1. Introduction
Duck Tembusu virus (DTMUV), family
Flaviviridae, genus
Flavivirus, is a newly emerging pathogen related to significant egg drop syndrome in laying ducks since 2010 [
1]. In addition to egg drop syndrome, acute anorexia, retarded growth and neurological dysfunction are also presented by DTMUV-infected young ducks. The disease follows a very acute course with a morbidity of 90% to 100% and a mortality of 10% to 30% [
2].
As a member of the family,
Flaviviridae, DTMUV is a single-stranded and positive-sense RNA virus, a length of 10,990 nucleotides. The whole open reading frame (ORF) encodes three structural proteins: the envelope (E); the core (C); and membrane (prM) proteins. The ORF also encodes seven non-structural (NS) proteins (NS1-NS2A-NS2B-NS3-NS4A-NS4B-NS5). Among these proteins, the E protein is the principal antigenic determinant that serves important functions in receptor binding and membrane fusion [
3]. Therefore, the E protein is a key target for vaccine and drug development. The emergence of DTMUV infection has seriously threatened the progression of the waterfowl industry in China. However, licensed DTMUV vaccine is currently unavailable. Hence, the development of a vaccine for ducks against DTMUV is critical for disease control.
Duck enteritis virus (DEV) belongs to anatid herpesvirus 1, and ducks, geese and swans are susceptible to it. The entire genome of DEV is approximately 160 kb and is composed of a unique long (UL), a unique short (US) and two inverted repeated sequences (IRS and TRS) [
4]. A live C-KCE vaccine strain attenuated in embryonated chicken egg has been developed; it is also one of the safest and most effective duck vaccines available. The DEV vaccine strain is an ideal viral vector for an exclusive avian influenza virus vaccine in broilers [
5,
6]. Hence, C-KCE may be a promising candidate viral vector for developing other bivalent vaccines.
In this study, we established a bacterial artificial chromosome (BAC) of the C-KCE strain. In addition, the E gene of DTMUV was accurately inserted into the C-KCE genome based on the mating-assisted genetically integrated cloning (MAGIC) strategy. Our data reflected that the E gene inserted into the C-KCE genome was robustly expressed under the promoter of human elongation factor 1a (hEF1a). We further demonstrated that the insertion of the E gene exerted no adverse effect on the parental virus C-KCE. In addition, ducks immunized with the C-KCE-E vaccine produced DTMUV-specific antibodies. These ducks were also completely protected against virulent DEV. Overall, this study provides valuable information to establish bivalent live attenuated vaccines against DTMUV and DEV.
2. Materials and Methods
2.1. Virus Strain and Cells
The attenuated duck enteritis virus C-KCE vaccine strain, obtained from the China Institute of Veterinary Drugs Control, was propagated and titrated in chicken embryo fibroblast (CEF) cells (made by ourselves) propagated in Earle’s minimal essential medium (EMEM, Biochrom, Berlin, Germany), which was supplemented with 100 μg/mL penicillin, 100 μg/mL streptomycin and 10% fetal bovine serum (FBS) at 37 °C under a 5% CO2 atmosphere, and a virulent DEV strain isolated from Hubei province was propagated and titrated in duck embryo fibroblast (DEF) cells. A virulent DTMUV strain, which adapted to BHK21 through serially passage (GenBank ID: KJ489355), also was isolated from Hubei province.
2.2. Plasmids and Bacterial Strains
All the plasmids and
E. coli strains were kindly donated by Dr. Lixin Ma. The mini-F plasmid pBlue-lox was maintained in
E. coli strain DH10B-IS2 (umuC:araC-ParaBAD-I-Sce-I-FRT), which has been constructed in Lixin Ma’s lab and expresses enzyme
I-SceI induced by 0.2% w/v L-arabinose [
7]. The plasmid, pML300, contained in DH10B-IS2 carries the
red and
gam recombinase gene stimulated by rhamnose and is unable to replicate when the host bacteria are grown at 42 °C [
8]. DH10b was used for generating the transfer vector, pRThGA-E. DH10b, but not DH10B-IS2, would offer a trans-acting factor, π, which could support the conditional origin of replication from R6K,
oriγ, which contained the donor vector, pRThGA1-E [
9]. Plasmid pCAGGS-NLS/cre expressing Cre was described previously [
10].
