Hemodynamic Effects of Anthrax Toxins in the Rabbit Model and the Cardiac Pathology Induced by Lethal Toxin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Telemetry Instrumentation and Data Acquisition
2.3. Challenge Procedure
2.4. Measurement of Cardiac Troponin I
2.5. Histological Analysis
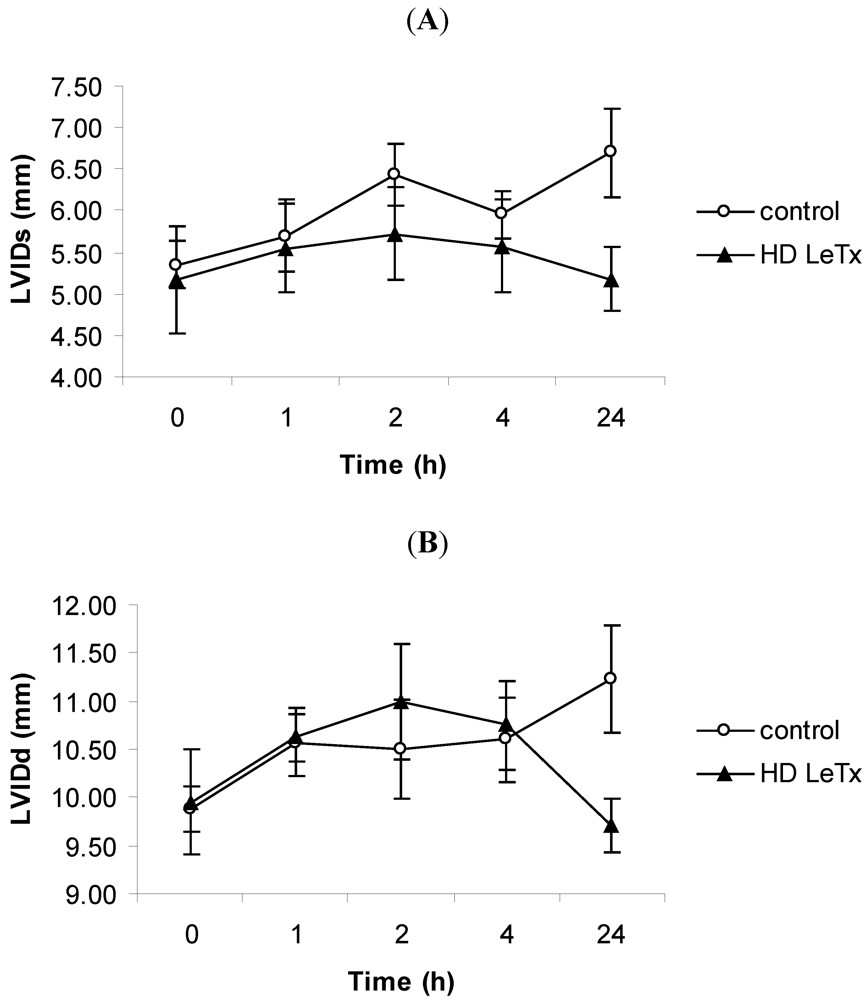
2.6. Echocardiography
2.7. Statistical Analysis
3. Results
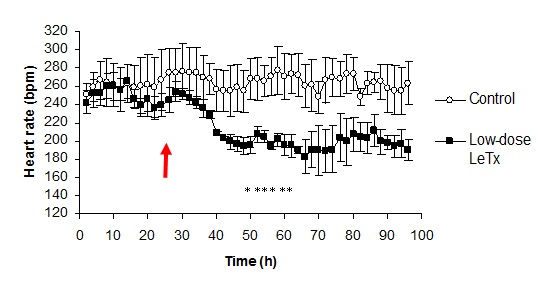
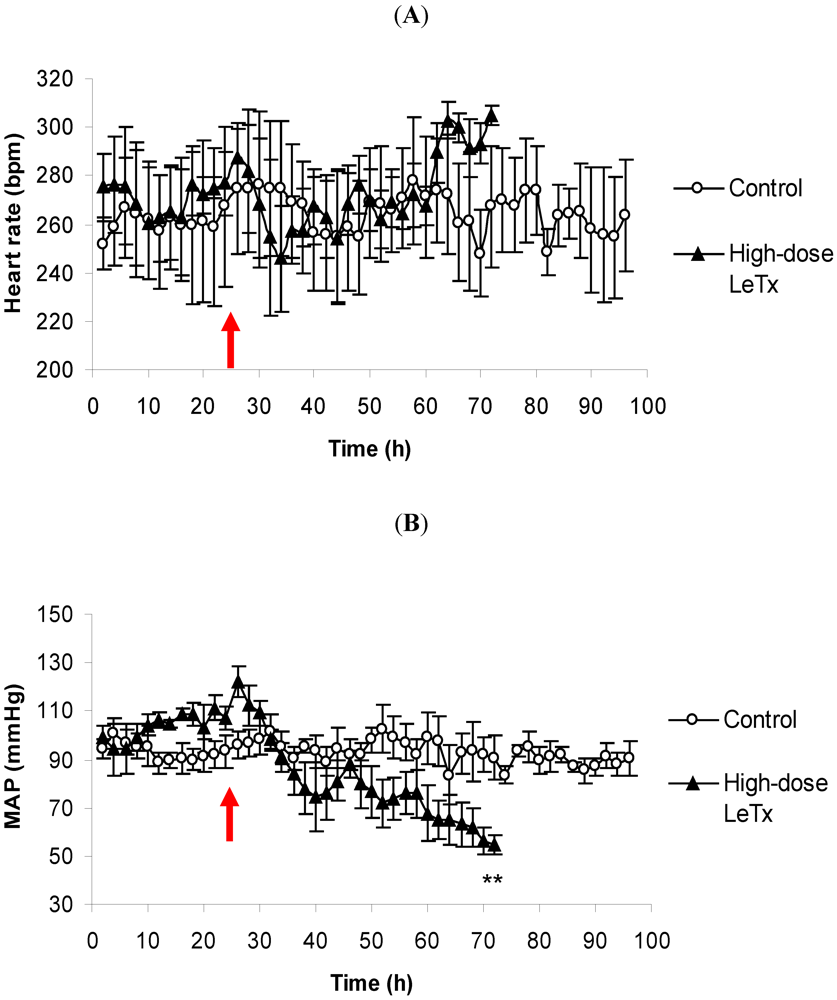
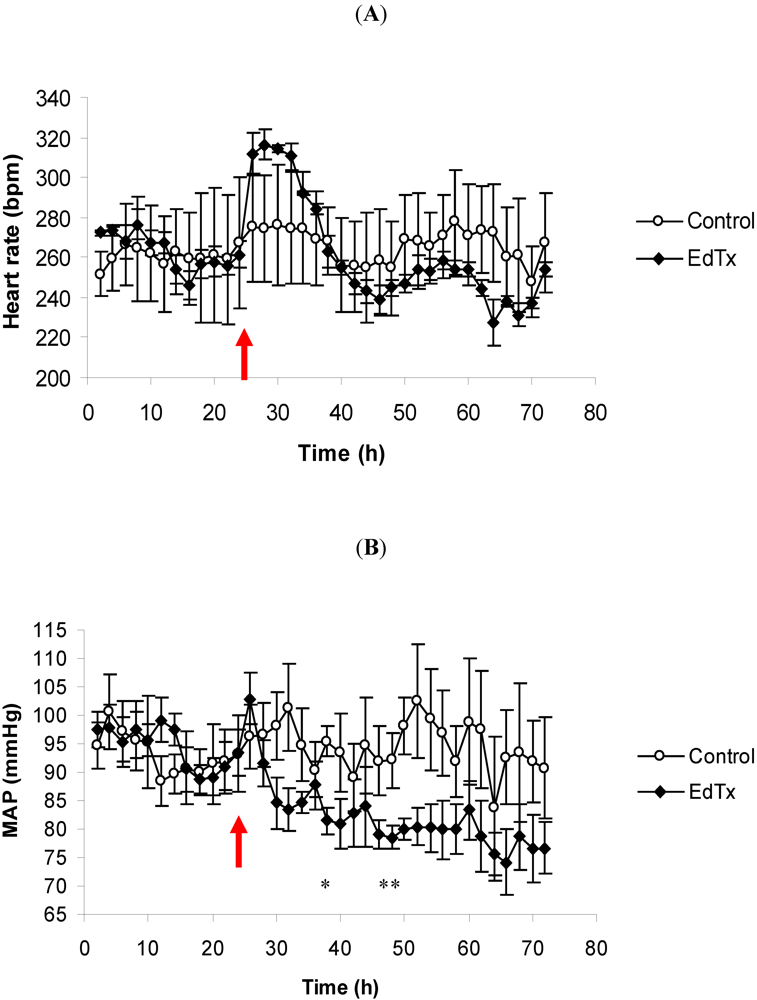
3.1. Changes in Heart Rate and Mean Arterial Pressure

3.2. Cardiac Pathology
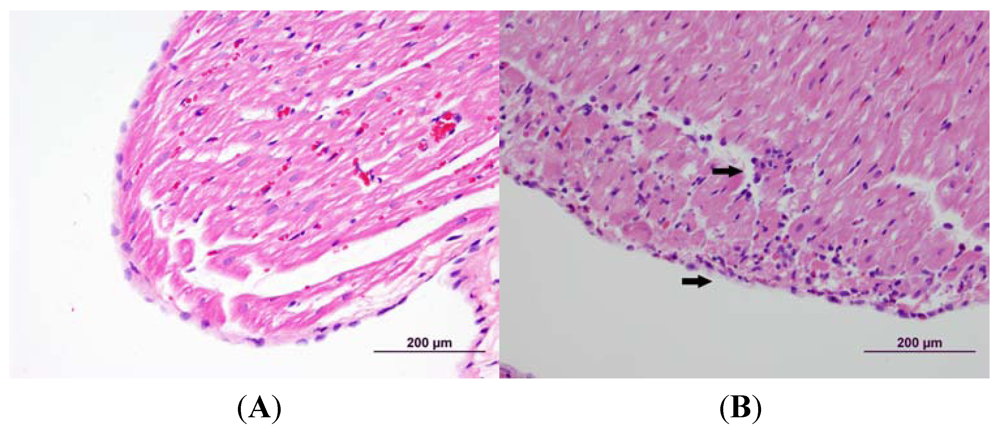
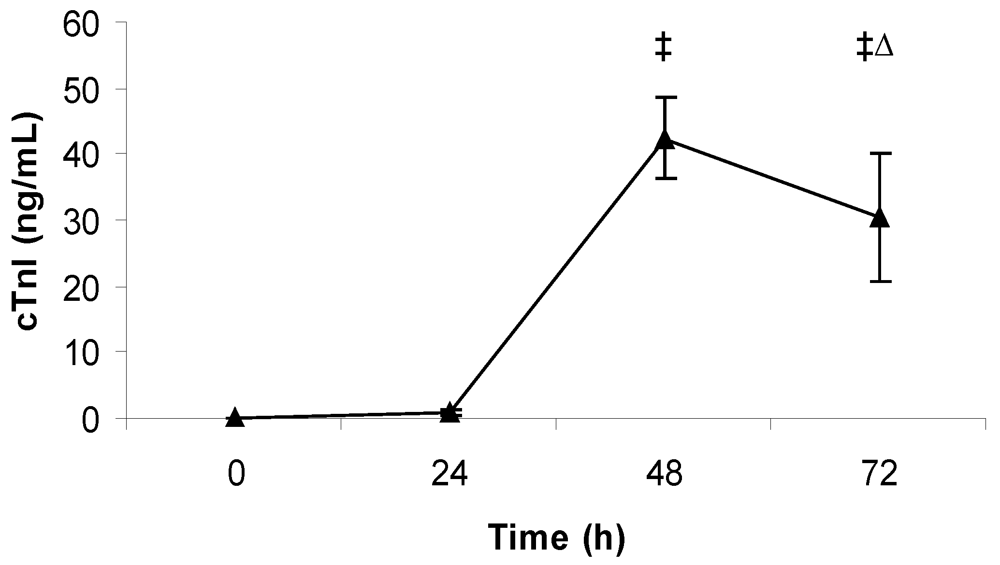
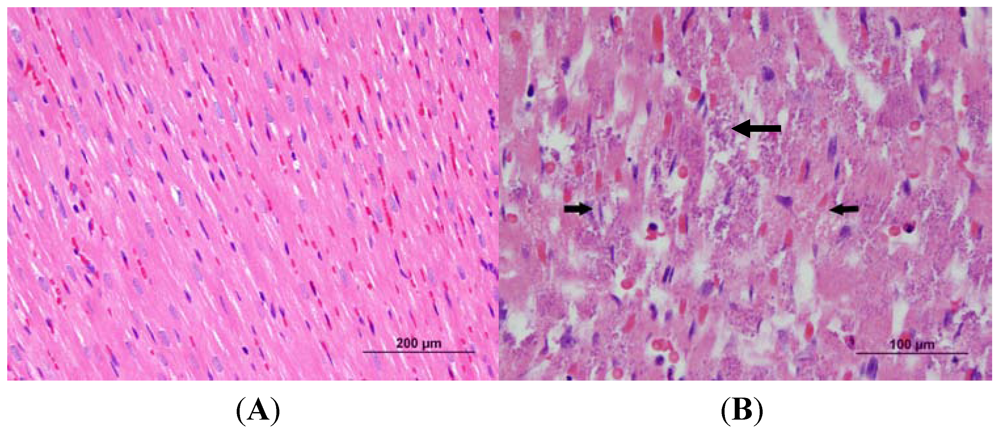
3.3. Echocardiography
4. Discussion
5. Conclusions
Acknowledgments
Conflict of Interest
- The authors declare no conflict of interest.
References
- Borio, L.; Frank, D.; Mani, V.; Chiriboga, C.; Pollanen, M.; Ripple, M.; Ali, S.; DiAngelo, C.; Lee, J.; Arden, J.; et al. Death due to bioterrorism-related inhalational anthrax: report of 2 patients. J. Am. Med. Assoc. 2001, 286, 2554–2559. [Google Scholar]
- Grinberg, L.M.; Abramova, F.A.; Yampolskaya, O.V.; Walker, D.H.; Smith, J.H. Quantitative pathology of inhalational anthrax I: Quantitative microscopic findings. Mod. Pathol. 2001, 14, 482–495. [Google Scholar]
