Occurrence of Ochratoxin A in Southern Spanish Generous Wines under the Denomination of Origin “Jerez-Xérès-Sherry and ‘Manzanilla’ Sanlúcar de Barrameda”
Abstract
:Abbreviations
1. Introduction
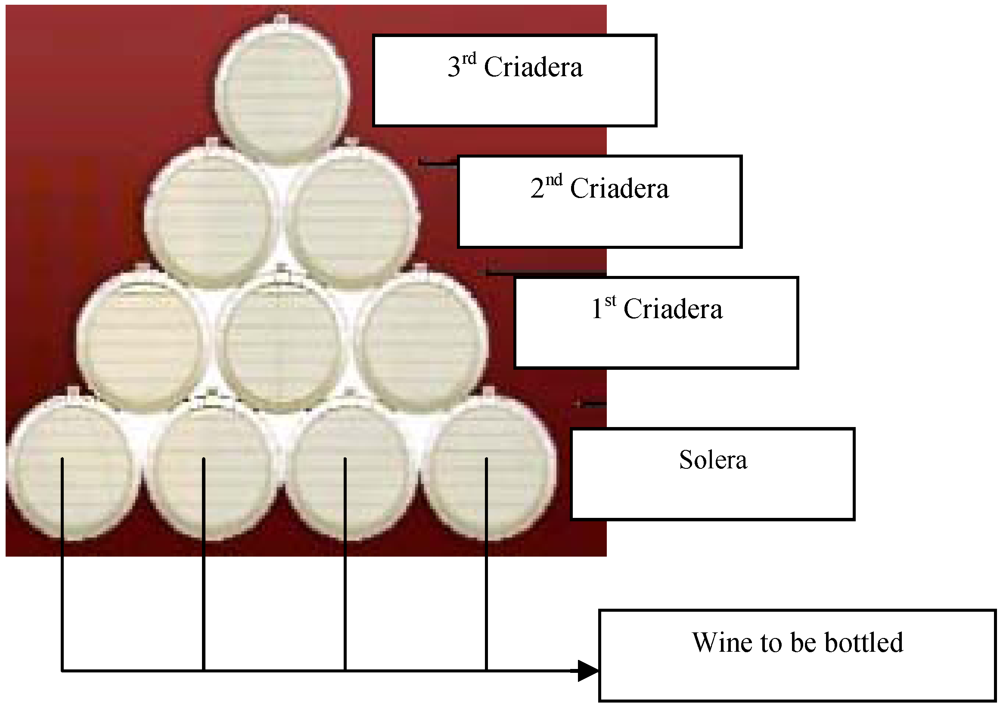
2. Material and Methods
2.1. Wine samples
2.2. Chemicals and reagents
2.3. Standard solutions
2.4. Wine sample preparation and IAC clean-up
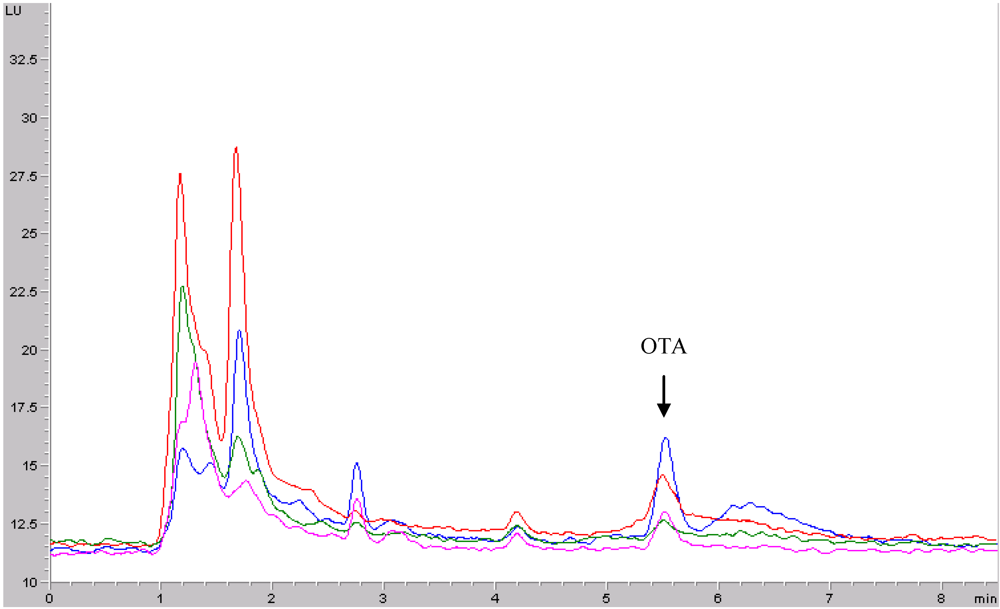
2.5. Apparatus and chromatographic conditions
3. Results and Discussion
3.1. Method validation
3.2. Sample analysis
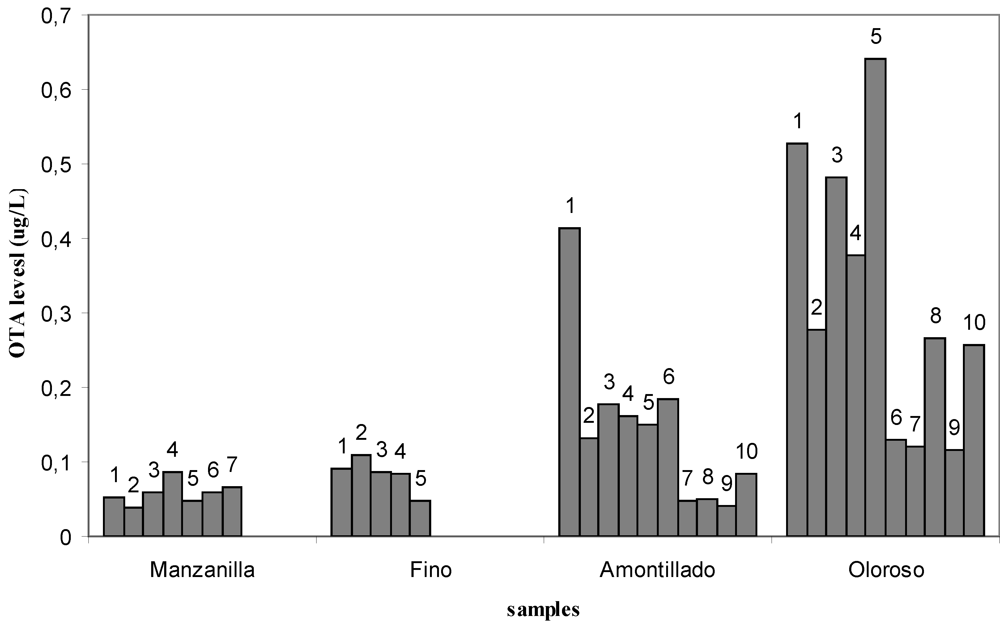
| Type of wine | Sample number | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| Manzanilla | 0.053 | 0.039 | 0.058 | 0.086 | 0.048 | 0.059 | 0.067 | <LOD | <LOD | <LOD |
| Fino | 0.092 | 0.110 | 0.087 | 0.084 | 0.047 | <LOD | <LOD | <LOD | <LOD | <LOD |
| Amontillado | 0.413 | 0.132 | 0.177 | 0.161 | 0.151 | 0.185 | 0.048 | 0.049 | 0.042 | 0.085 |
| Oloroso | 0.527 | 0.278 | 0.481 | 0.378 | 0.642 | 0.129 | 0.121 | 0.265 | 0.117 | 0.256 |
| Type of wine | Mean value | Median | Positive | Range | ||
|---|---|---|---|---|---|---|
| (μg/L) | (μg/L) | (%) | (μg/L) | |||
| Manzanilla | 0.042 | 0.051 | 70 | <LOD-0.086 | ||
| Fino | 0.044 | 0.026 | 50 | <LOD-0.110 | ||
| Amontillado | 0.144 | 0.142 | 100 | 0.042-0.413 | ||
| Oloroso | 0.319 | 0.272 | 100 | 0.117-0.642 | ||
| TOTAL | 0.138 | 0.086 | 80 | <LOD-0.642 | ||
| Origin | % Positive | Mean (μg/L) | LOD (μg/L) | Range (μg/L) | Reference |
|---|---|---|---|---|---|
| European and North African countries | 27 | 0.011 | 0.003 | <0.003-0.178 | [12] |
| Worldwide | 34 | 0.07 * | 0.01 | <0.01-1.20 | [14] |
| 25 | 0.108 | 0.01 | <0.01-1.36 | [11] | |
| -- | 0.059 | 0.05 | <0.05-0.18 | [23] | |
| -- e | 0.081 | <0.05-0.22 | |||
| European countries | 15 | 0.012 | 0.01 | <0.01-0.04 | [11] |
| Spain and other European countries | 65 | 0.020 | 0.003 | <0.003-0.267 | [24] |
| Spain | 100 c | 0.185 | 0.05 | 0.154-0.208 | [5] |
| 17 d | 0.192 | 0.192 | |||
| 10 | 0.18 | 0.05 | 0.05-1.13 | [25] | |
