Alphaviruses in Gene Therapy
Abstract
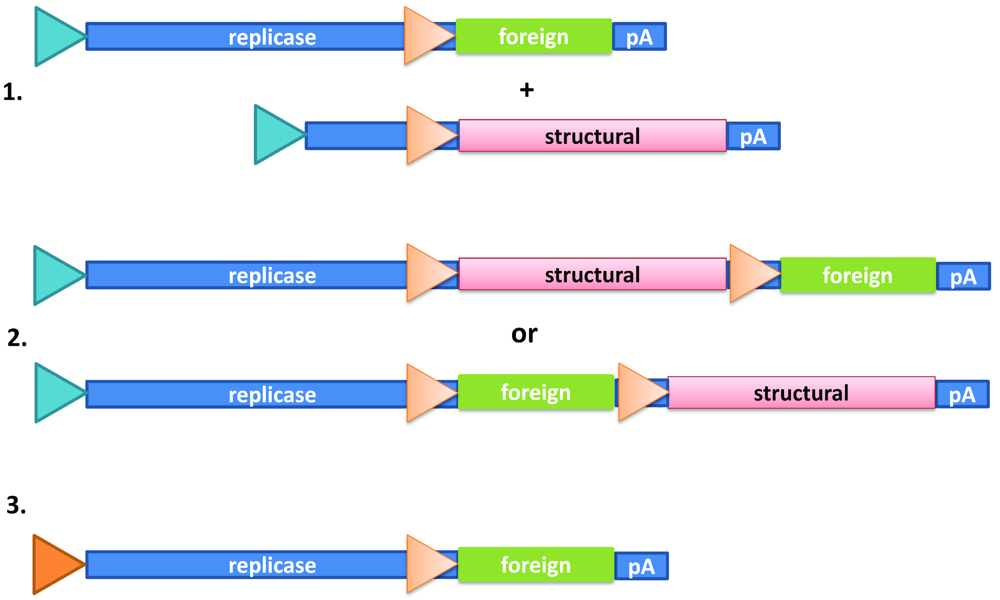
:1. Introduction
2. Alphaviruses in Gene Therapy
2.1. Vaccines
2.2. Cancer Therapy
2.3. Oncolytic Vectors
2.4. CNS Applications
2.5. Mutant Vectors
2.6. Targeting of Viral Particles
2.7. Clinical Trials for Alphaviruses
3. Conclusions
References
- Cavazzana-Calvo, M; Fischer, A. Gene therapy for severe combined immunedeficiency: are we there yet? J. Clin Invest 2007, 117, 1456–1465. [Google Scholar] [CrossRef]
- Check, E. Gene therapy: a tragic setback. Nature 2002, 402, 116–118. [Google Scholar] [CrossRef]
- Lundstrom, K. Latest development in viral vectors for gene therapy. Trends Biotechnol 2003, 21, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Ng, P; Graham, FL. Construction of first-generation adenoviral vectors. Methods Mol Med 2002, 69, 389–414. [Google Scholar] [PubMed]
- Rabinowitz, JE; Samulski, J. Adeno-associated virus expression systems for gene transfer. Curr Opin Biotechnol 1998, 9, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Osten, P; Grinevich, V; Cetin, A. Viral vectors:a wide range of choices and high levels of service. Handb Exp Pharmacol 2007, 178, 177–202. [Google Scholar] [PubMed]
- Strauss, JH; Strauss, EG. The alphaviruses: gene expression, replication and evolution. Microbiol Rev 1994, 58, 491–562. [Google Scholar] [PubMed]
- Liljeström, P; Garoff, H. A new generation of animal cell expression vectors based on the Semliki Forest virus replicon. Biotechnology(NY) 1991, 9, 1356–1361. [Google Scholar] [CrossRef]
- Xiong, C; Levis, R; Shen, P; Schlesinger, S; Rice, CM; Huang, HV. Sindbis virus: an efficient, broad host range vector for gene expression in animal cells. Science 1989, 243, 1188–1191. [Google Scholar] [PubMed]
- Davis, NL; Brown, KW; Johnston, RE. In vitro synthesis of infectious venezuelan equine encephalitis virus RNA from a cDNA clone: analysis of a viable deletion mutant. Virology 1989, 171, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Smerdou, C; Liljeström, P. Two-helper RNA system for production of recombinant Semliki forest virus particles. J Virol 1999, 73, 1092–1098. [Google Scholar] [PubMed]
- Lundstrom, K. Biology and application of alphaviruses in gene therapy. Gene Ther 2005, Suppl. 1, S92–S97. [Google Scholar] [CrossRef]
- Lundstrom, K. Semliki Forest virus vectors for rapid and high-level expression of integral membrane proteins. Biochim Biophys Acta 2001, 1601, 90–96. [Google Scholar]
- Lundstrom, K; Ehrengruber, MU. Semliki Forest virus (SFV) vectors in neurobiology and gene therapy. Methods Mol Med 2003, 76, 503–523. [Google Scholar] [PubMed]
- Ehrengruber, MU; Lundstrom, K; Schweitzer, C; Heuss, C; Schlesinger, S; Gähwiler, BH. Recombinant Semliki Forest virus and Sindbis virus efficiently infect neurons in hippocampal slice cultures. Proc Natl Acad Sci USA 1999, 96, 7041–7046. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K; Richards, JG; Pink, JR; Jenck, F. Efficient in vivo expression of a reporter gene in rat brain after injection of recombinant replication-deficient Semliki Forest virus. Gene Ther Mol Biol 1999, 3, 15–23. [Google Scholar]