- Jernigan, J.A.; Stephens, D.S.; Ashford, D.A.; Omenaca, C.; Topiel, M.S.; Galbraith, M.; Tapper, M.; Fisk, T.L.; Zaki, S.; Popovic, T.; et al. Bioterrorism-related inhalational anthrax: the first 10 cases reported in the United States. Emerg. Infect. Dis. 2001, 7, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Mina, B.; Dym, J.P.; Kuepper, F.; Tso, R.; Arrastia, C.; Kaplounova, I.; Faraj, H.; Kwapniewski, A.; Krol, C.M.; Grosser, M.; et al. Fatal inhalational anthrax with unknown source of exposure in a 61-year-old woman in New York City. J. Am. Med. Assoc. 2002, 287, 858–862. [Google Scholar]
- Mock, M.; Fouet, A. Anthrax. Annu. Rev. Microbiol. 2001, 55, 647–671. [Google Scholar]
- Brossier, F.; Mock, M. Toxins of Bacillus anthracis. Toxicon 2001, 39, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Duesbery, N.S.; Vande Woude, G.F. Anthrax lethal factor causes proteolytic inactivation of mitogen-activated protein kinase kinase. J. Appl. Microbiol. 1999, 87, 289–293. [Google Scholar]
- Mourez, M. Anthrax toxins. Rev. Physiol. Biochem. Pharmacol. 2004, 152, 135–164. [Google Scholar]
- Duesbery, N.S.; Vande Woude, G.F. Anthrax toxins. Cell. Mol. Life Sci. 1999, 55, 1599–1609. [Google Scholar]
- Collier, R.J.; Young, J.A. Anthrax toxin. Annu. Rev. Cell. Dev. Biol. 2003, 19, 45–70. [Google Scholar]
- Little, S.F.; Ivins, B.E. Molecular pathogenesis of Bacillus anthracis infection. Microbes Infect. 1999, 1, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Kirby, J.E. Anthrax lethal toxin induces human endothelial cell apoptosis. Infect. Immun. 2004, 72, 430–439. [Google Scholar]
