Assessing Environmental Risks for Established Invasive Weeds: Dalmatian (Linaria dalmatica) and Yellow (L. vulgaris) Toadflax in North America
Abstract
:1. Introduction
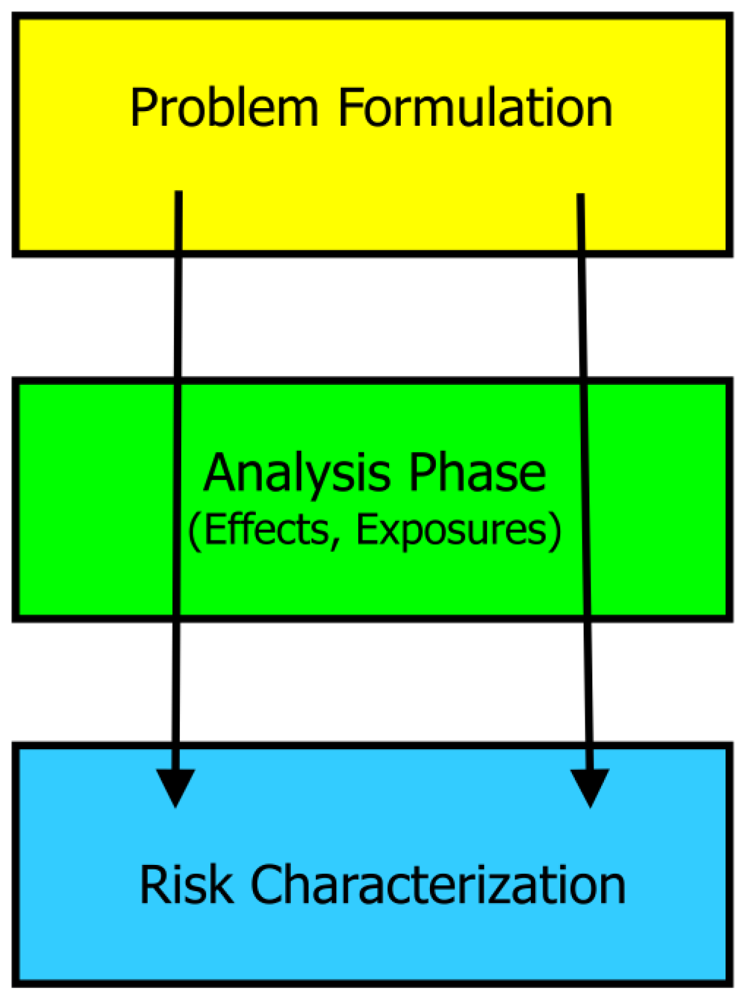
2. Approach
2.1. Problem Formulation
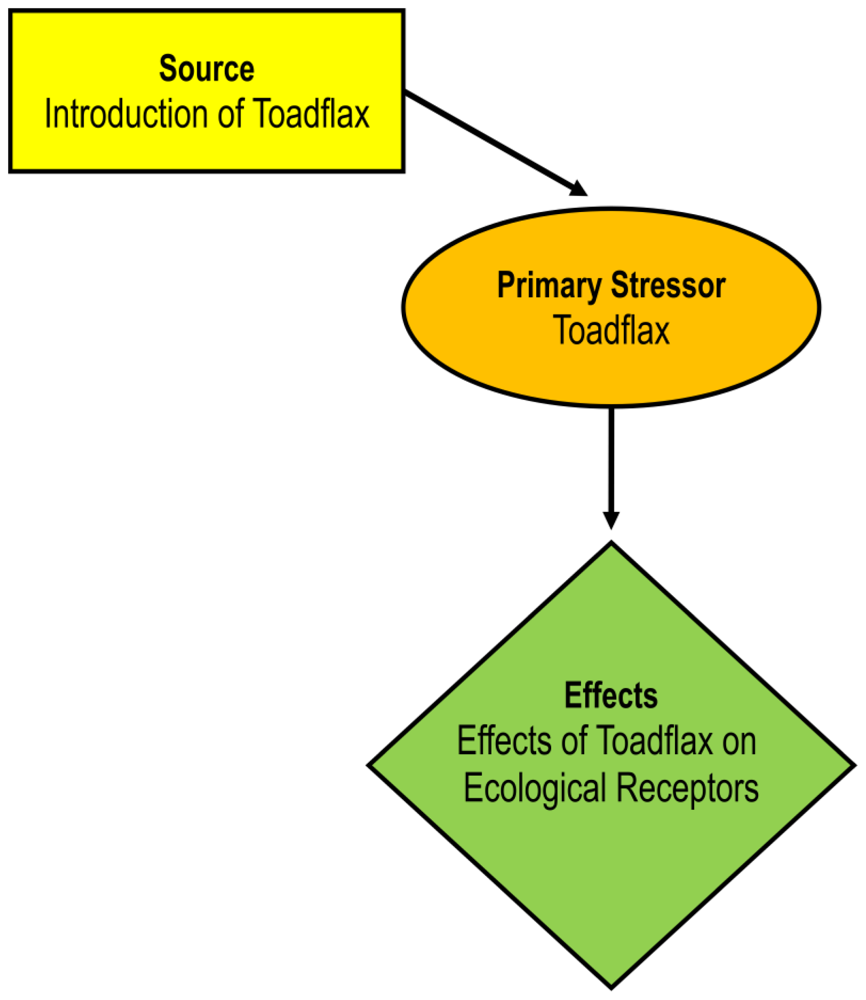
2.2. Description of the Primary Stressors
Species descriptions
Physiological range
Geographic range
North American invasion history
Hybridization
Biology/life cycle
Phytochemistry
2.3. Effects Assessment
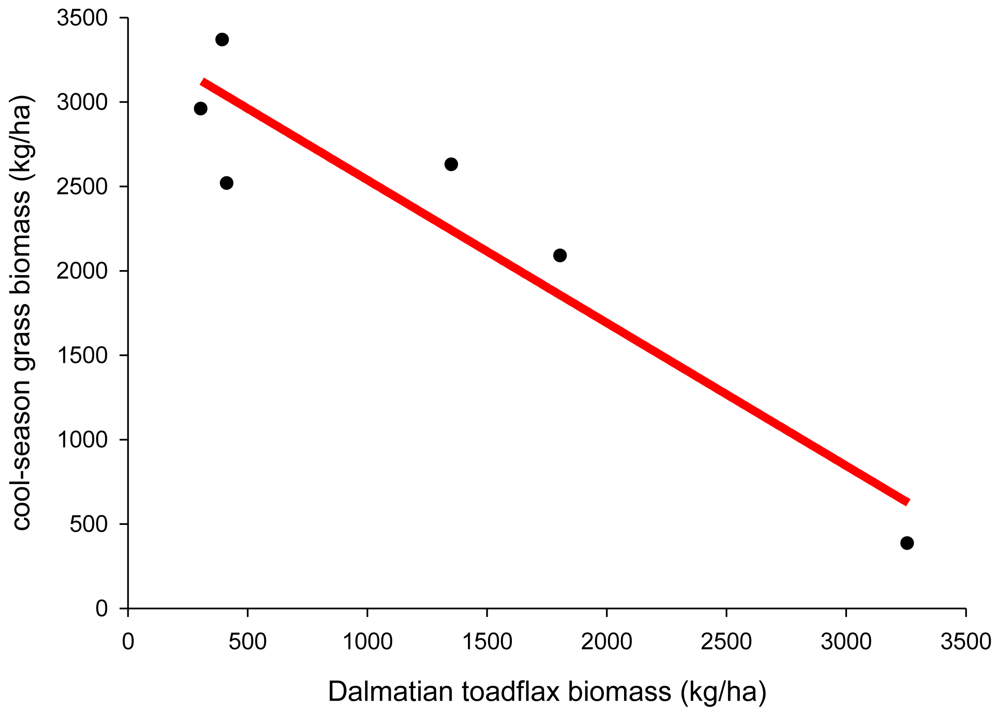
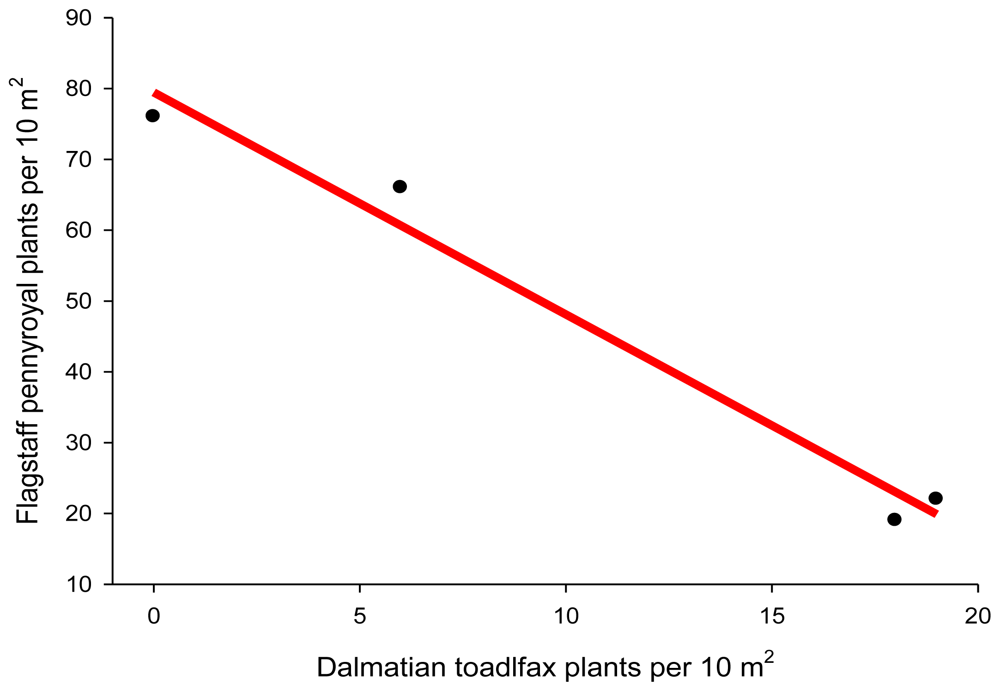
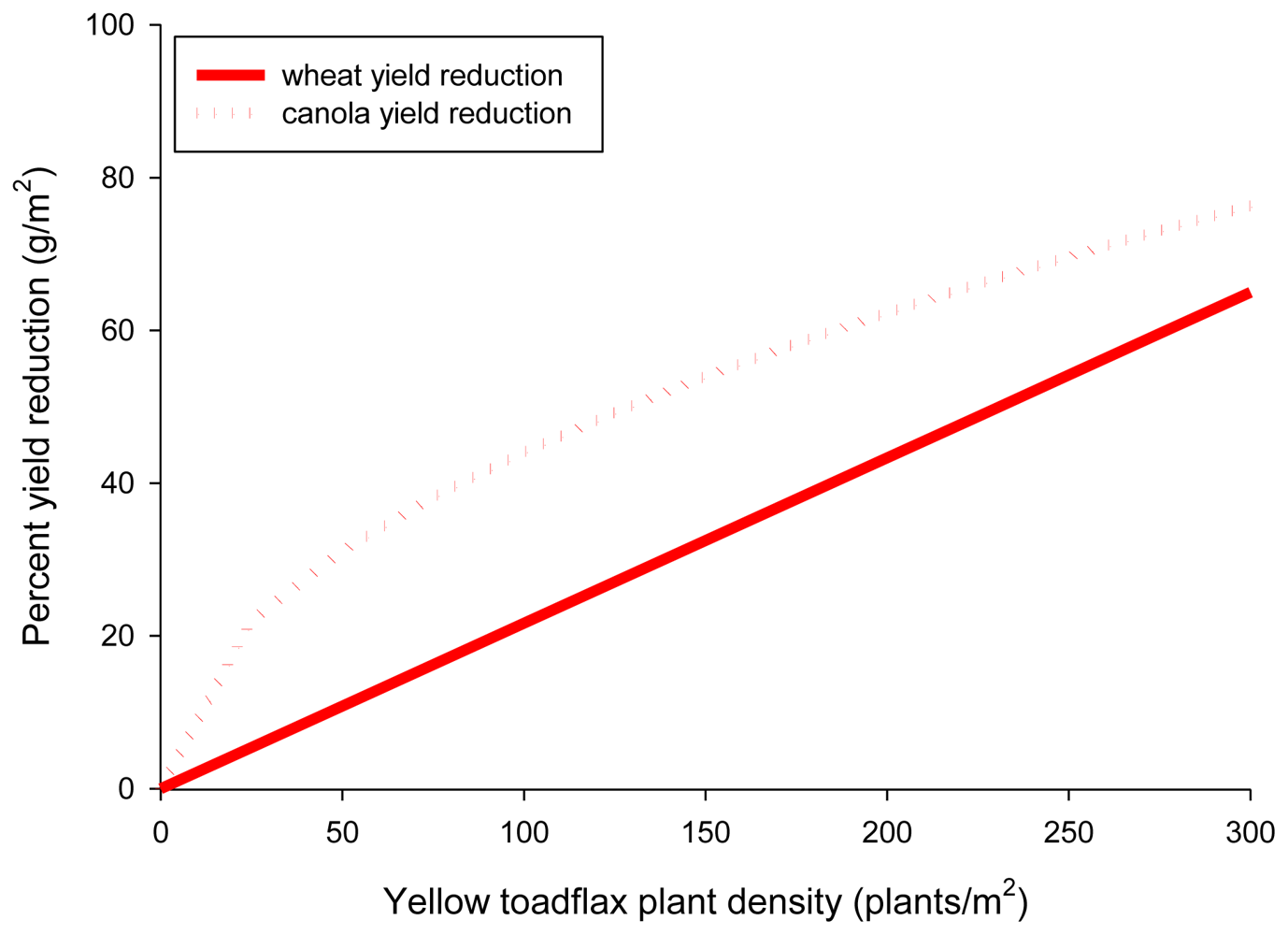
Plant displacement
Reservoirs of plant disease
Food resource and shelter—animals
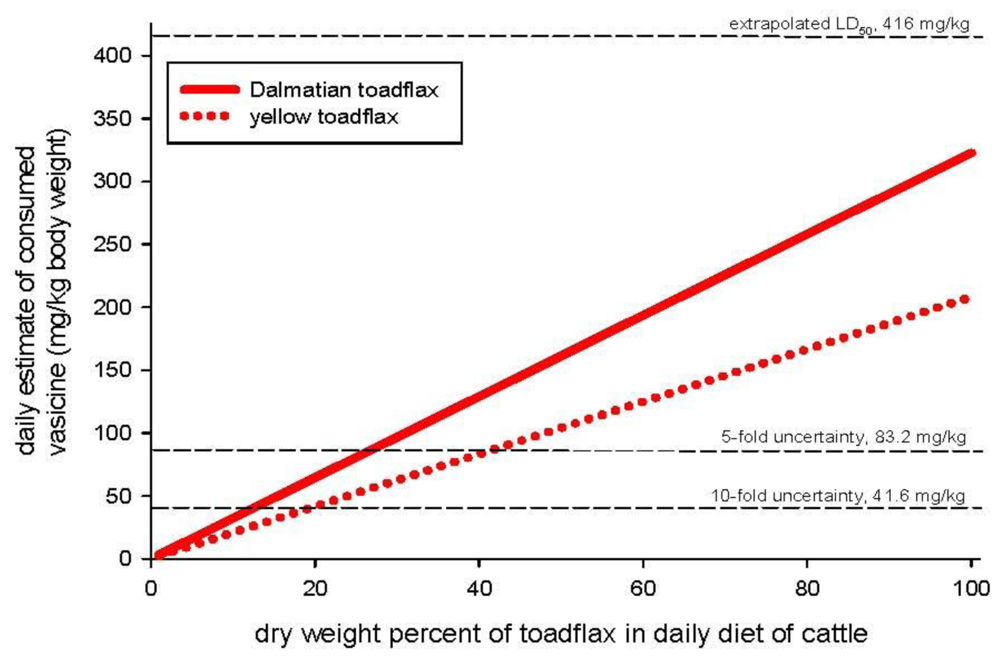
Animal toxicity
