CCR5 as a Natural and Modulated Target for Inhibition of HIV
Abstract
:1. CCR5 and the CCR5-Delta32 Deletion
2. A First Case of a “Natural” Gene Therapy
3. Benefits and Risks of Transplanting CCR5 Deficient Stem Cells
4. Challenges in Repeating the “Berlin Patient”
4.2. Cord Blood: An Alternative Stem Cell Source
- High accessibility of a haploidentical donor that bridges the time until the CCR5-negative UCB engrafts.
- Increased probability of finding matching UCB due to the reduction of the required CD34+ cell dose. Lowering the required cell dose from 2.5 to 1.0 × 107 TNC/kg will increase the probability from 27.9% to 82.1%.
- Reduced time of aplasia due to the haploidentical proportion of the transplant.
- Finally, after engraftment, the UCB unit will take over the myeloid function while the haploidentical part disappears after a period of time resulting in a 100% chimerism of CCR5 depleted and HIV resistant peripheral blood cells.
5. Limitations of the CCR5-Delta32 Approach
6. Modulation of CCR5 Expression
7. Addition of a Second Inhibitory Element
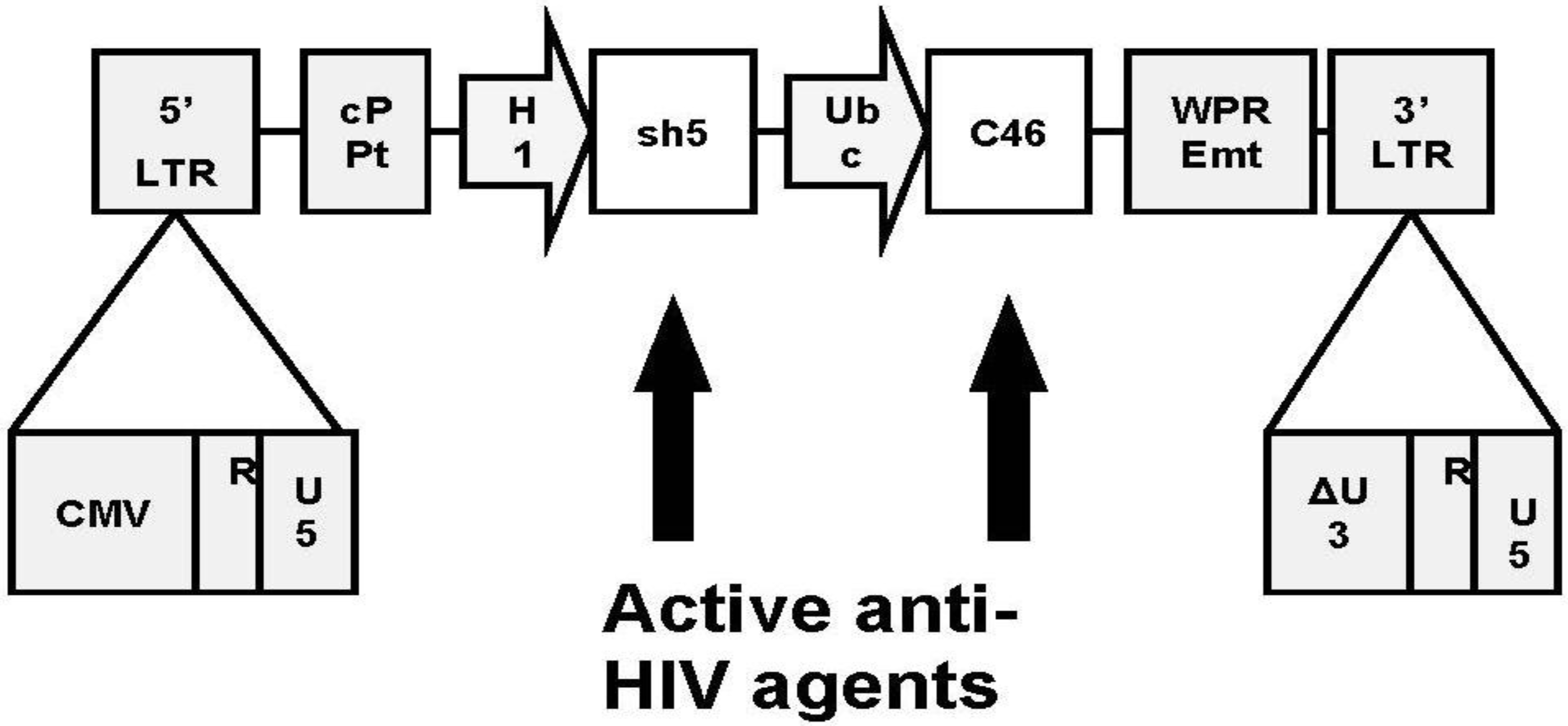
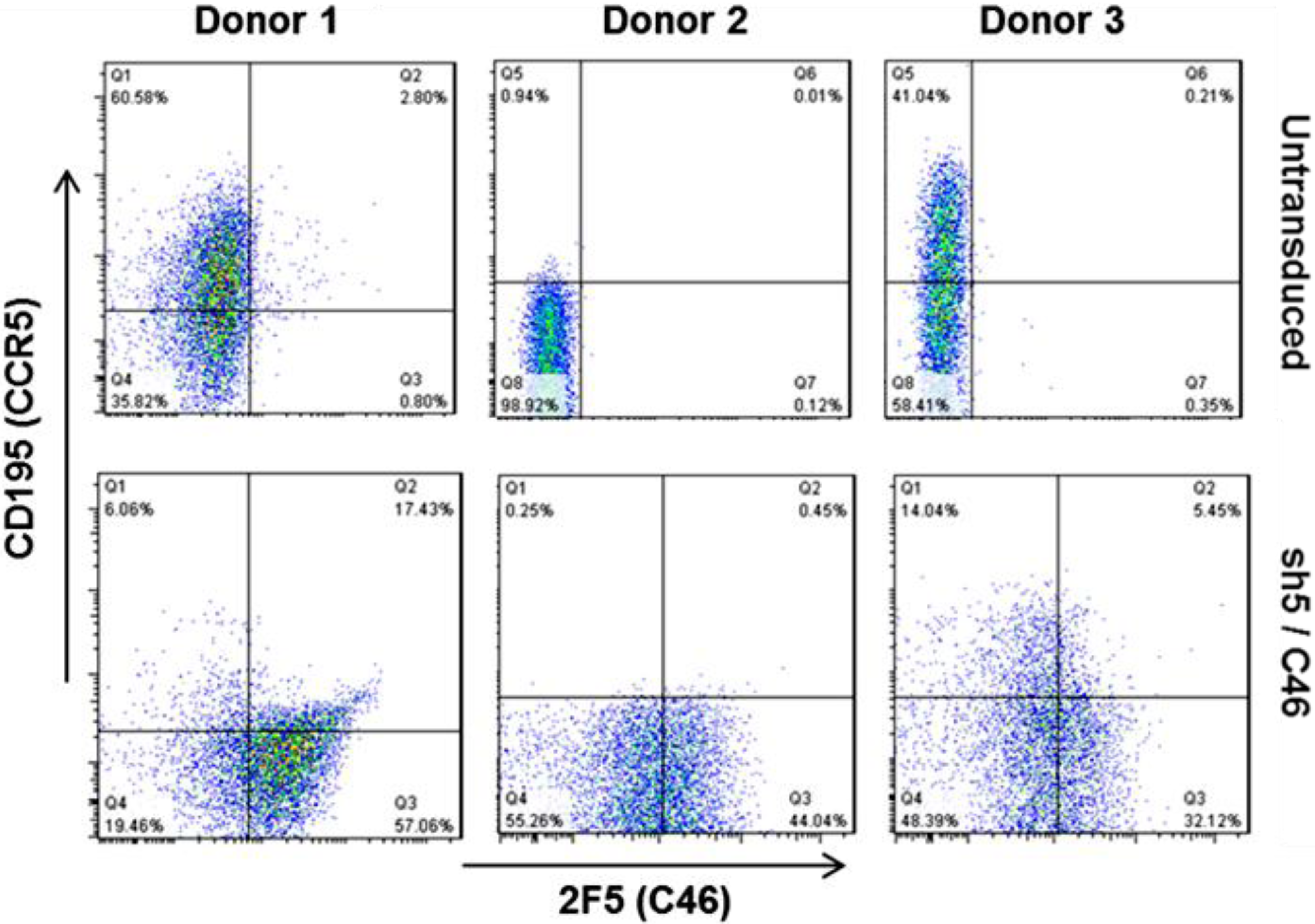
8. Clinical Trial Design
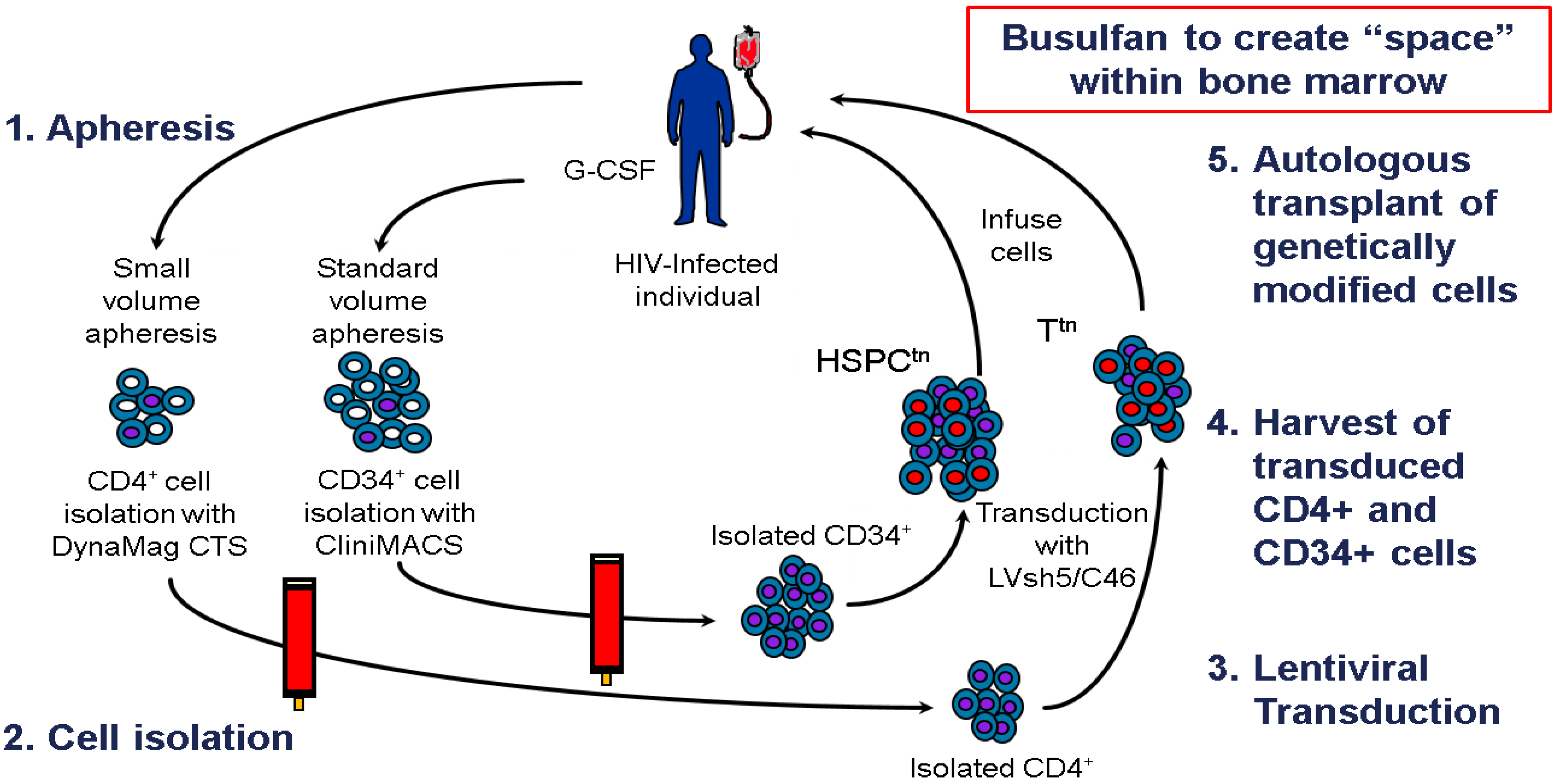
9. Conclusions: Tying It All Together
9.1. CCR5-Delta32 Compared with Down-Regulation of CCR5
9.2. Durability of Response
Acknowledgments
Conflicts of Interest
References and Notes
- Liu, R.; Paxton, W.A.; Choe, S.; Ceradini, D.; Martin, S.R.; Horuk, R.; MacDonald, M.E.; Stuhlmann, H.; Koup, R.A.; Landau, N.R. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell 1996, 86, 367–377. [Google Scholar] [CrossRef]
