Staphylococcal Enterotoxins
Abstract
:1. Introduction
1.1. Source
| Staphylococcal Enterotoxin | Feature | Binding to Class II MHC |
|---|---|---|
| SEA | Most common toxin associated with staphylococcal food poisoning | Alpha and beta chains [12] |
| SEB | Studied as a biological weapon | Alpha chain [13] |
| SEC | Commonly isolated from animals [14] | Outside the binding groove on the flanking helix from the α chain [15] |
| SED | Food poisoning [16] | Alpha and Beta chains [17] |
| SEE | Food poisoning [9] | Beta chain [18] |
| SEF | Associated with toxic shock syndrome [8] | Binds to alpha and beta chains [19] |
| SEG | Minor role in food poisoning [10] | SEB-like interaction with a chain [20] |
| SEH | Food poisoning [10,11] | Alpha chain [21] |
| SEI | Minor role in food poisoning [10] | Beta chain [22] |
1.2. Structure
1.3. Properties
2. SEs in Food-Borne Poisoning Associated Diarrhea
3. Staphylococcal Enterotoxins in Nosocomial and Antibiotic-Associated Diarrhea
4. Gastro-Intestinal Inflammatory Injury Associated with Enterotoxigenic Diarrheal Diseases

5. Mechanisms of Action
5.1. Emetic effect of SEs
5.2. SE superantigenic property in immunopathogenesis associated with staphylococcal diarrheal disease
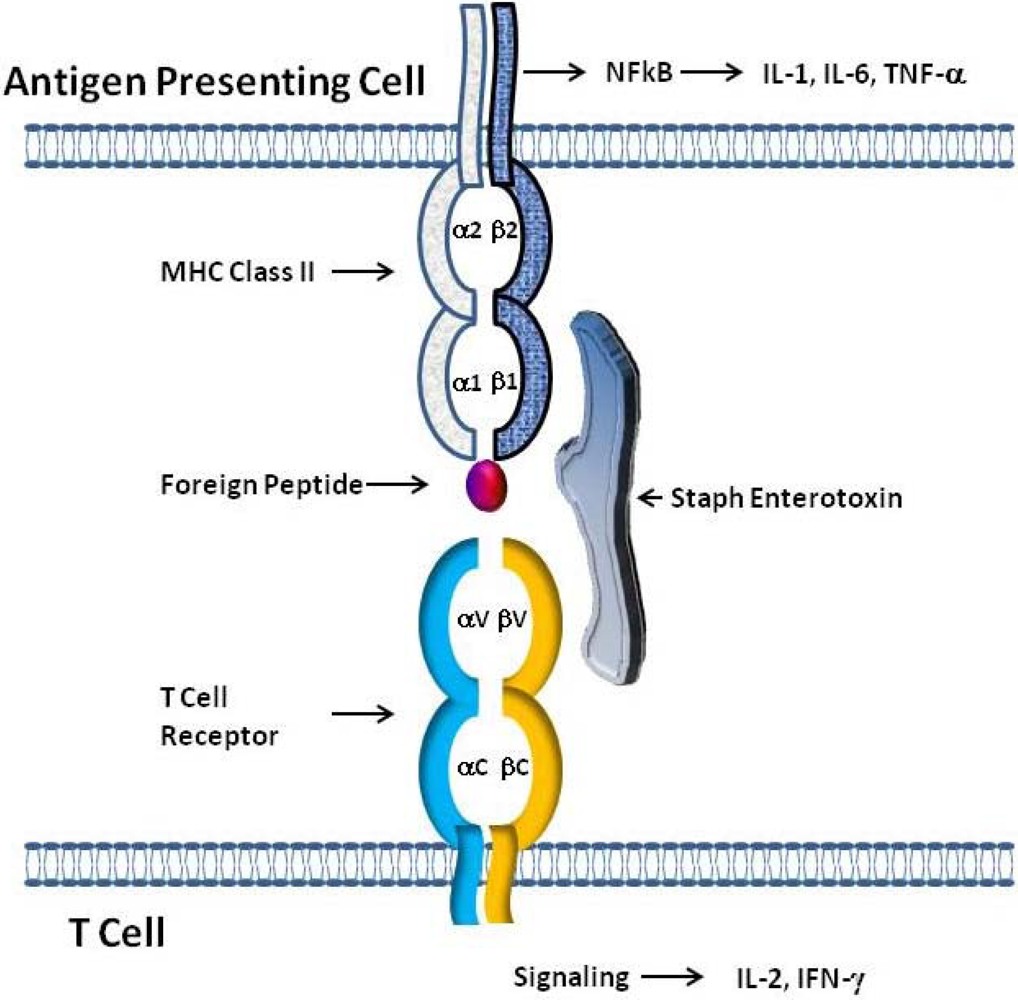
5.3. Effects on professional and non-professional APCs
6. In Vivo Modeling of SE Associated Diarrheal Diseases
7. Their Potential as Agents of Biological Warfare
8. Agents that Target the Superantigen Effect of SE
9. Concluding Remarks
Acknowledgements
References
- Kluytmans, J.; van Belkum, A.; Verbrugh, H. Nasal Carriage of Staphylococcus aureus: Epidemiology, Underlying Mechanisms, and Associated Risks. Clin. Microbiol. Rev. 1997, 10, 505–520. [Google Scholar]
- von Eiff, E.C.; Becker, K.; Machka, K.; Stammer, H.; Peters, G. Nasal Carriage As a Source of Staphylococcus aureus Bacteremia. Study Group. N. Engl. J. Med. 2001, 344, 11–16. [Google Scholar]
- Morandi, S.; Brasca, M.; Andrighetto, C.; Lombardi, A.; Lodi, R. Phenotypic and Genotypic Characterization of Staphylococcus aureus Strains From Italian Dairy Products. Int. J. Microbiol. 2009, 2009, 501362:1–501362:7. [Google Scholar]
- Zhang, S.; Iandolo, J.J.; Stewart, G.C. The Enterotoxin D Plasmid of Staphylococcus aureus Encodes a Second Enterotoxin Determinant (Sej). FEMS Microbiol. Lett. 1998, 168, 227–233. [Google Scholar]
- Lindsay, J.A.; Ruzin, A.; Ross, H.F.; Kurepina, N.; Novick, R.P. The Gene for Toxic Shock Toxin Is Carried by a Family of Mobile Pathogenicity Islands in Staphylococcus aureus. Mol. Microbiol. 1998, 29, 527–543. [Google Scholar]
- Varshney, A.K.; Mediavilla, J.R.; Robiou, N.; Guh, A.; Wang, X.; Gialanella, P.; Levi, M.H.; Kreiswirth, B.N.; Fries, B.C. Diverse Enterotoxin Gene Profiles Among Clonal Complexes of Staphylococcus aureus Isolates From the Bronx, New York. Appl. Environ. Microbiol. 2009, 75, 6839–6849. [Google Scholar]
- Ler, S.G.; Lee, F.K.; Gopalakrishnakone, P. Trends in Detection of Warfare Agents. Detection Methods for Ricin, Staphylococcal Enterotoxin B and T-2 Toxin. J. Chromatogr. A 2006, 1133, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bergdoll, M.S.; Crass, B.A.; Reiser, R.F.; Robbins, R.N.; Davis, J.P. A New Staphylococcal Enterotoxin, Enterotoxin F, Associated with Toxic-Shock-Syndrome Staphylococcus aureus Isolates. Lancet 1981, 1, 1017–1021. [Google Scholar]
- Morris, C.A.; Conway, H.D.; Everall, P.H. Food-Poisoning Due to Staphylococcal Enterotoxin E. Lancet 1972, 2, 1375–1376. [Google Scholar]
- Chen, T.R.; Chiou, C.S.; Tsen, H.Y. Use of Novel PCR Primers Specific to the Genes of Staphylococcal Enterotoxin G, H, I for the Survey of Staphylococcus aureus Strains Isolated From Food-Poisoning Cases and Food Samples in Taiwan. Int. J. Food Microbiol. 2004, 92, 189–197. [Google Scholar]
- Ikeda, T.; Tamate, N.; Yamaguchi, K.; Makino, S. Mass Outbreak of Food Poisoning Disease Caused by Small Amounts of Staphylococcal Enterotoxins A and H. Appl. Environ. Microbiol. 2005, 71, 2793–2795. [Google Scholar]