- Lundstrom, K; Wagner, R; Reinhart, C; Desmyter, A; Cherouati, N; Magnin, T; Zeder-Lutz, G; Courtot, M; Prual, C; André, N; Hassaine, G. ; Michel, H. ; Cambillau, C ; Pattus, F Structural genomics on membrane proteins: comparison of more than 100 GPCRs in 3 expression systems J Struct Funct Genomics 2006, 7, 77–91. [Google Scholar]
- Lundstrom, K. Alphavirus vectors for vaccine production and gene therapy. Expert Review of Vaccines 2003, 2, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, R; Zullo, SA; Ramsey, J; Yajima, N; Tsuchiya, N; Tanaka, R; Blaese, M; Xanthopoulos, KG. Marked enhancement of antitumor immune responses in mouse brain tumor models by genetically modified dendritic cells producing Semliki Forest virus-mediated interleukin-12. J Neurosurg 2002, 97, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Daemen, T; Regts, J; Holtrop, M; Wilschut, J. Immunization strategy against cervical cancer involving an alphavirus vector expressing high levels of a stable fusion protein of human papillomavirus 16 E6 and E7. Gene Ther 2002, 9, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Cheng, WF; Hung, CF; Hsu, KF; Chai, CY; He, L; Polo, JM; Slater, LA; Ling, M; Wu, TC. Cancer immunotherapy using Sindbis virus replicon particles encoding a VP22-antigen fusion. Hum Gene Ther 2002, 13, 553–568. [Google Scholar] [PubMed]
- Colmenero, P; Chen, M; Castaños-Velez, E; Liljeström, P; Jondal, M. Immunotherapy with recombinant SFV-replicons expressing the P815A tumor antigen or IL-12 induces tumor regression. Int J Cancer 2002, 98, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Ying, H; Zeng, G; Black, KL. Innovative cancer vaccine strategies based on the identification of tumour-associated antigens. Bio Drugs 2001, 15, 819–831. [Google Scholar]
- Leitner, WW; Hwang, LN; deVeer, MJ; Zhou, A; Silverman, RH; Williams, BR; Dubensky, TW; Ying, H; Restifo, NP. Alphavirus-based DNA vaccine breaks immunological tolerance by activating innate antiviral pathways. Nature Med 2003, 9, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Colmenero, P; Liljestrom, P; Jondal, M. Induction of P185 tumor immunity by recombinant Semliki Forest virus expressing the P1A gene. Gene Ther 1999, 6, 1728–1733. [Google Scholar] [CrossRef] [PubMed]
- Ying, H; Zaks, T.Z.; Wang, RF; Irvine, KR; Kammula, US; Marincola, FM; Leitner, WW; Restifo, NP. Cancer therapy using a self-replicating RNA vaccine. Nature Med 1999, 5, 823–827. [Google Scholar] [CrossRef] [PubMed]
- Velders, MP; McElhiney, S; Cassetti, MC; Eiben, GL; Higgins, T; Kovacs, GR; Elmishad, AG; Kast, WM; Smith, LR. Eradication of established tumors by vaccination with Venezuelan equine encephalitis virus replicon particles delivering human papillomavirus 16 E7 RNA. Cancer Res 2001, 61, 7861–7867. [Google Scholar] [PubMed]
- Yamanaka, R; Zullo, SA; Tanaka, R; Blaese, M; Xanthopoulos, KG. Enhancement of antitumor response in glioma models in mice by genetically modified dendritic cells pulsed with Semliki Forest virus-mediated complementary DNA. J Neurosurg 2001, 94, 478–481. [Google Scholar]
- Leslie, MC; Zhao, YJ; Lachman, LB; Hwu, P; Wu, GJ; Bar-Eli, M. Immunization against MUC18/MCAM, a novel antigen that drives melanoma invasion and metastasis. Gene Ther 2007, 14, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Moran, TP; Burgents, JE; Long, B; Ferrer, I; Jaffee, EM; Tisch, RM; Johnston, RE; Serody, JS. Alphaviral vector-transduced dendritic cells are successful therapeutic vaccines against neu-overexpressing tumors in wild-type mice. Vaccine 2007, 25, 6604–6612. [Google Scholar] [CrossRef] [PubMed]