Toxicity of crude plant material
Quinazoline alkaloids
Flavonoids
Iridoid glycosides
Human toxicity, allergenicity, and pharmacology
Erosion
Wildfire
2.4. Exposure Assessment
Plant displacement
Exposure to plant disease
Animal exposure
Quinazoline alkaloids
Flavonoids
Iridoid glycosides
Human exposure
2.5. Risk Characterization
2.6. Uncertainties
Acknowledgements
References
- McClay, A; Sissons, A; Wilson, C; Davis, S. Evaluation of the Australian weed risk assessment system for the prediction of plant invasiveness in Canada. Biol. Invasions 2010, 12, 4085–4098. [Google Scholar]
- Gordon, DR; Onderdonk, DA; Fox, AM; Stocker, RK. Consistent accuracy of the Australian weed risk assessment system across varied geographies. Divers. Distrib 2008, 14, 234–242. [Google Scholar]
- Pheloung, PC; Williams, PA; Halloy, SR. A weed risk assessment model for use as a biosecurity tool evaluating plant introductions. J. Environ. Manag 1999, 57, 239–251. [Google Scholar]
- SAAPCC, Weed Assessment Guide; South Australia Animal and Plant Control Commission: Adalaide, South Australia, 2000; p. 12.
- Richardson, DM; van Wilgen, BW. Invasive alien plants in South Africa: How well do we understand the ecological impacts? S. Afr. J. Sci 2004, 100, 45–52. [Google Scholar]
- USOSTP, Ecological Risk Assessment in the Federal Government, CENR/5-99/001; Committee on Environment and Natural Resources of the National Science and Technology Council: Washington, DC, USA, 1999; p. 219.