- Hudson, K.R.; Tiedemann, R.E.; Urban, R.G.; Lowe, S.C.; Strominger, J.L.; Fraser, J.D. Staphylococcal Enterotoxin A Has Two Cooperative Binding Sites on Major Histocompatibility Complex Class II. J. Exp. Med. 1995, 182, 711–720. [Google Scholar]
- Kim, J.; Urban, R.G.; Strominger, J.L.; Wiley, D.C. Toxic Shock Syndrome Toxin-1 Complexed with a Class II Major Histocompatibility Molecule HLA-DR1. Science 1994, 266, 1870–1874. [Google Scholar]
- Marr, J.C.; Lyon, J.D.; Roberson, J.R.; Lupher, M.; Davis, W.C.; Bohach, G.A. Characterization of Novel Type C Staphylococcal Enterotoxins: Biological and Evolutionary Implications. Infect. Immun. 1993, 61, 4254–4262. [Google Scholar]
- Redpath, S.; Alam, S.M.; Lin, C.M.; O'Rourke, A.M.; Gascoigne, N.R. Cutting Edge: Trimolecular Interaction of TCR with MHC Class II and Bacterial Superantigen Shows a Similar Affinity to MHC:Peptide Ligands. J. Immunol. 1999, 163, 6–10. [Google Scholar]
- Chang, H.C.; Bergdoll, M.S. Purification and Some Physicochemical Properties of Staphylococcal Enterotoxin D. Biochemistry 1979, 18, 1937–1942. [Google Scholar]
- Al-Daccak, R.; Mehindate, K.; Damdoumi, F.; Etongue-Mayer, P.; Nilsson, H.; Antonsson, P.; Sundstrom, M.; Dohlsten, M.; Sekaly, R.P.; Mourad, W. Staphylococcal Enterotoxin D Is a Promiscuous Superantigen Offering Multiple Modes of Interactions with the MHC Class II Receptors. J. Immunol. 1998, 160, 225–232. [Google Scholar]
- Karp, D.R.; Long, E.O. Identification of HLA-DR1 Beta Chain Residues Critical for Binding Staphylococcal Enterotoxins A and E. J. Exp. Med. 1992, 175, 415–424. [Google Scholar]
- Braunstein, N.S.; Weber, D.A.; Wang, X.C.; Long, E.O.; Karp, D. Sequences in Both Class II Major Histocompatibility Complex Alpha and Beta Chains Contribute to the Binding of the Superantigen Toxic Shock Syndrome Toxin 1. J. Exp. Med. 1992, 175, 1301–1305. [Google Scholar]
- Fernandez, M.M.; Bhattacharya, S.; De Marzi, M.C.; Brown, P.H.; Kerzic, M.; Schuck, P.; Mariuzza, R.A.; Malchiodi, E.L. Superantigen Natural Affinity Maturation Revealed by the Crystal Structure of Staphylococcal Enterotoxin G and Its Binding to T-Cell Receptor Vbeta8.2. Proteins 2007, 68, 389–402. [Google Scholar]
- Nilsson, H.; Bjork, P.; Dohlsten, M.; Antonsson, P. Staphylococcal Enterotoxin H Displays Unique MHC Class II-Binding Properties. J. Immunol. 1999, 163, 6686–6693. [Google Scholar]
- Fernandez, M.M.; Guan, R.; Swaminathan, C.P.; Malchiodi, E.L.; Mariuzza, R.A. Crystal Structure of Staphylococcal Enterotoxin I (SEI) in Complex with a Human Major Histocompatibility Complex Class II Molecule. J. Biol. Chem. 2006, 281, 25356–25364. [Google Scholar]
- Choi, Y.W.; Kotzin, B.; Herron, L.; Callahan, J.; Marrack, P.; Kappler, J. Interaction of Staphylococcus aureus Toxin "Superantigens" with Human T Cells. Proc. Natl. Acad. Sci. USA 1989, 86, 8941–8945. [Google Scholar]
- Balaban, N.; Rasooly, A. Staphylococcal Enterotoxins. Int. J. Food Microbiol. 2000, 61, 1–10. [Google Scholar]
- Schlievert, P.M.; Bohach, G.A.; Ohlendorf, D.H.; Stauffacher, C.V.; Leung, D.Y.; Murray, D.L.; Prasad, G.S.; Earhart, C.A.; Jablonski, L.M.; Hoffmann, M.L.; Chi, Y.I. Molecular Structure of Staphylococcus and Streptococcus Superantigens. J. Clin. Immunol. 1995, 15, 4S–10S. [Google Scholar]
- Jardetzky, T.S.; Brown, J.H.; Gorga, J.C.; Stern, L.J.; Urban, R.G.; Chi, Y.I.; Stauffacher, C.; Strominger, J.L.; Wiley, D.C. 3-Dimensional Structure of A Human Class-Ii Histocompatibility Molecule Complexed with Superantigen. Nature 1994, 368, 711–718. [Google Scholar]
- Watanabe, T.; Watanabe, M.; Ishii, Y.; Matsuba, H.; Kimura, S.; Fujita, T.; Kominami, E.; Katunuma, N.; Uchiyama, Y. An Immunocytochemical Study on Co-Localization of Cathepsin B and Atrial Natriuretic Peptides in Secretory Granules of Atrial Myoendocrine Cells of Rat Heart. J. Histochem. Cytochem. 1989, 37, 347–351. [Google Scholar]
- Singh, B.R.; Fu, F.N.; Ledoux, D.N. Crystal and Solution Structures of Superantigenic Staphylococcal Enterotoxins Compared. Nat. Struct. Biol. 1994, 1, 358–360. [Google Scholar]
