1. Introduction
Vitamin C (ascorbic acid) is one of the best known and most studied dietary antioxidants. Its antioxidant credentials are well-established
in vitro, since it can readily be demonstrated to prevent oxidation of lipids, DNA and other biological molecules. However, it is also possible for vitamin C to act as a pro-oxidant, through its ability to reduce transition metal ions, thus promoting the Fenton reaction which, acting on peroxides, produces highly reactive hydroxyl radicals [
1]. The effect of vitamin C
in vivo has been investigated in human trials with DNA oxidation as the measured endpoint. They include single dose intervention and longer trials, typically of a few weeks with daily supplementation. The first single-dose study was carried out by Green
et al. [
2], who administered 35 mg/kg of vitamin C to six healthy volunteers after an overnight fast and found a protection against γ radiation-induced DNA breaks in white blood cells, using the comet assay (described below). Since then, more than 20 intervention studies with vitamin C have been carried out, and they have been critically reviewed by Duarte and Lunec [
3] who find that most of the studies show either a decrease in DNA oxidation or no effect, while there are some that show an increase in DNA lesions. Møller and Loft [
4] also describe conflicting results with vitamin C, concluding that more studies with better designs to avoid bias should be made in order to understand the role of vitamin C in protecting against DNA oxidation. In a subsequent supplementation trial, Møller
et al. [
5] found that a slow release formulation of vitamin C had a more pronounced and sustained protective effect on the steady state level of DNA base oxidation than did a normal release formulation.
Herbert
et al. [
6] carried out a double-blind placebo-controlled trial giving different doses of vitamin C (0, 80, 200 and 400 mg/day) to four groups of 40 healthy volunteers over a period of 15 weeks (plus a 10 week washout period). They showed that vitamin C did not affect the intracellular level of 8-oxodGuo measured by HPLC. (This challenged an earlier report that vitamin C caused oxidation of bases in DNA [
7]).
Sram
et al. [
8] recently reviewed human studies based on measurement of a variety of biomarkers of genetic damage—including some trials aimed at specific groups with occupational/environmental exposure to genotoxins. Generally either a protective effect or no effect was seen, depending—they suggest—on factors such as individual diet-derived vitamin C concentrations, levels of exposure to xenobiotics, and oxidative stress.
Bjelakovic
et al. [
9] published a meta-analysis of randomized trials of antioxidant supplements aimed at primary and secondary prevention of various diseases, with mortality as the endpoint. They concluded that antioxidant supplements have no beneficial effects on mortality, though in the case of vitamin C the number of individuals sampled is still relatively small and a definitive statement cannot be made.
Reviews of this subject conclude that we still need more studies to clarify the effect of vitamin C in humans [
1,
8,
9,
10]. We considered that it would be useful to investigate effects of vitamin C on genetic stability in cell culture, and so have examined, in the human tumor cell line HeLa, potential pro-oxidant, DNA-damaging effects of the vitamin; protection against DNA breakage caused by H
2O
2; and protection against base oxidation induced by photosensitizer plus visible light. In addition, we investigate the possibility that vitamin C might influence the capacity of cells for DNA repair.
We applied the comet assay, as used in many human biomonitoring studies, to the measurement of DNA damage, both strand breaks (SBs) and oxidized bases (employing the enzyme formamidopyrimidine DNA glycosylase (FPG) which converts 8-oxoGua and breakdown products of damaged purines to SBs). We used two approaches to measure repair of DNA damage [
11]. If cells are subjected to damage (SBs or base oxidation), and then incubated to allow cellular repair, the residual lesions can be measured at intervals to show the kinetics of damage removal. Alternatively, in an
in vitro assay, a cell extract is incubated with substrate DNA containing specific damage. The ability of the extract to introduce DNA breaks in the substrate cells reflects the activity of the enzymes responsible for the initial steps of repair,
i.e., removing the lesion.
2. Materials and Methods
2.1. Cell Culture
HeLa cells (derived from human cervical cancer) were grown in DMEM supplemented with 10% fetal bovine serum and antibiotics (100 U/mL penicillin and 100 μg/mL streptomycin). According to the formulation for DMEM it does not contain vitamin C. Vitamin C was not detectable in the batch of serum used. Cells were maintained as monolayer cultures at 37 °C in a humidified atmosphere with 5% CO2 and routinely passaged by trypsinization when nearly confluent.