| Italy | 28 ª | 0.045 | 0.01 | <0.01-0.06 | [13] |
| 100 b | 0.535 | 0.10-0.97 | |||
| Morocco | 100 | 0.073 | 0.01f | 0.028-0.18 | [26] |
| Greece | 55 | 0.250 | 0.05 | <0.05-1.72 | [10] |
| 54 | 0.27 | 0.02 | <0.02-0.87 | [8] | |
| South Africa | 100 | 0.17 | 0.01 | 0.04-0.33 | [27] |
| Turkey | 85 | 0.108 | 0.006 | <0.006-0.618 | [28] |
4. Conclusions
Acknowledgements
References
- Burdaspal, P.A.; Legarda, T. Occurrence of ochratoxin A in sweet wines produced in Spain and other countries. Food Add. Contam. 2007, 24, 976–986. [Google Scholar] [CrossRef]
- Evaluation of Certain Food Additives and Contaminants: Sixty-Eighth Report of the Joint FAO/WHO Expert Committee on Food Additives. In Technical Report Series No.947; World Health Organization: Geneva, Switzerland, 2007.
- Pfohl-Leszkowicz, A. Ochratoxin A and aristolochic acid in the nephropathies and associated urothelial tract tumours development. Arh. Hig. Rada Toksikol. 2009, 60, 465–483. [Google Scholar] [PubMed]
- IARC. Monographs on Evaluation of Carcinogenic Risks to Humans. In Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins.; International Agency for Research on Cancer: Lyon, France, 1993; Volume 56, pp. 489–521. [Google Scholar]
- Lopez de Cerain, A.; Gonzalez-Peñas, E.; Jimenez, A.M.; Bello, J. Contribution to the study of ochratoxin A in Spanish wines. Food Add. Contam. 2002, 19, 1058–1064. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Manderville, R. Review on Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation No. 123/2005 of 26 January 2005 amending regulation No. 466/2001 as regards ochratoxin A. Off. J. Eur. Union 2005, L25, 3–5. [Google Scholar]
- Soufleros, E.H.; Tricard, C.; Bouloumpasi, E.C. Occurrence of ochratoxin A in Greek wines. J. Sci. Food Agric. 2003, 83, 173–179. [Google Scholar] [CrossRef]
- Zimmerli, B.; Dick, R. Determination of ochratoxin A at the ppt level in human blood, serum, milk and some foodstuffs by high-performance liquid chromatography with enhanced fluorescence detection and immunoaffinity column cleanup" methodology and Swiss data. J. Chromatogr. B 1995, 666, 85–99. [Google Scholar] [CrossRef]