- Hsu, P.N.; Huber, B.T. Superantigens. Gazing into the Crystal Ball. Curr. Biol. 1995, 5, 235–237. [Google Scholar]
- Schad, E.M.; Zaitseva, I.; Zaitsev, V.N.; Dohlsten, M.; Kalland, T.; Schlievert, P.M.; Ohlendorf, D.H.; Svensson, L.A. Crystal Structure of the Superantigen Staphylococcal Enterotoxin Type A. EMBO J. 1995, 14, 3292–3301. [Google Scholar]
- Papageorgiou, A.C.; Acharya, K.R.; Shapiro, R.; Passalacqua, E.F.; Brehm, R.D.; Tranter, H.S. Crystal Structure of the Superantigen Enterotoxin C2 From Staphylococcus aureus Reveals a Zinc-Binding Site. Structure 1995, 3, 769–779. [Google Scholar]
- Sundstrom, M.; Hallen, D.; Svensson, A.; Schad, E.; Dohlsten, M.; Abrahmsen, L. The Co-Crystal Structure of Staphylococcal Enterotoxin Type A with Zn2+ at 2.7 A Resolution. Implications for Major Histocompatibility Complex Class II Binding. J. Biol. Chem. 1996, 271, 32212–32216. [Google Scholar] [PubMed]
- Sundstrom, M.; Abrahmsen, L.; Antonsson, P.; Mehindate, K.; Mourad, W.; Dohlsten, M. The Crystal Structure of Staphylococcal Enterotoxin Type D Reveals Zn2+-Mediated Homodimerization. EMBO J. 1996, 15, 6832–6840. [Google Scholar]
- Papageorgiou, A.C.; Tranter, H.S.; Acharya, K.R. Crystal Structure of Microbial Superantigen Staphylococcal Enterotoxin B at 1.5 A Resolution: Implications for Superantigen Recognition by MHC Class II Molecules and T-Cell Receptors. J. Mol. Biol. 1998, 277, 61–79. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Llera, A.; Tsuchiya, D.; Leder, L.; Ysern, X.; Schlievert, P.M.; Karjalainen, K.; Mariuzza, R.A. Three-Dimensional Structure of the Complex Between a T Cell Receptor Beta Chain and the Superantigen Staphylococcal Enterotoxin B. Immunity 1998, 9, 807–816. [Google Scholar]
- Antonsson, P.; Wingren, A.G.; Hansson, J.; Kalland, T.; Varga, M.; Dohlsten, M. Functional Characterization of the Interaction Between the Superantigen Staphylococcal Enterotoxin A and the TCR. J. Immunol. 1997, 158, 4245–4251. [Google Scholar]
- Garcia, C.; Briggs, C.; Zhang, L.; Guan, L.; Gabriel, J.L.; Rogers, T.J. Molecular Characterization of the Putative T-Cell Receptor Cavity of the Superantigen Staphylococcal Enterotoxin B. Immunology 1998, 94, 160–166. [Google Scholar]
- Kappler, J.W.; Herman, A.; Clements, J.; Marrack, P. Mutations Defining Functional Regions of the Superantigen Staphylococcal Enterotoxin B. J. Exp. Med. 1992, 175, 387–396. [Google Scholar]
- Marrack, P.; Kappler, J. The Staphylococcal Enterotoxins and Their Relatives. Science 1990, 248, 705–711. [Google Scholar]
- Bohach, G.A. Staphylococcal Enterotoxins B and C. Structural Requirements for Superantigenic and Entertoxigenic Activities. Prep. Biochem. Biotechnol. 1997, 27, 79–110. [Google Scholar] [CrossRef] [PubMed]
- Rich, R.R.; Mollick, J.A.; Cook, R.G. Superantigens: Interaction of Staphylococcal Enterotoxins with MHC Class II Molecules. Trans. Am. Clin. Climatol. Assoc. 1990, 101, 195–204. [Google Scholar]
- Le Loir, Y.; Baron, F.; Gautier, M. Staphylococcus aureus and Food Poisoning. Genet. Mol. Res. 2003, 2, 63–76. [Google Scholar]
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-Related Illness and Death in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar]
- Buzby, J.C.; Roberts, T. Economic Costs and Trade Impacts of Microbial Foodborne Illness. World Health Stat. Q. 1997, 50, 57–66. [Google Scholar]
- Scherrer, D.; Corti, S.; Muehlherr, J.E.; Zweifel, C.; Stephan, R. Phenotypic and Genotypic Characteristics of Staphylococcus aureus Isolates From Raw Bulk-Tank Milk Samples of Goats and Sheep. Vet. Microbiol. 2004, 101, 101–107. [Google Scholar]
- Evenson, M.L.; Hinds, M.W.; Bernstein, R.S.; Bergdoll, M.S. Estimation of Human Dose of Staphylococcal Enterotoxin A From a Large Outbreak of Staphylococcal Food Poisoning Involving Chocolate Milk. Int. J. Food Microbiol. 1988, 7, 311–316. [Google Scholar]
- Atanassova, V.; Meindl, A.; Ring, C. Prevalence of Staphylococcus aureus and Staphylococcal Enterotoxins in Raw Pork and Uncooked Smoked Ham—a Comparison of Classical Culturing Detection and RFLP-PCR. Int. J. Food Microbiol. 2001, 68, 105–113. [Google Scholar]
