1. Introduction
The growing number of bone reconstructive surgery types, along with progressively prolonged life expectancy in developed countries, make research and the development of alternatives to natural bone grafts increasingly important. Bioactive scaffolds play an important role in tissue engineering [
1,
2].
A bone substitute material should display ‘bimodal’ behavior, which, in early differentiation stages, allows osteoblasts to build bridges between different sizes of grain and to integrate with other osteoblasts to support both proliferation and differentiation. New bone formation has been stimulated by the activation of mesenchymal stem cells and their absorption onto surfaces with nanoscale topographic features [
3]. The ultimate goal is to unite fully differentiated osteoblasts that support bone matrix production. This requires a porous structure with nanopores, micropores and macropores, all of which are involved in different stages of absorption, adhesion and bone material deposition on and within the bone substitute material [
4].
How to effectively improve the attachment of cells in the interior of large-sized scaffolds is still a significant bone tissue engineering challenge. Some studies have suggested that macropores with a pore diameter that falls within the 50–300 μm range are beneficial for cell attachment, proliferation and vascularization, while the micropores within the 0.5–10 μm range are desired to provide the effective delivery of nutrients and physical cues to enhance cell response. It now seems acceptable that a minimum interconnect size of ~100 μm is needed for mineralized tissue ingrowth [
1,
3,
4]. These results underline the need for developing new technologies to produce strong scaffolds with controlled porosity [
5,
6]. Part of this work focuses on the sintering of porous calcium silicophosphate scaffolds with macro- and microporosity by the polymer replication method.
Regarding silicon that contains calcium phosphate materials, the effect of Si on healthy bone and connective tissues is well-known. Silicon modifies the material properties and improves the biological activity of silicon that contains CaP materials. Thus, they have been widely studied as biomaterial for osseous repair [
7,
8].
The interest in rich-silico phosphate biomaterial is currently increasing because of its good bioactivity response and low cytotoxicity [
9,
10].
In this context, the compositions that belong to subsystem Nurse’s A-phase- silcocarnotite within system Ca
3(PO
4)
2–Ca
2SiO
4 are promising candidates for preparing new ceramic bone implants [
11]. Nurse’s A-phase is a solid solution with an approximate composition of 7CaOP
2O
52SiO
2 [
12,
13], which should not be confused with the mineral of the same composition identified by Nagelshmidt in 1937 [
14]. Silicocarnotite (5CaOP
2O
5SiO
2) can be defined as calcium silicophosphate with a carnotite structure [
7,
15].
The main purpose of the present study was to investigate the effect of pore morphology on its in vivo osteoconductivity and resorption process of new calcium silicophosphate ceramic scaffolds obtained by the polymer replication method.
3. Discussion
Interconnected Si-Ca-P porous scaffolds, where a porous polymer sponge was used as a template, were processed to be used as novel material with osteoconductive properties for bone reconstruction, with similar properties to autologous bone grafts. The main findings showed that the porous scaffold degraded over the experimental set points and allowed new bone tissue formation.
The porous bioactive scaffold also induced new bone tissue formation inside the material in three ways: (i) invasion of newly formed bone tissue on the material’s surface using the network of interconnected pores, with the material starting from the periphery toward its center; (ii) macrophage activity that precedes invasion or penetration of bone marrow, which provides a capillary axis accompanied with vascular cells, and an osteoblastic line; and (iii) the material per se is able to create the microenvironment required around it to locally carry out the osteogenic differentiation of the osteogenic precursor cells contained in the hematopoietic bone marrow.
Porous bioactive scaffolds are most interesting to be used as bone substitutes in the bone tissue engineering field [
16]. High bioactivity and adequate scaffold porosity are essential characteristics to stimulate osteoprogenitor cells and to support bone in-growth. Furthermore, resorption of the material with the same bone formation rate is required [
17]. Several in vivo studies have demonstrated that Si influences bone mineralization [
9,
10], metabolism [
18,
19], collagen synthesis [
20,
21] and crosslinking [
22]. These findings fall in line with the results of the current study, which revealed continuous newly bone tissue in-growth in the defect area, inside pores and in the spaces left by the degraded scaffold. After implantation, the dissolution of Si and Ca ions from the scaffold to bone tissue stimulates the formation of a carbonate-hydroxyapatite layer, which acts as a template for osteoblast growth and can affect osteogenesis [
3,
4,
23,
24]. High porosity and adequate pore sizes are essential factors for effective bone substitute material. Scaffolds with an optimal pore size allow bone in-growth and support neovascularization. Both the pore size and porosity of the bioactive scaffold used herein indicate its morphological characteristics, which make it suitable for being used as a bone graft. Moreover, the histological findings demonstrated that the scaffold degraded over time and that degradation happened according to the tissue in-growth rate. Besides adequate porosity, proper scaffold degradation is also essential for the process to happen since new bone tissue formation needs space to grow in [
3,
4,
23,
24].