2.2. Cell Treatment for DNA Damage and Protection
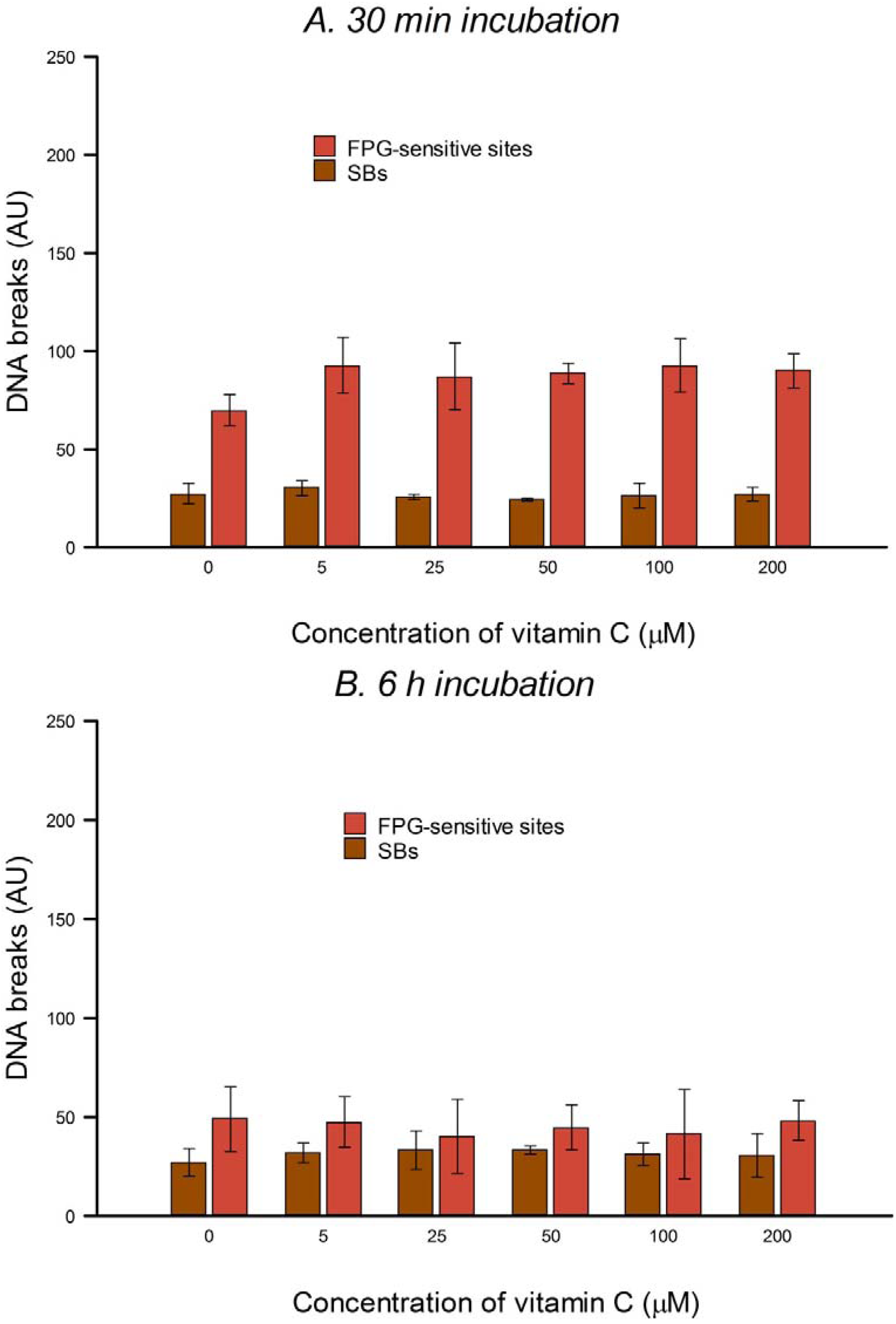
HeLa cells in culture medium were incubated with 0, 5, 25, 50, 100 or 200 µM vitamin C (ascorbic acid) for 30 min or 6 h (3 h incubation, followed by a wash with PBS and a further 3 h incubation with vitamin C at the same concentration) at 37 °C in the dark. The two consecutive 3 h treatments were designed to allow for the possible instability of vitamin C in solution. Vitamin C was dissolved in PBS in the dark just before use in each experiment. After treatment the comet assay was performed as described below. To check for DNA protection, after vitamin C treatment, cells were washed with PBS and then treated on ice with 25 µM H2O2 for 5 min to induce SBs, or with 1 µM of the photosensitizer Ro (Ro-19-8022, from F. Hoffmann-La Roche) plus 1.5 min visible light (500 W tungsten halogen lamp, at 33 cm on ice) to induce oxidized purines, mostly 8-oxoGua. After treatment the comet assay was performed (see below). Three independent experiments were carried out.