- Stefanaki, I.; Foufa, E.; Tsatsou-Dritsa, A.; Dais, P. Ochratoxin A concentrations in Greek domestic wines and dried vine fruits. Food Add. Contam. 2003, 20, 74–83. [Google Scholar] [CrossRef]
- Otteneder, H.; Majerus, P. Occurrence of ochratoxin A (OTA) in wines: Influence of the type of wine and its geographical origin. Food Add. Contam. 2000, 17, 793–798. [Google Scholar] [CrossRef]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: Occurrence and risk assessment. Food Add. Contam. 1996, 13, 655–668. [Google Scholar]
- Visconti, A.; Pascale, M.; Centonze, G. Determination of Ochratoxin A in wine by means of immunoaffinity column clean-up and high-performance liquid chromatography. J. Chromatog. A 1999, 864, 89–101. [Google Scholar] [CrossRef]
- Majerus, P.; Otteneder, H. Detection and occurrence of ochratoxin A in wine and grapejuice. Deut. Lebesnm. Rundsch. 1996, 92, 388–390. [Google Scholar]
- Brera, C.; Debegnach, F.; Minardi, V.; Prantera, E.; Pannunzi, E.; Faleo, S.; de Santis, B.; Miraglia, M. Ochratoxin A contamination in Italian wine samples and evaluation of the exposure in the Italian population. J. Agric. Food Chem. 2008, 56, 10611–10618. [Google Scholar]
- Altiokka, G.; Can, N.O.; Atkosar, Z.; Aboul-Enein, H.Y. Determination of ochratoxin A in Turkish wines. J. Food Drug Anal. 2009, 17, 467–473. [Google Scholar]
- Valero, A.; Marin, S.; Ramos, A.J.; Sanchis, V. Survey: Ochratoxin A in European special wines. Food Chem. 2008, 108, 593–599. [Google Scholar] [CrossRef]
- Meca, G.; Blaiotta, G.; Ritieni, A. Reduction of ochratoxin A during the fermentation of Italian red wine Moscato. Food Contr. 2010, 21, 579–583. [Google Scholar] [CrossRef]
- Valenta, H. Chromatographic methods for the determination of Ochratoxin A in animal and human tissues and fluids. J. Chromatog. A 1998, 815, 75–92. [Google Scholar] [CrossRef]
