trop. Med. Hyg. 2004, 71, 1–15. [Google Scholar]
- World Health Organization. World Malaria Report 2010. Available online: http://www.who.int/malaria/publications/atoz/artemisinin_resistance_containment_2011.pdf (accessed on 13 March 2011).
- Abdel-Sattar, E.; Maes, L.; Salama, M.M. In vitro activities of plant extracts from Saudi Arabia against malaria, leishmaniasis, sleeping sickness and Chagas disease. Phytother. Res. 2010, 24, 1322–1328. [Google Scholar] [CrossRef]
- Ayuko, T.A.; Njau, R.N.; Cornelius, W.; Leah, N.; Ndiege, I.O. In vitro antiplasmodial activity and toxicity assessment of plant extracts used in traditional malaria therapy in the Lake Victoria Region. Mem. Inst. Oswaldo Cruz. 2009, 104, 689–694. [Google Scholar] [CrossRef]
- Gao, B.; Xu, J.; Rodriguez Mdel, C.; Lanz-Mendoza, H.; Hernandez-Rivas, R.; Du, W.; Zhu, S. Characterization of two linear cationic antimalarial peptides in the scorpion Mesobuthus eupeus. Biochimie 2010, 92, 350–359. [Google Scholar] [CrossRef]
- Karunamoorthi, K.; Ilango, K.; Murugan, K. Laboratory evaluation of traditionally used plant-based insect repellent against the malaria vector Anopheles arabiensis Patton (Diptera: Culicidae). Parasitol. Res. 2010, 106, 1217–1223. [Google Scholar] [CrossRef]
- Milhous, W.K.; Weina, P.J. Plant science. The botanical solution for malaria. Science 2010, 327, 279–280. [Google Scholar] [CrossRef]
- Muller, G.C.; Beier, J.C.; Traore, S.F.; Toure, M.B.; Traore, M.M.; Bah, S.; Doumbia, S.; Schlein, Y. Successful field trial of attractive toxic sugar bait (ATSB) plant-spraying methods against malaria vectors in the Anopheles gambiae complex in Mali, West Africa. Malar. J. 2010, 9, 210. [Google Scholar] [CrossRef]
- Campbell, J.; Lamar, W. The Venomous Reptiles of the Western Hemisphere; Cornell University Press: Chichester, Scotland, UK, 2004. [Google Scholar]
- Alape-Giron, A.; Sanz, L.; Escolano, J.; Flores-Diaz, M.; Madrigal, M.; Sasa, M.; Calvete, J.J. Snake venomics of the lancehead pitviper Bothrops asper: Geographic, individual, and ontogenetic variations. J. Proteome Res. 2008, 7, 3556–3571. [Google Scholar] [CrossRef]
- Angulo, Y.; Lomonte, B. Biochemistry and toxicology of toxins purified from the venom of the snake Bothrops asper. Toxicon 2009, 54, 949–957. [Google Scholar] [CrossRef]
- Six, D.A.; Dennis, E.A. The expanding superfamily of phospholipase A(2) enzymes: Classification and characterization. Biochim. Biophys. Acta 2000, 1488, 1–19. [Google Scholar] [CrossRef]
- Talvinen, K.A.; Nevalainen, T.J. Cloning of a novel phospholipase A2 from the cnidarian Adamsia carciniopados. Comp. Biochem. Physiol. B 2002, 132, 571–578. [Google Scholar] [CrossRef]
- Andriao-Escarso, S.H.; Soares, A.M.; Rodrigues, V.M.; Angulo, Y.; Diaz, C.; Lomonte, B.; Gutierrez, J.M.; Giglio, J.R. Myotoxic phospholipases A(2) in bothrops snake venoms: Effect of chemical modifications on the enzymatic and pharmacological properties of bothropstoxins from Bothrops jararacussu. Biochimie 2000, 82, 755–763. [Google Scholar] [CrossRef]