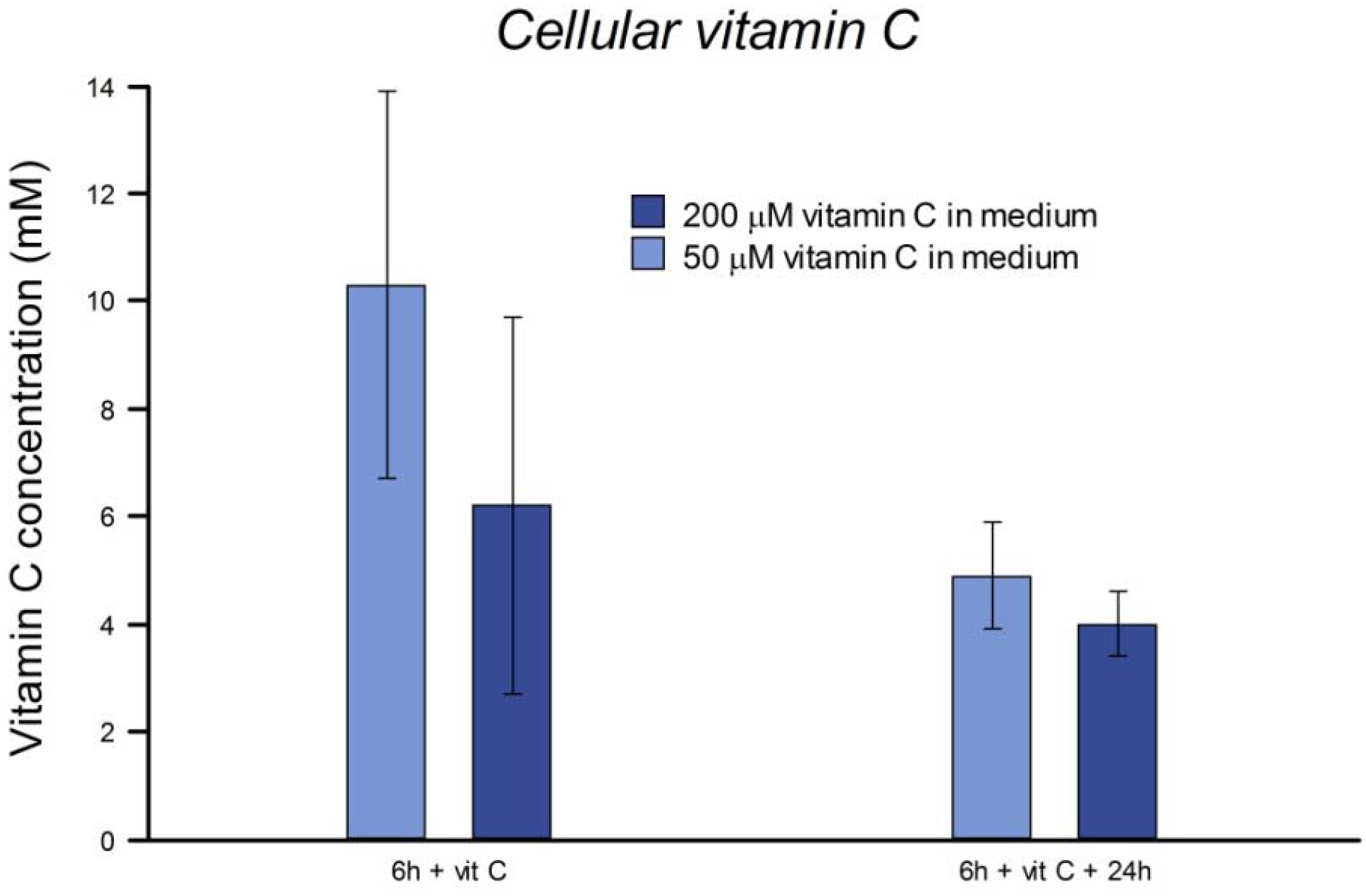
2.3. Measuring Uptake of Vitamin C
Near-confluent cultures of HeLa cells in 60 mm dishes were incubated for 6 h (3 h + wash + 3 h, as above) with vitamin C at 50 or 200 µM. At the end of this incubation, or after a further 24 h incubation in fresh medium without vitamin C, cells were washed with PBS and scraped with a silicone rubber scraper into suspension in PBS. A cell count was carried out. The suspension was centrifuged (400× g, 5 min) and the pellet resuspended in 250 µL of PBS to which an equal volume of 10% metaphosphoric acid was added before storing at −20 °C.