- Hütter, G.; Nowak, D.; Mossner, M.; Ganepola, S.; Mussig, A.; Allers, K.; Schneider, T.; Hofmann, J.; Kücherer, C.; Blau, O.; et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N. Engl. J. Med. 2009, 360, 692–698. [Google Scholar] [CrossRef]
- Allers, K.; Hütter, G.; Hofmann, J.; Loddenkemper, C.; Rieger, K.; Thiel, E.; Schneider, T. Evidence for the cure of HIV infection by CCR5Delta32/Delta32 stem cell transplantation. Blood 2011, 117, 2791–2799. [Google Scholar] [CrossRef]
- Fischereder, M.; Luckow, B.; Hocher, B.; Wuthrich, R.P.; Rothenpieler, U.; Schneeberger, H.; Panzer, U.; Stahl, R.A.; Hauser, I.A.; Budde, K.; et al. CC chemokine receptor 5 and renal-transplant survival. Lancet 2001, 357, 1758–1761. [Google Scholar] [CrossRef]
- Heidenhain, C.; Puhl, G.; Moench, C.; Lautem, A.; Neuhaus, P. Chemokine receptor 5Delta32 mutation reduces the risk of acute rejection in liver transplantation. Ann. Transplant. 2009, 14, 36–44. [Google Scholar]
- Schnickel, G.T.; Bastani, S.; Hsieh, G.R.; Shefizadeh, A.; Bhatia, R.; Fishbein, M.C.; Belperio, J.; Ardehali, A. Combined CXCR3/CCR5 blockade attenuates acute and chronic rejection. J. Immunol. 2008, 180, 4714–4721. [Google Scholar]
- Bogunia-Kubik, K.; Duda, D.; Suchnicki, K.; Lange, A. CCR5 deletion mutation and its association with the risk of developing acute graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Haematologica 2006, 91, 1628–1634. [Google Scholar]
- Prahalad, S. Negative association between the chemokine receptor CCR5-Delta32 polymorphism and rheumatoid arthritis: A meta-analysis. Genes Immun. 2006, 7, 264–268. [Google Scholar] [CrossRef]