- Casman, E.P. Staphylococcal Enterotoxin. Ann. N. Y. Acad. Sci. 1965, 128, 124–131. [Google Scholar]
- Flemming, K.; Ackermann, G. Prevalence of Enterotoxin Producing Staphylococcus aureus in Stools of Patients with Nosocomial Diarrhea. Infection 2007, 35, 356–358. [Google Scholar]
- Boyce, J.M.; Havill, N.L. Nosocomial Antibiotic-Associated Diarrhea Associated with Enterotoxin-Producing Strains of Methicillin-Resistant Staphylococcus aureus. Am. J. Gastroenterol. 2005, 100, 1828–1834. [Google Scholar]
- Ehlert, K. Methicillin-Resistance in Staphylococcus aureus - Molecular Basis, Novel Targets and Antibiotic Therapy. Curr. Pharm. Des. 1999, 5, 45–55. [Google Scholar]
- CDC NNIS System. National Nosocomial Infections Surveillance (NNIS) System Report, Data Summary From January 1992 Through June 2004, Issued October 2004. Am. J. Infect. Control 2004, 32, 470–485. [CrossRef] [PubMed]
- Schmitz, F.J.; MacKenzie, C.R.; Geisel, R.; Wagner, S.; Idel, H.; Verhoef, J.; Hadding, U.; Heinz, H.P. Enterotoxin and Toxic Shock Syndrome Toxin-1 Production of Methicillin Resistant and Methicillin Sensitive Staphylococcus aureus Strains. Eur. J. Epidemiol. 1997, 13, 699–708. [Google Scholar]
- Banwell, J.G.; Sherr, H. Effect of Bacterial Enterotoxins on the Gastrointestinal Tract. Gastroenterology 1973, 65, 467–497. [Google Scholar]
- Merrill, T.G.; Sprinz, H. The Effect of Staphylococcal Enterotoxin on the Fine Structure of the Monkey Jejunum. Lab. Invest. 1968, 18, 114–123. [Google Scholar]
- Shemano, I.; Hitchens, J.T.; Beiler, J.M. Paradoxical Intestinal Inhibitory Effects of Staphylococcal Enterotoxin. Gastroenterology 1967, 53, 71–77. [Google Scholar]
- Stiles, J.W.; Denniston, J.C. Response of the Rhesus Monkey, Macaca Mulatta, to Continuously Infused Staphylococcal Enterotoxin B. Lab. Invest. 1971, 25, 617–625. [Google Scholar]
- Van Prohaska, J. Role of Staphylococcal Enterotoxin in the Induction of Experimental Ileitis. Ann. Surg. 1963, 158, 492–497. [Google Scholar]
- Tranter, H.S. Foodborne Staphylococcal Illness. Lancet 1990, 336, 1044–1046. [Google Scholar]
- Taylor, S.L.; Schlunz, L.R.; Beery, J.T.; Cliver, D.O.; Bergdoll, M.S. Emetic Action of Staphylococcal Enterotoxin A on Weanling Pigs. Infect. Immun. 1982, 36, 1263–1266. [Google Scholar]
- van Gessel, Y.A.; Mani, S.; Bi, S.; Hammamieh, R.; Shupp, J.W.; Das, R.; Coleman, G.D.; Jett, M. Functional Piglet Model for the Clinical Syndrome and Postmortem Findings Induced by Staphylococcal Enterotoxin B. Exp. Biol. Med. (Maywood) 2004, 229, 1061–1071. [Google Scholar] [PubMed]
- Beery, J.T.; Taylor, S.L.; Schlunz, L.R.; Freed, R.C.; Bergdoll, M.S. Effects of Staphylococcal Enterotoxin A on the Rat Gastrointestinal Tract. Infect. Immun. 1984, 44, 234–240. [Google Scholar]
- Lu, J.; Philpott, D.J.; Saunders, P.R.; Perdue, M.H.; Yang, P.C.; McKay, D.M. Epithelial Ion Transport and Barrier Abnormalities Evoked by Superantigen-Activated Immune Cells Are Inhibited by Interleukin-10 but Not Interleukin-4. J. Pharmacol. Exp. Ther. 1998, 287, 128–136. [Google Scholar]
- McKay, D.M.; Benjamin, M.A.; Lu, J. CD4+ T Cells Mediate Superantigen-Induced Abnormalities in Murine Jejunal Ion Transport. Am. J. Physiol. 1998, 275, G29–G38. [Google Scholar]
- McKay, D.M.; Botelho, F.; Ceponis, P.J.; Richards, C.D. Superantigen Immune Stimulation Activates Epithelial STAT-1 and PI 3-K: PI 3-K Regulation of Permeability. Am. J. Physiol. Gastrointest. Liver Physiol. 2000, 279, G1094–G1103. [Google Scholar]
- Pender, S.L.; Breese, E.J.; Gunther, U.; Howie, D.; Wathen, N.C.; Schuppan, D.; MacDonald, T.T. Suppression of T Cell-Mediated Injury in Human Gut by Interleukin 10: Role of Matrix Metalloproteinases. Gastroenterology 1998, 115, 573–583. [Google Scholar]
- Pinchuk, I.V.; Beswick, E.J.; Saada, J.I.; Suarez, G.; Winston, J.; Mifflin, R.C.; Di Mari, J.F.; Powell, D.W.; Reyes, V.E. Monocyte Chemoattractant Protein-1 Production by Intestinal Myofibroblasts in Response to Staphylococcal Enterotoxin a: Relevance to Staphylococcal Enterotoxigenic Disease. J. Immunol. 2007, 178, 8097–8106. [Google Scholar]