- NRC, Risk Assessment in the Federal Government: Managing the Process; National Research Council, National Academy Press: Washington, DC, USA, 1983.
- Keller, RP; Lodge, DM; Finnoff, DC. Risk assessment for invasive species produces net bioeconomic benefits. Proc. Natl. Acad. Sci. USA 2007, 104, 203–207. [Google Scholar]
- Alex, JF. The taxonomy, history, and distribution of Linaria dalmatica. Can. J. Bot 1962, 40, 295–307. [Google Scholar]
- Robocker, WC. Life History, Ecology, and Control of Dalmatian Toadflax; Technical Bulletin 79; Washington Agricultural Experiment Station, Washington State University: Pullman, WA, USA, 1974; p. 20. [Google Scholar]
- Vujnovic, K; Wein, RW. The biology of Canadian weeds. 106. Linaria dalmatica (L.) Mill. Can. J. Plant Sci 1997, 77, 483–491. [Google Scholar]
- Rice, PM. INVADERS Database System; Division of Biological Sciences, University of Montana: Missoula, MT, USA, 2011. Available online: http://invader.dbs.umt.edu accessed on 19 June 2011.
- USDA, NRCS. The PLANTS Database; National Plant Data Team: Greensboro, NC, USA, 2011. Available online: http://plants.usda.gov accessed on 19 June 2011.
- USDA, ARS, National Genetic Resources Program. Germplasm Resources Information Network (GRIN); National Germplasm Resources Laboratory: Beltsville, MD, USA, 2011. Available online: http://www.ars-grin.gov accessed on 19 June 2011.
- ITIS. Scrophulariaceae—Taxonomic Serial No. 33005; Integrated Taxonomic Information System, 2011. Available online: http://www.itis.gov accessed on 19 June 2011.
