A Randomized Steady-State Bioavailability Study of Synthetic versus Natural (Kiwifruit-Derived) Vitamin C
Abstract
:1. Introduction
2. Study Design and Methods
2.1. Participants
2.2. Study Design
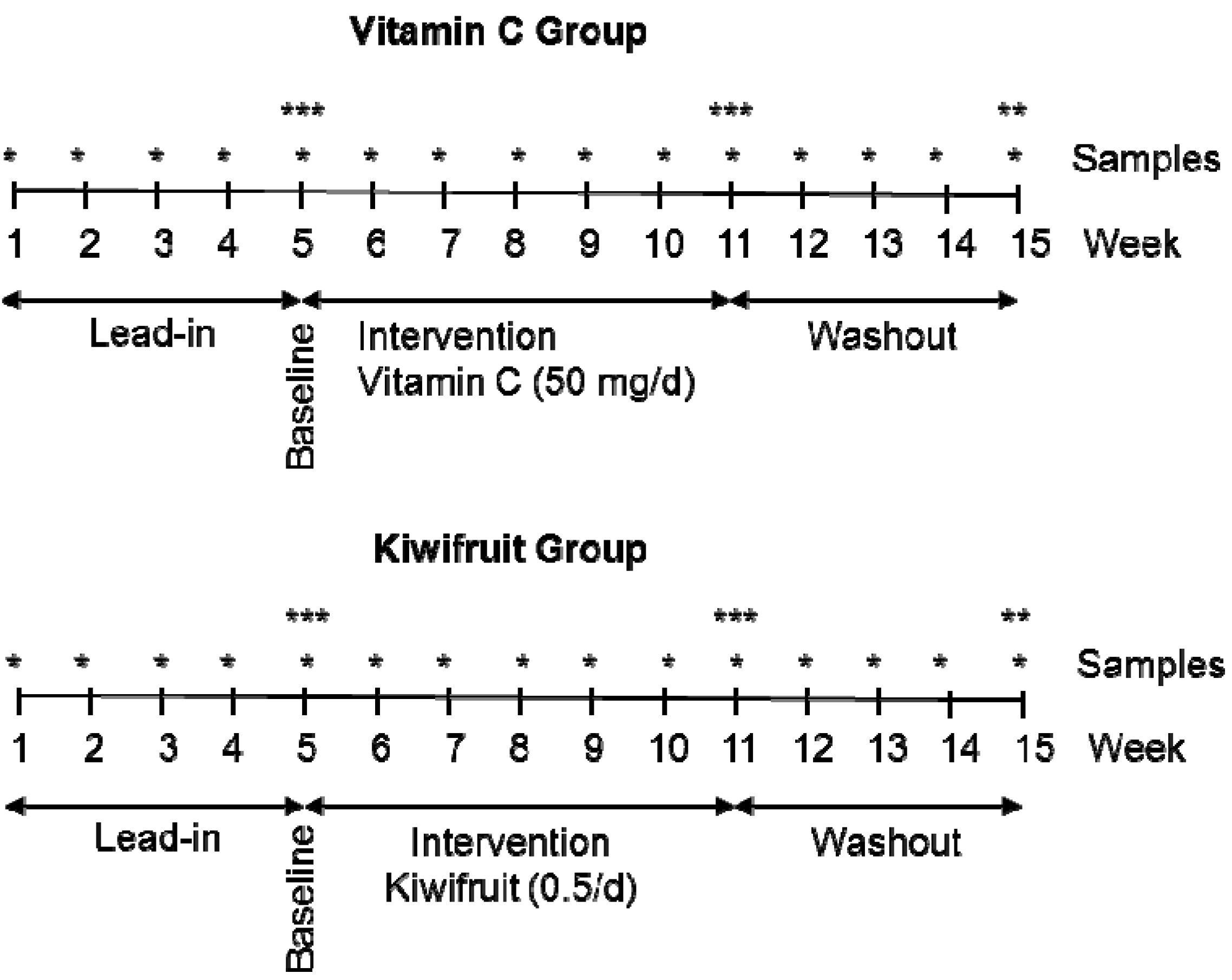
2.3. Interventions
2.4. Sample Collection and Processing
2.4.1. Plasma, Urine and Semen
2.4.2. Mononuclear Leukocytes and Neutrophils
2.4.3. Skeletal Muscle Tissue
2.5. Analysis of Vitamin C by HPLC
2.6. Analysis of Food and Beverage Records
2.7. Statistical Analysis
3. Results
3.1. Participant Characteristics
| Screened a (n = 134) | Vitamin C Group a (n = 18) | Kiwifruit Group a (n = 18) | |
|---|---|---|---|
| Age (years) | 21 ± 3 | 21 ± 3 | 22 ± 4 |
| Weight (kg) | 81 ± 16 | 84 ± 19 | 89 ± 23 |
| Height (cm) | 182 ± 7 | 181 ± 7 | 181 ± 7 |
| BMI (kg/m2) | 24 ± 4 | 26 ± 5 | 27 ± 6 * |
| Ascorbate (µmol/L) | 48 ± 16 | 31 ± 11 ** | 34 ± 10 ** |
3.2. Dietary Intake of Vitamin C
| Vitamin C group a (50 mg/day) | Kiwifruit group a (half/day) | Between group | |||||
|---|---|---|---|---|---|---|---|
| Baseline | Intervention | Washout | Baseline | Intervention | Washout | Intervention P value b | |
| 31.2 ± 3.1 | 76.7 ± 2.6 *** | 29.1 ± 2.9 | 28.6 ± 3.1 | 73.4 ± 4.2 *** | 31.4 ± 5.1 | 0.512 | |
| Plasma (µmol/L) | 23.5 ± 2.5 | 51.3 ± 3.5 *** | 34.3 ± 4.4 * | 22.7 ± 2.5 | 45.5 ± 2.5 *** | 30.5 ± 3.1 | 0.860 |
| Urine (µmol/24 h) | 42.8 ± 9.2 | 104.2 ± 27.6 * | 59.5 ± 19.9 | 32.3 ± 8.1 | 70.5 ± 17.0 * | 64.7 ± 29.8 | 0.503 |
| Semen (µmol/L) | 284.8 ±27.0 | 321.0 ± 33.3 | 264.4 ± 31.1 * | 326.3 ± 47.5 | 378.8 ± 42.7 | 237.7 ± 19.6 * | 0.676 |
| Mononuclear cells (nmol/108 cells) | 38.7 ± 6.2 | 84.5 ± 6.3 *** | 78.5 ± 4.3 ** | 60.5 ± 6.1 | 90.9 ± 6.1 ** | 75.2 ± 5.2 | 0.227 |
| Neutrophils (nmol/108 cells) | 21.9 ± 3.1 | 39.6 ± 3.8 ** | 24.9 ± 2.1 | 13.7 ± 2.4 | 30.4 ± 2.5 *** | 24.8 ± 1.7 *** | 0.798 |
| Skeletal muscle (nmol/g) | 14.5 ± 2.0 | 61.3 ± 3.5 *** | nd | 15.1 ± 2.5 | 52.8 ± 5.0 *** | nd | 0.429 |
3.3. Vitamin C Status of Plasma, Urine and Semen

3.4. Vitamin C Status of Leukocytes and Skeletal Muscle Tissue