The frozen, acidified samples were thawed, and centrifuged (3500× g at 4 °C for 10 min). A volume of 100 μL clear supernatant was mixed with 400 μL of the mobile phase (2% acetonitrile in 2.5 mM NaH2PO4, 2.5 mM dodecyltrimethyl ammonium chloride and 1.25 mM Na2EDTA in Milli-Q water) for direct determination of vitamin C by high-performance liquid chromatography (HPLC). For separation of vitamin C from interfering sample constituents, a Chromolith Performance RP18-e, 4.6 mm × 100 mm column was used, with a Chromolith Performance RP18-e, 4.6 mm × 10 mm guard column (Phenomenex, Torrance, USA). The injection volume was 5 μL and the flow rate was 6.0 mL/min. A variable wavelength ultraviolet (UV) detector was used at 264 nm.
2.4. Treatment of Cells for Cellular Repair Assay
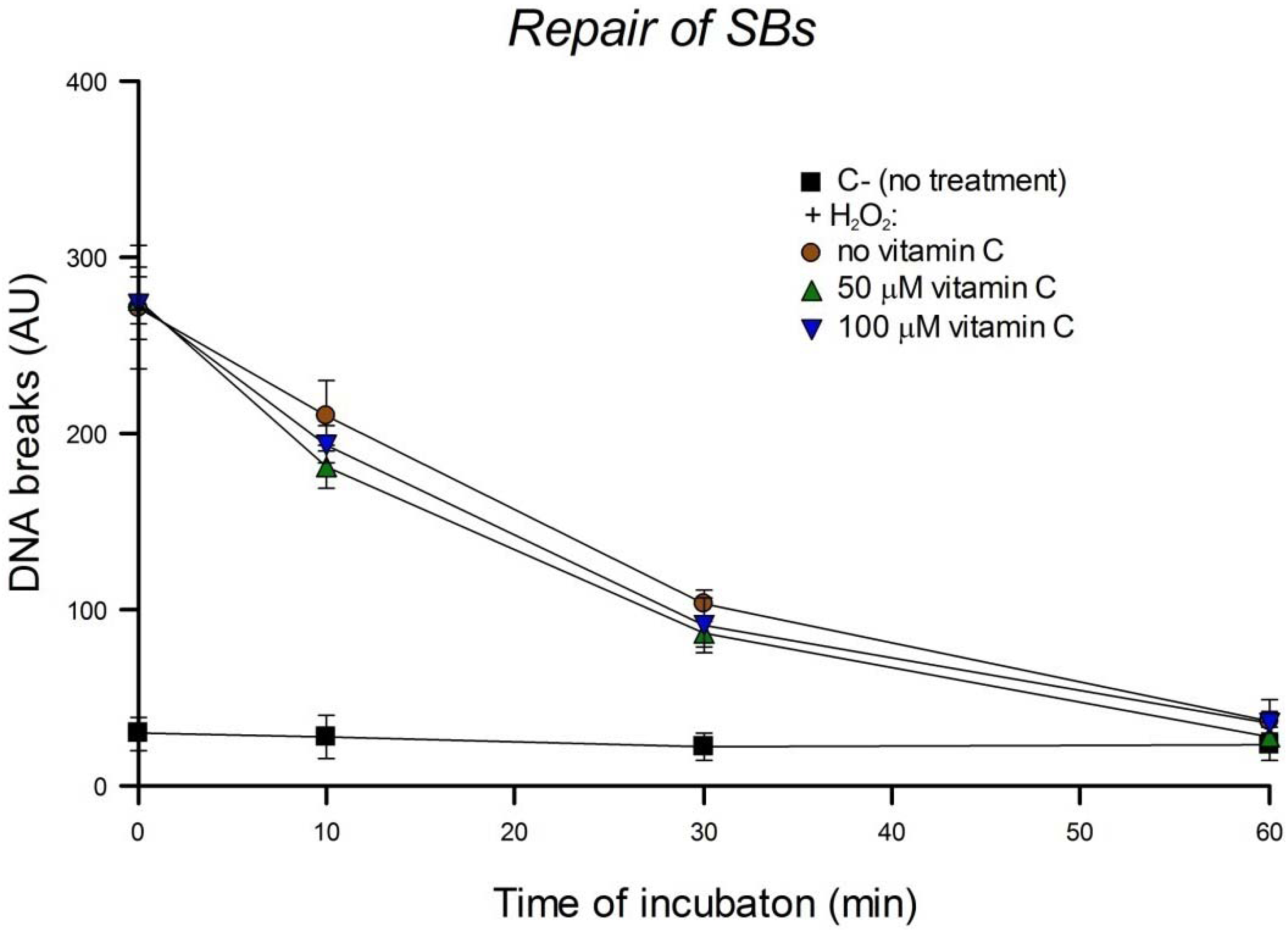
Repair of DNA damage can be studied by treating cells with a DNA-damaging agent, incubating, and measuring the damage remaining at intervals. Using this cellular repair assay we investigated the kinetics of DNA SB rejoining and base excision repair (BER). To check the influence of vitamin C on the repair of SBs, HeLa cells were pre-incubated with 0, 50 or 100 µM vitamin C overnight. Then cells were washed with PBS, treated with 0 or 100 µM H2O2 for 5 min on ice, and incubated for 10, 30 or 60 min in culture medium. To check the influence of vitamin C on the repair of oxidized bases, pre-incubation overnight with 0, 50 or 100 µM vitamin C was followed by a wash with PBS, treatment with 0 or 1 µM Ro plus 5 min of