2.3. Generation of pBlue-lox-SORF3-US2-Amp Insertion Plasmid and of Donor Plasmid pRThGA1-E
Plasmid pBlue-lox-SORF3-US2-Amp contains two copies of the
Pac I restriction site, an enhanced red fluorescent protein (RFP) gene and its cassette, two copies of the direct orientation 34-bp
Loxp and two copies of the reverse complement, 18-bp
I-sceI. For insertion of the 8.28-kb spanning BAC mini-F plasmid into the C-KCE genome, a 277-bp inter-genic region between the SORF3 and US2 genes was found to be suitable. Briefly, the SORF3 upstream (partial) region and the inter-genic region were amplified as a 530-bp fragment using primers SORF3-F/SORF3-R (
Table 1). The inter-genic region and downstream US2 region were then amplified as a 1.0-kb fragment using primers US2-F/US2-R (
Table 1). The RFP gene under the control of the immediate early promoter of human cytomegalovirus (PHCMV) was amplified from pRTRA as a 1.8-kb fragment using primers Red-F/Red-R (
Table 1). The three PCR products described above were used as the templates for “a ligation PCR” using primers US2-F/Red-R (
Table 1); then, a 3.3-kb fragment was cloned into the
Sal I/
Not I sites of pBlue-lox, resulting in pBlue-lox-SORF3-US2. To increase the copy number of pBlue-lox, the ampicillin resistance gene replicon fragment was amplified from pcdna3.1 (+) with the primers, Amp-F/Amp-R (
Table 1), and inserted into
PacI-digested pBlue-lox-gB-UL26 to obtain plasmid pBlue-lox-SORF3-US2-Amp.
Table 1.
Primers used for generating pBAC-C-KCE, donor plasmid pRThGA and identification of the pBAC-C-KCE-E.
Table 1.
Primers used for generating pBAC-C-KCE, donor plasmid pRThGA and identification of the pBAC-C-KCE-E.
| Purpose and Primer | Sequence (5’→3’) | Sequence Designation, Restriction Enzyme Site and Introduction Sequence |
|---|
| E Gene Clone a |
| E-1F | aaaggatccatgttcagctgtctggggatgcag | BamH I site (bold) |
| E-1R | aaggaattcggcattgacatttactgccaggaa | EcoR I site (bold) |
| BAC Insertion b |
| US2-F | aaagtcgacataacttcgtatagcatacattatacgaagttatcacgtcttcccgcgaggcc | Sal I site (bold), Lox p sequence (underline) |
| US2-R | ttaattaacgcggacaaaacgacgattac | Pac I site (bold) |
| SORF3-F | gtaatcgtcgttttgtccgcgttaattaatgaaaaagacggcggtacaat | Pac I site (bold) |
| SORF3-R | aagaatgcattcggcctgg | |
| Red-F | accagccagctgcgcttgctcgtggggtgtggtgcttttggt | |
| Red-R | tcgagcggccgctagggataacagggtaatccccaccttatatattctttcccaccctcgaagagcgttc | Not I site (bold), I-isce I sequence (underline) |
| Amp-F | aaattaattaaggggataacgcaggaaagaac | Pac I site (bold) |
| Amp-R | aaattaattaaacgtcaggtggcacttttcg | Pac I site (bold) |
| Modification pRThGA c |
| pCA-I-SceI-H1-F | aaatagggataacagggtaatgttgagcctttttgtggagtgggttaaattgtactagcgcgtttcgctttgcagtacatctacgtattagtcatcgctatta | I-isce I sequence (bold), Homology arm H1 (underline) |
| pCA-I-SceI-H2-R | aaatagggataacagggtaattagcatgcataacttcgtataatgtatgctatacgaagttatgcggccgc
cacacaggaaacagctatgaccatgattac | I-isce I sequence (bold), Homology arm H2 (underline) |
| Amp t-I-SceI-F | aaaattaccctgttatccctacacgttaagggattttggtcat | I-isce I sequence (bold) |
| OriT-R6K-I-SceI-R | aaaattaccctgttatcccta | I-isce I sequence (bold) |
| Identification E d |
| E-2F | atgttcagctgtctggggatgcag | |
| E-2R | ggcattgacatttactgccaggaa | |
Fragments from human elongation factor 1a (hEF1a) promoter to BGH ploy A were amplified from vector pEF6-v5/his with the primers, pCA-I-SceI-H1-F/pCA-I-SceI-H2-R, flanked by 50-bp homology arms and
I-sceI restriction sites. The fragment cut by
I-sceI was ligated into the pRThGA vector (also cut by the same enzymes), resulting in the recombinant plasmid, pRThGA1. Genomic RNA of DTMUV was extracted from DTMUV virions. An RT-PCR fragment encoding the E gene was amplified using primers E1-F/E1-R (
Table 1). A start codon was added. The PCR product was digested with
BamH I and
EcoR I and ligated to pRThGA, resulting in pRThGA-E.
2.4. Construction of a C-KCE BAC Clone
Prior to the attachment and penetration of C-KCE virus at a multiplicity of infection (MOI) of 50, the virus was incubated to CEFs for 2 h at 37 °C; pBlue-lox-SORF3-US2-Amp linearized with
Pac I (
Figure 1B) was transfected by calcium phosphate precipitation as described earlier [
11]. When the complete cytopathic effect was observed, the total supernatant was harvested; the infected virus was diluted and then plated on the fresh CEFs and overlaid with DMEM-FBS containing 0.5% methylcellulose. When red fluorescent plaques were observed, plaque-purification was carried out as described earlier [
10] to obtain a fluorescent plaque population, termed vBAC-C-KCE. Circular viral DNA was extracted from CEFs by the method of Hirt [
12]. A total 5 µg of genomic DNA was used to electroporate
DH10B-IS2 with 0.1-cm cuvettes under the following conditions: at 1.5 kV, a resistance of 200 Ω and a capacitance of 25 μF.