- Popov, S.G.; Villasmil, R.; Bernardi, J.; Grene, E.; Cardwell, J.; Wu, A.; Alibek, D.; Bailey, C.; Alibek, K. Lethal toxin of Bacillus anthracis causes apoptosis of macrophages. Biochem. Biophys. Res. Commun. 2002, 293, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Pulendran, B. Anthrax lethal toxin: a weapon of multisystem destruction. Cell. Mol. Life Sci. 2004, 61, 2859–2865. [Google Scholar]
- Baldari, C.T.; Tonello, F.; Paccani, S.R.; Montecucco, C. Anthrax toxins: A paradigm of bacterial immune suppression. Trends Immunol. 2006, 27, 434–440. [Google Scholar]
- Cui, X.; Moayeri, M.; Li, Y.; Li, X.; Haley, M.; Fitz, Y.; Correa-Araujo, R.; Banks, S.M.; Leppla, S.H.; Eichacker, P.Q. Lethality during continuous anthrax lethal toxin infusion is associated with circulatory shock but not inflammatory cytokine or nitric oxide release in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, R699–R709. [Google Scholar]
- Culley, N.C.; Pinson, D.M.; Chakrabarty, A.; Mayo, M.S.; Levine, S.M. Pathophysiological manifestations in mice exposed to anthrax lethal toxin. Infect. Immun. 2005, 73, 7006–7010. [Google Scholar]
- Firoved, A.M.; Miller, G.F.; Moayeri, M.; Kakkar, R.; Shen, Y.; Wiggins, J.F.; McNally, E.M.; Tang, W.J.; Leppla, S.H. Bacillus anthracis edema toxin causes extensive tissue lesions and rapid lethality in mice. Am. J. Pathol. 2005, 167, 1309–1320. [Google Scholar] [CrossRef] [PubMed]
- Moayeri, M.; Haines, D.; Young, H.A.; Leppla, S.H. Bacillus anthracis lethal toxin induces TNF-alpha-independent hypoxia-mediated toxicity in mice. J. Clin. Invest. 2003, 112, 670–682. [Google Scholar] [PubMed]