- Barbosa, P.S.; Martins, A.M.; Havt, A.; Toyama, D.O.; Evangelista, J.S.; Ferreira, D.P.; Joazeiro, P.P.; Beriam, L.O.; Toyama, M.H.; Fonteles, M.C.; et al. Renal and antibacterial effects induced by myotoxin I and II isolated from Bothrops jararacussu venom. Toxicon 2005, 46, 376–386. [Google Scholar] [CrossRef]
- Costa Torres, A.F.; Dantas, R.T.; Toyama, M.H.; Diz Filho, E.; Zara, F.J.; Rodrigues de Queiroz, M.G.; Pinto Nogueira, N.A.; Rosa de Oliveira, M.; de Oliveira Toyama, D.; Monteiro, H.S.; et al. Antibacterial and antiparasitic effects of Bothrops marajoensis venom and its fractions: Phospholipase A2 and L-amino acid oxidase. Toxicon 2010, 55, 795–804. [Google Scholar] [CrossRef]
- Evangelista, I.L.; Martins, A.M.; Nascimento, N.R.; Havt, A.; Evangelista, J.S.; de Noroes, T.B.; Toyama, M.H.; Diz-Filho, E.B.; toyama Dde, O.; Fonteles, M.C.; et al. Renal and cardiovascular effects of Bothrops marajoensis venom and phospholipase A2. Toxicon 2010, 55, 1061–1070. [Google Scholar] [CrossRef]
- Gutierrez, J.M.; Lomonte, B. Phospholipase A2 myotoxins from Bothrops snake venoms. Toxicon 1995, 33, 1405–1424. [Google Scholar] [CrossRef]
- Harris, J.B.; Grubb, B.D.; Maltin, C.A.; Dixon, R. The neurotoxicity of the venom phospholipases A(2), notexin and taipoxin. Exp. Neurol. 2000, 161, 517–526. [Google Scholar] [CrossRef]
- Kini, R.M. Excitement ahead: Structure, function and mechanism of snake venom phospholipase A2 enzymes. Toxicon 2003, 42, 827–840. [Google Scholar] [CrossRef]
- Kini, R.M.; Evans, H.J. Structure-function relationships of phospholipases. The anticoagulant region of phospholipases A2. J. Biol. Chem. 1987, 262, 14402–14407. [Google Scholar]
- Landucci, E.C.; de Castro, R.C.; Toyama, M.; Giglio, J.R.; Marangoni, S.; de Nucci, G.; Antunes, E. Inflammatory oedema induced by the lys-49 phospholipase A(2) homologue piratoxin-i in the rat and rabbit. Effect of polyanions and p-bromophenacyl bromid. Biochem. Pharmacol. 2000, 59, 1289–1294. [Google Scholar]
- Murakami, M.T.; Arruda, E.Z.; Melo, P.A.; Martinez, A.B.; Calil-Elias, S.; Tomaz, M.A.; Lomonte, B.; Gutierrez, J.M.; Arni, R.K. Inhibition of myotoxic activity of Bothrops asper myotoxin II by the anti-trypanosomal drug suramin. J. Mol. Biol. 2005, 350, 416–426. [Google Scholar] [CrossRef]
- Quintana, J.C.; Chacon, A.M.; Vargas, L.; Segura, C.; Gutierrez, J.M.; Alarcon, J.C. Antiplasmodial effect of the venom of Crotalus durissus cumanensis, crotoxin complex and Crotoxin B. Acta Trop. 2012, 124, 126–132. [Google Scholar] [CrossRef]
- Deregnaucourt, C.; Schrevel, J. Bee venom phospholipase A2 induces stage-specific growth arrest of the intraerythrocytic Plasmodium falciparum via modifications of human serum components. J. Biol. Chem. 2000, 275, 39973–39980. [Google Scholar] [CrossRef]
- Guillaume, C.; Deregnaucourt, C.; Clavey, V.; Schrevel, J. Anti-Plasmodium properties of group IA, IB, IIA and III secreted phospholipases A2 are serum-dependent. Toxicon 2004, 43, 311–318. [Google Scholar] [CrossRef]