- Tutin, TG; Heywood, VH; Burges, NA; Moore, DM; Valentine, DH; Walters, SM; Webb, DA. Flora Europaea; Volume 3, Cambridge University Press: London, UK, 1972. [Google Scholar]
- Saner, MA; Clements, DR; Hall, MR; Doohan, DJ; Crompton, CW. The biology of Canadian weeds. 105. Linaria vulgaris Mill. Can. J. Plant Sci 1995, 75, 525–537. [Google Scholar]
- Albach, DC; Meudt, HM; Oxelman, B. Piecing together the “new” Plantaginaceae. Am. J. Bot 2005, 92, 297–315. [Google Scholar]
- Olmstead, RG; DePamphilis, CW; Wolfe, AD; Young, ND; Elisons, WJ; Reeves, PA. Disintegration of the Scrophulariaceae. Am. J. Bot 2001, 88, 348–361. [Google Scholar]
- Ghebrehiwet, M; Bremer, B; Thulin, M. Phylogeny of the tribe Antirrhineae (Scrophulariaceae) based on morphological and ndhF sequence data. Plant Syst. Evol 2000, 220, 223–239. [Google Scholar]
- Kartesz, JT. Scrophulariaceae (the Figwort Family); The Biota of North America Program (BONAP)—Texas A&M University Bioinformatics Working Group: Chapel Hill, NC, USA, 1996. Available online: http://www.csdl.tamu.edu/FLORA/bonapfams/bonxxscr.htm accessed on 19 June 2011.
- Allen, K; Hansen, K. Geography of exotic plants adjacent to campgrounds, Yellowstone National Park, USA. Great Basin Nat 1999, 59, 315–322. [Google Scholar]
- Sutton, J; Stohlgren, T; Beck, K. Predicting yellow toadflax infestations in the Flat Tops Wilderness of Colorado. Biol. Invasions 2007, 9, 783–793. [Google Scholar]
- Pauchard, A; Alaback, PB; Edlund, EG. Plant invasions in protected areas at multiple scales: Linaria vulgaris (Scrophulariaceae) in the West Yellowstone area. West. N. Am. Nat 2003, 63, 416–428. [Google Scholar]
- Leeson, JY; Thomas, AG; Beckie, HJ; Hall, LM; Brenzil, C; Van Acker, RC; Brown, KR; Andrews, T. Group 2 Herbicide Use in the Prairie Provinces. Proceedings of 2004 Canadian Weed Science Society (CWSS) Annual Conference, Winnipeg, MB, Canada, 28–30 November 2004, Turnbull, G, Ed.; CWSS: Sainte-Anne-de-Bellevue, QC, Canada, 2005. [Google Scholar]
- Davidson, A; Moxley, GL. Flora of Southern California; Times-Mirror Press: Los Angeles, CA, USA, 1923. [Google Scholar]
- Macoun, WT. List of Herbaceous Perennials Tested in the Arboretum and Botanic Garden, Central Experimental Farm, Ottawa, Canada with Descriptions of Flowers, and Other Notes. Can. Bull. 5, Second Series; Government Printing Bureau: Ottawa, ON, Canada, 1908. [Google Scholar]
- Mack, RN. Plant naturalizations and invasions in the eastern United States: 1634–1860. Ann. Mo. Bot. Gard 2003, 90, 77–90. [Google Scholar]
- Lajeunesse, S. Dalmatian and Yellow Toadflax. In Biology and Management of Noxious Rangeland Weeds; Sheley, RL, Petroff, JK, Eds.; Oregon State University Press: Corvallis, OR, USA, 1999; pp. 202–216. [Google Scholar]
- Pennell, FW. The Scrophulariaceae of eastern temperate North America. Acad. Nat. Sci. Philadelphia Monogr 1935, 1, 297–312. [Google Scholar]
- Mitich, LW. Yellow toadflax. Weed Technol 1993, 7, 791–793. [Google Scholar]
- Rousseau, C. Histoire, habitat et distribution de 220 plantes introduites au Quebec. Nat. Can 1968, 95, 49–169. [Google Scholar]