4. Discussion
5. Conclusions
Acknowledgements
Conflicts of Interest
References
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar]
- Tsukaguchi, H.; Tokui, T.; Mackenzie, B.; Berger, U.V.; Chen, X.Z.; Wang, Y.; Brubaker, R.F.; Hediger, M.A. A family of mammalian Na+-dependent l-ascorbic acid transporters. Nature 1999, 399, 70–75. [Google Scholar] [CrossRef]
- Savini, I.; Rossi, A.; Pierro, C.; Avigliano, L.; Catani, M.V. SVCT1 and SVCT2: Key proteins for vitamin C uptake. Amino Acids 2008, 34, 347–355. [Google Scholar] [CrossRef]
- Englard, S.; Seifter, S. The biochemical functions of ascorbic acid. Annu. Rev. Nutr. 1986, 6, 365–406. [Google Scholar] [CrossRef]
- Nishiyama, I.; Yamashita, Y.; Yamanaka, M.; Shimohashi, A.; Fukuda, T.; Oota, T. Varietal difference in vitamin C content in the fruit of kiwifruit and other actinidia species. J. Agric. Food Chem. 2004, 52, 5472–5475. [Google Scholar]
- Vissers, M.C.M.; Bozonet, S.M.; Pearson, J.F.; Braithwaite, L.J. Dietary ascorbate affects steady state tissue levels in vitamin C-deficient mice: Tissue deficiency after sub-optimal intake and superior bioavailability from a food source (kiwifruit). Am. J. Clin. Nutr. 2011, 93, 292–301. [Google Scholar] [CrossRef]
- Beker, B.Y.; Sonmezoglu, I.; Imer, F.; Apak, R. Protection of ascorbic acid from copper(II)-catalyzed oxidative degradation in the presence of flavonoids: Quercetin, catechin and morin. Int. J. Food Sci. Nutr. 2011, 62, 504–512. [Google Scholar] [CrossRef]
- Clemetson, C.A.; Andersen, L. Plant polyphenols as antioxidants for ascorbic acid. Ann. N. Y. Acad. Sci. 1966, 136, 341–476. [Google Scholar] [CrossRef]
- Fiorentino, A.; D’Abrosca, B.; Pacifico, S.; Mastellone, C.; Scognamiglio, M.; Monaco, P. Identification and assessment of antioxidant capacity of phytochemicals from kiwi fruits. J. Agric. Food Chem. 2009, 57, 4148–4155. [Google Scholar]
- Tanaka, K.; Hashimoto, T.; Tokumaru, S.; Iguchi, H.; Kojo, S. Interactions between vitamin C and vitamin E are observed in tissues of inherently scorbutic rats. J. Nutr. 1997, 127, 2060–2064. [Google Scholar]