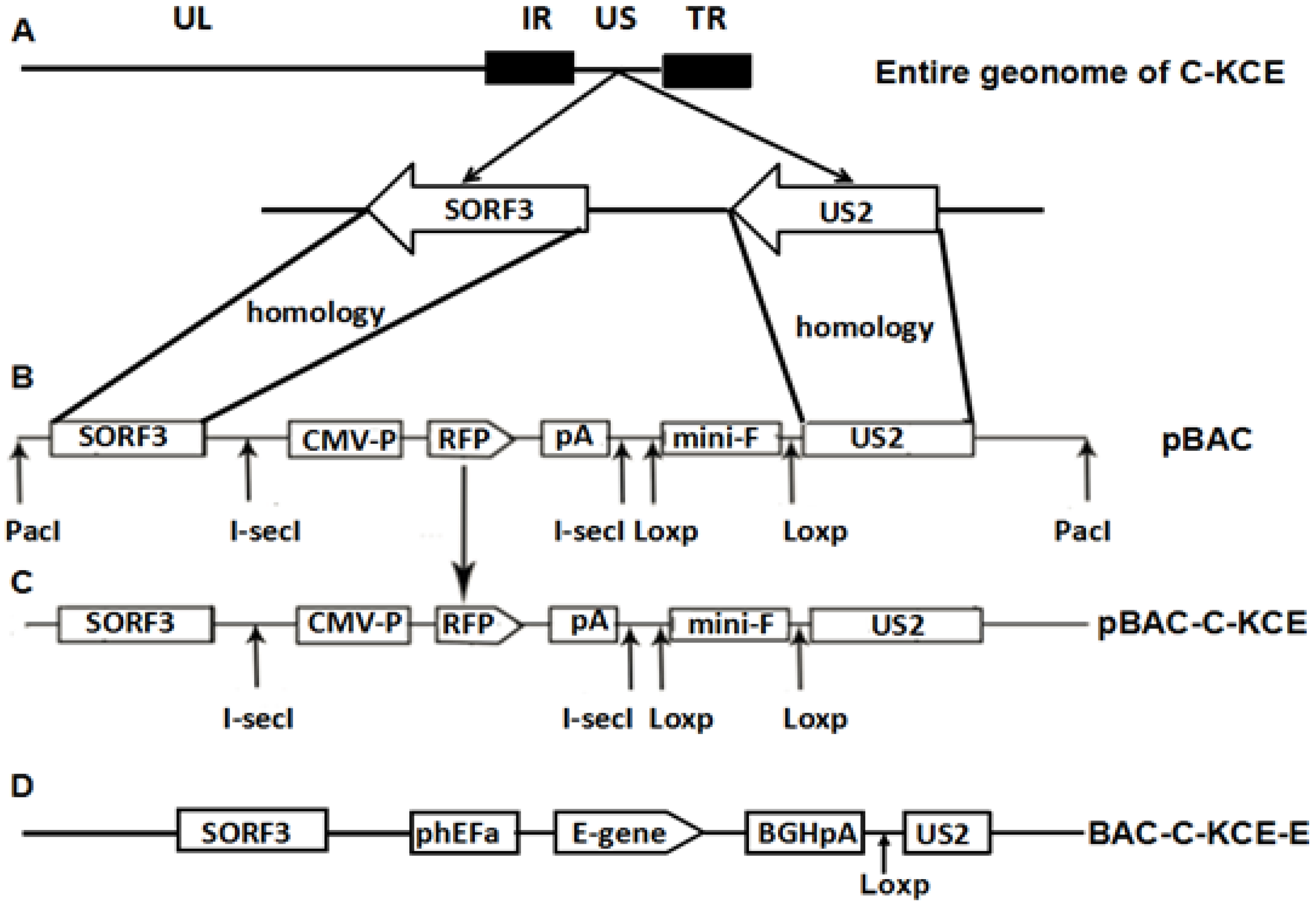
Figure 1.
(A) The organization of the 158-kbp attenuated commercial duck enteritis virus (DEV) vaccine strain (C-KCE); (B) the organization of plasmid pBlue-lox-SORF3-US2-Amp digested by PacI contains an enhanced red fluorescent protein gene and its expression cassette, two copies of the direct orientation 34-bp Loxp and two copies of the reverse complement, 18-bp I-sceI; (C) after homologous recombination, pBlue-lox-SORF3-US2-Amp was inserted into the genome of C-KCE with the red fluorescent protein as a selection marker; (D) the organization of the C-KCE-E.