- Chater, AO; Valdés, B; Webb, DA. Linaria Miller. In Flora Europeae; Tutin, TG, Heywood, VH, Burges, NA, Moore, DM, Valentine, DH, Walters, SM, Webb, DA, Eds.; Cambridge University Press: Cambridge, UK, 1972; Volume 3, pp. 226–236. [Google Scholar]
- Jankó, B. Variation-pattern investigations in natural Linaria populations. Acta Bot 1964, 10, 257–274. [Google Scholar]
- Bruun, HG. Genetical notes on Linaria, I-II. Hereditas 1937, 22, 395–401. [Google Scholar]
- Docherty, Z. Self-incompatibility in Linaria. Heredity 1982, 49, 349–352. [Google Scholar]
- Olsson, U. A biometric study of the pollen morphology of Linaria vulgaris (L.) Miller and L. repens (L.) Miller (Schrophulariaceae) and their hybrid progeny in F1 and F2 generations. Grana 1974, 14, 92–99. [Google Scholar]
- Olsson, U. On the hereditary transmission of microstructures of testa in reciprocal crosses between Linaria vulgaris and L. repens. Hereditas 1975, 79, 251–254. [Google Scholar]
- Ward, SM; Fleischmann, CE; Turner, MF; Sing, SE. Hybridization between invasive populations of Dalmatian toadflax (Linaria dalmatica) and yellow toadflax (Linaria vulgaris). Invasive Plant Sci. Mana 2009, 2, 369–378. [Google Scholar]
- Arnold, RM. Pollination, predation and seed set in Linaria vulgaris (Scrophulariaceae). Am. Midlands Nat 1982, 107, 360–369. [Google Scholar]
- Nadeau, LB; King, JR. Seed dispersal and seedling establishment of Linaria vulgaris Mill. Can. J. Plant Sci 1991, 71, 771–782. [Google Scholar]
- Robocker, WC. Seed characteristics and seedling emergence of Dalmatian toadflax. Weed Sci 1970, 18, 720–725. [Google Scholar]
- Lajeunesse, SE; Fay, PK; Cooksey, D; Lacey, JR; Nowierski, RM; Zamora, D. Dalmatian and Yellow Toadflax: Weeds of Pasture and Rangeland; Extension Service, Montana State University: Bozeman, MT, USA, 1993. [Google Scholar]
- Clements, DR; Cavers, PB. Seasonal seed viability patterns and the role of incomplete seed development in the life history strategy of Linaria vulgaris. Nat. Can 1990, 117, 189–198. [Google Scholar]
- Rose, KK; Whitson, TD; Koch, DW. The Competitive Effects of Five Cool-Season Grasses on Dalmatian Toadflax (Linaria Genistifolia Ssp. Dalmatica). Proceedings of the Western Society of Weed Science, Colorado Springs, CO, USA, 8–11 March 1999, Christianson, K, Ed.; WSWS: Colorado Springs, CO, USA; pp. 64–67.
- Coupland, RT; Zilke, S; Selleck, GW. Spread of toadflax in Saskatchewan. Can. J. Plant Sci 1963, 43, 214–221. [Google Scholar]
- Bakshi, TS; Coupland, RT. Vegetative propagation in Linaria vulgaris. Can. J. Bot 1960, 38, 243–249. [Google Scholar]
- Charlton, WA. The root system of Linaria vulgaris Mill. I. Morphology and anatomy. Can. J. Bot 1966, 44, 1111–1116. [Google Scholar]
- Nadeau, LB; King, JR; Harker, KN. Comparison of growth of seedling and plants grown from root peices of yellow toadflax (Linaria vulgaris). Weed Sci 1992, 40, 43–47. [Google Scholar]
- Zilke, S; Coupland, RT. The effect of one cultivation on the density of toadflax (Linaria vulgaris Mill.). In Res. Rep. National Weed Committee (West. Sec.); Canada Department Agriculture: Ottawa, ON, Canada, 1954; p. 105. [Google Scholar]
- Nadeau, LB; Dale, MRT; King, JR. The development of spatial pattern in shoots of Linaria vulgaris (Scrophulariaceae) growing on fallow land or in a barley crop. Can. J. Bot 1991, 69, 2539–2544. [Google Scholar]
