Impact of Short Term Consumption of Diets High in Either Non-Starch Polysaccharides or Resistant Starch in Comparison with Moderate Weight Loss on Indices of Insulin Sensitivity in Subjects with Metabolic Syndrome
Abstract
:1. Introduction
2. Experimental Section
2.1. Kinetic Measurements
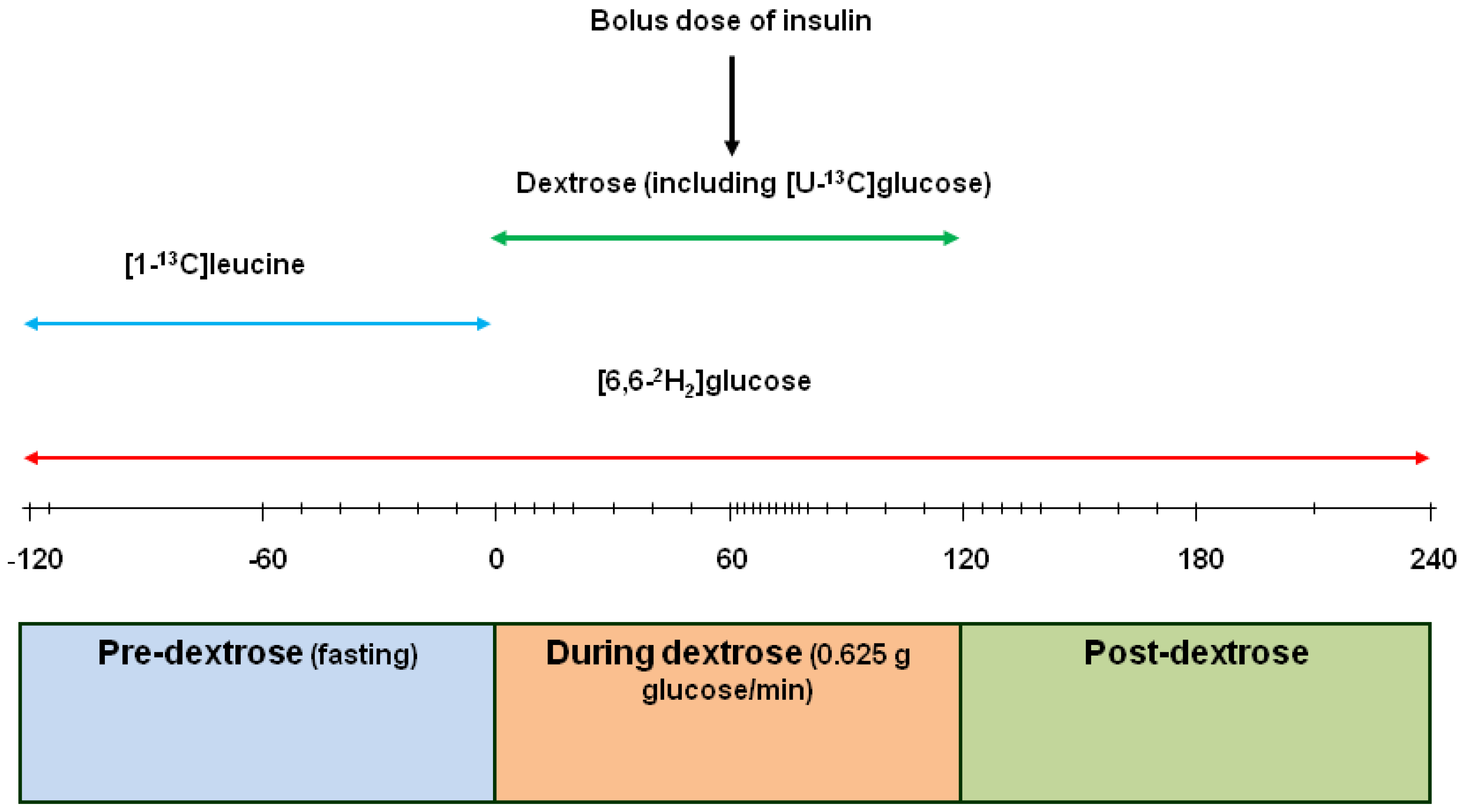
2.2. Laboratory Analyses
2.3. Calculations

2.4. Insulin Sensitivity
2.4.1. Rationale

2.4.2. Estimation of Compartmental Model Parameters from Bolus Dose [U-13C]glucose

2.4.3. Adapting the 3-Pool Model Parameters to a 2-Pool Model
2.4.4. Concentrations of Various Glucose Species
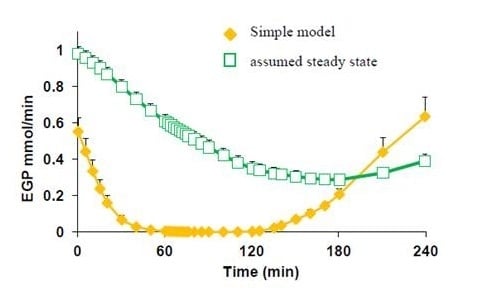
2.4.5. Formal Model for Estimation of ILR and EGP during Dextrose Challenge
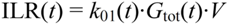
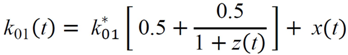
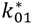 (min–1) the fractional removal rate constant at baseline. It is also assumed that
(min–1) the fractional removal rate constant at baseline. It is also assumed that
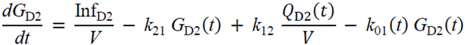
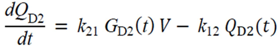
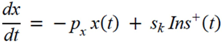
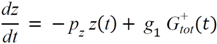
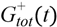 = Gtot(t) − Gtot(0), if Gtot(t) > Gtot(0); and
= Gtot(t) − Gtot(0), if Gtot(t) > Gtot(0); and 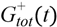 = 0, for Gtot(t) ≤ Gtot(0). Ins+ were defined similarly. The use of z(t) in Equation (1) acts as inhibition on the fractional removal rate constant k01 under excess glucose load, where it is assumed that the maximum inhibition is limited to k01 being reduced by half. The rate of inhibition is modelled by means of parameter g1 (L/mmol/min) in Equation (5). The use of x(t) in equation (1) acts as enhancer of glucose removal due to high insulin concentrations, where the enhancement rate is modelled by means of sk (L/mU/min2) in Equation (4). This equation shows that for high insulin concentrations x(t) will be large and hence ILR will be enhanced. The parameters px (min−1) and pz (min−1) model the gradual decay of the enhanced (or suppressed) removal with time.