Figure 1.
(A) The organization of the 158-kbp attenuated commercial duck enteritis virus (DEV) vaccine strain (C-KCE); (B) the organization of plasmid pBlue-lox-SORF3-US2-Amp digested by PacI contains an enhanced red fluorescent protein gene and its expression cassette, two copies of the direct orientation 34-bp Loxp and two copies of the reverse complement, 18-bp I-sceI; (C) after homologous recombination, pBlue-lox-SORF3-US2-Amp was inserted into the genome of C-KCE with the red fluorescent protein as a selection marker; (D) the organization of the C-KCE-E.
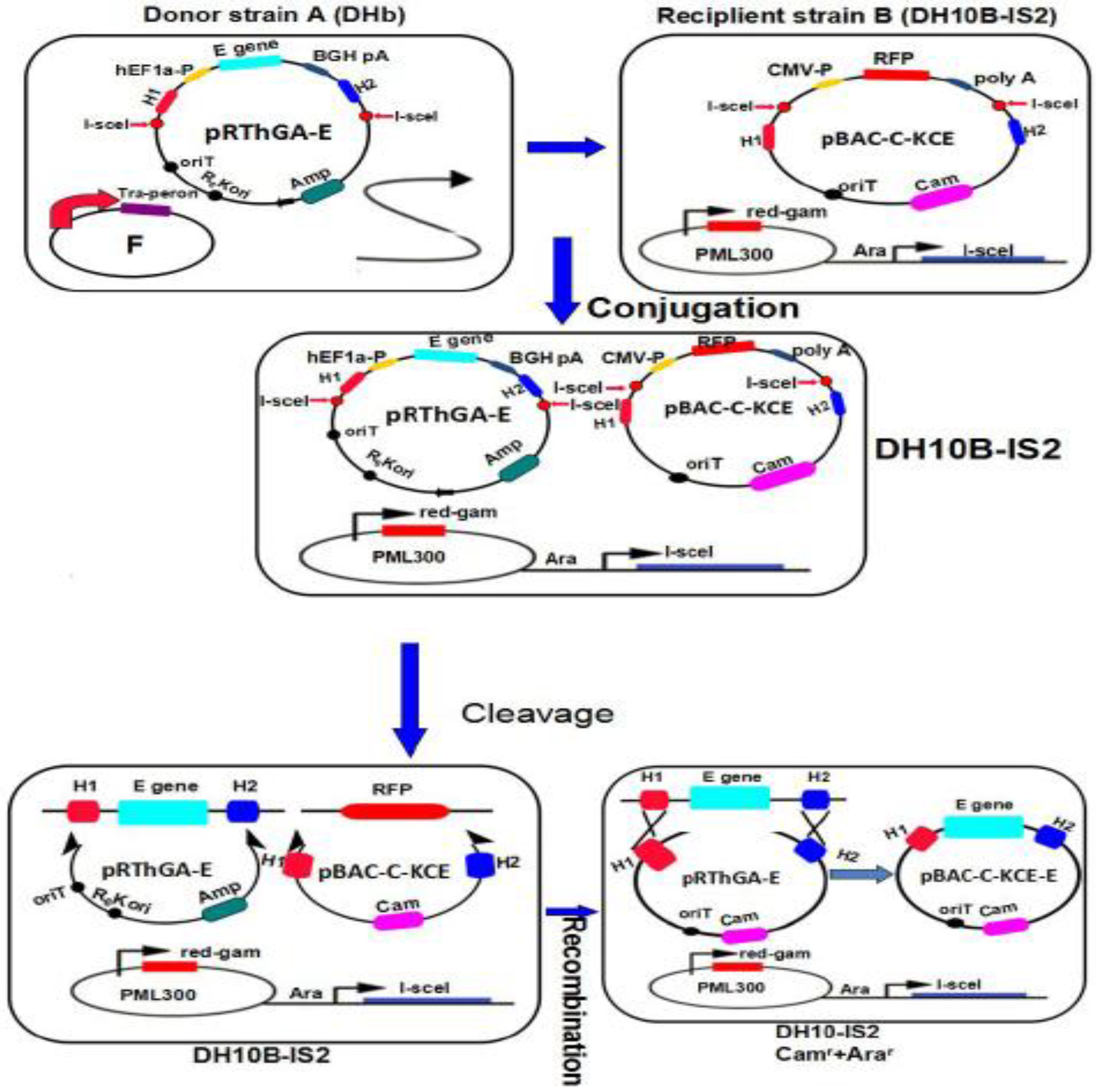
2.5. Generating the Recombinant pBAC-C-KCE-E Vector by MAGIC and Deleting the BAC Vector
E. coli DH10b containing the donor plasmid, pRThGA-E, was grown in LB broth containing 100 μg/mL ampicillin. The recipient strain, DH10B-IS2, containing the plasmid, pML300, and the recipient plasmid, pBAC-C-KCE, was grown in LB containing 50 μg/mL spectinomycin, 34 μg/mL chloramphenicol, 100 μg/mL streptomycin and 0.2% w/v glucose. The procedures of MAGIC were performed as described previously [
8] with slight modifications (
Figure 4). The positive clone was termed pBAC-C-KCE-E. To delete the BAC vector sequences, pC-KCE-BAC–E was co-transfected along with pCAGGS-NLS/cre into CEFs. The deleted BAC vector, termed the C-KCE-E virus, was purified by plaque.