- Benjamin, M.A.; Lu, J.; Donnelly, G.; Dureja, P.; McKay, D.M. Changes in Murine Jejunal Morphology Evoked by the Bacterial Superantigen Staphylococcus aureus Enterotoxin B Are Mediated by CD4+ T Cells. Infect. Immun. 1998, 66, 2193–2199. [Google Scholar]
- Hu, D.L.; Omoe, K.; Shimoda, Y.; Nakane, A.; Shinagawa, K. Induction of Emetic Response to Staphylococcal Enterotoxins in the House Musk Shrew (Suncus Murinus). Infect. Immun. 2003, 71, 567–570. [Google Scholar]
- Hu, D.L.; Zhu, G.; Mori, F.; Omoe, K.; Okada, M.; Wakabayashi, K.; Kaneko, S.; Shinagawa, K.; Nakane, A. Staphylococcal Enterotoxin Induces Emesis Through Increasing Serotonin Release in Intestine and It Is Downregulated by Cannabinoid Receptor 1. Cell. Microbiol. 2007, 9, 2267–2277. [Google Scholar]
- Hu, D.L.; Omoe, K.; Sashinami, H.; Shinagawa, K.; Nakane, A. Immunization with a Nontoxic Mutant of Staphylococcal Enterotoxin A, SEAD227A, Protects Against Enterotoxin-Induced Emesis in House Musk Shrews. J. Infect. Dis. 2009, 199, 302–310. [Google Scholar]
- Thibodeau, J.; Cloutier, I.; Lavoie, P.M.; Labrecque, N.; Mourad, W.; Jardetzky, T.; Sekaly, R.P. Subsets of HLA-DR1 Molecules Defined by SEB and TSST-1 Binding. Science 1994, 266, 1874–1878. [Google Scholar]
- Panina-Bordignon, P.; Fu, X.T.; Lanzavecchia, A.; Karr, R.W. Identification of HLA-DR Alpha Chain Residues Critical for Binding of the Toxic Shock Syndrome Toxin Superantigen. J. Exp. Med. 1992, 176, 1779–1784. [Google Scholar]
- Al-Daccak, R.; Mehindate, K.; Poubelle, P.E.; Mourad, W. Signalling Via MHC Class II Molecules Selectively Induces IL-1 Beta Over IL-1 Receptor Antagonist Gene Expression. Biochem. Biophys. Res. Commun. 1994, 201, 855–860. [Google Scholar]
- Assenmacher, M.; Lohning, M.; Scheffold, A.; Manz, R.A.; Schmitz, J.; Radbruch, A. Sequential Production of IL-2, IFN-Gamma and IL-10 by Individual Staphylococcal Enterotoxin B-Activated T Helper Lymphocytes. Eur. J. Immunol. 1998, 28, 1534–1543. [Google Scholar]
- Carlsson, R.; Fischer, H.; Sjogren, H.O. Binding of Staphylococcal Enterotoxin A to Accessory Cells Is a Requirement for Its Ability to Activate Human T Cells. J. Immunol. 1988, 140, 2484–2488. [Google Scholar]
- Carlsson, R.; Sjogren, H.O. Kinetics of IL-2 and Interferon-Gamma Production, Expression of IL-2 Receptors, and Cell Proliferation in Human Mononuclear Cells Exposed to Staphylococcal Enterotoxin A. Cell. Immunol. 1985, 96, 175–183. [Google Scholar]
- Cameron, S.B.; Nawijn, M.C.; Kum, W.W.; Savelkoul, H.F.; Chow, A.W. Regulation of Helper T Cell Responses to Staphylococcal Superantigens. Eur. Cytokine Netw. 2001, 12, 210–222. [Google Scholar]
- Grumann, D.; Scharf, S.S.; Holtfreter, S.; Kohler, C.; Steil, L.; Engelmann, S.; Hecker, M.; Volker, U.; Broker, B.M. Immune Cell Activation by Enterotoxin Gene Cluster (Egc)-Encoded and Non-Egc Superantigens From Staphylococcus aureus. J. Immunol. 2008, 181, 5054–5061. [Google Scholar]
- Hamad, A.R.; Marrack, P.; Kappler, J.W. Transcytosis of Staphylococcal Superantigen Toxins. J. Exp. Med. 1997, 185, 1447–1454. [Google Scholar]
- Muraille, E.; De Trez, C.; Pajak, B.; Brait, M.; Urbain, J.; Leo, O. T Cell-Dependent Maturation of Dendritic Cells in Response to Bacterial Superantigens. J. Immunol. 2002, 168, 4352–4360. [Google Scholar]
- Desouza, I.A.; Hyslop, S.; Franco-Penteado, C.F.; Ribeiro-DaSilva, G. Mouse Macrophages Release a Neutrophil Chemotactic Mediator Following Stimulation by Staphylococcal Enterotoxin Type A. Inflamm. Res. 2001, 50, 206–212. [Google Scholar]
- Desouza, I.A.; Hyslop, S.; Franco-Penteado, C.F.; Ribeiro-DaSilva, G. Evidence for the Involvement of a Macrophage-Derived Chemotactic Mediator in the Neutrophil Recruitment Induced by Staphylococcal Enterotoxin B in Mice. Toxicon 2002, 40, 1709–1717. [Google Scholar]