= 0, for Gtot(t) ≤ Gtot(0). Ins+ were defined similarly. The use of z(t) in Equation (1) acts as inhibition on the fractional removal rate constant k01 under excess glucose load, where it is assumed that the maximum inhibition is limited to k01 being reduced by half. The rate of inhibition is modelled by means of parameter g1 (L/mmol/min) in Equation (5). The use of x(t) in equation (1) acts as enhancer of glucose removal due to high insulin concentrations, where the enhancement rate is modelled by means of sk (L/mU/min2) in Equation (4). This equation shows that for high insulin concentrations x(t) will be large and hence ILR will be enhanced. The parameters px (min−1) and pz (min−1) model the gradual decay of the enhanced (or suppressed) removal with time.2.4.6. Simple Model for Estimation of ILR and EGP during Dextrose Challenge
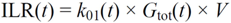
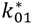 ・ Gtot(t)). The changes in the GD2 concentration and pool size are modelled as described in Equations (2) and (3).
・ Gtot(t)). The changes in the GD2 concentration and pool size are modelled as described in Equations (2) and (3).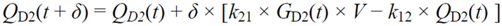
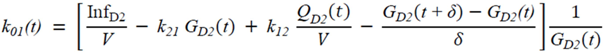
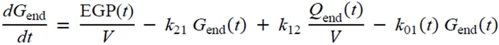
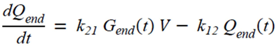
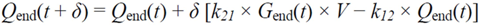
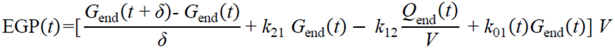
2.5. Statistical Analysis
3. Results
3.1. Fasted Parameters (Table 1)
| Parameter | M | NSP | RS | WL | SED | P4 Diet | P5 Time |
|---|---|---|---|---|---|---|---|
| Glucose mM | 6.13 | 6.01 | 5.91 | 6.03 | 0.127 | 0.392 | 0.011 |
| Insulin pM | 94.9 a | 95.0 a | 81.8 a | 59.4 b | 1.109 | <0.001 | 0.214 |
| C-peptide pM | 1505 | ND | ND | 1085 | 83.1 | <0.001 | |
| EGP μmol/min 2 | 1041 a | 995 a | 996 a | 923 b | 30.6 | 0.006 | <0.001 |
| EGPμmol/min/kg FFM 3 | 15.13 a | 14.56 a,b | 14.65 a | 13.72 b | 0.444 | 0.027 | <0.001 |
| HOMA2-IR | 1.83 a | 1.83 a | 1.58 a | 1.16 b | 0.129 | <0.001 | |
| HOMA2-%B | 93.2 a | 96.0 a | 90.7 a | 69.1 b | 5.15 | <0.001 | |
| HOMA2-%S | 71.9 a | 80.6 a | 80.3 a | 130.1 b | 14.07 | <0.001 | |
| HIS | 14.2 a | 15.9 a | 16.2 a | 28.3 b | 3.12 | <0.001 |
3.2. Basal Glucose and Leucine Metabolism
| AA 3 | Maint | NSP | RS | WL | SED | P 4 Diet |
|---|---|---|---|---|---|---|
| ILRmop μmol/min | 200.6 a | 188.3 b | 198.0 a | 177.2 c | 4.91 | <0.001 |
| Ala μmol/kg | 404.3 a | 382.5 a,b | 362.8 b | 299.1 c | 16.31 | <0.001 |
| Arg μmol/kg | 101.9 a | 97.5 a,b | 94.7 b,c | 89.1 c | 3.53 | 0.007 |
| Asp μmol/kg | 4.0 | 3.7 | 3.4 | 3.0 | 0.40 | 0.076 |
| Cys μmol/kg | 300.1 a | 302.1 a | 287.9 a,b | 298.3 a | 3.63 | 0.002 |
| Gln μmol/kg | 587.4 | 593.5 | 578.8 | 566.3 | 13.70 | 0.233 |
| Glu μmol/kg | 98.0 a | 93.6 a | 90.1 a | 70.8 b | 5.73 | <0.001 |
| Gly μmol/kg | 169.6 | 174.7 | 164.3 | 162.8 | 5.85 | 0.176 |
| Hcy μmol/kg | 6.7 a | 6.4 a,b | 6.1 b | 6.5 a | 0.19 | 0.017 |
| His μmol/kg | 79.5 a | 77.3 a,b | 75.4 b,c | 72.4 c | 1.64 | <0.001 |
| Ile μmol/kg | 73.4 | 78.0 | 75.6 | 74.5 | 3.06 | 0.485 |
| Leu μmol/kg | 146.6 | 141.3 | 140.3 | 137.9 | 4.44 | 0.258 |
| Lys μmol/kg | 180.6 | 176.9 | 177.1 | 179.9 | 4.99 | 0.833 |