2.6. Detection the Expression of E Protein
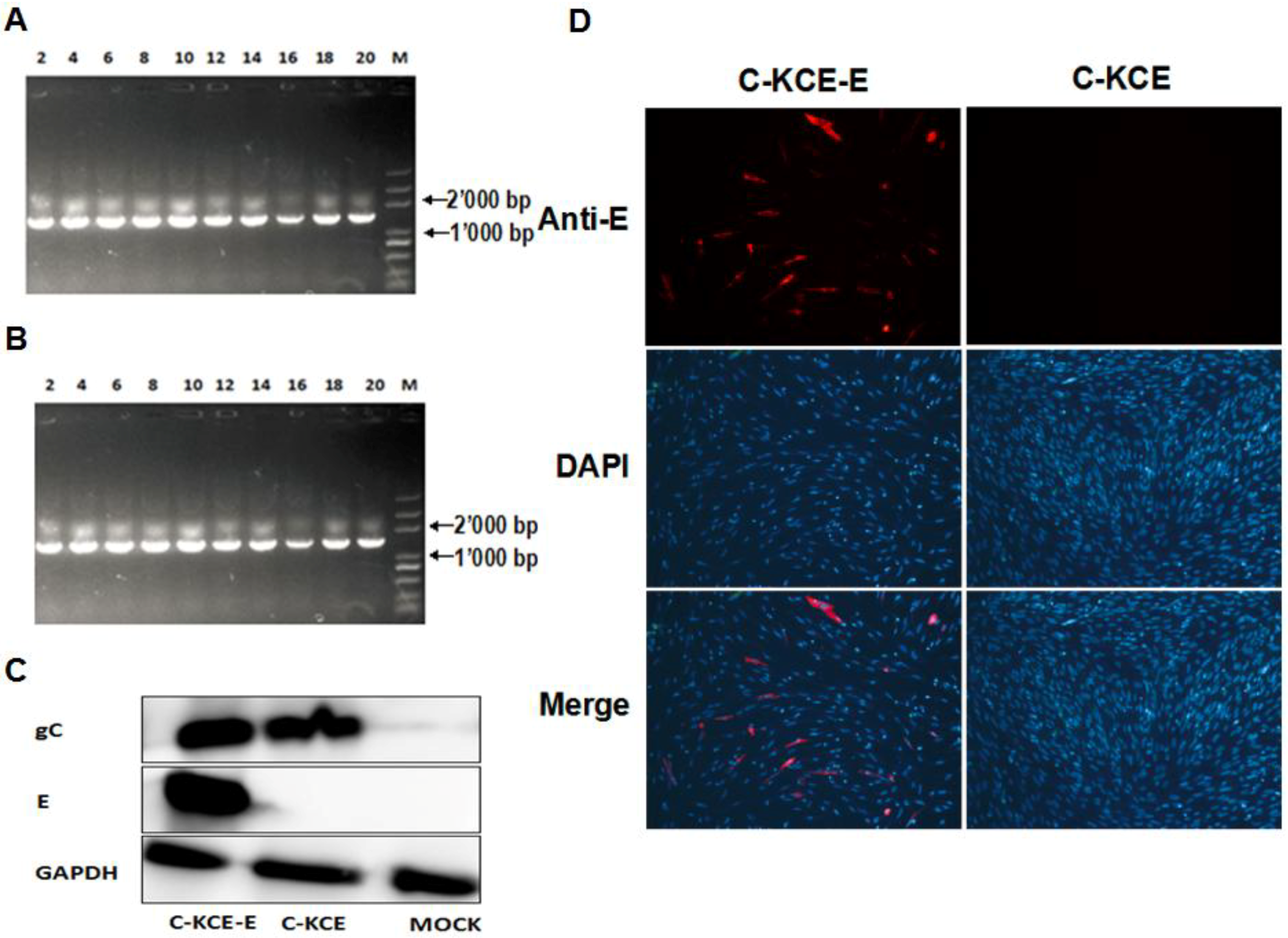
E protein expression in the C-KCE-E was evaluated by immunofluorescence (IFA) and western blot. The monoclonal antibodies (mAbs) against E and the polyclonal antibodies (pAbs) against gC were produced as described previously [
13,
14]. For IFA, the CEFs were infected at an MOI of 1 with C-KCE or C-KCE-E. The mAbs against E (made by us) was used as the primary antibodies. The secondary antibodies were fluorescein isothiocyanate-conjugated goat anti-mouse IgGs (for E detection) (Santa Cruz). The CEFs nuclei were stained with 4’-6-diamidino-2-phenylindole (DAPI). Additionally, the cells were observed with a laser-scanning confocal microscope (Carl Zeiss, Heidenheim, Germany). The results were analyzed using the software, Image J (NIH, Bethesda, MD, USA). For western blot analysis, E expression was carried out in CEFs infected with C-KCE-E and C-KCE at an MOI of 1. mAbs against E, pAbs against gC (made by us) and mAbs against GAPDH (Santa Cruz, CA, USA) for the control were used as primary antibodies; goat HRP-conjugated anti-rabbit or anti-mouse IgGs were used as secondary antibodies. The bands were visualized using ECL detection reagents (Thermo, Waltham, MA, USA), according to the manufacturer’s instructions.