- Latocha, P.; Krupa, T.; Wolosiak, R.; Worobiej, E.; Wilczak, J. Antioxidant activity and chemical difference in fruit of different Actinidia sp. Int. J. Food Sci. Nutr. 2010, 61, 381–394. [Google Scholar] [CrossRef]
- Carr, A.C.; Pullar, J.M.; Moran, S.; Vissers, M.C.M. Bioavailability of vitamin C from kiwifruit in non-smoking males: Determination of “healthy” and “optimal” intakes. J. Nutr. Sci. 2012, 1, e14. [Google Scholar] [CrossRef]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef]
- Mangels, A.R.; Block, G.; Frey, C.M.; Patterson, B.H.; Taylor, P.R.; Norkus, E.P.; Levander, O.A. The bioavailability to humans of ascorbic acid from oranges, orange juice and cooked broccoli is similar to that of synthetic ascorbic acid. J. Nutr. 1993, 123, 1054–1061. [Google Scholar]
- Hartzler, E.R. The availability of ascorbic acid in papayas and guavas. J. Nutr. 1945, 30, 355–365. [Google Scholar]
- Clayton, M.M.; Borden, R.A. The availability for human nutrition of the vitamin C in raw cabbage and home-canned tomato juice. J. Nutr. 1943, 25, 349–369. [Google Scholar]
- Todhunter, E.N.; Fatzer, A.S. A comparison of the utilization by college women of equivalent amounts of ascorbic acid (vitamin C) in red raspberries and in crystalline form. J. Nutr. 1940, 19, 121–130. [Google Scholar]
- Hawley, E.E.; Stephens, D.J.; Anderson, G. The excretion of vitamin C in normal individuals following a comparable quantitative administration in the form of orange juice, cevitamic acid by mouth and cevitamic acid intravenously. J. Nutr. 1936, 11, 135–145. [Google Scholar]
- Pelletier, O.; Keith, M.O. Bioavailability of synthetic and natural ascorbic acid. J. Am. Diet. Assoc. 1974, 64, 271–275. [Google Scholar]
- Carr, A.C.; Bozonet, S.M.; Pullar, J.M.; Simcock, J.W.; Vissers, M.C. Human skeletal muscle ascorbate is highly responsive to changes in vitamin C intake and plasma concentrations. Am. J. Clin. Nutr. 2013, 97, 800–807. [Google Scholar] [CrossRef]
- Diet Cruncher, version 1.6; Nutricomp: Dunedin, New Zealand, 2003.