Several techniques have been used to assess the biomaterial-to-bone tissue interface. Many evaluations of the interface using LM, SEM, and TEM have shown how the same structure is represented differently depending on the examination method selected [
25,
26]. The traditional method used during bone regeneration, histological staining followed by examination under a light microscope, provides substantial information. However, its low spatial resolution does not provide ultrastructural information. The precision and reliability of a histomorphometric study of newly formed bone depends on the correct identification and ultrastructural characterization of all the cellular components that could play a role in the osseointegration process. LM lacks the resolving power required for detailed structural analyses. Electron microscopy techniques can help characterize the morphological changes that occur during osseointegration. Despite certain limitations [
27], transmission electron microscopy has been successfully used to describe the cellular components of newly formed bone.
SEM, used to examine bone-to-biomaterial interfaces, was first reported by Jasty et al. [
28]. This method has been employed mainly for descriptive studies performed with calcified tissue [
29,
30]. SEM images can be used to highlight contrasts between areas of different chemical compositions, and this technique is especially effective whenever average atomic numbers of the components of each region vary. Since SEM signal intensity depends on the sample’s mean atomic number, the SEM technique not only serves to distinguish inorganic features, but also offers the interesting possibility of identifying ultrastructural cell components and their micromorphological details.
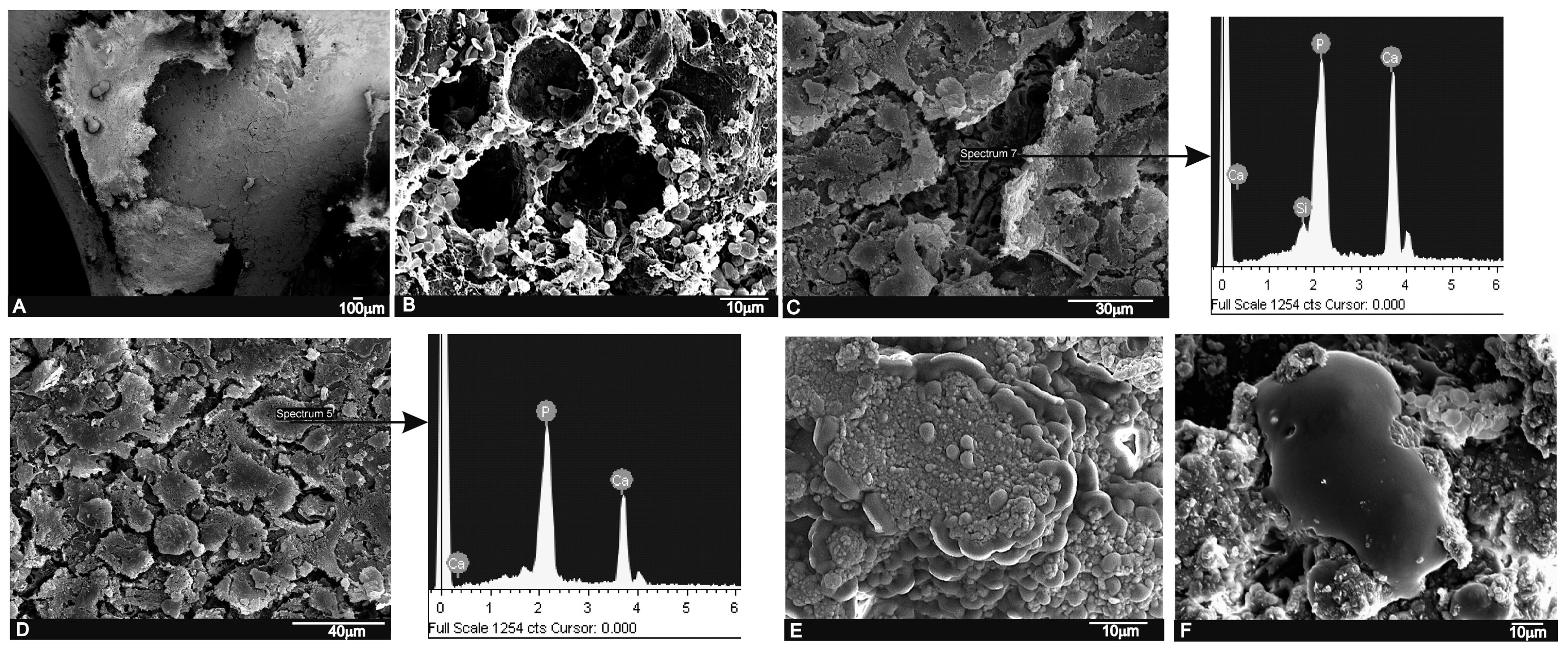
In the present study, the LM, SEM and EDS analyses revealed a close relation between the newly formed bone matrix and the scaffold particle surface. The elemental analysis of bone tissue demonstrated the presence of calcium and phosphorus, which indicated the presence of mineralized bone tissue on the particle surface (
Table 2 and
Table 3). This observation suggests that the scaffold surface could provide an optimal stratum for bone tissue in-growth. The SEM analysis also showed that the new bone matrix had grown over the scaffold surface, and had completely penetrated the deep central zones through its porous structure. After six months of implantation, the scaffold implant had well integrated into the host tissue, and had formed an irregular surface boundary caused by the material’s gradual degradation (
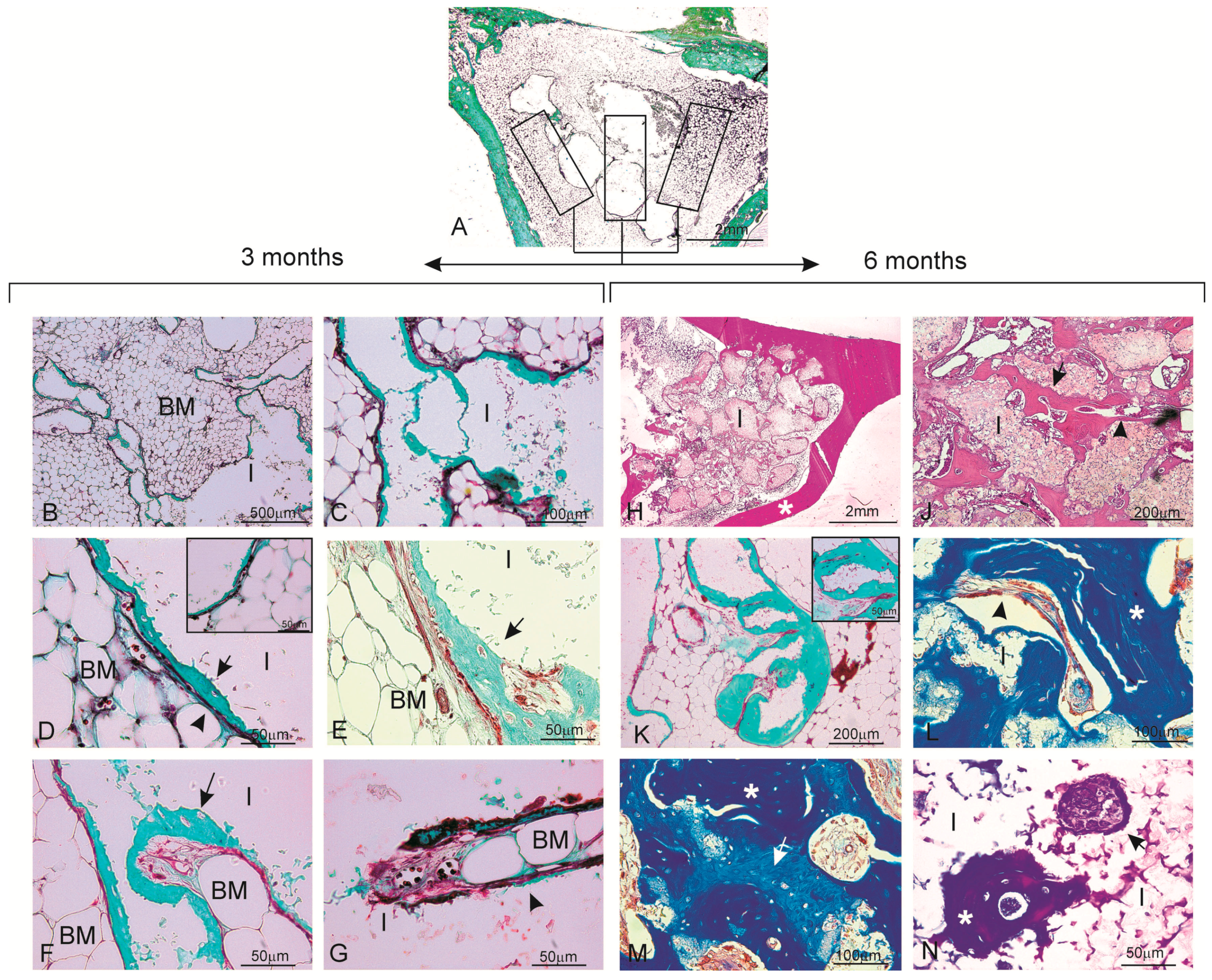
Figure 6 and
Figure 7). The interface developed between the implant and the surrounded tissue was characterized by the intermittent presence of the calcium phosphate phase, which corresponded to new bone tissue in structure and morphology terms.
The new bone ingrowths in the implant were more evident at six months and advanced into the spaces between the exposed scaffold particles in the implant to form a characteristic interlocking pattern at the interface as the process moved further into the implant. SEM (