2.7. Stability and Growth Properties of the Recovered Virus C-KCE-E
To analyze the genetic stability of the C-KCE-E, the virus was grown on CEFs sequentially for 20 passages, and virus DNA was extracted and analyzed using primers E-2F/E-2R (
Table 1). To compare the growth between C-KCE and C-KCE-E, assays of multi-step growth kinetics and measurements of plaque size were performed as described earlier [
15].
2.8. Animal Experiments
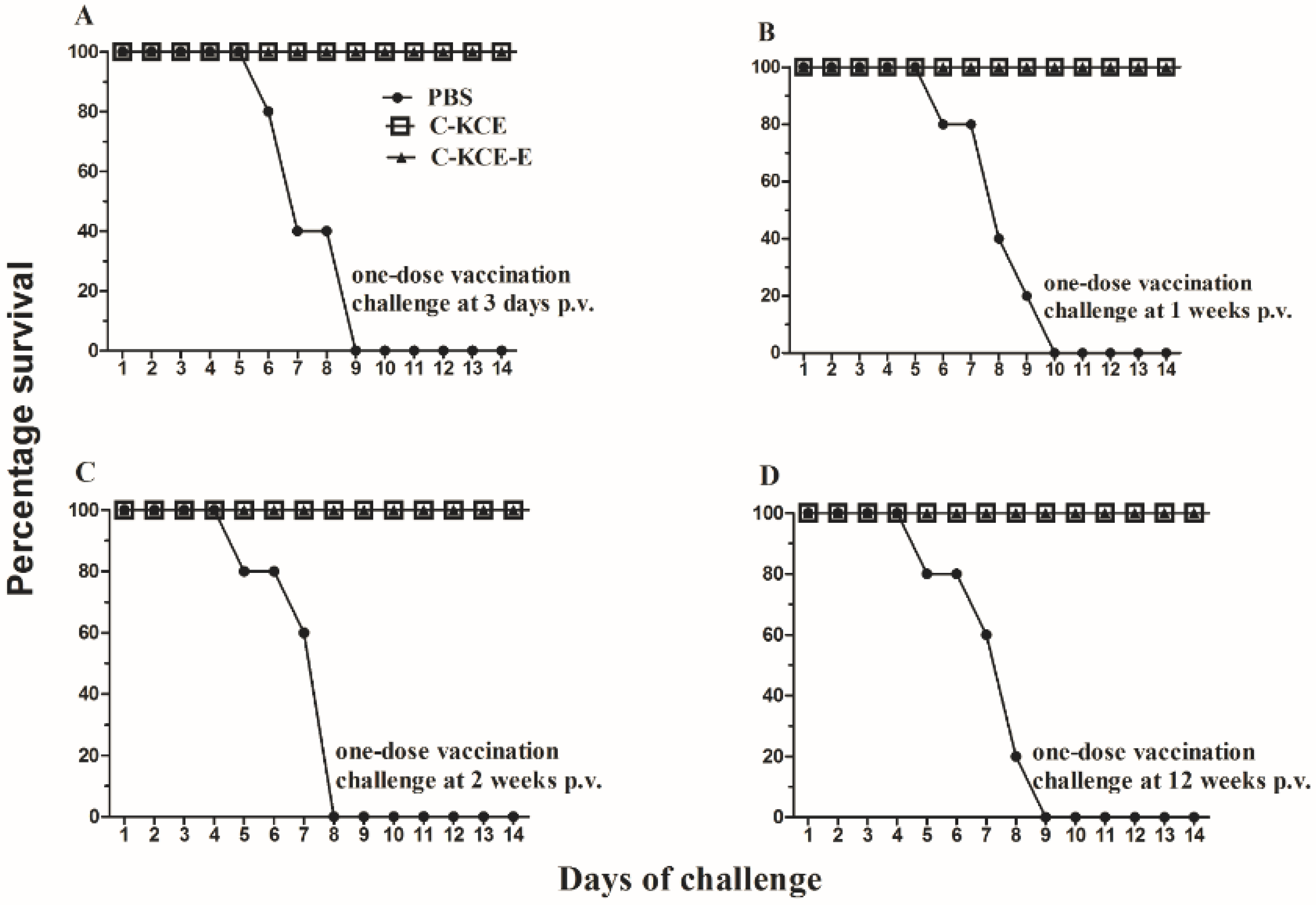
Specific-pathogen-free (SPF) ducks were obtained from Harbin Veterinary Research Institute, China. Ducks were inoculated subcutaneously with 0.1 mL of PBS-diluted C-KCE or C-KCE-E and then intramuscularly challenged with lethal virulent DEV at different time points post-vaccination (p.v.). Ducks were observed daily for signs of disease and death after post-challenge (p.c.).
