Ochratoxin A and Aflatoxins in Liquorice Products
Abstract
:1. Introduction
| OTA (µg kg-1) | AFB1 (µg kg-1) | Sum of AFB1, AFB2, AFG1 and AFG2(µg kg-1) | |
|---|---|---|---|
| Cereals for direct human consumption | 3.0 | 2.0 | 4.0 |
| Groundnuts, nuts and dried fruit for direct human consumption | - | 2.0 | 4.0 |
| Spices (Capsicum spp., Piper spp., Myristica fragrans, Zingiber officinale, Curcuma longa) | 30* | 5.0 | 10.0 |
| Dried wine fruit | 10.0 | - | - |
| Wine and grape juice | 2.0 | - | - |
| Roasted coffee | 5.0 | - | - |
| Soluble coffee | 10.0 | - | - |
| Processed cereal-based foods and baby foods for infants and young children | 0.50 | 0.1 | |
| Liquorice root | 20 | ||
| Liquorice extract | 80 |
2. Results and Discussion
2.1. Performance of the analytical method
| Recovery (%) | ||||
|---|---|---|---|---|
| Spike level (µg kg-1) | Number of analyses | Mean ± standard deviation | Range | |
| OTA | ||||
| Dried liquorice extract | 2 | 4 | 92.2 ± 1.7 | 90.1–94.2 |
| 10 | 4 | 91.8 ± 1.5 | 90.0–93.6 | |
| Liquorice confectionery | 2 | 4 | 91.0 ± 1.8 | 89.1–93.3 |
| 10 | 4 | 90.6 ± 1.6 | 88.7–92.5 | |
| AFB1 | ||||
| Dried liquorice extract | 2 | 4 | 90.6 ± 1.8 | 88.4–92.5 |
| 10 | 4 | 90.9 ± 1.6 | 89.0–92.8 | |
| Liquorice confectionery | 2 | 4 | 89.7 ± 2.3 | 87.5–91.8 |
| 10 | 4 | 89.5 ± 2.0 | 87.3–91.5 | |
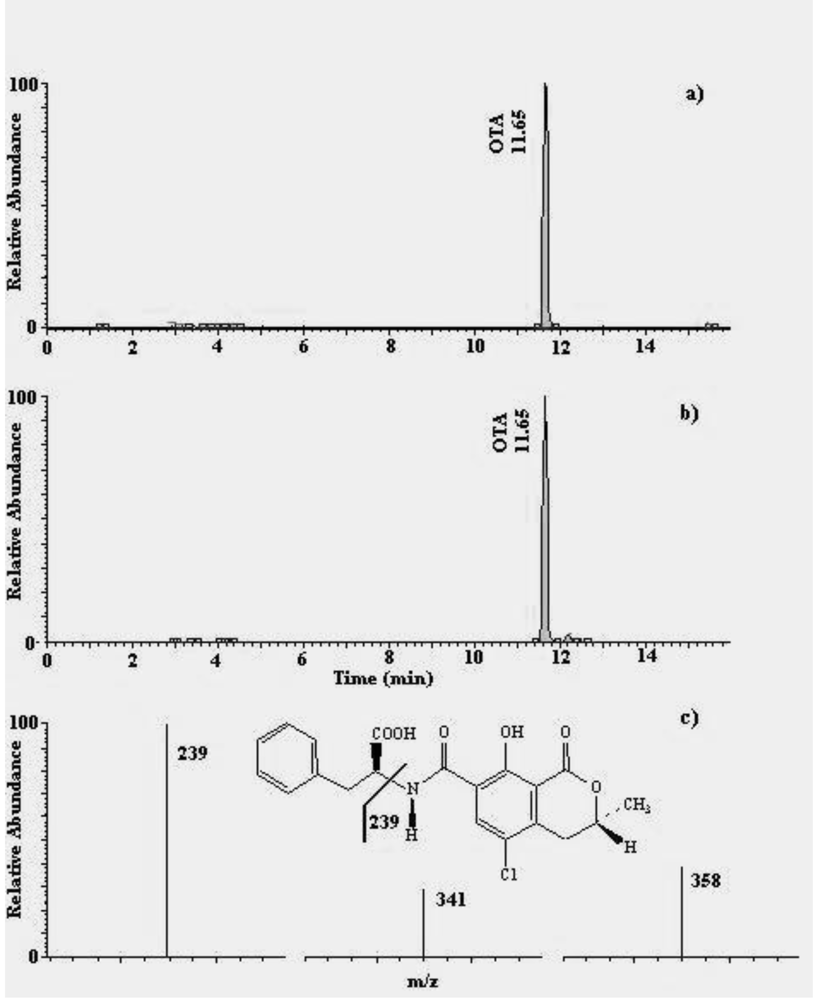
2.2. Ochratoxin A
| n | Positives > LOD (>LOQ) | Incidence of positives (%) | Mean of positives (µg kg-1) | Mean of all* (µg kg-1) | Median (µg kg-1) | Maximum value (µg kg-1) | |
|---|---|---|---|---|---|---|---|
| OTA | |||||||
| Dried liquorice extract | 28 | 28 (27) | 100 | 89.6 | 89.6 | 26.3 | 990.1 |
| Liquorice confectionery | 54 | 33 (20) | 61.1 | 1.53 | 0.96 | 0.17 | 8.3 |
| AFB1 | |||||||
| Dried liquorice extract | 28 | 5 (4) | 17.9 | 1.57 | 0.38 | <LOD | 2.4 |
| Liquorice confectionery | 54 | 8 (5) | 14.8 | 2.06 | 0.41 | <LOD | 7.7 |
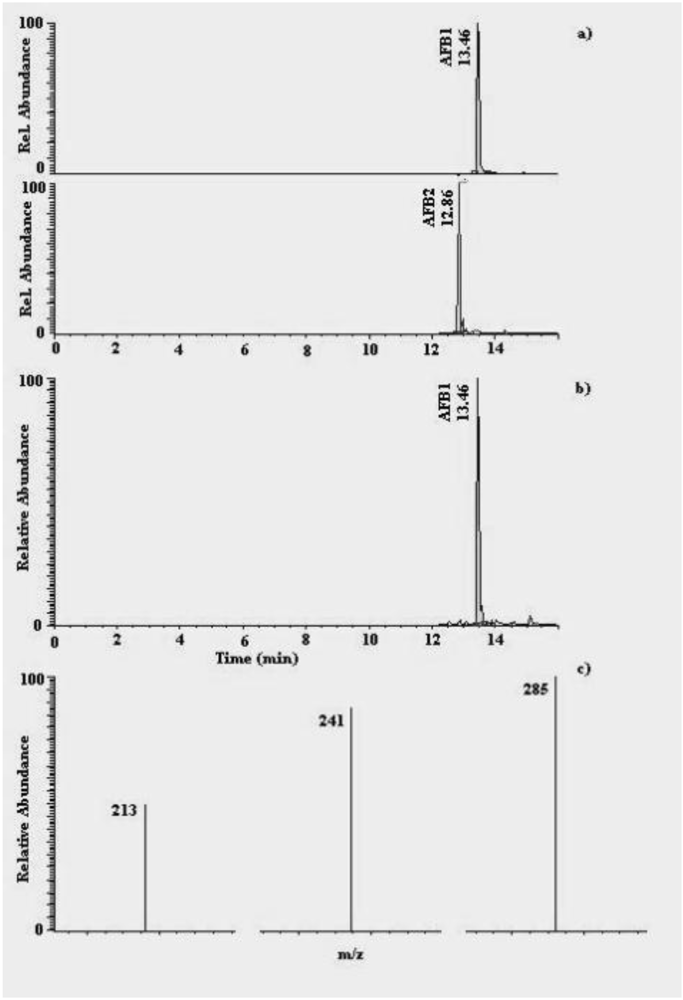
2.3. Aflatoxins
2.4. Estimation of OTA dietary intakes by the consumption of liquorice products
3. Experimental Section
3.1. Samples
3.2. Reagents and standards
3.3. Analysis for OTA
3.3.1. HPLC-FLD analysis
3.3.2. HPLC-MS/MS analysis
3.4. Analysis for AFs
3.4.1. HPLC-FLD analysis
3.4.2. HPLC-MS/MS analysis
4. Conclusions
References
- Frisvad, J.C.; Thrane, U.; Samson, R.A. Mycotoxin producers. In Food Mycology. A Multifaceted Approach to Fungi and Food; Dijksterhuis, J., Samson, R.A., Eds.; CRC Press,Taylor & Francis Group: Boca Raton, FL, USA, 2007; pp. 135–159. [Google Scholar]