- SigmaStat, version 11; Systat Software Inc.: San Jose, CA, USA, 2009.
- Wilson, H.K.; Price-Jones, C.; Hughes, R.E. The influence of an extract of orange peel on the growth and ascorbic acid metabolism of young guinea-pigs. J. Sci. Food Agric. 1976, 27, 661–666. [Google Scholar]
- Douglass, C.D.; Kamp, G.H. The effect of orally administered rutin on the adrenal ascorbic acid level in guinea pigs. J. Nutr. 1959, 67, 531–536. [Google Scholar]
- Papageorge, E.; Mitchell, G.L., Jr. The effect of oral administration of rutin on blood, liver and adrenal ascorbic acid and on liver and adrenal cholesterol in guinea pigs. J. Nutr. 1949, 37, 531–540. [Google Scholar]
- Cotereau, H.; Gabe, M.; Gero, E.; Parrot, J.L. Influence of vitamin P (vitamin C2) upon the amount of ascorbic acid in the organs of the guinea pig. Nature 1948, 161, 557–558. [Google Scholar] [CrossRef]
- Hughes, R.E.; Hurley, R.J.; Jones, P.R. The retention of ascorbic acid by guinea-pig tissues. Br. J. Nutr. 1971, 26, 433–438. [Google Scholar] [CrossRef]
- Todhunter, E.N.; Robbins, R.C.; Ivey, G.; Brewer, W. A comparison of the utilization by guinea pigs of equivalent amounts of ascorbic acid (vitamin C) in lemon juice and in crystalline form. J. Nutr. 1940, 19, 113–120. [Google Scholar]
- Corpe, C.P.; Lee, J.H.; Kwon, O.; Eck, P.; Narayanan, J.; Kirk, K.L.; Levine, M. 6-Bromo-6-deoxy-l-ascorbic acid: An ascorbate analog specific for Na+-dependent vitamin C transporter but not glucose transporter pathways. J. Biol. Chem. 2005, 280, 5211–5220. [Google Scholar]
- Washko, P.W.; Wang, Y.; Levine, M. Ascorbic acid recycling in human neutrophils. J. Biol. Chem. 1993, 268, 15531–15535. [Google Scholar]
- Park, J.B.; Levine, M. Intracellular accumulation of ascorbic acid is inhibited by flavonoids via blocking of dehydroascorbic acid and ascorbic acid uptakes in HL-60, U937 and Jurkat cells. J. Nutr. 2000, 130, 1297–1302. [Google Scholar]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radi. Biol. Med. 2006, 41, 1727–1746. [Google Scholar] [CrossRef]
- Savini, I.; Catani, M.V.; Duranti, G.; Ceci, R.; Sabatini, S.; Avigliano, L. Vitamin C homeostasis in skeletal muscle cells. Free Radic. Biol. Med. 2005, 38, 898–907. [Google Scholar] [CrossRef]
- Low, M.L.; Sandoval, D.; Aviles, E.; Perez, F.; Nualart, F.; Henriquez, J.P. The ascorbic acid transporter SVCT2 is expressed in slow-twitch skeletal muscle fibres. Histochem. Cell. Biol. 2009, 131, 565–574. [Google Scholar] [CrossRef]
- Jacob, R.A.; Pianalto, F.S.; Agee, R.E. Cellular ascorbate depletion in healthy men. J. Nutr. 1992, 122, 1111–1118. [Google Scholar]