- Tournier, J.N.; Quesnel-Hellmann, A.; Cleret, A.; Vidal, D.R. Contribution of toxins to the pathogenesis of inhalational anthrax. Cell. Microbiol. 2007, 9, 555–565. [Google Scholar]
- Cui, X.; Li, Y.; Li, X.; Laird, M.W.; Subramanian, M.; Moayeri, M.; Leppla, S.H.; Fitz, Y.; Su, J.; Sherer, K.; Eichacker, P.Q. Bacillus anthracis edema and lethal toxin have different hemodynamic effects but function together to worsen shock and outcome in a rat model. J. Infect. Dis. 2007, 195, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Watson, L.E.; Kuo, S.R.; Katki, K.; Dang, T.; Park, S.K.; Dostal, D.E.; Tang, W.J.; Leppla, S.H.; Frankel, A.E. Anthrax toxins induce shock in rats by depressed cardiac ventricular function. PLoS One 2007, 2, e466. [Google Scholar]
- Watson, L.E.; Mock, J.; Lal, H.; Lu, G.; Bourdeau, R.W.; Tang, W.J.; Leppla, S.H.; Dostal, D.E.; Frankel, A.E. Lethal and edema toxins of anthrax induce distinct hemodynamic dysfunction. Front. Biosci. 2007, 12, 4670–4675. [Google Scholar]
- Lawrence, W.S.; Hardcastle, J.M.; Brining, D.L.; Weaver, L.E.; Ponce, C.; Whorton, E.B.; Peterson, J.W. The physiologic responses of Dutch belted rabbits infected with inhalational anthrax. Comp. Med. 2009, 59, 257–265. [Google Scholar]
- Mabry, R.; Brasky, K.; Geiger, R.; Carrion, R.; Hubbard, G.B.; Leppla, S.; Patterson, J.L.; Georgiou, G.; Iverson, B.L. Detection of anthrax toxin in the serum of animals infected with Bacillus anthracis by using engineered immunoassays. Clin. Vaccine Immunol. 2006, 13, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Molin, F.D.; Fasanella, A.; Simonato, M.; Garofolo, G.; Montecucco, C.; Tonello, F. Ratio of lethal and edema factors in rabbit systemic anthrax. Toxicon 2008, 52, 824–828. [Google Scholar]