- Passero, L.F.; Laurenti, M.D.; Tomokane, T.Y.; Corbett, C.E.; Toyama, M.H. The effect of phospholipase A2 from Crotalus durissus collilineatus on Leishmania (Leishmania) amazonensis infection. Parasitol. Res. 2008, 102, 1025–1033. [Google Scholar] [CrossRef]
- Lomonte, B.; Rangel, J. Snake venom Lys49 myotoxins: From phospholipases A(2) to non-enzymatic membrane disruptors. Toxicon 2012, 60, 520–530. [Google Scholar] [CrossRef]
- Nuñez, V.; Arce, V.; Gutierrez, J.M.; Lomonte, B. Structural and functional characterization of myotoxin I, a Lys49 phospholipase A2 homologue from the venom of the snake Bothrops atrox. Toxicon 2004, 44, 91–101. [Google Scholar] [CrossRef]
- Santamaria, C.; Larios, S.; Quiros, S.; Pizarro-Cerda, J.; Gorvel, J.P.; Lomonte, B.; Moreno, E. Bactericidal and antiendotoxic properties of short cationic peptides derived from a snake venom Lys49 phospholipase A2. Antimicrob. Agents Chemother. 2005, 49, 1340–1345. [Google Scholar] [CrossRef]
- Hill, D.A.; Desai, S.A. Malaria parasite mutants with altered erythrocyte permeability: A new drug resistance mechanism and important molecular tool. Future Microbiol. 2010, 5, 81–97. [Google Scholar] [CrossRef]
- Vial, H.J.; Ancelin, M.L. Malarial lipids. In Malaria: Parasite Biology, Pathogenesis, and Protection; Sherman, I.W., Ed.; ASM Press: Washington, DC, USA, 1998; pp. 159–175. [Google Scholar]
- Moll, G.N.; Vial, H.J.; van der Wiele, F.C.; Ancelin, M.L.; Roelofsen, B.; Slotboom, A.J.; de Haas, G.H.; van Deenen, L.L.; Op den Kamp, J.A. Selective elimination of malaria infected erythrocytes by a modified phospholipase A2 in vitro. Biochim. Biophys. Acta 1990, 1024, 189–192. [Google Scholar] [CrossRef]
- Lizano, S.; Lambeau, G.; Lazdunski, M. Cloning and cDNA sequence analysis of Lys(49) and Asp(49) basic phospholipase A(2) myotoxin isoforms from Bothrops asper. Int. J. Biochem. Cell Biol. 2001, 33, 127–132. [Google Scholar] [CrossRef]
- Soares, A.M.; Rodrigues, V.M.; Homsi-Brandeburgo, M.I.; Toyama, M.H.; Lombardi, F.R.; Arni, R.K.; Giglio, J.R. A rapid procedure for the isolation of the Lys-49 myotoxin II from Bothrops moojeni (caissaca) venom: Biochemical characterization, crystallization, myotoxic and edematogenic activity. Toxicon 1998, 36, 503–514. [Google Scholar] [CrossRef]
- Diaz, C.; Lomonte, B.; Zamudio, F.; Gutierrez, J.M. Purification and characterization of myotoxin IV, a phospholipase A2 variant, from Bothrops asper snake venom. Nat. Toxins 1995, 3, 26–31. [Google Scholar] [CrossRef]
- Arni, R.K.; Ward, R.J.; Gutierrez, J.M.; Tulinsky, A. Structure of a calcium-independent phospholipase-like myotoxic protein from Bothrops asper venom. Acta Crystallogr. D 1995, 51, 311–317. [Google Scholar] [CrossRef]
- Rodrigues, V.M.; Soares, A.M.; Mancin, A.C.; Fontes, M.R.; Homsi-Brandeburgo, M.I.; Giglio, J.R. Geographic variations in the composition of myotoxins from Bothrops neuwiedi snake venoms: Biochemical characterization and biological activity. Comp. Biochem. Physiol. A 1998, 121, 215–222. [Google Scholar] [CrossRef]