- Sandford, A.J.; Zhu, S.; Bai, T.R.; Fitzgerald, J.M.; Pare, P.D. The role of the C-C chemokine receptor-5 Delta32 polymorphism in asthma and in the production of regulated on activation, normal T cells expressed and secreted. J. Allergy Clin. Immunol. 2001, 108, 69–73. [Google Scholar] [CrossRef]
- Atzeni, F.; Boiardi, L.; Casali, B.; Farnetti, E.; Nicoli, D.; Sarzi-Puttini, P.; Pipitone, N.; Olivieri, I.; Cantini, F.; La Corte, R.; et al. CC chemokine receptor 5 polymorphism in Italian patients with Behcet’s disease. Rheumatology 2012, 51, 2141–2145. [Google Scholar] [CrossRef]
- Yang, B.; Houlberg, K.; Millward, A.; Demaine, A. Polymorphisms of chemokine and chemokine receptor genes in Type 1 diabetes mellitus and its complications. Cytokine 2004, 26, 114–121. [Google Scholar] [CrossRef]
- Glass, W.G.; McDermott, D.H.; Lim, J.K.; Lekhong, S.; Yu, S.F.; Frank, W.A.; Pape, J.; Cheshier, R.C.; Murphy, P.M. CCR5 deficiency increases risk of symptomatic West Nile virus infection. J. Exp. Med. 2006, 203, 35–40. [Google Scholar] [CrossRef]
- Hütter, G.; Thiel, E. Allogeneic transplantation of CCR5-deficient progenitor cells in a patient with HIV infection: An update after 3 years and the search for patient no. 2. AIDS 2009, 25, 273–274. [Google Scholar]
- Petz, L.D.; Redei, I.; Bryson, Y.; Regan, D.; Kurtzberg, J.; Shpall, E.; Gutman, J.; Querol, S.; Clark, P.; Tonai, R.; et al. Hematopoietic cell transplantation with cord blood for cure of HIV infections. Biol. Blood Marrow. Transplant. 2012, 19, 393–397. [Google Scholar]