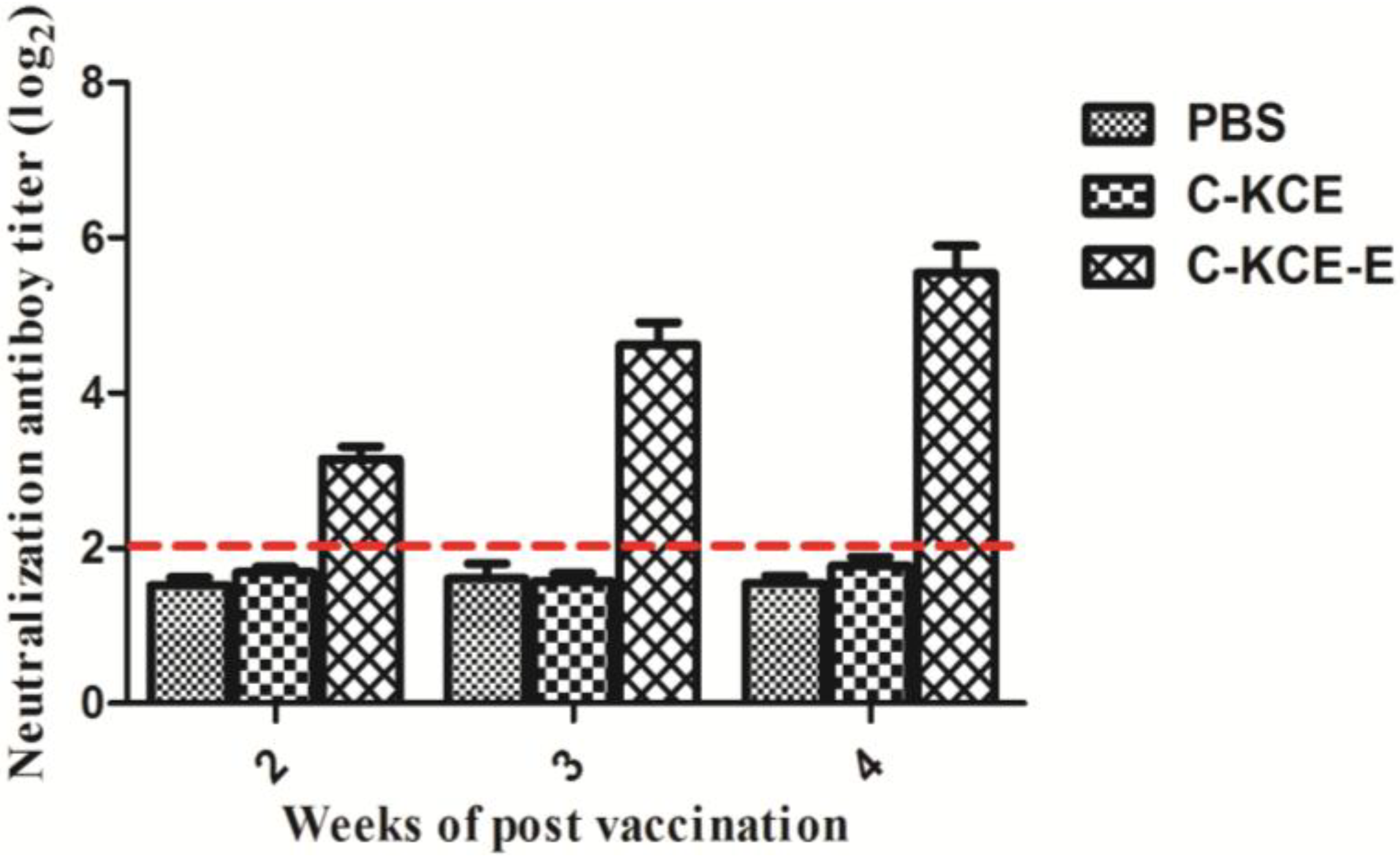
2.9. Neutralization Tests
At Weeks 2, 3 and 4 postvaccination, ducks that received the 10
5 PFU C-KCE-E, 10
5 PFU C-KCE or PBS were sacrificed humanely. Prior to the test, the serum samples obtained from the three groups were inactivated at 56 °C for 30 min. The test was performed using 9-day-old SPF chicken embryonated eggs as previously described [
13].