light, and incubation for 1, 2, 4, 6, 8, or 24 h in culture medium. In another experimental design, HeLa cells in culture medium were pre-incubated with a higher concentration (200 µM) of vitamin C but for only 30 min. Then cells were washed with PBS, treated with 1 µM Ro plus 2.5 min of light, and incubated for 3 h in culture medium including 0 or 200 µM vitamin C. They were then washed with PBS and incubated again in culture medium with the same concentration of vitamin C for a further 3 h. The comet assay was performed after each time of incubation. The concentration of vitamin C used in these experiments was not genotoxic. Three independent experiments were performed.
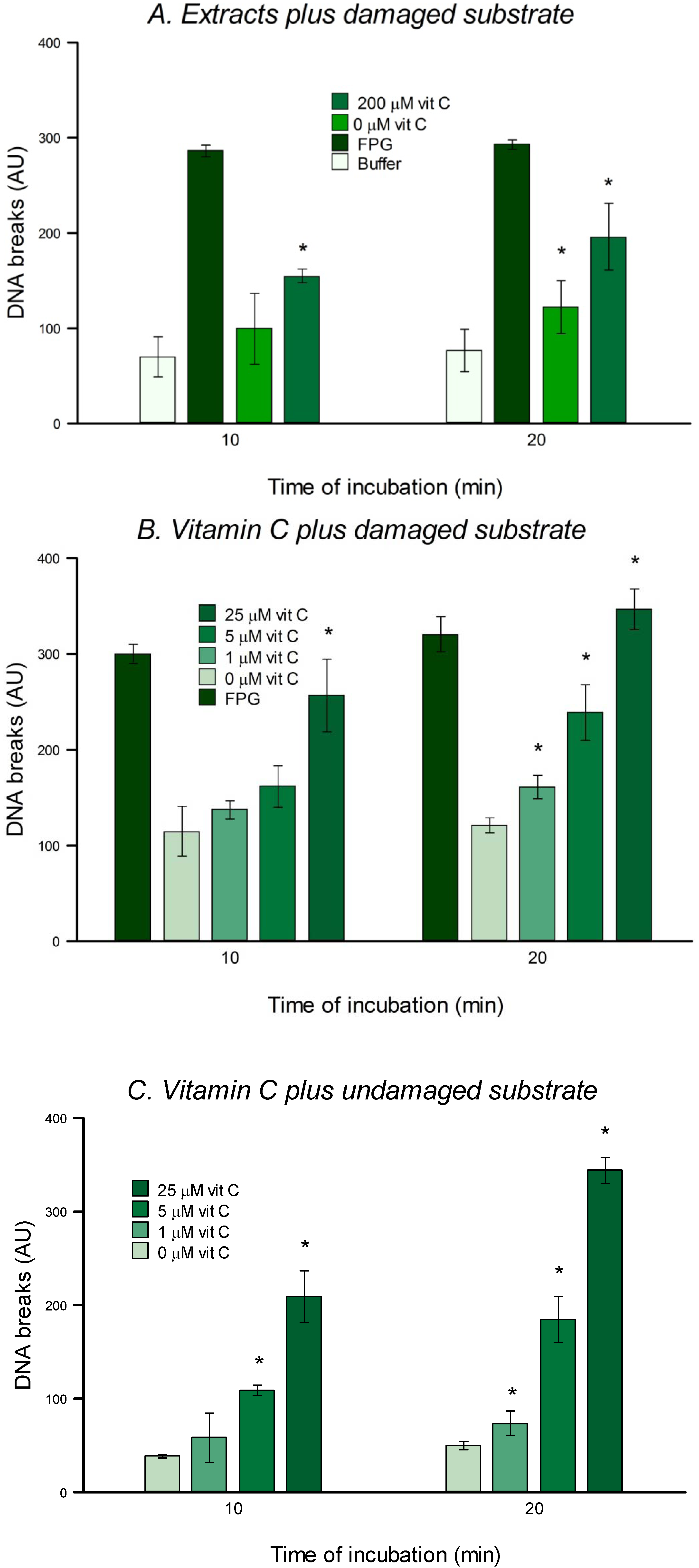
2.6. In Vitro Assay for DNA Repair
The in vitro repair assay is used to check the ability of cell extracts to carry out the first steps of BER. Gel-embedded nucleoids from HeLa cells carrying oxidized bases (substrate) were incubated with extracts from HeLa cells pre-incubated with/without vitamin C and with different concentrations of vitamin C directly.
2.6.1. Preparation of Substrate Cells
HeLa cells were grown to 80% confluence, washed with PBS and treated with 1 µM Ro plus 4 min of light. Then they were washed with cold PBS, detached by trypsinization, centrifuged for 5 min at 800× g at 4 °C and resuspended in freezing medium (DMEM supplemented with 20% fetal bovine serum, and 10% DMSO) at a concentration of 1 × 106 cells per mL. Aliquots of 0.7 mL were frozen slowly and stored at −80 °C.