- Kwon, M.; Balsalobre, P.; Serrano, D.; Perez Corral, A.; Buno, I.; Anguita, J.; Gayoso, J.; Diez-Martin, J.L. Single cord blood combined with HLA-mismatched third party donor cells: Comparable results to matched unrelated donor transplantation in high-risk patients with hematologic disorders. Biol. Blood Marrow. Transplant. 2012, 19, 143–149. [Google Scholar]
- Liu, H.; Rich, E.S.; Godley, L.; Odenike, O.; Joseph, L.; Marino, S.; Kline, J.; Nguyen, V.; Cunningham, J.; Larson, R.A.; et al. Reduced-intensity conditioning with combined haploidentical and cord blood transplantation results in rapid engraftment, low GVHD, and durable remissions. Blood 2011, 118, 6438–6445. [Google Scholar] [CrossRef]
- Kwon, M.; Kuball, J.H.; Balsalobre, P.; Serrano, D.; Buno, I.; Gayoso, J.; van Besien, K.M.; Petz, L.D.; Diez-Martin, J.L.; Wensing, A. A Single Cord Blood Transplantation Combined with a HLA Mismatched Third Party Donor for a Patuient with Burkitt Lymphoma and HIV Infection. In Proceedings of the 10th International Cord Blood Symposium, San Francisco, CA, USA, 7–9 June 2012.
- Kuball, J. (Ed.) Towards the next generation of transplantation: HIV positive patients. In Proceedings of the 10th International Cord Blood Symposium, San Francisco, CA, USA, 7–9 June 2012.
- Díez-Martín, J.L. Personal communication, Hematology Department, HGU Gregorio Marañón: Madrid, Spain, 2012.