2.10. Statistical Analysis
All experiments were reproducible and performed in triplicate. Statistical analyses were conducted by a one-way ANOVA test to compare the data of the difference groups using GraphPad Prism version 5.0 (GraphPad Software, La Jolla, CA, USA). p-values of <0.05 were considered statistically significant.
4. Discussion
The newly emerged duck Tembusu viral disease caused by DTMUV severely threatens the progression of the duck industry. Similar to the other members of the
Flaviviridae family, DTMUV may also pose a potential threat to mammals, even including humans [
16,
17]. Close contact between humans and ducks or duck products is inevitable. Therefore, a vaccine against DMUV should be developed for potential public health concerns and for the duck industry.
Vaccination is the most effective method to control this pathogen. Currently, no licensed DTMUV vaccine is available. Only FX2010-180p attenuated in embryonated chicken egg can be utilized for a live attenuated vaccine candidate [
18]. In this study, we established a BAC clone of the DEV attenuated strain C-KCE. The E gene of DTMUV was inserted into the C-KCE genome based on the MAGIC strategy. Although C-KCE-E contained an E gene and its cassette, the insertion had an adverse effect of this insertion on C-KCE replication, egress or viral pathogenesis in our assays. This study is the first to demonstrate the potential of C-KCE-E as a bivalent live attenuated vaccine against DTMUV and DEV.
BAC, which is the most popular platform to generate recombinant virus, has been extensively studied in many viruses. The BAC of C-KCE can be used to develop a bivalent vaccine, as well as to facilitate research on DEV pathogenesis. For instance, the DEV virulent strain 2085 is the first established infectious BAC to evaluate the function of the gC gene [
10]. To establish a BAC clone of C-KCE, the BAC vector should be inserted into a large inter-genic region of the C-KCE genome. The full genome of C-KCE has been sequenced recently in our lab (GenBank ID:KF263690.1). The sequencing revealed that C-KCE and other herpesvirus type 1 share the similar molecular characterization in their genomes. The gene junction of SORF3 and US2 has been proven to be suitable for foreign gene insertion [
19]. Therefore, the same location was selected for constructing the BAC of C-KCE. The expression of the neighboring genes could not be interfered at this junction. In addition to the BAC strategy, there are several other routine strategies that can be used to generate recombinant herpesvirus, such as cosmid. Compared with BAC, cosmid has limited capacity. Thus, inserting several immunogenic foreign genes to develop a polyvalent vaccine is difficult. More importantly, transfection of the cosmid vectors into target cells requires several rounds of homologous recombination to recover the virus; thus, accuracy is not guaranteed [
20].
We accurately and efficiently inserted the E gene into pBAC-C-KCE based on the MAGIC method. To the best of our knowledge, the strategy described here is the first case to use MAGIC for the construction of recombinant DEV. The efficiency of the recombination can reach 90%, because the
red and
gam system is extremely highly efficient and because arabinose can function as a selection marker [
7]. Utilizing MAGIC, we successfully constructed and obtained the recombinant C-KCE-E within two weeks of acquiring the E gene. Therefore, the approach is a highly-efficient, time-saving and labor-saving method for generating recombinant DEV.
Generally, the level of expression has two important determinants, including the inserted gene itself and its promoter [
21]. Sonoda et al. compared the protective efficacy of the chicken β-actin promoter and the SV40 promoter for expressing the F gene of Newcastle disease virus inserted between the US10 of Marek's disease virus. They found that the chicken β-actin promoter performed better than the SV40 promoter and that the expression of F from the chicken β-actin promoter offered better protection against virulent MDV challenge [
22]. Thus, we concluded that enhancing the expression level of the E gene is critical to inducing a higher level of protective neutralization antibodies against DTMUV. For this purpose, we chose the hEF1α promoter, driving a high-level of protein expression across a wide range of species and cell types. Indeed, the results of western blot and IFA manifested that the E protein was robustly expressed during C-KCE-E replication.
Neutralizing the antibody against DTMUV is the hallmark of protective immunity. As shown in the animal tests, C-KCE-E vaccination can induce DTMUV-specific antibodies at three weeks p.v. Considering that DTMUV challenge was not performed in this study, we were uncertain about the efficacy of the vaccine against a lethal DTMUV challenge in ducks. The DEV challenge test showed that the ducks immunized with C-KCE or C-KCE-E survived. Therefore, we concluded that the inserted gene did not alter the protective efficacy of the parental virus C-KCE.
In summary, we successfully developed one recombinant DEV delivering E of DTMUV. In our future studies, we will determine whether or not C-KCE-E can provide optimum protection against DTMUV infection in ducks. If the result is positive, C-KCE-E will be an extremely cost-effective option to control the devastating effect of DEV and DTMUV in ducks.