| Met μmol/kg | 29.1 a | 27.1 b | 25.9 b,c | 24.3 c | 0.87 | <0.001 |
| Phe μmol/kg | 65.9 a | 61.1 b | 62.4 b | 59.5 b | 1.44 | <0.001 |
| Pro μmol/kg | 229.3a | 203.6a | 179b | 171.5b | 7.83 | <0.001 |
| Ser μmol/kg | 85.7 a | 85.1 a | 82.7 a | 94.1 b | 2.36 | <0.001 |
| Thr μmol/kg | 117.4 | 114.6 | 117.2 | 119.5 | 5.19 | 0.829 |
| Trp μmol/kg | 58.6 a | 58.9 a | 59.9 a | 54.0 b | 1.50 | 0.001 |
| Tyr μmol/kg | 85.2 a | 78.7 b | 77.8 b | 72.0 c | 2.54 | <0.001 |
| Val μmol/kg | 248.2 | 242.3 | 244.1 | 253.7 | 6.61 | 0.336 |
3.3. Bolus [13C]Glucose Injections
3.4. Responses to Dextrose and Post-Dextrose Recovery
| Time (min) | M | NSP | RS | WL | SED | P3 Time | P3 Diet | P3 T × D | |
|---|---|---|---|---|---|---|---|---|---|
| Glucose | 0–60 | 10.49 | 10.46 | 10.46 | 10.74 | 0.38 | <0.001 | 0.450 | 0.209 |
| 60–120 | 13.67 | 14.04 | 13.84 | 14.74 | |||||
| 120–180 | 8.34 | 8.56 | 8.51 | 8.90 | |||||
| 180–240 | 4.81 | 4.63 | 4.67 | 4.79 | |||||
| Insulin | 0–60 | 294 a | 287 a,b | 294 a | 199 b | 45.6 | <0.001 | <0.001 | 0.001 |
| 60–120 | 689 a | 352 a | 634 a | 437 b | |||||
| 120–180 | 572 a | 601 a | 544 a | 353 b | |||||
| 180–240 | 214 a | 185 a,b | 161 a,b | 107 b | |||||
| Cpep:Ins | 0–60 | 8.61 a | 11.37 b | 2.596 | 0.028 | <0.001 | 0.017 | ||
| 60–120 | 5.99 | 8.66 | |||||||
| 120–180 | 8.94 | 12.59 | |||||||
| 180-240 | 15.70 a | 26.26 b |
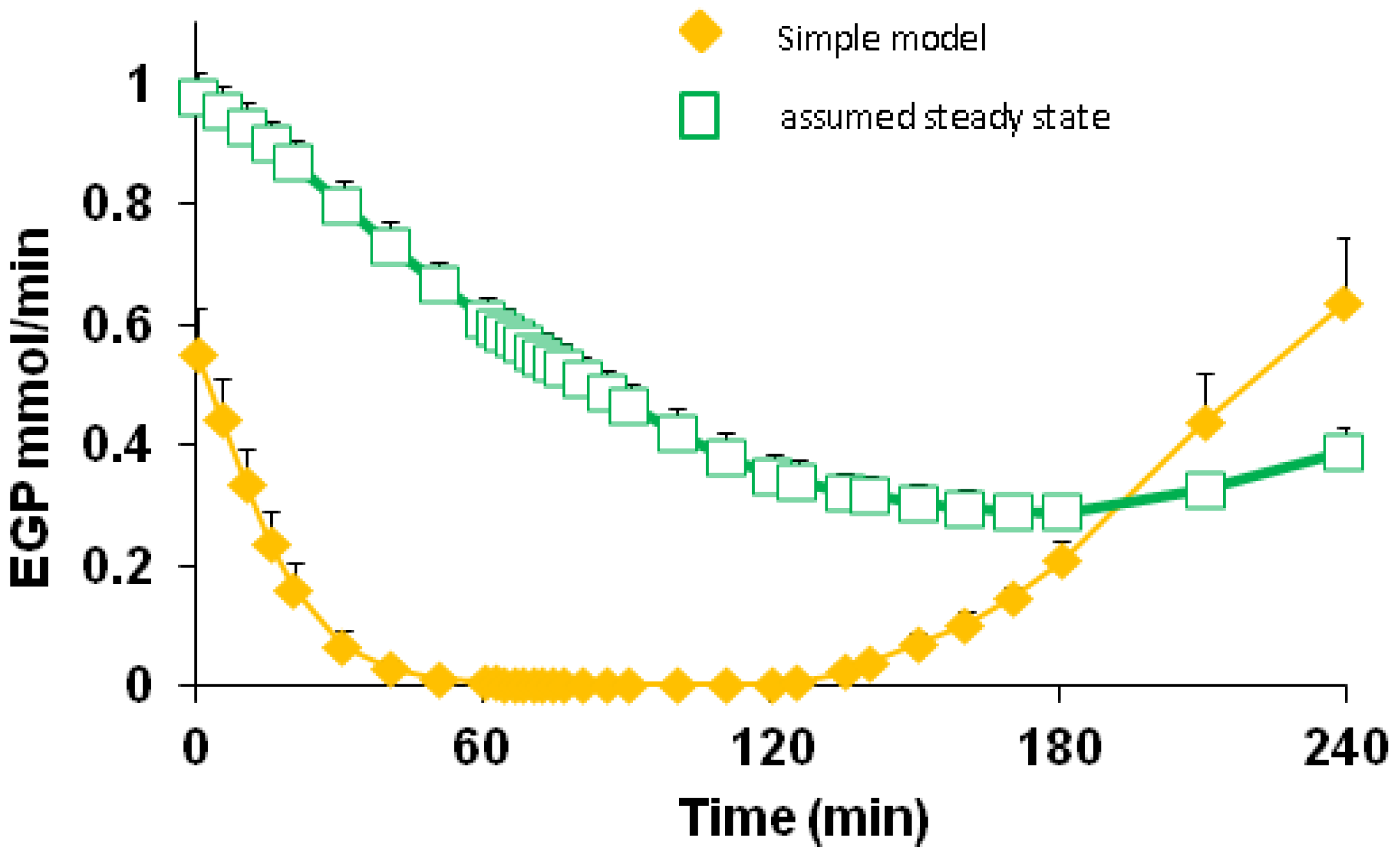
3.5. Endogenous Glucose Production and Glucose ILR
| Variable2 | Time (min) | M | NSP | RS | WL | SED | P3 Time | P3 Diet | P3 T × D |
|---|---|---|---|---|---|---|---|---|---|
| EGP | 0–60 | 0.149 | 0.146 | 0.169 | 0.173 | 0.036 | <0.001 | 0.601 | 0.966 |
| 60–120 | 0.002 | 0.006 | 0.013 | 0.015 | |||||
| 120–180 | 0.076 | 0.083 | 0.101 | 0.128 | |||||
| 180–240 | 0.428 | 0.373 | 0.389 | 0.418 | |||||
| ILR | 0–60 | 1.95 a | 1.85 a,b | 1.94 a | 1.64 b | 0.11 | <0.001 | 0.830 | 0.012 |
| 60–120 | 3.74 a | 3.74 a | 3.73 a | 3.72 a | |||||
| 120–180 | 3.07 a | 3.23 a,b | 3.20 a | 3.42 b | |||||
| 180–240 | 1.40 a | 1.38 a | 1.38 a | 1.54 a | |||||
| k01 | 0–60 | 0.0124 a | 0.0117 a | 0.0122 a | 0.0101 b | 0.0011 | <0.001 | 0.929 | 0.047 |
| 60–120 | 0.0184 a | 0.0182 a | 0.0182 a | 0.0172 a | |||||
| 120–180 | 0.0261 a | 0.0277 a | 0.0268 a | 0.0278 a | |||||
| 180–240 | 0.0194 a | 0.0203 a | 0.0207 a | 0.0219 b | |||||
| ILR/Ins | 0–60 | 8.83 a | 9.33 a | 8.67 a | 13.50 a | 2.834 | <0.001 | 0.003 | <0.001 |
| 60–120 | 0.045 a | 0.049 a | 0.046 a | 0.075 a | |||||
| 120–180 | 9.17 a | 9.34 a | 9.84 a | 17.84 b | |||||
| 180–240 | 11.84 a | 13.50 a | 13.00 a | 27.84 b |
3.6. Insulin Sensitivity
| Parameter | M | NSP | RS | WL | SED | P 3 Diet |
|---|---|---|---|---|---|---|
| px (/min) | 16.71 | 12.40 | 15.13 | 10.61 | 3.38 | 0.286 |
| sk (L/mU/min2) | 7.43 a | 8.06 a | 8.20 a | 10.93 b | 1.17 | 0.024 |
| pz (/min) | 1.30 | 29.80 | 32.45 | 1.56 | 23.53 | 0.867 |
| g1 (L/mmol//min) | 153 | 122 | 157 | 225 | 54.2 | 0.240 |
4. Discussion
4.1. Protein Dynamics and Aminoacidaemia
4.2. Endogenous Glucose Production
4.3. C-Peptide and Insulin
4.4. Diet and Insulin Sensitivity
5. Conclusions
Acknowledgements
Conflict of Interest
References