- Byrne, B.; Madrigal-Estebas, L.; McEvoy, A.; Carton, J.; Doherty, D.G.; Whelan, A.; Feighery, C.; O'Donoghue, D.P.; O'Farrelly, C. Human Duodenal Epithelial Cells Constitutively Express Molecular Components of Antigen Presentation but Not Costimulatory Molecules. Hum. Immunol. 2002, 63, 977–986. [Google Scholar]
- Fromont, G.; Cerf-Bensussan, N.; Patey, N.; Canioni, D.; Rambaud, C.; Goulet, O.; Jan, D.; Revillon, Y.; Ricour, C.; Brousse, N. Small Bowel Transplantation in Children: an Immunohistochemical Study of Intestinal Grafts. Gut 1995, 37, 783–790. [Google Scholar]
- Krakauer, T. Costimulatory Receptors for the Superantigen Staphylococcal Enterotoxin B on Human Vascular Endothelial Cells and T Cells. J. Leukoc. Biol. 1994, 56, 458–463. [Google Scholar]
- Fujisawa, N.; Hayashi, S.; Kurdowska, A.; Noble, J.M.; Naitoh, K.; Miller, E.J. Staphylococcal Enterotoxin A-Induced Injury of Human Lung Endothelial Cells and IL-8 Accumulation Are Mediated by TNF-Alpha. J. Immunol. 1998, 161, 5627–5632. [Google Scholar]
- Fischer, H.; Dohlsten, M.; Lindvall, M.; Sjogren, H.O.; Carlsson, R. Binding of Staphylococcal Enterotoxin A to HLA-DR on B Cell Lines. J. Immunol. 1989, 142, 3151–3157. [Google Scholar]
- Huvenne, W.; Callebaut, I.; Plantinga, M.; Vanoirbeek, J.A.; Krysko, O.; Bullens, D.M.; Gevaert, P.; Van Cauwenberge, P.; Lambrecht, B.N.; Ceuppens, J.L.; Bachert, C.; Hellings, P.W. Staphylococcus aureus Enterotoxin B Facilitates Allergic Sensitization in Experimental Asthma. Clin. Exp. Allergy 2010, 40, 1079–1090. [Google Scholar]
- Bachert, C.; Zhang, N.; Patou, J.; Van Zele, T.; Gevaert, P. Role of Staphylococcal Superantigens in Upper Airway Disease. Curr. Opin. Allergy Clin. Immunol. 2008, 8, 34–38. [Google Scholar]
- Bachert, C.; Gevaert, P.; Zhang, N.; Van Zele, T.; Perez-Novo, C. Role of Staphylococcal Superantigens in Airway Disease. Chem. Immunol. Allergy 2007, 93, 214–236. [Google Scholar]
- Krakauer, T. Chemotherapeutics Targeting Immune Activation by Staphylococcal Superantigens. Med. Sci. Monit. 2005, 11, RA290–RA295. [Google Scholar]
- Normann, S.J.; Jaeger, R.F.; Johnsey, R.T. Pathology of Experimental Enterotoxemia. The in vivo Localization of Staphylococcal Enterotoxin B. Lab. Invest. 1969, 20, 17–25. [Google Scholar] [PubMed]
- Reck, B.; Scheuber, P.H.; Londong, W.; Sailer-Kramer, B.; Bartsch, K.; Hammer, D.K. Protection Against the Staphylococcal Enterotoxin-Induced Intestinal Disorder in the Monkey by Anti-Idiotypic Antibodies. Proc. Natl. Acad. Sci. USA 1988, 85, 3170–3174. [Google Scholar]
- Sheahan, D.G.; Jervis, H.R.; Takeuchi, A.; Sprinz, H. The Effect of Staphylococcal Enterotoxin on the Epithelial Mucosubstances of the Small Intestine of Rhesus Monkeys. Am. J. Pathol. 1970, 60, 1–18. [Google Scholar]
- Kohrman, K.A.; Kirkland, J.J.; Danneman, P.J. Response of Various Animal Species to Experimental Infection with Different Strains of Staphylococcus aureus. Rev. Infect. Dis. 1989, 11 (Suppl. 1), S231–S236. [Google Scholar] [PubMed]
- Kocandrle, V.; Houttuin, E.; Prohaska, J.V. Acute Hemodynamic and Gastrointestinal Changes Produced by Staphylococcal Exotoxin and Enterotoxin in Dogs. J. Surg. Res. 1966, 6, 50–57. [Google Scholar]
- Sullivan, R. Effects of Enterotoxin B on Intestinal Transport in Vivo. Proc. Soc. Exp. Biol. Med. 1969, 131, 1159–1162. [Google Scholar]
- Cheng, S.; Smart, M.; Hanson, J.; David, C.S. Characterization of HLA DR2 and DQ8 Transgenic Mouse with a New Engineered Mouse Class II Deletion, Which Lacks All Endogenous Class II Genes. J. Autoimmun. 2003, 21, 195–199. [Google Scholar]
- Gill, D.M. Bacterial Toxins: a Table of Lethal Amounts. Microbiol. Rev. 1982, 46, 86–94. [Google Scholar]
- Rajagopalan, G.; Sen, M.M.; Singh, M.; Murali, N.S.; Nath, K.A.; Iijima, K.; Kita, H.; Leontovich, A.A.; Gopinathan, U.; Patel, R.; David, C.S. Intranasal Exposure to Staphylococcal Enterotoxin B Elicits an Acute Systemic Inflammatory Response. Shock 2006, 25, 647–656. [Google Scholar]
- Weng, C.F.; Komisar, J.L.; Hunt, R.E.; Johnson, A.J.; Pitt, M.L.; Ruble, D.L.; Tseng, J. Immediate Responses of Leukocytes, Cytokines and Glucocorticoid Hormones in the Blood Circulation of Monkeys Following Challenge with Aerosolized Staphylococcal Enterotoxin B. Int. Immunol. 1997, 9, 1825–1836. [Google Scholar]