- Fraga, C.G.; Motchnik, P.A.; Shigenaga, M.K.; Helbock, H.J.; Jacob, R.A.; Ames, B.N. Ascorbic acid protects against endogenous oxidative DNA damage in human sperm. Proc. Natl. Acad. Sci. USA 1991, 88, 11003–11006. [Google Scholar]
- Kandar, R.; Drabkova, P.; Hampl, R. The determination of ascorbic acid and uric acid in human seminal plasma using an HPLC with UV detection. J. Chromatogr. B 2011, 879, 2834–2839. [Google Scholar] [CrossRef]
- Jacob, R.A.; Kelley, D.S.; Pianalto, F.S.; Swendseid, M.E.; Henning, S.M.; Zhang, J.Z.; Ames, B.N.; Fraga, C.G.; Peters, J.H. Immunocompetence and oxidant defense during ascorbate depletion of healthy men. Am. J. Clin. Nutr. 1991, 54, 1302S–1309S. [Google Scholar]
- Dawson, E.B.; Harris, W.A.; Rankin, W.E.; Charpentier, L.A.; McGanity, W.J. Effect of ascorbic acid on male fertility. Ann. N. Y. Acad. Sci. 1987, 498, 312–323. [Google Scholar] [CrossRef]
- Beck, K.; Conlon, C.A.; Kruger, R.; Coad, J.; Stonehouse, W. Gold kiwifruit consumed with an iron-fortified breakfast cereal meal improves iron status in women with low iron stores: A 16-week randomised controlled trial. Br. J. Nutr. 2011, 105, 101–109. [Google Scholar] [CrossRef]
- Hallberg, L.; Brune, M.; Rossander, L. Effect of ascorbic acid on iron absorption from different types of meals. Studies with ascorbic-acid-rich foods and synthetic ascorbic acid given in different amounts with different meals. Hum. Nutr. Appl. Nutr. 1986, 40, 97–113. [Google Scholar]
- Stonehouse, W.; Gammon, C.S.; Beck, K.L.; Conlon, C.A.; von Hurst, P.R.; Kruger, R. Kiwifruit: Our daily prescription for health. Can. J. Physiol. Pharmacol. 2013, 91, 442–447. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carr, A.C.; Bozonet, S.M.; Pullar, J.M.; Simcock, J.W.; Vissers, M.C.M. A Randomized Steady-State Bioavailability Study of Synthetic versus Natural (Kiwifruit-Derived) Vitamin C. Nutrients 2013, 5, 3684-3695. https://doi.org/10.3390/nu5093684
Carr AC, Bozonet SM, Pullar JM, Simcock JW, Vissers MCM. A Randomized Steady-State Bioavailability Study of Synthetic versus Natural (Kiwifruit-Derived) Vitamin C. Nutrients. 2013; 5(9):3684-3695. https://doi.org/10.3390/nu5093684
Chicago/Turabian StyleCarr, Anitra C., Stephanie M. Bozonet, Juliet M. Pullar, Jeremy W. Simcock, and Margreet C. M. Vissers. 2013. "A Randomized Steady-State Bioavailability Study of Synthetic versus Natural (Kiwifruit-Derived) Vitamin C" Nutrients 5, no. 9: 3684-3695. https://doi.org/10.3390/nu5093684
APA StyleCarr, A. C., Bozonet, S. M., Pullar, J. M., Simcock, J. W., & Vissers, M. C. M. (2013). A Randomized Steady-State Bioavailability Study of Synthetic versus Natural (Kiwifruit-Derived) Vitamin C. Nutrients, 5(9), 3684-3695. https://doi.org/10.3390/nu5093684