2.6.2. Preparation of Cell Extracts
Extracts were prepared from HeLa cells incubated with 0, 50, 100 or 200 µM vitamin C for 6 h (3 h + wash + 3 h). They were washed with cold PBS, trypsinized, centrifuged at 800× g for 5 min at 4 °C and resuspended in cold PBS at a concentration of 1.25 × 106 cells per mL. Then the cell suspension was split into aliquots of 1 mL and centrifuged at 14,000× g for 5 min at 4 °C. The supernatant of each aliquot was completely discarded and the dry pellets were frozen in liquid nitrogen and stored at −80 °C. After thawing the pellets were resuspended in 32 µL of extraction buffer A (45 mM HEPES, 0.4 M KCl, 1 mM EDTA, 0.1 mM dithiothreitol and 10% glycerol, pH 7.8) containing 0.25% of Triton X-100, mixed using vortex at high speed for 5–10 s, incubated for 5 min on ice and centrifuged at 14,000× g for 5 min at 4 °C. Then 27 µL of the supernatant was mixed with 110 µL of cold reaction buffer F.
2.6.3. The Reaction
The substrate cells were thawed, diluted in cold PBS, centrifuged at 800× g for 5 min at 4 °C and resuspended in PBS at a concentration of 1 × 106 cells per mL. As described in section 2.4 gels were made, put in lysis for 1 h and washed 3 times (5 min each time) with buffer F. Then 30 µL of extract (experimental treatment), FPG (positive control), or buffer F (negative control) were placed on each gel (two gels per condition) and covered with a cover slip. Slides were placed in a moist box and incubated for 10 and 20 min. Afterwards the rest of the comet assay was performed as described above.