- Willett, W.C.; Dietz, W.H.; Colditz, G.A. Guidelines for healthy weight. N. Engl. J. Med. 1999, 341, 427–434. [Google Scholar] [CrossRef]
- Haslam, D.W.; James, W.P. Obesity. Lancet 2005, 366, 1197–1209. [Google Scholar] [CrossRef]
- Counterweight Project Team. Influence of body mass index on prescribing costs and potential cost savings of a weight management programme in primary care. J. Health Serv. Res. Policy 2008, 13, 158–166. [CrossRef]
- Nuttall, F.Q.; Gannon, M.C.; Saeed, A.; Jordan, K.; Hoover, H. The metabolic response of subjects with type 2 diabetes to a high-protein, weight-maintenance diet. J. Clin. Endocrinol. Metab. 2003, 88, 3577–3583. [Google Scholar] [CrossRef]
- Sullivan, V.K. Prevention and treatment of the metabolic syndrome with lifestyle intervention: Where do we start? J. Am. Diet. Assoc. 2006, 106, 668–671. [Google Scholar] [CrossRef]
- Fappa, E.; Yannakoulia, M.; Pitsavos, C.; Skoumas, I.; Valourdou, S.; Stefanadis, C. Lifestyle intervention in the management of metabolic syndrome: Could we improve adherence issues? Nutrition 2008, 24, 286–291. [Google Scholar] [CrossRef]
- Muscelli, E.; Camastra, S.; Catalano, C.; Galvan, A.Q.; Ciociaro, D.; Baldi, S.; Ferrannini, E. Metabolic and cardiovascular assessment in moderate obesity: Effect of weight loss. J. Clin. Endocrinol. Metab. 1997, 82, 2937–2943. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Kelley, D.E.; Wing, R.R.; Meier, A.; Thaete, F.L. Effects of weight loss on regional fat distribution and insulin sensitivity in obesity. Diabetes 1999, 48, 839–847. [Google Scholar] [CrossRef]
- Skov, A.R.; Toubro, S.; Ronn, B.; Holm, L.; Astrup, A. Randomized trial on protein vs. carbohydrate in ad libitum fat reduced diet for the treatment of obesity. Int. J. Obes. Relat. Metab. Disord. 1999, 23, 528–536. [Google Scholar]
- Weigle, D.S.; Breen, P.A.; Matthys, C.C.; Callahan, H.S.; Meeuws, K.E.; Burden, V.R.; Purnell, J.Q. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am. J. Clin. Nutr. 2005, 82, 41–48. [Google Scholar]
- Johnstone, A.M.; Horgan, G.W.; Murison, S.D.; Bremner, D.M.; Lobley, G.E. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am. J. Clin. Nutr. 2008, 87, 44–55. [Google Scholar]
- Wolever, T.M.; Gibbs, A.L.; Mehling, C.; Chiasson, J.L.; Connelly, P.W.; Josse, R.G.; Leiter, L.A.; Maheux, P.; Rabasa-Lhoret, R.; Rodger, N.W.; Ryan, E.A. The Canadian Trial of Carbohydrates in Diabetes (CCD), a 1-y controlled trial of low-glycemic-index dietary carbohydrate in type 2 diabetes: No effect on glycated hemoglobin but reduction in C-reactive protein. Am. J. Clin. Nutr. 2008, 87, 114–125. [Google Scholar]
- Robertson, M.D. Metabolic cross talk between the colon and the periphery: Implications for insulin sensitivity. Proc. Nutr. Soc. 2007, 66, 351–361. [Google Scholar] [CrossRef]
- Wursch, P.; Pi-Sunyer, F.X. The role of viscous soluble fiber in the metabolic control of diabetes. A review with special emphasis on cereals rich in beta-glucan. Diabetes Care 1997, 1774–1780. [Google Scholar]
- Morita, T.; Kasaoka, S.; Hase, K.; Kiriyama, S. Psyllium shifts the fermentation site of high-amylose cornstarch toward the distal colon and increases fecal butyrate concentration in rats. J. Nutr. 1999, 129, 2081–2087. [Google Scholar]