- Marquardt, R.; Frohlich, A. A review of recent advances in understanding ochratoxicosis. J. Anim. Sci. 1992, 70, 3968–3988. [Google Scholar]
- Pleština, R. Nephrotoxicity of ochratoxin A. Food Addit. Contam. 1996, 13, 49–50. [Google Scholar] [PubMed]
- Schlatter, Ch.; Studer-Rohr, J.; Rásonyi, Th. Carcinogenicity and kinetic aspects of ochratoxin A. Food Addit. Contam. 1996, 13, 43–44. [Google Scholar] [PubMed]
- Joint FAO/WHO Expert Committee on Food Additives (JEFCA). Ochratoxin A. In Safety Evaluation of Certain Mycotoxins, Prepared by the Fifty-sixth Meeting of JEFCA. WHO food additives series 47/FAO food and nutrition 74 International Programme on Chemial Safety (IPCS); WHO: Geneva, Switzerland, 2001; p. 281. [Google Scholar]
- Benford, D.; Boyle, C.; Dekant, W.; Fuchs, R.; Gaylor, D.; Hard, G.; Mc Gregor, D.; Pitt, J.; Pleština, R.; Shephard, G.; Solfrizzo, M.; Vergere, P.; Walker, R. Ochratoxin A. Safety evaluation of certain mycotoxins in food. In WHO Food Additives; World Health Organization: Geneva, Switzerland, 2001; Volume Series 47, p. 281. [Google Scholar]
- Thirumala Devi, K.; Mayo, M.; Gopal-Reddy Emanuel, K.; Larondelle, Y.; Reddy, D.; Reddy, G. Occurrence of ochratoxin A in black pepper, coriander, ginger and turmeric in India. Food Addit. Contam. 2001, 18, 830–835. [Google Scholar]
- Pietri, A.; Bertuzzi, T.; Gualla, A.; Piva, G. Occurrence of ochratoxin A in raw ham muscle and in pork products from northern Italy. Ital. J. Food Sci. 2006, 1, 1–8. [Google Scholar]
- Zimmerli, B.; Dick, R. Ochratoxin A in table wine and grape-juice: occurrence and risk assessment. Food Addit. Contam. 1996, 13, 655–688. [Google Scholar]
- Shephard, G.S. Impact of mycotoxins on human health in developing countries. Food Addit. Contam. 2008, 25, 146–151. [Google Scholar]
- Fernandez-Pinto, V.; Patriarca, A.; Locani, O.; Vaamonde, G. Natural co-occurrence of aflatoxin and cyclopiazonic acid in peanuts grown in Argentina. Food Addit. Contam. 2001, 18, 1017–1020. [Google Scholar]
- Pietri, A.; Bertuzzi, T.; Pallaroni, L.; Piva, G. Occurrence of mycotoxins and ergosterol in maize harvested over 5 years in Northern Italy. Food Addit. Contam. 2004, 21 (5), 479–487. [Google Scholar]
- Galvano, F.; Ritieni, A.; Piva, G.; Pietri, A. Mycotoxins in the human food chain. In Mycotoxins Blue Book; Diaz, D., Ed.; Nottingham University Press: Nottingham, UK, 2005; pp. 187–224. [Google Scholar]
- Castegnaro, M.; Wild, C. IARC activities in mycotoxins research. Nat. Toxins 1995, 3, 327–331. [Google Scholar]