- Rusnak, J.M.; Kortepeter, M.; Ulrich, R.; Poli, M.; Boudreau, E. Laboratory Exposures to Staphylococcal Enterotoxin B. Emerg. Infect. Dis. 2004, 10, 1544–1549. [Google Scholar]
- Silverman, S.J.; Espeseth, D.A.; Schantz, E.J. Effect of Formaldehyde on the Immunochemical and Biological Activity of Staphylococcal Enterotoxin B. J. Bacteriol. 1969, 98, 437–442. [Google Scholar]
- Tseng, J.; Komisar, J.L.; Trout, R.N.; Hunt, R.E.; Chen, J.Y.; Johnson, A.J.; Pitt, L.; Ruble, D.L. Humoral Immunity to Aerosolized Staphylococcal Enterotoxin B (SEB), a Superantigen, in Monkeys Vaccinated with SEB Toxoid-Containing Microspheres. Infect. Immun. 1995, 63, 2880–2885. [Google Scholar]
- Stiles, B.G.; Garza, A.R.; Ulrich, R.G.; Boles, J.W. Mucosal Vaccination with Recombinantly Attenuated Staphylococcal Enterotoxin B and Protection in a Murine Model. Infect. Immun. 2001, 69, 2031–2036. [Google Scholar]
- Boles, J.W.; Pitt, M.L.; LeClaire, R.D.; Gibbs, P.H.; Torres, E.; Dyas, B.; Ulrich, R.G.; Bavari, S. Generation of Protective Immunity by Inactivated Recombinant Staphylococcal Enterotoxin B Vaccine in Nonhuman Primates and Identification of Correlates of Immunity. Clin. Immunol. 2003, 108, 51–59. [Google Scholar]
- Kaferstein, F. Foodborne Diseases in Developing Countries: Aetiology, Epidemiology and Strategies for Prevention. Int. J. Environ. Health Res. 2003, 13 (Suppl. 1), S161–S168. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Kotler, D.P.; Schlievert, P.M.; Sordillo, E.M. Staphylococcal Enterocolitis: Forgotten but Not Gone? Dig. Dis. Sci. 2010, 55, 1200–1207. [Google Scholar] [CrossRef] [PubMed]
- Maudsley, J.; Stone, S.P.; Kibbler, C.C.; Iliffe, S.R.; Conaty, S.J.; Cookson, B.D.; Duckworth, G.J.; Johnson, A.; Wallace, P.G. The Community Prevalence of Methicillin-Resistant Staphylococcus aureus (MRSA) in Older People Living in Their Own Homes: Implications for Treatment, Screening and Surveillance in the UK. J. Hosp. Infect. 2004, 57, 258–262. [Google Scholar]
- Cooper, B.S.; Medley, G.F.; Stone, S.P.; Kibbler, C.C.; Cookson, B.D.; Roberts, J.A.; Duckworth, G.; Lai, R.; Ebrahim, S. Methicillin-Resistant Staphylococcus aureus in Hospitals and the Community: Stealth Dynamics and Control Catastrophes. Proc. Natl. Acad. Sci. USA 2004, 101, 10223–10228. [Google Scholar]
- Much, P.; Pichler, J.; Kasper, S.S.; Allerberger, F. Foodborne Outbreaks, Austria 2007. Wien. Klin. Wochenschr. 2009, 121, 77–85. [Google Scholar]
- Murray, R.J. Recognition and Management of Staphylococcus aureus Toxin-Mediated Disease. Intern. Med. J. 2005, 35 (Suppl. 2), S106–S119. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.P.; Chesney, P.J.; Wand, P.J.; LaVenture, M. Toxic-Shock Syndrome: Epidemiologic Features, Recurrence, Risk Factors, and Prevention. N. Engl. J. Med. 1980, 303, 1429–1435. [Google Scholar]
- Gbaguidi-Haore, H.; Thouverez, M.; Couetdic, G.; Cholley, P.; Talon, D.; Bertrand, X. Usefulness of Antimicrobial Resistance Pattern for Detecting PVL- or TSST-1-Producing Meticillin-Resistant Staphylococcus aureus in a French University Hospital. J. Med. Microbiol. 2009, 58, 1337–1342. [Google Scholar]
- Fraser, J.D.; Proft, T. The Bacterial Superantigen and Superantigen-Like Proteins. Immunol. Rev. 2008, 225, 226–243. [Google Scholar]
- Larkin, E.A.; Carman, R.J.; Krakauer, T.; Stiles, B.G. Staphylococcus aureus: the Toxic Presence of a Pathogen Extraordinaire. Curr. Med. Chem. 2009, 16, 4003–4019. [Google Scholar]
- Yanagisawa, C.; Hanaki, H.; Natae, T.; Sunakawa, K. Neutralization of Staphylococcal Exotoxins in Vitro by Human-Origin Intravenous Immunoglobulin. J. Infect. Chemother. 2007, 13, 368–372. [Google Scholar]
- Basma, H.; Norrby-Teglund, A.; Guedez, Y.; McGeer, A.; Low, D.E.; El Ahmedy, O.; Schwartz, B.; Kotb, M. Risk Factors in the Pathogenesis of Invasive Group A Streptococcal Infections: Role of Protective Humoral Immunity. Infect. Immun. 1999, 67, 1871–1877. [Google Scholar]