- Nijhuis, M.; Kwon, M.; Kuball, J.; Ellerbroek, P.; Berenguer, J.; Stam, A.; Symons, J.; Richman, D.; Diez-Martin, J.L.; Wensing, A. (Eds.) Early viral dynamics after cord blood stem cell transplantation (with and without CCR5delta32) combined with HLA mismatched donor in two HIV-patients. In Proceedings of the 20th Conference on Retroviruses and Opportunistic Infections, Atlanta, GA, USA, 3–6 March 2013.
- Symons, J.; Deeks, S.; Hütter, G.; Wensing, A.; Martin, J.; van Ham, P.; Vandekerckhove, L.; Nijhuis, M. The cure of the 'Berlin patient': why did pre-existing X4-variants not emergence after allogeneic CCR5-Delta 32 SCT? JIAS 2012, 15, 17–18. [Google Scholar]
- Yukl, S.A.; Boritz, E.; Busch, M.; Bentsen, C.; Chun, T.W.; Douek, D.; Eisele, E.; Haase, A.; Ho, Y.C.; Hütter, G.; et al. Challenges in detecting HIV persistence during potentially curative interventions: A study of the Berlin patient. PLOS Pathog. 2013, 9, e1003347. [Google Scholar] [CrossRef]
- Henrich, T.J.; Hu, Z.; Li, J.Z.; Sciaranghella, G.; Busch, M.P.; Keating, S.M.; Gallien, S.; Lin, N.H.; Giquel, F.F.; Lavoie, L.; et al. Long-term reduction in peripheral blood HIV type 1 reservoirs following reduced-intensity conditioning allogeneic stem cell transplantation. J. Infect. Dis. 2013, 207, 1694–1702. [Google Scholar] [CrossRef]
- Henrich, T.; Hanhauser, E.; Sirignano, M.; Davis, B.; Lee, T.-H.; Keating, S.; Busch, M.; Marty, F.; LaCasce, A.; Armand, P.; et al. In depth investigation of peripheral and gut HIV-1 reservoirs, HIV-specific cellular immunity, and host microchimerism following allogeneic hematopoetic stem cell transplantation. In Proceedings of the 7th International AIDS Society Conference on HIV Pathogenesis, Treatment and Prevention, Kuala Lumpur, Malaysia, 30 June–3 July 2013.