- Durso, RJ; Andjelic, S; Gardner, JP; Margitich, DJ; Donovan, GP; Arrigale, RR; Wang, X; Maughan, MF; Talarico, TL; Olmsted, RA; Heston, WD; Maddon, PJ; Olson, WC. A novel alphavirus vaccine encoding prostate-specific membrane antigen elicits potent cellular and humoral immune responses. Clin Cancer Res 2007, 13, 3999–4008. [Google Scholar] [CrossRef] [PubMed]
- Riezebos-Brilman, A; Regts, J; Chen, M; Wilschut, J; Daemen, T. Augmentation of alphavirus vector-induced human papilloma virus-specific immune and anti-tumour responses by co-expression of interleukin-12. Vaccine 2009, 27, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Asselin-Paturel, C; Lassau, N; Guinebretière, JM; Zhang, J; Gay, F; Bex, F; Hallez, S; Leclere, J; Peronneau, P; Mami-Chouaib, F; Chouaib, S. Transfer of the murine interleukin-12 gene in vivo by a Semliki Forest virus vector induces B16 tumor regression through inhibition of tumor blood vessel formation monitored by Doppler ultrasonography. Gene Ther 1999, 6, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Murphy, AM; Morris-Downes, MM; Sheahan, BJ; Atkins, GJ. Inhibition of human lung carcinoma cell growth by apoptosis induction using Semliki Forest virus recombinant particles. Gene Ther 2000, 7, 1477–1482. [Google Scholar] [CrossRef] [PubMed]
- Smyth, JW; Fleeton, MN; Sheahan, BJ; Atkins, GJ. Treatment of rapidly growing K-BALB and CT26 mouse tumours using Semliki Forest virus and its derived vector. Gene Ther 2005, 12, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Chikkanna-Gowda, CP; Sheahan, BJ; Fleeton, MN; Atkins, GJ. Regression of mouse tumours and inhibition of metastases following administration of a Semliki Forest virus vector with enhanced expression of IL-12. Gene Ther 2005, 12, 1253–1263. [Google Scholar] [CrossRef] [PubMed]
- Chikkanna-Gowda, CP; McNally, S; Sheahan, BJ; Fleeton, MN; Atkins, GJ. Inhibition of murine K-BALB and CT26 tumour growth using a Semliki Forest virus vector with enhanced expression of IL-18. Oncol Rep 2006, 16, 713–719. [Google Scholar] [PubMed]
- Lyons, JA; Sheahan, BJ; Galbraith, SE; Mehra, R; Atkins, GJ; Fleeton, MN. Inhibition of angiogenesis by a Semliki Forest virus vector expressing VEGFR-2 reduces tumour growth and metastasis in mice. Gene Ther 2007, 14, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Vähä-Koskela, MJ; Kallio, JP; Jansson, LC; Heikkilä, JE; Zakhartchenko, VA; Kallajoki, MA; Kähäri, VM; Hinkkanen, AE. Oncolytic, capacity of attenuated replicative semliki forest virus in human melanoma xenografts in severe combined immunodeficient mice. Cancer Res 2006, 66, 7185–7194. [Google Scholar] [CrossRef] [PubMed]
- Ketola, A; Hinkkanen, A; Yongabi, F; Furu, P; Määttä, AM; Liimatainen, T; Pirinen, R; Björn, M; Hakkarainen, T; Mäkinen, K; Wahlfors, J; Pellinen, R. Oncolytic Semliki forest virus vector as a novel candidate against unresectable osteosarcoma. Cancer Res 2008, 68, 8342–8350. [Google Scholar] [CrossRef] [PubMed]