Figure 7) showed a massive bone colonization of the implant through the original scaffold pores, caused by the structure’s gradual dissolution. Due to these advanced processes, the scaffold material’s free particles were found in many areas across the restructuring implant. Densities at the bone-ceramic interface and inside the material gradually and significantly reduced. This indicates that the resortive process went from the periphery to the center, and it initiated in an early material implantation stage by a cellular mechanism (macrophage cells). At the end of the study (six months), the central part of the implanted material remained partially degraded as it was in the histological and radiological images (
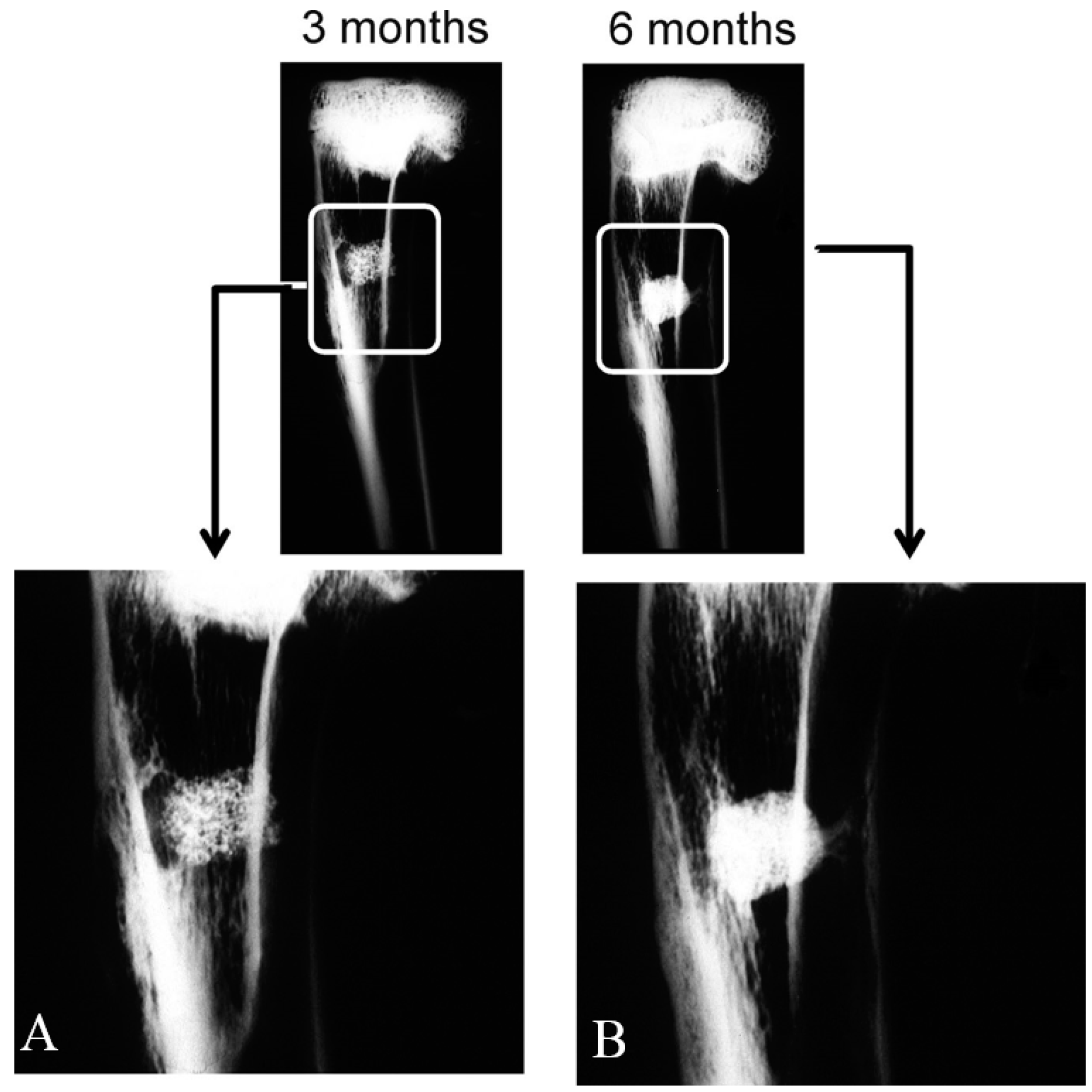
Figure 2 and
Figure 3). However, a lot of new bone was observed in bone defects treated with the scaffold, in which 62.18% ± 2.28% of the bone defect was filled by the newly formed bone after six months of implantation (
Table 2).
We conclude that the results of this initial research confirmed our hypothesis that the high resorbable porous bioactive calcium silicophosphate scaffold has an adequate porosity structure and is able to support bone tissue in-growth by new bone, while gradually being resorbed by the cell-mediated process at the same time. Thus, it constitutes a promising alternative to be used as bone grafts for tissue engineering. Future research should be conducted using other scaffolds made from standard biomaterials, such as Si-HA or Si-TCP. The ceramic’s biological performance should be investigated in different bone defect models and animals, and probably with long-term assays, as proposed in International Standard ISO-10993-5.
4. Materials and Methods
4.1. Biomaterial