- Kitchen, S.G.; Shimizu, S. Stem cell-based anti-HIV gene therapy. Virology 2011, 411, 260–272. [Google Scholar] [CrossRef]
- An, D.S.; Qin, F.X.; Auyeung, V.C.; Mao, S.H.; Kung, S.K.; Baltimore, D.; Chen, I.S.Y. Optimization and functional effects of stable short hairpin RNA expression in primary human lymphocytes via lentiviral vectors. Molecular therapy. J. Am. Soc. Gene Ther. 2006, 14, 494–504. [Google Scholar] [CrossRef]
- An, D.S.; Donahue, R.E.; Kamata, M.; Poon, B.; Metzger, M.; Mao, S.H.; Bonifacion, A.; Krouse, A.E.; Darlix, J.-L.; Baltimore, D.; et al. Stable reduction of CCR5 by RNAi through hematopoietic stem cell transplant in non-human primates. Proc. Natl. Acad. Sci. USA 2007, 104, 13110–13115. [Google Scholar] [CrossRef]
- Qin, X.F.; An, D.S.; Chen, I.S.; Baltimore, D. Inhibiting HIV-1 infection in human T cells by lentiviral-mediated delivery of small interfering RNA against CCR5. Proc. Nat. Acad. Sci. USA 2003, 100, 183–188. [Google Scholar] [CrossRef]
- Liang, M.; Kamata, M.; Chen, K.N.; Pariente, N.; An, D.S.; Chen, I.S. Inhibition of HIV-1 infection by a unique short hairpin RNA to chemokine receptor 5 delivered into macrophages through hematopoietic progenitor cell transduction. J. Gene Med. 2010, 12, 255–265. [Google Scholar] [CrossRef]
- Shimizu, S.; Hong, P.; Arumugam, B.; Pokomo, L.; Boyer, J.; Koizumi, N.; Kittipongdaja, P.; Chen, A.; Bristol, G.; Galic, Z.; et al. A highly efficient short hairpin RNA potently down-regulates CCR5 expression in systemic lymphoid organs in the hu-BLT mouse model. Blood 2010, 115, 1534–1544. [Google Scholar] [CrossRef]
- Egelhofer, M.; Brandenburg, G.; Martinius, H.; Schult-Dietrich, P.; Melikyan, G.; Kunert, R.; Baum, C.; Ingrid, C.; Alexandrov, A.; von Laer, D. Inhibition of human immunodeficiency virus type 1 entry in cells expressing gp41-derived peptides. J. Virol. 2004, 78, 568–575. [Google Scholar] [CrossRef]
- Zahn, R.C.; Hermann, F.G.; Kim, E.Y.; Rett, M.D.; Wolinsky, S.M.; Johnson, R.P.; Villinger, F.; von Laer, D.; Schmitz, J.E. Efficient entry inhibition of human and nonhuman primate immunodeficiency virus by cell surface-expressed gp41-derived peptides. Gene Ther. 2008, 15, 1210–1222. [Google Scholar] [CrossRef]
- Van Lunzen, J.; Glaunsinger, T.; Stahmer, I.; von Baehr, V.; Baum, C.; Schilz, A.; Kuehlcke, K.; Naundorf, S.; Martinius, H.; Hermann, F.; et al. Transfer of autologous gene-modified T cells in HIV-infected patients with advanced immunodeficiency and drug-resistant virus. Mol. Ther. 2007, 15, 1024–1033. [Google Scholar]
- Younan, P.M.; Polacino, P.; Kowalski, J.P.; Peterson, C.W.; Maurice, N.J.; Williams, N.P.; Ho, O.; Trobridge, G.D.; Von Laer, D.; Prlic, M.; et al. Positive selection of mC46-expressing CD4+ T cells and maintenance of virus specific immunity in a primate AIDS model. Blood 2013, 122, 179–187. [Google Scholar] [CrossRef]
- Sallusto, F.; Lenig, D.; Forster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999, 401, 708–712. [Google Scholar] [CrossRef]
- Scholler, J.; Brady, T.L.; Binder-Scholl, G.; Hwang, W.T.; Plesa, G.; Hege, K.M.; Vogel, A.N.; Kalos, M.; Riley, J.L.; Deeks, S.G.; et al. Decade-long safety and function of retroviral-modified chimeric antigen receptor T cells. Sci. Transl. Med. 2012, 4, 132ra53. [Google Scholar]