- Pohland, A.E.; Schüller, P.L.; Steyn, P.S.; van Egmond, H. Physicochemical data for selected mycotoxins. Pure Appl. Chem. 1982, 54, 2219–2284. [Google Scholar] [CrossRef]
- Jimenez, A.M.; Lopez de Cerain, A.; Gonzalez-Peñas, E.; Bello, J. A high-performance liquid-chromatographic method for the determination of ochratoxin A in human plasma. Chromatographia 1999, 50, 457–460. [Google Scholar] [CrossRef]
- Amézqueta, S.; González-Peñas, E.; Murillo, M.; López de Cerain, A. Validation of a high-performance liquid chromatography analytical method for ochratoxin A quantification in cocoa beans. Food Add. Contam. 2004, 21, 1096–1106. [Google Scholar] [CrossRef]
- Chiodini, A.M.; Scherpenisse, P.; Bergwerff, A.A. Ochratoxin A contents in Wine: Comparison of organically and conventionally produced products. J. Agri. Food Chem. 2006, 54, 7399–7404. [Google Scholar]
- Burdaspal, P.A.; Legarda, T.M. Ochratoxin A in wines and grape products originated from Spain and other European countries. Alimentaria 1999, 36, 107–113. [Google Scholar]
- Belli, N.; Marin, S.; Duaigues, A.; Ramos, A.J.; Sanchis, V. Ochratoxin A in wines, musts and grape juices from Spain. J. Sci. Food Agric. 2004, 84, 541–546. [Google Scholar] [CrossRef]
- Filali, A.; Ouammi, L.; Betbeder, A.M.; Baudrimont, I.; Soulaymani, R.; Benayada, A.; Creppy, E.E. Ochratoxin A in beverages from Morocco: A preliminary survey. Food Add. Contam. 2001, 18, 565–568. [Google Scholar]
- Shephard, G.S.; Fabiani, A.; Stockenstrom, S.; Mshicileli, N.; Sewram, V. Quantitation of Ochratoxin A in South African Wines. J. Agric. Food Chem. 2003, 51, 1102–1106. [Google Scholar]
- Var, I.; Kabak, B. Occurrence of ochratoxin A in Turkish wines. Microchem. J. 2007, 86, 241–247. [Google Scholar] [CrossRef]
- Var, I.; Erginkaya, Z.; Kabak, B. Reduction of ochratoxin A levels in white wine by yeast treatments. J. Inst. Brew. 2009, 115, 30–34. [Google Scholar]
- Bizaj, E.; Mavri, J.; Cus, F.; Raspor, A. Removal of ochratoxin A in saccharomyces cerevisiae liquid cultures. S. Afr. J. Eonol. Vitic. 2009, 30, 151–155. [Google Scholar]