In this study porous scaffolds, which corresponded to the 28.39 wt % Nurse’A—71.61 wt % Silicocarnotite composition, were produced by the polymer replication method.
Nurse’s A (7CaOP2O52SiO2) and Silicocarnotite (5CaOP2O5SiO2) ceramic powders synthesized previously in our laboratory were used as starting materials. Details of the technique and the characterization of the starting materials can be found in previous publications [
7,
8,
13]. First, the desired proportions of each component were weighed on an analytical balance and powder was thoroughly attrition-milled in alcoholic media (isopropilic alcohol) using ZrO
2-Y
2O
3 balls (1 mm diameter) for 4 h. The resulting particle size was 2.1 μm measured with laser scattering particle size equipment (Mastersizer, Malvern, UK). Ceramic slurry was prepared with 60% solid contents in water media. Dolapix CE-64 (Zschimmer Schwartz, Lahnstein, Germany) was added as a defloculant (1 wt %) and Optapix PAF-35 (Zschimmer Schwartz, Lahnstein, Germany) as a binder (3 wt %). The powder:water ratio was 60:40.
Scaffolds were prepared using polyurethane sponges with open cells (60 ppi) as a template. Sponges were impregnated with ceramic slurry and sintered at 1450 °C for 2 h with 5 °C/min as the heating and cooling rates. Then, powder was turned off and samples were allowed to cool inside the furnace for 24 h. Next cylindrical scaffolds (5 ± 1 mm diameter and 6 ± 0.3 mm long) were cleaned and washed several times in sterile PBS solution, dried at 37 °C, and finally sterilized by gas plasma (Sterrad-100S, Irvine, CA, USA) and kept in individual packages under sterile conditions until implantation.