- Deeks, S.G.; Wagner, B.; Anton, P.A.; Mitsuyasu, R.T.; Scadden, D.T.; Huang, C.; Macken, C.; Richman, D.D.; Christopherson, C.; June, C.H.; et al. A phase II randomized study of HIV-specific T-cell gene therapy in subjects with undetectable plasma viremia on combination antiretroviral therapy. Mol. Ther. 2002, 5, 788–797. [Google Scholar] [CrossRef]
- Edelstein, M.L.; Abedi, M.R.; Wixon, J. Gene therapy clinical trials worldwide to 2007—An update. J. Gene Med. 2007, 9, 833–842. [Google Scholar] [CrossRef]
- Frecha, C.; Levy, C.; Cosset, F.L.; Verhoeyen, E. Advances in the field of lentivector-based transduction of T and B lymphocytes for gene therapy. Mol. Ther. 2010, 18, 1748–1757. [Google Scholar] [CrossRef]
- Humeau, L.M.; Binder, G.K.; Lu, X.; Slepushkin, V.; Merling, R.; Echeagaray, P.; Pereira, M.; Slepushkina, T.; Barnett, S.; Dropulic, L.K.; et al. Efficient lentiviral vector-mediated control of HIV-1 replication in CD4 lymphocytes from diverse HIV+ infected patients grouped according to CD4 count and viral load. Mol. Ther. 2004, 9, 902–913. [Google Scholar] [CrossRef]
- June, C.H. Disruption of CCR5 in Zinc Finger Nuclease-treated CD4 T Cells: Phase I Trials. In Proceedings of the 18th Conference on Retroviruses and Opportunistic Infections, Boston, MA, USA, 27 February–2 March 2011. Abstract 165.
- Tebas, P.; Stein, S.; Zifchak, L.; Seda, A.; Binder, G.; Aberra, F.; Collman, G.; McGarrity, B.; June, C. Prolonged control of viremia after transfer of autologous CD4 T cells genetically modified with a lentiviral vector expressing long antisense to HIV env (VRX496). In Proceedings of the 17th Conference on Retroviruses and Opportunistic Infections, San Francisco, CA, USA, 16–19 February 2010.
- Levine, B.L.; Bernstein, W.B.; Aronson, N.E.; Schlienger, K.; Cotte, J.; Perfetto, S.; Humphries, M.J.; Ratto-Kim, S.; Birx, D.L.; Steffens, C.; et al. Adoptive transfer of costimulated CD4+ T cells induces expansion of peripheral T cells and decreased CCR5 expression in HIV infection. Nat. Med. 2002, 8, 47–53. [Google Scholar] [CrossRef]
- Levine, B.L.; Humeau, L.M.; Boyer, J.; MacGregor, R.R.; Rebello, T.; Lu, X.; Binder, G.K.; Slepushkin, V.; Lemiale, F.; Mascola, J.R.; et al. Gene transfer in humans using a conditionally replicating lentiviral vector. Proc. Natl. Acad. Sci. USA 2006, 103, 17372–17377. [Google Scholar] [CrossRef]
- Levine, B.L. T lymphocyte engineering ex vivo for cancer and infectious disease. Expert. Opin. Biol. Ther. 2008, 8, 475–489. [Google Scholar] [CrossRef]
- Macpherson, J.L.; Boyd, M.P.; Arndt, A.J.; Todd, A.V.; Fanning, G.C.; Ely, J.A.; Elliott, F.; Knop, A.; Raponi, M.; Murray, J.; et al. Long-term survival and concomitant gene expression of ribozyme-transduced CD4+ T-lymphocytes in HIV-infected patients. J. Gene Med. 2005, 7, 552–564. [Google Scholar] [CrossRef]
- Mitsuyasu, R.T.; Anton, P.A.; Deeks, S.G.; Scadden, D.T.; Connick, E.; Downs, M.T.; Bakker, A.; Roberts, M.R.; June, C.H.; Jalali, S.; Lin, A.A.; et al. Prolonged survival and tissue trafficking following adoptive transfer of CD4zeta gene-modified autologous CD4(+) and CD8(+) T cells in human immunodeficiency virus-infected subjects. Blood 2000, 96, 785–793. [Google Scholar]