2.6.4. Treatment of Nucleoids with Vitamin C
In order to check the direct effect of vitamin C on DNA, non-damaged HeLa cell nucleoids, or substrate nucleoids prepared as described in
Section 2.6.1, were treated for 10 or 20 min with vitamin C dissolved in buffer A at 0, 1, 5 or 25 µM and diluted with buffer F, as described in
Section 2.6.2. The reaction was carried out as described in
Section 2.6.3. The final concentrations of vitamin C were therefore 0, 0.25, 1.25, and 6.25 μM.
2.7. Statistical Analysis
The statistical analysis was performed by using the software SPSS 11.0. Data are presented with descriptive analysis (mean ± SD for 3 independent experiments). The comparisons between total comet scores of the different groups were performed by the non-parametric Kruskal-Wallis test followed by Mann-Whitney U test when the first one showed the presence of differences. Probability p ≤ 0.05 was accepted as the level of significance.
4. Discussion
In vitro, vitamin C is capable of acting as a pro-oxidant, particularly in combination with metal ions [
14], and there are concerns that it might lead to excessive DNA oxidation in humans. There are reports of increases in oxidized bases in the DNA of white blood cells after supplementation of normal subjects with vitamin C [
7] or with vitamin C plus ferrous sulfate [
15]. However, these studies were carried out using GC-MS to measure oxidized bases—a technique now recognized as being particularly prone to artifact and therefore unsuitable for determining low level base oxidation [
16].
The saturation plasma level for vitamin C in humans is around 80 μM, and this concentration is approached with daily intake of 200 mg/day [
17]—an intake that can feasibly be attained by a recommended intake of several portions of fruits and vegetables each day. The justification for exceeding in our experiments the maximum vitamin C concentration found in plasma is that vitamin C is reportedly not stable in cell culture medium; 40% depletion was detected after 2 h of incubation [
18], and only about 30% remained at 6 h.
In vivo, in subjects supplemented with 200 mg of vitamin C per day, active accumulation by lymphocytes results in intracellular levels saturating at 3.5 mM [
17]. Welch
et al. [
19] reported a linear accumulation of vitamin C up to about 0.5 mM in 4 h in human fibroblasts incubated in 50 μM vitamin C. We found that the concentration of vitamin C inside HeLa cells after culture in medium with the vitamin was in the mM range, and was still high after 24 h. Although they have undergone many changes in their several decades of continuous culture from their origin in tumor tissue, HeLa cells evidently retain the active transport mechanisms responsible in normal cells for uptake of vitamin C [
19], and so we regard them as suitable for an investigation of intracellular effects of vitamin C.
In the present study, in spite of the high intracellular concentration of vitamin C, there was no increase in SBs or FPG-sensitive sites. Others have reported no increase in SBs and/or oxidized bases in various cell types after incubation with vitamin C—in human HepG2 liver-derived cells with 10 μM vitamin C [
20], in primary human fibroblasts incubated with 100 μM vitamin C [
21], and most recently in HL-60 cells (human leukemic cell line) incubated with concentrations of 50–250 μM vitamin C [
22,
23].
In our experiments, preincubation with up to 200 μM vitamin C had no significant effect on the level of H
2O
2-induced breaks or 8-oxoGua induced by treatment with Ro plus light. There are mixed reports on the effect of pre-incubation with vitamin C on the DNA damage inflicted by genotoxic agents. Vitamin C pre-incubation (30 min, 10 or 50 μM) decreased the SBs caused by streptozotocin in lymphocytes or HeLa cells [
24], and protected against the effect of NiCl
2 in lymphocytes [
25]. Duarte and Jones [
21] preincubated primary human fibroblasts for 12 h with 20, 100 or 500 μM vitamin C, and—also using the comet assay—found an increase in H
2O
2-induced damage. Carbofuran-induced damage to human lymphocyte DNA was decreased by preincubation with 30 μM vitamin C [
26]. Similar protection was afforded by 10–50 μM vitamin C pretreatment against damage induced by 2-hydroxyethyl methacrylate and urethane dimethacrylate (used in dental treatment) [
27,
28].