- Cain, A.E.; Tanner, D.M.; Khalil, R.A. Endothelin-1-induced enhancement of coronary smooth muscle contraction via MAPK-dependent and MAPK-independent [Ca(2+)](i) sensitization pathways. Hypertension 2002, 39, 543–549. [Google Scholar]
- Streefkerk, J.O.; Hoogaars, W.M.; Christoffels, V.M.; Sand, C.; Pfaffendorf, M.; Peters, S.L.; van Zwieten, P.A. Vasopressin-induced vasoconstriction is dependent on MAPKerk1/2 phosphorylation. Fundam. Clin. Pharmacol. 2004, 18, 45–50. [Google Scholar]
- Sweeny, D.A.; Cui, X.; Solomon, S.B.; Vitberg, D.A.; Migone, T.S.; Scher, D.; Danner, R.L.; Natanson, C.; Subramanian, G.M.; Eichacker, P.Q. Anthrax lethal and edema toxins produce different patterns of cardiovascular and renal dysfunction and synergistically decrease survival in canines. J. Investig. Dermatol. 2010, 202, 1885–1896. [Google Scholar]
- Hicks, C.W.; Li, Y.; Okugawa, S.; Solomon, S.B.; Moayeri, M.; Leppla, S.H.; Mohanty, A.; Subramanian, G.M.; Mignone, T.S.; Fitz, Y.; Cui, X.; Eichacker, P.Q. Anthrax edema toxin has cAMP-mediated stimulatory effects and high-dose lethal toxin has depressant effects in an isolated perfused rat heart model. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H1108–H1118. [Google Scholar]