- Morgan, R.A.; Walker, R.; Carter, C.S.; Natarajan, V.; Tavel, J.A.; Bechtel, C.; Herpin, B.; Muul, L.; Zheng, Z.; Jaqannatha, S.; et al. Preferential survival of CD4+ T lymphocytes engineered with anti-human immunodeficiency virus (HIV) genes in HIV-infected individuals. Hum. Gene Ther. 2005, 16, 1065–1074. [Google Scholar] [CrossRef]
- Morgan, R.A.; Dudley, M.E.; Wunderlich, J.R.; Hughes, M.S.; Yang, J.C.; Sherry, R.M.; Royal, R.E.; Topalian, S.L.; Kammula, T.U.; Restifo, N.P.; et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science 2006, 314, 126–129. [Google Scholar] [CrossRef]
- Ranga, U.; Woffendin, C.; Verma, S.; Xu, L.; June, C.H.; Bishop, D.K.; Nabel, G.J. Enhanced T cell engraftment after retroviral delivery of an antiviral gene in HIV-infected individuals. Proc. Natl. Acad. Sci. USA 1998, 95, 1201–1206. [Google Scholar] [CrossRef]
- Rossi, J.J.; June, C.H.; Kohn, D.B. Genetic therapies against HIV. Nat. Biotechnol. 2007, 25, 1444–1454. [Google Scholar] [CrossRef]
- Amado, R.G.; Mitsuyasu, R.T.; Rosenblatt, J.D.; Ngok, F.K.; Bakker, A.; Cole, S.; Chorn, N.; Lin, L.S.; Bristol, G.; Boyd, M.P.; et al. Anti-human immunodeficiency virus hematopoietic progenitor cell-delivered ribozyme in a phase I study: myeloid and lymphoid reconstitution in human immunodeficiency virus type-1-infected patients. Hum. Gene Ther. 2004, 15, 251–262. [Google Scholar] [CrossRef]
- Kohn, D.B. Gene therapy using hematopoietic stem cells. Curr. Opin. Mol. Ther. 1999, 1, 437–442. [Google Scholar]
- Mitsuyasu, R.T.; Merigan, T.C.; Carr, A.; Zack, J.A.; Winters, M.A.; Workman, C.; Bloch, M.; Lalezari, J.; Becker, S.; Thornton, L.; et al. Phase 2 gene therapy trial of an anti-HIV ribozyme in autologous CD34+ cells. Nat. Med. 2009, 15, 285–292. [Google Scholar] [CrossRef]
- Podsakoff, G.M.; Engel, B.C.; Kohn, D.B. Perspectives on gene therapy for immune deficiencies. Biol. Blood Marrow. Transplant. 2005, 11, 972–976. [Google Scholar] [CrossRef]
- Muul, L.M.; Tuschong, L.M.; Soenen, S.L.; Jagadeesh, G.J.; Ramsey, W.J.; Long, Z.; Carter, C.S.; Garabedian, E.K.; Alleyne, M.; Brown, M.; Bernstein, W.; et al. Persistence and expression of the adenosine deaminase gene for 12 years and immune reaction to gene transfer components: Long-Term results of the first clinical gene therapy trial. Blood 2003, 101, 2563–2569. [Google Scholar] [CrossRef]
- Seymour, L.W.; Thrasher, A.J. Gene therapy matures in the clinic. Nat. Biotechnol. 2012, 30, 588–593. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Burke, B.P.; Boyd, M.P.; Impey, H.; Breton, L.R.; Bartlett, J.S.; Symonds, G.P.; Hütter, G. CCR5 as a Natural and Modulated Target for Inhibition of HIV. Viruses 2014, 6, 54-68. https://doi.org/10.3390/v6010054
Burke BP, Boyd MP, Impey H, Breton LR, Bartlett JS, Symonds GP, Hütter G. CCR5 as a Natural and Modulated Target for Inhibition of HIV. Viruses. 2014; 6(1):54-68. https://doi.org/10.3390/v6010054
Chicago/Turabian StyleBurke, Bryan P., Maureen P. Boyd, Helen Impey, Louis R. Breton, Jeffrey S. Bartlett, Geoff P. Symonds, and Gero Hütter. 2014. "CCR5 as a Natural and Modulated Target for Inhibition of HIV" Viruses 6, no. 1: 54-68. https://doi.org/10.3390/v6010054