- Giacco, R.; Parillo, M.; Rivellese, A.A.; Lasorella, G.; Giacco, A.; D’Episcopo, L.; Riccardi, G. Long-term dietary treatment with increased amounts of fiber-rich low-glycemic index natural foods improves blood glucose control and reduces the number of hypoglycemic events in type 1 diabetic patients. Diabetes Care 2000, 23, 1461–1466. [Google Scholar] [CrossRef]
- Robertson, M.D.; Currie, J.M.; Morgan, L.M.; Jewell, D.P.; Frayn, K.N. Prior short-term consumption of resistant starch enhances postprandial insulin sensitivity in healthy subjects. Diabetologia 2003, 46, 659–665. [Google Scholar]
- Robertson, M.D.; Bickerton, A.S.; Dennis, A.L.; Vidal, H.; Frayn, K.N. Insulin-sensitizing effects of dietary resistant starch and effects on skeletal muscle and adipose tissue metabolism. Am. J. Clin. Nutr. 2005, 82, 559–567. [Google Scholar]
- Krebs, M.; Brehm, A.; Krssak, M.; Anderwald, C.; Bernroider, E.; Nowotny, P.; Roth, E.; Chandramouli, V.; Landau, B.R.; Waldhausl, W.; Roden, M. Direct and indirect effects of amino acids on hepatic glucose metabolism in humans. Diabetologia 2003, 46, 917–925. [Google Scholar] [CrossRef]
- Chevalier, S.; Burgess, S.C.; Malloy, C.R.; Gougeon, R.; Marliss, E.B.; Morais, J.A. The greater contribution of gluconeogenesis to glucose production in obesity is related to increased whole-body protein catabolism. Diabetes 2006, 55, 675–681. [Google Scholar] [CrossRef]
- Veldhorst, M.A.; Westerterp-Plantenga, M.S.; Westerterp, K.R. Gluconeogenesis and energy expenditure after a high-protein, carbohydrate-free diet. Am. J. Clin. Nutr. 2009, 90, 519–526. [Google Scholar] [CrossRef]
- Schutz, Y. Protein turnover, ureagenesis and gluconeogenesis. Int. J. Vitam. Nutr Res. 2011, 81, 101–107. [Google Scholar] [CrossRef]
- Walker, A.W.; Ince, J.; Duncan, S.H.; Webster, L.M.; Holtrop, G.; Ze, X.; Brown, D.; Stares, M.D.; Scott, P.; Bergerat, A.; et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME. J. 2011, 5, 220–230. [Google Scholar] [CrossRef]
- McIntosh, F.M.; Maison, N.; Holtrop, G.; Young, P.; Stevens, V.J.; Ince, J.; Johnstone, A.M.; Lobley, G.E.; Flint, H.J.; Louis, P. Phylogenetic distribution of genes encoding beta-glucuronidase activity in human colonic bacteria and the impact of diet on faecal glycosidase activities. Environ. Microbiol. 2012, 14, 1876–1887. [Google Scholar] [CrossRef]
- O’Sullivan, K.R.; Cho, S.S. Fibre recommendations throughout the world. Int. J. Food Sci. Nutr. 1998, 49, S13–S21. [Google Scholar]
- Johnstone, A.M.; Lobley, G.E.; Horgan, G.W.; Bremner, D.M.; Fyfe, C.L.; Morrice, P.C.; Duthie, G.G. Effects of a high-protein, low-carbohydrate v. high-protein, moderate-carbohydrate weight-loss diet on antioxidant status, endothelial markers and plasma indices of the cardiometabolic profile. Br. J. Nutr. 2011, 106, 282–291. [Google Scholar] [CrossRef]
- Calder, A.G.; Smith, A. Stable isotope ratio analysis of leucine and ketoisocaproic acid in blood plasma by gas chromatography/mass spectrometry. Use of tertiary butyldimethylsilyl derivatives. Rapid Commum. Mass Spectrom. 1988, 2, 14–16. [Google Scholar] [CrossRef]