- International Agency for Research on Cancer (IARC). Some naturally occurring substances, food items and constituents, heterocyclic aromatic amines and mycotoxins. In IARC Monographs on Evaluation of Carcinogenic Risk to Humans; IARC Press: Lyon, France, 1993; Volume 56, p. 245. [Google Scholar]
- Commission of the European Communities. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364, 5–24. [Google Scholar]
- Commission of the European Communities. Commission Regulation (EC) No 105/2010 of 5 February 2010 setting maximum levels for certain contaminants in foodstuffs as regards ochratoxin A. Off. J. Eur. Union 2010, L35, 7–8. [Google Scholar]
- European Food safety Authority (EFSA). Opinion of the scientific panel on contaminants in the food chain on a request from the commission related to ochratoxin A in food. Question No EFSA-Q-2005-154, adopted on 4 April 2006. EFSA J. 2006, 365, 1–56. [Google Scholar]
- Bresch, H.; Urbanek, M.; Hell, K. Ochratoxin A in coffee, tea and beer. Archiv für Lebensmittelhygiene 2000, 51, 89–94. [Google Scholar]
- Bresch, H.; Urbanek, M.; Nusser, M. Ochratoxin A in food containing liquorice. Nahrung 2000, 44, 276–278. [Google Scholar]
- Majerus, P.; Max, M.; Klaffke, H.; Palavinskas, R. Ochratoxin A in Süßholz, Lakritze und daraus hergestellten Erzeugnissen. Deutsche Lebensmittel-Rundschau 2000, 96, 451–454. [Google Scholar]
- Arino, A.; Herrera, M.; Estopañan, G.; Juan, T. High levels of ochratoxin A in licorice and derived products. Int. J. Microbiol. 2007, 114, 366–369. [Google Scholar]
- Trucksess, M.W.; Scott, P.M. Mycotoxins in botanicals and dried fruits: A review. Food Addit. Contam. 2008, 25, 181–192. [Google Scholar]
- Herrera, M.; Herrera, A.; Arino, A. Estimation of dietary intake of ochratoxin A from liquorice confectionery. Food Chem. Toxicol. 2009, 47, 2002–2006. [Google Scholar]
- Kabelitz, L.; Sievers, H. Contaminants of medicinal and food herbs with a view to EU regulations. Innovations Food Technol. 2004, 25–27. [Google Scholar]
- Arino, A.; Herrera, M.; Langa, E.; Raso, J.; Herrera, A. Ochratoxin A in liquorice as affected by processing methods. Food Addit. Contam. 2007, 24 (9), 987–992. [Google Scholar]
- Bertuzzi, T.; Gualla, A.; Fortunati, P.; Pietri, A. Indagine sulla presenza di aflatossine e ocratossina A in prodotti a base di liquirizia. In Rapporti ISTISAN, II Conference “Le micotossine nella filiera agro-alimentare”, Rome, Italy, 16-18/10/2006; 07/37, pp. 139–142.