The microstructure of the scaffolds was characterized by scanning electron microscopy (SEM-Hitachi S-3500N, Ibaraki, Japan). The chemical composition was qualitatively determined by an Energy Dispersive X-ray Spectroscopy system (EDS-INCA system, by Oxford Instruments Analytical, High Wycombe, UK).
4.2. Animals and Surgical Procedure
Mature male New Zealand (NZ) rabbits (n = 8, 4.0 ± 0.3 kg) were used. Animals were divided and randomly assigned (simple randomization) into two time groups of four animals (n1 = 4, n2 = 4) according to the previously set 3- and 6-month periods. Animals were not placed on a special diet, but received feed and water ad libitum. Animals were housed in individual standard steel cages and maintained in a 12:12 h light dark cycle under controlled environmental conditions. All the animals were allowed 1 week from their arrival to facilitate acclimation.
The study protocol was examined and approved by the Institutional Ethic and Animal Experimentation Committee of the Miguel Hernandez University according to Spanish Government Guidelines and European Community Guidelines for animal care (authorized no. 2014/VSC/PEA/00056 tipo2).
Animals were pre-medicated and anesthetized by an intramuscular (im) injection of Atropine sulfate, 0.3 mg·kg−1 and hydrochloride of clorpromazine, 10 mg·kg−1, and then hydrochlorate of ketamine, 50 mg·kg−1 and xylazine 5 mg·kg−1. As an antibiotic prophylaxis, a single dose of Enrofloxacin 2.5 mg·kg−1 im (Virbac, Barcelona, Spain) was provided. Both legs were shaved and washed with Chlorhexidine® (Bohm SA, Madrid, Spain). Then Betadine® (Meda Manufacturing, Bourdeaux, France) was applied and the surgical area was covered with a sterile drape. Afterward, a longitudinal incision (1.2–1.5 cm long) was made along the medial aspect in the proximal metaphysial area of each tibia. Subcutaneous tissue, fascia and periostium were dissected to expose the medial surface of the tibia. An end-cutting bur (5 mm diameter) was connected to a micromotor at low revolutions with continuous saline irrigation/suction to avoid overheating and thermal damage to bone, and was used to create an unicortical bone defect (5 mm diameter) to avoid invading the medullary cavity. Sufficient hemostasis was achieved. Then, the defect was thoroughly washed several times with physiological saline before performing implantation and removing bone debris to avoid it entering the defect. Porous cylindrical implants were press-fit placed into the defects to ensure initial stability. Identical osseous defect at the contralateral tibia remained empty (ungrafted) as a control.
Afterward, wounds were carefully and closed sutured by a meticulous technique (anatomical layers) with continuous absorbable sutures (Vicryl™ 3/0 (Agatho AB, Lidingo, Sweden) for deep planes and Vicryl Rapid™ 3/0 for skin). Then, a single local anesthetic (0.2 mL·kg−1, chlorhydrate lidocaine 2%, subcutaneously) was provided routinely at the surgery site to avoid immediate postoperative pain. On the first three post-surgery days, animals were given a subcutaneous injection of 0.1 mL·kg−1 of Tolphenamic acide twice a day (every 12 h) as an analgesic control. They were allowed to freely move (limb loading) immediately after restoring from anesthesia. They all survived the 3- and 6-month study periods, and surgical wounds healed with no complications or infection. Therefore, all the animals were included in the study. Total limb loading was allowed after restoring from anesthesia.
At the end of the 3- and 6-month periods, animals were euthanized under sedation (hydrochlorate of ketamine, 50 mg·kg−1 im), with an overdose (0.5 mL) of pentobarbital sodium (Dolethal™, Lab. Vetoquinal, Cedex, France) intracardiacally. To remove the implanted area, the same surgical procedure as that described above was followed.
4.3. Radiographic Imaging
Two X-ray images (standard antero-posterior and lateral projection angles) were obtained of the area of the bone-containing implants, which was cleaned of adherent soft tissue by the Kodak RVG 6100 Digital Radiography System (Kodak DS, Rochester, NY, USA) with an X-ray taken at 32 kV, 40 mA by automatic light metering. Radiographs of all the specimens were taken. Images were used to observe changes in the morphology and radiological density (radiopacity) descriptive levels of the material in the medullar and cortical areas where defects were created.