- Calder, A.G.; Garden, K.E.; Anderson, S.E.; Lobley, G.E. Quantitation of blood and plasma amino acids using isotope dilution electron impact gas chromatography/mass spectrometry with U-(13)C amino acids as internal standards. Rapid Commum. Mass Spectrom. 1999, 13, 2080–2083. [Google Scholar]
- Wilson, F.A.; van den Borne, J.J.; Calder, A.G.; O'Kennedy, N.; Holtrop, G.; Rees, W.D.; Lobley, G.E. Tissue methionine cycle activity and homocysteine metabolism in female rats: Impact of dietary methionine and folate plus choline. Am. J. Physiol Endocrinol. Metab. 2009, 296, E702–E713. [Google Scholar] [CrossRef]
- Patterson, B.W.; Carraro, F.; Wolfe, R.R. Measurement of 15N enrichment in multiple amino acids and urea in a single analysis by gas chromatography/mass spectrometry. Biol. Mass Spectrom. 1993, 22, 518–523. [Google Scholar] [CrossRef]
- Levy, J.C.; Matthews, D.R.; Hermans, M.P. Correct homeostasis model assessment (HOMA) evaluation uses the computer program. Diabetes Care 1998, 21, 2191–2192. [Google Scholar] [CrossRef]
- The Oxford Centre for Diabetes. Endocrinology & Metabolism, Diabetes Trial Unit. HOMA Calculator. Available online: http://www.dtu.ox.ac.uk/homacalculator/index.php (accessed on 16 July 2012).
- Matsuda, M.; Defronzo, R.A. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 1999, 22, 1462–1470. [Google Scholar] [CrossRef]
- Wolever, T.M.; Jenkins, D.J.; Jenkins, A.L.; Josse, R.G. The glycemic index: Methodology and clinical implications. Am. J. Clin. Nutr. 1991, 54, 846–854. [Google Scholar]
- Basu, R.; Di Camillo, B.; Toffolo, G.; Basu, A.; Shah, P.; Vella, A.; Rizza, R.; Cobelli, C. Use of a novel triple-tracer approach to assess postprandial glucose metabolism. Am. J. Physiol Endocrinol. Metab. 2003, 284, E55–E69. [Google Scholar]
- Bluck, L.J.; Clapperton, A.T.; Coward, W.A. 13C- and 2H-labelled glucose compared for minimal model estimates of glucose metabolism in man. Clin. Sci. (Lond.) 2005, 109, 513–521. [Google Scholar] [CrossRef]
- Vicini, P.; Zachwieja, J.J.; Yarasheski, K.E.; Bier, D.M.; Caumo, A.; Cobelli, C. Glucose production during an IVGTT by deconvolution: validation with the tracer-to-tracee clamp technique. Am. J. Physiol. 1999, 276, E285–E294. [Google Scholar]
- Shipley, R.A.; Clark, R.E. Tracer Methods for in Vivo Kinetics. Theory and Applications; Academic Press: London, UK, 1972. [Google Scholar]
- Ferrannini, E.; Smith, J.D.; Cobelli, C.; Toffolo, G.; Pilo, A.; Defronzo, R.A. Effect of insulin on the distribution and disposition of glucose in man. J. Clin. Invest. 1985, 76, 357–364. [Google Scholar] [CrossRef]
- Toffolo, G.; Basu, R.; Dalla, M.C.; Rizza, R.; Cobelli, C. Assessment of postprandial glucose metabolism: Conventional dual- vs. triple-tracer method. Am. J. Physiol Endocrinol. Metab. 2006, 291, E800–E806. [Google Scholar] [CrossRef]
- Vicini, P.; Caumo, A.; Cobelli, C. The hot IVGTT two-compartment minimal model: Indexes of glucose effectiveness and insulin sensitivity. Am. J. Physiol. 1997, 273, E1024–E1032. [Google Scholar]
- The R Project for Statistical Computing. Available online: http://www.r-project.org/ (accessed on 6 November 2012).
- Statistical Libraries. GLIM4 Macro Libraries. Available online: http://www.commanster.eu/rcode.html (accessed on 6 November 2012).