- Määttä, AM; Mäkinen, K; Ketola, A; Liimatainen, T; Yongabi, FN; Vähä-Koskela, M; Pirinen, R; Rautsi, O; Pellinen, R; Hinkkanen, A; Wahlfors, J. Replication competent Semliki Forest virus prolongs survival in experimental lung cancer. Int J Cancer 2008, 123, 1704–1711. [Google Scholar] [CrossRef] [PubMed]
- Atkins, GJ; Sheahan, BJ; Liljestrom, P. The molecular pathogenesis of Semliki Forest virus: a model virus made useful? J. Gen Virol 1999, 80, 2287–2297. [Google Scholar]
- Ehrengruber, MU; Renggli, M; Raineteau, O; Hennou, S; Vähä-Koskela, MJ; Hinkkanen, AE; Lundstrom, K. Semliki Forest virus A7[74) transduces hippocampal neurons and glial cells in a temperature-dependent dual manner. J Neurovirol 2003, 9, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Graham, A; Walker, R; Baird, P; Hahn, CN; Fazakerley, JK. CNS gene therapy applications of the Semliki Forest virus 1 vector are limited by neurotoxicity. Mol Ther 2006, 13, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Sammin, DJ; Butler, D; Atkins, GJ; Sheahan, BJ. Cell death mechanisms in the olfactory bulb of rats infected intranasally with Semliki forest virus. Neuropathol Appl Neurobiol 1999, 25, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Jerusalmi, A; Morris-Downes, MM; Sheahan, BJ; Atkins, GJ. Effect of intranasal administration of Semliki Forest virus recombinant particles expressing reporter and cytokine genes on the progression of experimental autoimmune encephalomyelitis. Mol Ther 2003, 8, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Quinn, K. Effect of intranasal administration of Semliki Forest virus recombinant particles expressing interferon-β on the progression of experimental autoimmune encephalomyelitis. Mol Med Rep 2008, 1, 335–342. [Google Scholar]
- Vähä-Koskela, MJ; Kuusinen, TI; Holmlund-Hampf, JC; Furu, PT; Heikkilä, JE; Hinkkanen, AE. Semliki Forest virus vectors expressing transforming growth factor beta inhibit experimental autoimmune encephalomyelitis in Balb/c mice. Biochem Biophys Res Commun 2007, 355, 776–781. [Google Scholar] [CrossRef] [PubMed]
- Nygårdas, PT; Grönberg, SA; Heikkilä, J; Joronen, K; Sorsa, T; Hinkkanen, AE. Treatment of experimental autoimmune encephalomyelitis with a neurotropic alphavirus vector expressing tissue inhibitor of metalloproteinase-2. Scand J Immunol 2004, 60, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Dryga, SA; Dryga, OA; Schlesinger, S. Identification of mutations in a Sindbis virus variant able to establish persistent infection in BHK cells: the importance of a mutation in the nsP2 gene. Virology 1997, 228, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Agapov, EV; Frolov, I; Lindenbach, BD; Prágai, BM; Schlesinger, SRiceCM. Noncytopathic Sindbis virus RNA vectors for heterologous gene expression. Proc Natl Acad Sci USA 1998, 95, 12989–12994. [Google Scholar] [CrossRef] [PubMed]
- Boorsma, M; Nieba, L; Koller, D; Bachmann, MF; Bailey, JE; Renner, WA. A temperature-regulated replicon-based DNA expression system. Nat Biotechnol 2000, 18, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Perri, S; Driver, DA; Gardner, JP; Sherrill, S; Belli, BA; Dubensky, TWJr; Polo, JM. Replicon vectors derived from Sindbis virus and Semliki forest virus that establish persistent replication in host cells. J Virol 2000, 74, 9802–9807. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K; Rotmann, D; Hermann, D; Schneider, EM; Ehrengruber, MU. Novel mutant Semliki Forest virus vectors: gene expression and localization studies in neuronal cells. Histochem Cell Biol 2001, 115, 83–91. [Google Scholar] [PubMed]