- Commission of the European Communities. Commission Decision (EC) No 657/2002 of 12 August 2002 concerning the performance of analytical methods and the interpretation of results. Off. J. Eur. Union 2002, L221, 8–36. [Google Scholar]
- Becker, M.; Degelmann, P.; Herderich, M.; Schreier, P.; Humpf, H. Column liquid chromatography-electrospray ionisation-tandem mass spectrometry for the analysis of ochratoxin. J. Chromatogr. A 1998, 818, 260–264. [Google Scholar]
- Plattner, R.D.; Bennet, G.A.; Stubblefield, R.D. Identification of aflatoxins in dust and urine by liquid chromatography-electrospray ionization tandem mass spectrometry. J. Assoc. Off. Anal. Chem. 1984, 67 (4), 734–738. [Google Scholar]
- Edinboro, L.E.; Karnes, H.T. Determination of aflatoxin B1 in sidestream cigarettes smoke by immunoaffinity column extraction coupled with liquid chromatography/mass spectrometry. J. Chromatogr. A 2005, 1083 (1-2), 127–132. [Google Scholar] [CrossRef]
- Lattanzio, V.M.T.; Solfrizzo, M.; Powers, S.; Visconti, A. Simultaneous determination of aflatoxins, ochratoxin A and Fusarium toxins in maize by liquid chromatography/tandem mass spectrometry after multitoxin immunoaffinity cleanup. Rapid Commun. Mass Spectrom. 2007, 21, 3253–3261. [Google Scholar]
- Fiore, C.; Eisenhut, M.; Ragazzi, E.; Zanchin, G.; Armanini, D. A history of the therapeutic use of liquorice in Europe. J. Ethnopharmacol. 2005, 99, 317–324. [Google Scholar]
- SCF. Reports of the Scientific Committee on Food (29th series), Commission of the European Communities, Food Science and Techniques. Report No EUR 14482 EN; CEC: Luxembourg, 1991. [Google Scholar]
- Størmer, F.; Reistad, R.; Alexander, J. Glycyrrhizinic acid in liquorice-evaluation of health hazard. Food Chem. Toxicol. 1993, 31, 303–312. [Google Scholar]
- SCF. Opinion of the Scientific Committee on food on glycyrrhizinic acid and its ammonium salt (expressed on 4 April 2003). Directorate-Scientific Opinions SCF/CS/ADD/EDUL/225 Final-10 April 2003; European Commission: Brussels, Belgium.
- A.O.A.C. Natural Toxins: Ochratoxins. In Official Methods of Analysis, 16th ed; International Association of Official Analytical Chemists: Arlington, VA, USA, 1995; Volume Chapter 49, p. 38. [Google Scholar]
- A.O.A.C. Natural Toxins: Aflatoxins. In Official Methods of Analysis, 18th ed; International Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2005; Volume Chapter 49, p. 4. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pietri, A.; Rastelli, S.; Bertuzzi, T. Ochratoxin A and Aflatoxins in Liquorice Products. Toxins 2010, 2, 758-770. https://doi.org/10.3390/toxins2040758
Pietri A, Rastelli S, Bertuzzi T. Ochratoxin A and Aflatoxins in Liquorice Products. Toxins. 2010; 2(4):758-770. https://doi.org/10.3390/toxins2040758
Chicago/Turabian StylePietri, Amedeo, Silvia Rastelli, and Terenzio Bertuzzi. 2010. "Ochratoxin A and Aflatoxins in Liquorice Products" Toxins 2, no. 4: 758-770. https://doi.org/10.3390/toxins2040758