- Rigden, D.J.; Hwa, L.W.; Marangoni, S.; Toyama, M.H.; Polikarpov, I. The structure of the D49 phospholipase A2 piratoxin III from Bothrops pirajai reveals unprecedented structural displacement of the calcium-binding loop: Possiblerelationship to cooperative substrate binding. Acta Crystallogr. D 2003, 59, 255–262. [Google Scholar] [CrossRef]
- Higuchi, D.A.; Barbosa, C.M.; Bincoletto, C.; Chagas, J.R.; Magalhaes, A.; Richardson, M.; Sanchez, E.F.; Pesquero, J.B.; Araujo, R.C.; Pesquero, J.L. Purification and partial characterization of two phospholipases A2 from Bothrops leucurus (white-tailed-jararaca) snake venom. Biochimie 2007, 89, 319–328. [Google Scholar] [CrossRef]
- Ponce-Soto, L.A.; Lomonte, B.; Gutierrez, J.M.; Rodrigues-Simioni, L.; Novello, J.C.; Marangoni, S. Structural and functional properties of BaTX, a new Lys49 phospholipase A2 homologue isolated from the venom of the snake Bothrops alternatus. Biochim. Biophys. Acta 2007, 1770, 585–593. [Google Scholar] [CrossRef]
- Vadas, P. Group II phospholipases A2 are indirectly cytolytic in the presence of exogenous phospholipid. Biochim. Biophys. Acta 1997, 1346, 193–197. [Google Scholar] [CrossRef]
- Otero, R.; Nunez, V.; Osorio, R.G.; Gutierrez, J.M.; Giraldo, C.A.; Posada, L.E. Ability of six Latin American antivenoms to neutralize the venom of mapana equis (Bothrops atrox) from Antioquia and Choco (Colombia). Toxicon 1995, 33, 809–815. [Google Scholar] [CrossRef]
- Otero-Patino, R. Epidemiological, clinical and therapeutic aspects of Bothrops asper bites. Toxicon 2009, 54, 998–1011. [Google Scholar] [CrossRef]
- Otero-Patino, R.; Cardoso, J.L.; Higashi, H.G.; Nunez, V.; Diaz, A.; Toro, M.F.; Garcia, M.E.; Sierra, A.; Garcia, L.F.; Moreno, A.M.; et al. A randomized, blinded, comparative trial of one pepsin-digested and two whole IgG antivenoms for Bothrops snake bites in Uraba, Colombia. The Regional Group on Antivenom Therapy Research (REGATHER). Am. J. Trop. Med. Hyg. 1998, 58, 183–189. [Google Scholar]
- Gutierrez, J.M.; Ownby, C.L.; Odell, G.V. Pathogenesis of myonecrosis induced by crude venom and a myotoxin of Bothrops asper. Exp. Mol. Pathol. 1984, 40, 367–379. [Google Scholar] [CrossRef]
- Gutierrez, J.M.; Ownby, C.L.; Odell, G.V. Isolation of a myotoxin from Bothrops asper venom: Partial characterization and action on skeletal muscle. Toxicon 1984, 22, 115–128. [Google Scholar] [CrossRef]
- Trebien, H.A.; Calixto, J.B. Pharmacological evaluation of rat paw oedema induced by Bothrops jararaca venom. Agents Actions 1989, 26, 292–300. [Google Scholar] [CrossRef]
- Choi, S.J.; Parent, R.; Guillaume, C.; Deregnaucourt, C.; Delarbre, C.; Ojcius, D.M.; Montagne, J.J.; Celerier, M.L.; Phelipot, A.; Amiche, M.; et al. Isolation and characterization of Psalmopeotoxin I and II: Two novel antimalarial peptides from the venom of the tarantula Psalmopoeus cambridgei. FEBS Lett. 2004, 572, 109–117. [Google Scholar] [CrossRef]