- Lundstrom, K; Abenavoli, A; Malgaroli, A; Ehrengruber, MU. Novel Semliki Forest virus vectors with reduced cytotoxicity and temperature sensitivity for long-term enhancement of transgene expression. Mol Ther 2003, 7, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Casales, E; Rodriguez-Madoz, JR; Ruiz-Guillen, M; Razquin, N; Cuevas, Y; Prieto, J; Smerdou, C. Development of a new noncytopathic Semliki Forest virus vector providing high expression levels and stability. Virology 2008, 376, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Ohno, K; Sawai, K; Iijima, Y; Levin, B; Meruelo, D. Cell-specific targeting of Sindbis virus vectors displaying IgG-binding domains of protein A. Nat Biotechnol 1997, 15, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Tseng, JC; Levin, B; Hurtado, A; Yee, H; Perez, deCastroI; Jimenez, M; Shamamian, P; Jin, R; Novick, RP; Pellicer, A; Meruelo, D. Systemic tumor targeting and killing by Sindbis viral vectors. Nat Biotechnol 2004, 22, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K; Boulikas, T. Breakthrough in cancer therapy: encapsulation of drugs and viruses. Curr Drug Discov 2002, 11, 19–23. [Google Scholar]
- 15 4 2009. http://clinicaltrials.gov/ct2/results?term=alphavirus.
- Lundstrom, K; Boulikas, T. Viral and non-viral vectors in gene therapy: technology development and clinical trials. Technol Cancer Res Treat 2003, 2, 471–486. [Google Scholar] [PubMed]
- Ren, H; Boulikas, T; Lundstrom, K; Söling, A; Warnke, PC; Rainov, NG. Immunogene therapy of recurrent glioblastoma multiforme with a liposomally encapsulated replication-incompetent Semliki forest virus vector carrying the human interleukin-12 gene--a phase I/II clinical protocol. J Neurooncol 2003, 64, 147–154. [Google Scholar] [PubMed]
| Target | Gene | Vector/Delivery | Response | Ref |
|---|---|---|---|---|
| Brain tumor | IL–12 | SFV/particles | Immunogenicity | [19] |
| Cervical cancer | HPV E6–E7 | SFV/particles | Tumor protection | [20] |
| Glioma | B16, 203 | SFV/particles | Tumor protection | [28] |
| Tumor | β–gal | SFV/RNA | Tumor protection | [26] |
| Tumor | HPV E7 | VEE/particles | Tumor protection | [27] |
| Tumor | HPVE7–VP22 | SIN/particles | CD8+ T–cell response | [21] |
| Tumor | P815A | SFV/particles | Tumor protection | [22] |
| Tumor antigen | MHC Class II | SFV/particles, DNA | Immunogenicity | [23] |
| Tumor antigen | P185 | SFV/particles | CTL, tumor protection | [25] |
| Tumor antigen | Tyr–related prot–1 | SIN/DNA | Antitumor activity | [24] |
| Melanoma | MUC18 | SIN/DNA | Tumor protection | [29] |
| Tumor | Neu | VEE/particles | Tumor protection | [30] |
| Prostate cancer | PSMA | VEE/particles | Immunogenicity | [31] |
Share and Cite
Lundstrom, K. Alphaviruses in Gene Therapy. Viruses 2009, 1, 13-25. https://doi.org/10.3390/v1010013
Lundstrom K. Alphaviruses in Gene Therapy. Viruses. 2009; 1(1):13-25. https://doi.org/10.3390/v1010013
Chicago/Turabian StyleLundstrom, Kenneth. 2009. "Alphaviruses in Gene Therapy" Viruses 1, no. 1: 13-25. https://doi.org/10.3390/v1010013