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Felig, P. Amino acid metabolism in man. Annu. Rev. Biochem. 1975, 44, 933–955. [Google Scholar] [CrossRef]
- Basu, R.; Schwenk, W.F.; Rizza, R.A. Both fasting glucose production and disappearance are abnormal in people with “mild” and “severe” type 2 diabetes. Am. J. Physiol Endocrinol. Metab. 2004, 28, E55–E62. [Google Scholar] [CrossRef]
- Gastaldelli, A.; Toschi, E.; Pettiti, M.; Frascerra, S.; Quinones-Galvan, A.; Sironi, A.M.; Natali, A.; Ferrannini, E. Effect of physiological hyperinsulinemia on gluconeogenesis in nondiabetic subjects and in type 2 diabetic patients. Diabetes 2001, 50, 1807–1812. [Google Scholar] [CrossRef]
- Boden, G.; Cheung, P.; Stein, T.P.; Kresge, K.; Mozzoli, M. FFA cause hepatic insulin resistance by inhibiting insulin suppression of glycogenolysis. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E12–E19. [Google Scholar]
- Robertson, M.D.; Wright, J.W.; Loizon, E.; Debard, C.; Vidal, H.; Shojaee-Moradie, F.; Russell-Jones, D.; Umpleby, A.M. Insulin-sensitizing effects on muscle and adipose tissue after dietary fiber intake in men and women with metabolic syndrome. J. Clin. Endocrinol. Metab. 2012, 97, 3326–3332. [Google Scholar] [CrossRef]
- Weickert, M.O.; Roden, M.; Isken, F.; Hoffmann, D.; Nowotny, P.; Osterhoff, M.; Blaut, M.; Alpert, C.; Gogebakan, O.; Bumke-Vogt, C.; et al. Effects of supplemented isoenergetic diets differing in cereal fiber and protein content on insulin sensitivity in overweight humans. Am. J. Clin. Nutr. 2011, 94, 459–471. [Google Scholar] [CrossRef]
- Viljanen, A.P.; Iozzo, P.; Borra, R.; Kankaanpaa, M.; Karmi, A.; Lautamaki, R.; Jarvisalo, M.; Parkkola, R.; Ronnemaa, T.; Guiducci, L.; et al. Effect of weight loss on liver free fatty acid uptake and hepatic insulin resistance. J. Clin. Endocrinol. Metab. 2009, 94, 50–55. [Google Scholar]
- Wahren, J.; Efendic, S.; Luft, R.; Hagenfeldt, L.; Bjorkman, O.; Felig, P. Influence of somatostatin on splanchnic glucose metabolism in postabsorptive and 60-hour fasted humans. J. Clin. Invest. 1977, 59, 299–307. [Google Scholar] [CrossRef]
- Laffel, L. Ketone bodies: A review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab Res. Rev. 1999, 15, 412–426. [Google Scholar] [CrossRef]
- Hollenbeck, C.B.; Coulston, A.M.; Reaven, G.M. To what extent does increased dietary fiber improve glucose and lipid metabolism in patients with noninsulin-dependent diabetes mellitus (NIDDM)? Am. J. Clin. Nutr. 1986, 43, 16–24. [Google Scholar]
- Gatenby, S.J.; Ellis, P.R.; Morgan, L.M.; Judd, P.A. Effect of partially depolymerized guar gum on acute metabolic variables in patients with non-insulin-dependent diabetes. Diabet. Med. 1996, 13, 358–364. [Google Scholar]
- Nuttall, F.Q.; Mooradian, A.D.; Gannon, M.C.; Billington, C.; Krezowski, P. Effect of protein ingestion on the glucose and insulin response to a standardized oral glucose load. Diabetes Care 1984, 7, 465–470. [Google Scholar]
- Kelley, D.E.; Wing, R.; Buonocore, C.; Sturis, J.; Polonsky, K.; Fitzsimmons, M. Relative effects of calorie restriction and weight loss in noninsulin-dependent diabetes mellitus. J. Clin. Endocrinol. Metab. 1993, 77, 1287–1293. [Google Scholar] [CrossRef]
- McLaughlin, T.; Carter, S.; Lamendola, C.; Abbasi, F.; Yee, G.; Schaaf, P.; Basina, M.; Reaven, G. Effects of moderate variations in macronutrient composition on weight loss and reduction in cardiovascular disease risk in obese, insulin-resistant adults. Am. J. Clin. Nutr. 2006, 8, 813–821. [Google Scholar]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Faber, O.K.; Hagen, C.; Binder, C.; Markussen, J.; Naithani, V.K.; Blix, P.M.; Kuzuya, H.; Horwitz, D.L.; Rubenstein, A.H.; Rossing, N. Kinetics of human connecting peptide in normal and diabetic subjects. J. Clin. Invest. 1978, 62, 197–203. [Google Scholar] [CrossRef]
- McLaughlin, T.; Schweitzer, P.; Carter, S.; Yen, C.G.; Lamendola, C.; Abbasi, F.; Reaven, G. Persistence of improvement in insulin sensitivity following a dietary weight loss programme. Diabetes Obes. Metab. 2008, 10, 1186–1194. [Google Scholar]
- Niskanen, L.; Uusitupa, M.; Sarlund, H.; Siitonen, O.; Paljarvi, L.; Laakso, M. The effects of weight loss on insulin sensitivity, skeletal muscle composition and capillary density in obese non-diabetic subjects. Int. J. Obes. Relat. Metab. Disord. 1996, 20, 154–160. [Google Scholar]