When vitamin C is present simultaneously with a genotoxic agent (rather than during a preincubation), potentiation of the damaging effect of the latter may be seen—even when the effect of preincubation is a protective one. Such a dual effect was reported by Wozniak and Blasiak [
25] for NiCl
2. Vanadyl sulfate led to more DNA breaks in HeLa cells and lymphocytes when they were co-incubated with vitamin C for 1 h [
29], perhaps as a result of a metal ion-vitamin C interaction. Both DNA breaks and oxidized purines were induced when HL-60 cells were incubated with vitamin C and copper (II) sulfate, but no such damage was caused by vitamin C alone [
23]. Vitamin C was unable to protect human submandibular gland and oral epithelial cells against DNA damage induced by bracken fern, and in fact showed a synergistic effect on DNA breakage [
30]. In contrast, co-incubation of HepG2 cells with both vitamin C and
N-nitrosamines resulted in fewer DNA breaks/oxidized bases [
20]—perhaps an effect of vitamin C mediated through xenobiotic metabolizing enzymes.
To summarize our results relating to DNA damage, in HeLa cells—which clearly are able to take up and accumulate vitamin C—we find no sign of either pro- or anti-oxidant effects.
There are some human intervention studies that suggest a possible regulatory role for vitamin C in the repair of oxidized DNA—a topic highlighted by Sram
et al. in their recent review [
8] as deserving further attention. Cooke
et al. [
31] measured 8-oxodGuo in lymphocyte DNA, serum and urine from 30 healthy volunteers involved in a 6 week placebo and 6 week vitamin C (500 mg/day) supplementation study (plus a washout period). They found a decrease in the levels of 8-oxodGuo in DNA and an increase in urine and serum. In addition to reservations about the less than ideal study design and the likely errors in the measurement of 8-oxodGuo, the interpretation of the results is questionable, since the presence of 8-oxodGuo in serum or urine does not directly reflect OGG activity; the base, not the nucleoside, is excised during BER. Tarng
et al. [
32] supplemented chronic hemodialysis patients with 300 mg of vitamin C, and found a significant upregulation of
hOGG1 mRNA at 24 h after vitamin C administration. Astley
et al. [
33] took lymphocytes from human volunteers after 3 weeks of supplementation with the modest dose of 60 mg vitamin C per 2 days. There was no significant effect on plasma vitamin C, and no change in repair capacity (detected as repair synthesis on an oxidatively damaged plasmid template).
Supplementation for 3 weeks with green kiwifruits led to a significant increase in plasma vitamin C, decreases in endogenous base oxidation and H
2O
2-induced damage
ex vivo, and an enhancement of lymphocyte BER on a substrate containing 8-oxoGua [
34]; however, golden kiwifruits modestly increased plasma vitamin C and showed protection against DNA oxidation but no effect on DNA repair [
35]. Guarnieri
et al. [
36] supplemented volunteers with 0.5 g/day vitamin C in normal or slow release form (together with vitamin E); only the slow release form stimulated BER capacity in lymphocytes.
Reports in the literature on effects of vitamin C on DNA repair in cells in culture are sparse. Konopacka
et al. [
37] irradiated mouse lymphocytes with 2 Gy of γ-rays, and then incubated them with or without vitamin C, vitamin E and β-carotene; rejoining of strand breaks was faster in the presence of antioxidants.
We report here that vitamin C has no effect on cellular repair of SBs or oxidized bases in HeLa cells. The
in vitro test for 8-oxoGua DNA glycosylase (OGG) activity could not be carried out, since it was evident that vitamin C itself can cause damage to nucleoid DNA (
Figure 6), presumably acting as a pro-oxidant (as mentioned in the Introduction). This cleavage could be mediated via production of hydroxyl radicals in the presence of DNA-bound transition metal such as copper [
14].
The reason for native nuclear DNA being resistant to attack by vitamin C, while nucleoid DNA is susceptible, is not clear, though it may be that this apparently pro-oxidant effect of vitamin C is prevented by the high concentrations of the thiol-rich antioxidant glutathione present in the intact nucleus. Bergstrom
et al. [
22,
23] report the oxidation of dGuo
in vitro by vitamin C. To summarize, we have found no evidence in HeLa cells for pro-oxidant effects of vitamin C at extracellular concentrations up to 200 μM, but also no evidence for antioxidant protection. Nor did we see any effect on the repair of SBs or oxidized bases. Vitamin C at low concentration is, however, capable of damaging nucleoid DNA. The same strategy was used in our laboratory to study the antioxidant/pro-oxidant effects of β-cryptoxanthin [
13]. Our results showed that this carotenoid does not cause DNA damage but, in contrast to vitamin C, it protects HeLa cells from damage induced by H
2O
2 or photosensitizer plus light. In addition, it enhances DNA repair, as measured with the same assays as were used in the present report.