4.4. Histological and Histomorphometric Analysis
After 3 and 6 months, the implants together with the surrounding tissues were removed and fixed in 10% neutral buffered formalin and decalcified. The decalcification method utilized Osteomoll Merck KbaA (Darmstadt, Germany) that contained HCl (10%) and CH2O (4%), immersing samples for 17 days, and the solution was renewed every 24 h. Subsequently, all the samples were paraffin embedded, sectioned at 5 μm, and stained using hematoxylin-eosin (H-E, red stain) and Masson’s trichrome (MT) stain. The entire circumference of each section (containing bone, scaffold particles, and connective tissue) was traced manually to create an individual region of interest (ROI).
Histomorphometric evaluations consisted of taking measurements of the area of material in relation to the total measurement area. These were carried out using Image J software (developed by the National Institute of Health (NIH), Bethesda, MD, USA). Examinations were performed under a Nikon Elipse 80i microscope (Teknooptik AB, Huddinge, Sweden), equipped with an Easy Image 2000 system (Teknooptik AB, Huddinge, Sweden). Images were generated using a Leica Z6 APO microscope connected to a Leica DC 500 (Barcelona, Spain) digital camera. After calibrating the system and digitalizing images, interactive measurements of the areas of interest were obtained with the Leica QWin V3 image analysis software (Barcelona, Spain). The histomorphometric analysis produced one BIC measurement, measured as the percentage of the circumference, and length of the cylinder that came into contact with new bone. In the same way, the cortical bone defect in the control group was also evaluated.
4.5. Scanning Electron Microscopy Study
To assess the continuing effect of the implant in the medullary cavity surrounded by hematopoietic bone marrow from an ultrastructural point of view, cross-sections of the non-decalcified tissues were also examined for the ultrastructural study in SEM-EDS. Therefore, some sections (1–2 mm) in the implantation area were fixed by immersion in 3% glutaraldehyde after being soaked in buffer solution for 4 h, postfixed in 1% osmium tetroxide for 1 h at 4 °C, and washed in 0.1 M cacodilate solution and dehydrated in graded ethanol solutions series, and embedded in hydroxyethyl methacrylate resin. They were then polished with 1-μm diamond pastes for the SEM-EDS analyses. Back-scattered SEM imaging was employed to highlight the contrasts among the resin, bone and the biomaterial.
4.6. Statistical Analysis
A statistical analysis was performed with the PASW Statistics v.20.0.0 software (SPSS Inc., Armonk, NY, USA). Sample size was pre-calculated using the statistical method provided by the software. Values were recorded as means ± standard deviation and medians. The pre-statistical analysis of sample distribution was performed to evaluate normality. Kolmogorov and Smirnov’s test values were normal. A non-parametric Friedman Test for the related samples was applied to the comparison of the medians and to quantify any relationships between differences (p < 0.05).
5. Conclusions
The biocompatibility of this novel porous calcium silicophosphate, developed by the polymer replication method, was high and caused no local or systemic immune inflammatory response, and no fibrosis was developed between the ceramic and bone after its intramedullary implantation into a rabbit tibia.
The results indicate that this material provides an optimal microenvironment for the osteogenic differentiation of the undifferentiated osteoblastic precursor cells contained in hematopoietic bone marrow. Their suitable interconnected network porous structure facilitates colonization by new bone tissue and bone marrow. Presence of the vascular capillaries that surround the material promotes this process by providing all the necessary nutrients.
This confirms that the porous calcium silicophosphate obtained in this work favors and supports the integration of bone by combining osteoconductive behavior with the enhanced bone tissue in-growth of the open-pore structure. The dynamic biodegradation of the ceramic with time was also documented. Densities were reduced throughout the study as a result of the simultaneous phagocytic activity of macrophages.
Finally, we therefore conclude that this novel porous calcium silicophosphate ceramic scaffold implanted into the medullary cavity is biocompatible, bioresorbable, osteoconductive and has osteogenic potential. Hence, it can be considered a potential substitute of bone tissue, and is suitable for clinical applications in filling bone defects and being used as a scaffold or matrix for bone tissue engineering.