- Robinson, R.B.; Siegelbaum, S.A. Hyperpolarization-activated cation currents: From molecules to physiological function. Annu. Rev. Physiol. 2003, 65, 453–480. [Google Scholar]
- Namm, D.H.; Leader, J.P. Occurrence and function of cyclic nucleotides in blood vessels. Blood Vessel. 1976, 13, 24–47. [Google Scholar]
- Purcell, N.H.; Wilkins, B.J.; York, A.; Saba-El-Leil, M.K.; Meloche, S.; Robbins, J.; Molkentin, J.D. Genetic inhibition of cardiac ERK1/2 promotes stress-induced apoptosis and heart failure but has no effect on hypertrophy in vivo. Proc. Natl. Acad. Sci. USA 2007, 104, 14074–14079. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lawrence, W.S.; Marshall, J.R.; Zavala, D.L.; Weaver, L.E.; Baze, W.B.; Moen, S.T.; Whorton, E.B.; Gourley, R.L.; Peterson, J.W. Hemodynamic Effects of Anthrax Toxins in the Rabbit Model and the Cardiac Pathology Induced by Lethal Toxin. Toxins 2011, 3, 721-736. https://doi.org/10.3390/toxins3060721
Lawrence WS, Marshall JR, Zavala DL, Weaver LE, Baze WB, Moen ST, Whorton EB, Gourley RL, Peterson JW. Hemodynamic Effects of Anthrax Toxins in the Rabbit Model and the Cardiac Pathology Induced by Lethal Toxin. Toxins. 2011; 3(6):721-736. https://doi.org/10.3390/toxins3060721
Chicago/Turabian StyleLawrence, William S., Jeffrey R. Marshall, Diana L. Zavala, Lori E. Weaver, Wallace B. Baze, Scott T. Moen, Elbert B. Whorton, Randy L. Gourley, and Johnny W. Peterson. 2011. "Hemodynamic Effects of Anthrax Toxins in the Rabbit Model and the Cardiac Pathology Induced by Lethal Toxin" Toxins 3, no. 6: 721-736. https://doi.org/10.3390/toxins3060721