- Huwig, A.; Freimund, S.; Kappeli, O.; Dutler, H. Mycotoxin detoxification of animal feed by different adsorbents. Toxicol. Lett. 2001, 122, 179–188. [Google Scholar] [CrossRef]
- Stander, M.A.; Bornscheuer, U.T.; Henke, E.; Steyn, P.S. Screening of commercial hydrolases for the degradation of ochratoxin A. J. Agric. Food Chem. 2000, 48, 5736–5739. [Google Scholar]
- Deberghes, P.; Deffieux, G.; Gharbi, A.; Betbeder, A.M.; Boisard, F.; Blanc, R.; Delaby, J.F.; Creppy, E.E. Detoxification de l’ochratoxine A par des moyens physiques, chimiques et enzymatiques. Hum. Ochratoxicosis Pathol. 1993, 231, 75–82. [Google Scholar]
- Karlovsky, P. Biological detoxification of fungal toxins and its use in plant breeding, feed and food production. Nat. Toxins 1999, 7, 1–23. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Murillo-Arbizu, M.T.; Amézqueta, S.; González-Peñas, E.; De Cerain, A.L. Occurrence of Ochratoxin A in Southern Spanish Generous Wines under the Denomination of Origin “Jerez-Xérès-Sherry and ‘Manzanilla’ Sanlúcar de Barrameda”. Toxins 2010, 2, 1054-1064. https://doi.org/10.3390/toxins2051054
Murillo-Arbizu MT, Amézqueta S, González-Peñas E, De Cerain AL. Occurrence of Ochratoxin A in Southern Spanish Generous Wines under the Denomination of Origin “Jerez-Xérès-Sherry and ‘Manzanilla’ Sanlúcar de Barrameda”. Toxins. 2010; 2(5):1054-1064. https://doi.org/10.3390/toxins2051054
Chicago/Turabian StyleMurillo-Arbizu, Mª Teresa, Susana Amézqueta, Elena González-Peñas, and Adela López De Cerain. 2010. "Occurrence of Ochratoxin A in Southern Spanish Generous Wines under the Denomination of Origin “Jerez-Xérès-Sherry and ‘Manzanilla’ Sanlúcar de Barrameda”" Toxins 2, no. 5: 1054-1064. https://doi.org/10.3390/toxins2051054
APA StyleMurillo-Arbizu, M. T., Amézqueta, S., González-Peñas, E., & De Cerain, A. L. (2010). Occurrence of Ochratoxin A in Southern Spanish Generous Wines under the Denomination of Origin “Jerez-Xérès-Sherry and ‘Manzanilla’ Sanlúcar de Barrameda”. Toxins, 2(5), 1054-1064. https://doi.org/10.3390/toxins2051054