- Shinohara, L.; de Freitas, S.F.; da Silva, R.J.; Guimaraes, S. In vitro effects of Crotalus durissus terrificus and Bothrops jararaca venoms on Giardia duodenalis trophozoites. Parasitol. Res. 2006, 98, 339–344. [Google Scholar] [CrossRef]
- Adade, C.M.; Cons, B.L.; Melo, P.A.; Souto-Padron, T. Effect of Crotalus viridis viridis snake venom on the ultrastructure and intracellular survival of Trypanosoma cruzi. Parasitology 2011, 138, 46–58. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Shevchenko, A.; Wilm, M.; Vorm, O.; Mann, M. Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal. Chem. 1996, 68, 850–858. [Google Scholar] [CrossRef]
- Matrix Science. Mascot MS/MS Ion Search. Available online: http://www.matrixscience.com/cgi/search_form.pl?FORMVER=2&SEARCH=MIS (accessed on 12 May 2012).
- NCBI. Standard Protein Blast. Available online: http://blast.ncbi.nlm.nih.gov/Blast.cgi?PAGE=Proteins (accessed on 15 May 2012).
- Lomonte, B.; Gutierrez, J.M.; Romero, M.; Nunez, J.; Tarkowski, A.; Hanson, L.A. An MTT-based method for the in vivo quantification of myotoxic activity of snake venoms and its neutralization by antibodies. J. Immunol. Methods 1993, 161, 231–237. [Google Scholar] [CrossRef]
- Habermann, E.; Hardt, K.L. A sensitive and specific plate test for the quantitation of phospholipases. Anal. Biochem. 1972, 50, 163–173. [Google Scholar]
- Gutierrez, J.M.; Chaves, F.; Rojas, E.; Elizondo, J.; Avila, C.; Cerdas, L. Production of monovalent anti-Bothrops asper antivenom: Development of immune response in horses and neutralizing ability. Rev. Biol. Trop. 1988, 36, 511–517. [Google Scholar]
- Trager, W.; Jenson, J.B. Cultivation of malarial parasites. Nature 1978, 273, 621–622. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Castillo, J.C.Q.; Vargas, L.J.; Segura, C.; Gutiérrez, J.M.; Pérez, J.C.A. In Vitro Antiplasmodial Activity of Phospholipases A2 and a Phospholipase Homologue Isolated from the Venom of the Snake Bothrops asper. Toxins 2012, 4, 1500-1516. https://doi.org/10.3390/toxins4121500
Castillo JCQ, Vargas LJ, Segura C, Gutiérrez JM, Pérez JCA. In Vitro Antiplasmodial Activity of Phospholipases A2 and a Phospholipase Homologue Isolated from the Venom of the Snake Bothrops asper. Toxins. 2012; 4(12):1500-1516. https://doi.org/10.3390/toxins4121500
Chicago/Turabian StyleCastillo, Juan Carlos Quintana, Leidy Johana Vargas, Cesar Segura, José María Gutiérrez, and Juan Carlos Alarcón Pérez. 2012. "In Vitro Antiplasmodial Activity of Phospholipases A2 and a Phospholipase Homologue Isolated from the Venom of the Snake Bothrops asper" Toxins 4, no. 12: 1500-1516. https://doi.org/10.3390/toxins4121500
APA StyleCastillo, J. C. Q., Vargas, L. J., Segura, C., Gutiérrez, J. M., & Pérez, J. C. A. (2012). In Vitro Antiplasmodial Activity of Phospholipases A2 and a Phospholipase Homologue Isolated from the Venom of the Snake Bothrops asper. Toxins, 4(12), 1500-1516. https://doi.org/10.3390/toxins4121500