- Wolever, T.M.; Mehling, C.; Chiasson, J.L.; Josse, R.G.; Leiter, L.A.; Maheux, P.; Rabasa-Lhoret, R.; Rodger, N.W.; Ryan, E.A. Low glycaemic index diet and disposition index in type 2 diabetes (the Canadian trial of carbohydrates in diabetes): A randomised controlled trial. Diabetologia 2008, 51, 1607–1615. [Google Scholar] [CrossRef]
- Anderson, G.H.; Cho, C.E.; Akhavan, T.; Mollard, R.C.; Luhovyy, B.L.; Finocchiaro, E.T. Relation between estimates of cornstarch digestibility by the Englyst in vitro method and glycemic response, subjective appetite, and short-term food intake in young men. Am. J. Clin. Nutr. 2010, 9, 932–939. [Google Scholar]
- Robertson, M.D. Dietary-resistant starch and glucose metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 362–367. [Google Scholar] [CrossRef]
- Dixon, L.B.; Subar, A.F.; Peters, U.; Weissfeld, J.L.; Bresalier, R.S.; Risch, A.; Schatzkin, A.; Hayes, R.B. Adherence to the USDA Food Guide, DASH Eating Plan, and Mediterranean dietary pattern reduces risk of colorectal adenoma. J. Nutr. 2007, 137, 2443–2450. [Google Scholar]
- Johnston, K.L.; Thomas, E.L.; Bell, J.D.; Frost, G.S.; Robertson, M.D. Resistant starch improves insulin sensitivity in metabolic syndrome. Diabet. Med. 2010, 27, 391–397. [Google Scholar]
- Bodinham, C.L.; Frost, G.S.; Robertson, M.D. Acute ingestion of resistant starch reduces food intake in healthy adults. Br. J. Nutr. 2010, 103, 917–922. [Google Scholar] [CrossRef]
- Bodinham, C.L.; Smith, L.; Wright, J.; Frost, G.S.; Robertson, M.D. Dietary fibre improves first-phase insulin secretion in overweight individuals. PLoS One 2012, 7, e40834. [Google Scholar]
- Maki, K.C.; Pelkman, C.L.; Finocchiaro, E.T.; Kelley, K.M.; Lawless, A.L.; Schild, A.L.; Rains, T.M. Resistant starch from high-amylose maize increases insulin sensitivity in overweight and obese men. J. Nutr. 2012, 142, 717–723. [Google Scholar] [CrossRef]
- Todesco, T.; Rao, A.V.; Bosello, O.; Jenkins, D.J. Propionate lowers blood glucose and alters lipid metabolism in healthy subjects. Am. J. Clin. Nutr. 1991, 54, 860–865. [Google Scholar]
- Clark, M.J.; Robien, K.; Slavin, J.L. Effect of prebiotics on biomarkers of colorectal cancer in humans: A systematic review. Nutr. Rev. 2012, 70, 436–443. [Google Scholar] [CrossRef]
- Sims, E.A. Are there persons who are obese, but metabolically healthy? Metabolism 2001, 50, 1499–1504. [Google Scholar] [CrossRef]
- Brochu, M.; Tchernof, A.; Dionne, I.J.; Sites, C.K.; Eltabbakh, G.H.; Sims, E.A.; Poehlman, E.T. What are the physical characteristics associated with a normal metabolic profile despite a high level of obesity in postmenopausal women? J. Clin. Endocrinol. Metab. 2001, 86, 1020–1025. [Google Scholar] [CrossRef]
- Karelis, A.D.; Faraj, M.; Bastard, J.P.; St Pierre, D.H.; Brochu, M.; Prud’homme, D.; Rabasa-Lhoret, R. The metabolically healthy but obese individual presents a favorable inflammation profile. J. Clin. Endocrinol. Metab. 2005, 90, 4145–4150. [Google Scholar] [CrossRef]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev. 2001, 81, 1031–1064. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lobley, G.E.; Holtrop, G.; Bremner, D.M.; Calder, A.G.; Milne, E.; Johnstone, A.M. Impact of Short Term Consumption of Diets High in Either Non-Starch Polysaccharides or Resistant Starch in Comparison with Moderate Weight Loss on Indices of Insulin Sensitivity in Subjects with Metabolic Syndrome. Nutrients 2013, 5, 2144-2172. https://doi.org/10.3390/nu5062144
Lobley GE, Holtrop G, Bremner DM, Calder AG, Milne E, Johnstone AM. Impact of Short Term Consumption of Diets High in Either Non-Starch Polysaccharides or Resistant Starch in Comparison with Moderate Weight Loss on Indices of Insulin Sensitivity in Subjects with Metabolic Syndrome. Nutrients. 2013; 5(6):2144-2172. https://doi.org/10.3390/nu5062144
Chicago/Turabian StyleLobley, Gerald E., Grietje Holtrop, David M. Bremner, A. Graham Calder, Eric Milne, and Alexandra M. Johnstone. 2013. "Impact of Short Term Consumption of Diets High in Either Non-Starch Polysaccharides or Resistant Starch in Comparison with Moderate Weight Loss on Indices of Insulin Sensitivity in Subjects with Metabolic Syndrome" Nutrients 5, no. 6: 2144-2172. https://doi.org/10.3390/nu5062144
APA StyleLobley, G. E., Holtrop, G., Bremner, D. M., Calder, A. G., Milne, E., & Johnstone, A. M. (2013). Impact of Short Term Consumption of Diets High in Either Non-Starch Polysaccharides or Resistant Starch in Comparison with Moderate Weight Loss on Indices of Insulin Sensitivity in Subjects with Metabolic Syndrome. Nutrients, 5(6), 2144-2172. https://doi.org/10.3390/nu5062144