- Darenberg, J.; Soderquist, B.; Normark, B.H.; Norrby-Teglund, A. Differences in Potency of Intravenous Polyspecific Immunoglobulin G Against Streptococcal and Staphylococcal Superantigens: Implications for Therapy of Toxic Shock Syndrome. Clin. Infect. Dis. 2004, 38, 836–842. [Google Scholar]
- Hu, D.L.; Cui, J.C.; Omoe, K.; Sashinami, H.; Yokomizo, Y.; Shinagawa, K.; Nakane, A. A Mutant of Staphylococcal Enterotoxin C Devoid of Bacterial Superantigenic Activity Elicits a Th2 Immune Response for Protection Against Staphylococcus aureus Infection. Infect. Immun. 2005, 73, 174–180. [Google Scholar]
- Arad, G.; Levy, R.; Hillman, D.; Kaempfer, R. Superantigen Antagonist Protects Against Lethal Shock and Defines a New Domain for T-Cell Activation. Nat. Med. 2000, 6, 414–421. [Google Scholar]
- Lehnert, N.M.; Allen, D.L.; Allen, B.L.; Catasti, P.; Shiflett, P.R.; Chen, M.; Lehnert, B.E.; Gupta, G. Structure-Based Design of a Bispecific Receptor Mimic That Inhibits T Cell Responses to a Superantigen. Biochemistry 2001, 40, 4222–4228. [Google Scholar]
- Yang, X.; Buonpane, R.A.; Moza, B.; Rahman, A.K.; Wang, N.; Schlievert, P.M.; McCormick, J.K.; Sundberg, E.J.; Kranz, D.M. Neutralization of Multiple Staphylococcal Superantigens by a Single-Chain Protein Consisting of Affinity-Matured, Variable Domain Repeats. J. Infect. Dis. 2008, 198, 344–348. [Google Scholar]
- Buonpane, R.A.; Churchill, H.R.; Moza, B.; Sundberg, E.J.; Peterson, M.L.; Schlievert, P.M.; Kranz, D.M. Neutralization of Staphylococcal Enterotoxin B by Soluble, High-Affinity Receptor Antagonists. Nat. Med. 2007, 13, 725–729. [Google Scholar]
- Liu, D.; Zienkiewicz, J.; DiGiandomenico, A.; Hawiger, J. Suppression of Acute Lung Inflammation by Intracellular Peptide Delivery of a Nuclear Import Inhibitor. Mol. Ther. 2009, 17, 796–802. [Google Scholar]
- Krakauer, T.; Buckley, M.; Issaq, H.J.; Fox, S.D. Rapamycin Protects Mice From Staphylococcal Enterotoxin B-Induced Toxic Shock and Blocks Cytokine Release in vivo and in Vivo. Antimicrob. Agents Chemother. 2010, 54, 1125–1131. [Google Scholar]
- Tilahun, A.Y.; Theuer, J.E.; Patel, R.; David, C.S.; Rajagopalan, G. Detrimental Effect of the Proteasome Inhibitor, Bortezomib in Bacterial Superantigen- and Lipopolysaccharide-Induced Systemic Inflammation. Mol. Ther. 2010, 18, 1143–1154. [Google Scholar]
- Miethke, T.; Wahl, C.; Heeg, K.; Echtenacher, B.; Krammer, P.H.; Wagner, H. T Cell-Mediated Lethal Shock Triggered in Mice by the Superantigen Staphylococcal Enterotoxin B: Critical Role of Tumor Necrosis Factor. J. Exp. Med. 1992, 175, 91–98. [Google Scholar]
- Stiles, B.G.; Campbell, Y.G.; Castle, R.M.; Grove, S.A. Correlation of Temperature and Toxicity in Murine Studies of Staphylococcal Enterotoxins and Toxic Shock Syndrome Toxin 1. Infect. Immun. 1999, 67, 1521–1525. [Google Scholar]
- Hayworth, J.L.; Kasper, K.J.; Leon-Ponte, M.; Herfst, C.A.; Yue, D.; Brintnell, W.C.; Mazzuca, D.M.; Heinrichs, D.E.; Cairns, E.; Madrenas, J.; Hoskin, D.W.; McCormick, J.K.; Haeryfar, S.M. Attenuation of Massive Cytokine Response to the Staphylococcal Enterotoxin B Superantigen by the Innate Immunomodulatory Protein Lactoferrin. Clin. Exp. Immunol. 2009, 157, 60–70. [Google Scholar]
- Perez-Bosque, A.; Miro, L.; Polo, J.; Russell, L.; Campbell, J.; Weaver, E.; Crenshaw, J.; Moreto, M. Dietary Plasma Protein Supplements Prevent the Release of Mucosal Proinflammatory Mediators in Intestinal Inflammation in Rats. J. Nutr. 2010, 140, 25–30. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pinchuk, I.V.; Beswick, E.J.; Reyes, V.E. Staphylococcal Enterotoxins. Toxins 2010, 2, 2177-2197. https://doi.org/10.3390/toxins2082177
Pinchuk IV, Beswick EJ, Reyes VE. Staphylococcal Enterotoxins. Toxins. 2010; 2(8):2177-2197. https://doi.org/10.3390/toxins2082177
Chicago/Turabian StylePinchuk, Irina V., Ellen J. Beswick, and Victor E. Reyes. 2010. "Staphylococcal Enterotoxins" Toxins 2, no. 8: 2177-2197. https://doi.org/10.3390/toxins2082177