- Openshaw, HT. The quinazoline alkaloids. Alkaloids 1953, 3, 101–118. [Google Scholar]
- Gröger, D; Johne, S. Üeber das vorkommen von paganin in Linaria arten. Planta Med 1965, 13, 182–188. [Google Scholar]
- Harkiss, KJ. Studies in the Scrophulariaceae. Part VI Quantitative determination of choline in Antirrhinum majus L., A. orontium L., and Linaria vulgaris Mill. Planta Med 1972, 21, 353–357. [Google Scholar]
- Johne, S. Quinazoline alkaloids. In The Alkaloids; Brossi, A, Ed.; Academic Press: New York Ny, USA, 1986; Volume 29, pp. 99–140. [Google Scholar]
- D’yakonov, AL; Telezhenetskaya, MV. Quinazoline alkaloids in nature. Chem. Nat. Compd 1997, 33, 221–267. [Google Scholar]
- Hua, H; Cheng, M; Li, X; Pei, Y. A new pyrroloquinazoline alkaloid from Linaria vulgaris. Chem. Pharm. Bull 2002, 50, 1393–1394. [Google Scholar]
- Bartik, M; Piskac, A. Veterinary Toxicology; Elsevier Scientific: Amsterdam, The Netherlands, 1981. [Google Scholar]
- Smirnova, LP; Boryaev, KI; Ban’kovskii, AI. Acacetin and its glycosides in plants of the genus Linaria. Chem. Nat. Compd 1974, 10, 100. [Google Scholar]
- Kiryanov, GI; Manamshjan, TA; Polyakov, VY; Fais, D; Chentsov, JS. Levels of granular organization of the chromatin fiber. FEBS Lett 1976, 67, 323–327. [Google Scholar]
- Pethes, E; Marczal, G; Kery, A; Petho, M. Flavonoids as biologically active agents and their occurrence in the Scrophulariaceae family. Acta pharm. Hung 1974, 44, 83–89. [Google Scholar]
- Valdés, B. Flavonoid pigments in flower and leaf of the genus Linaria (Scrophulariaceae). Phytochemistry 1970, 9, 1253–1260. [Google Scholar]
- Sticher, O. Isolation of antirrhinoside from Linaria vulgaris. Phytochem. Rep 1971, 10, 1974–1975. [Google Scholar]
- Bianco, A; Guiso, M; Ballero, M; Foddai, S; Nicoletti, M; Piccin, A; Serafini, M; Tomassini, L. Glycosidic monoterpenes from Linaria capraria. Nat. Prod. Res 2004, 18, 241–246. [Google Scholar]
- Serafini, M; Foddai, S; Ballero, M; Guiso, M; Bianco, A. The occurrence of iridoid glycosides in Cymbalaria spp. in Italy. Nat. Prod. Res 2004, 18, 325–327. [Google Scholar]
- Bowers, MD; Collinge, SK. Fate of iridoid glycosides in different life stages of the Buckeye, Junonia coenia (Lepidoptera: Nymphalidae). J. Chem. Ecol 1992, 18, 817–831. [Google Scholar]
- Jamieson, MA; Bowers, MD. Iridoid glycoside variation in the invasive plant Dalmatian toadflax, Linaria dalmatica (Plantaginaceae), and sequestration by the biological control agent, Calophasia lunula. J. Chem. Ecol 2010, 36, 70–79. [Google Scholar]
- Baig, MN; Darwent, AL; Harker, KN; O’Donovan, JT. Preharvest applications of glycophosate for yellow toadflax (Linaria vulgaris) control. Weed Technol 1999, 13, 777–782. [Google Scholar]
- Harker, NK; O’Donovan, JT; Blackshaw, RE. Integrated management of yellow toadflax in a zero tillage regime. In Bringing Conservation Technology to the Farm; Lafond, GP, Plas, HM, Smith, EG, Eds.; Proceedings of the Conservation Workshop, Saskatoon, SK, Canada, 1995; pp. 79–82. [Google Scholar]
- Darwent, AL; Lobay, W; Yarish, W; Harris, P. Distribution and importance in northwestern Alberta of toadflax and its insect enemies. Can. J. Plant Sci 1975, 55, 157–162. [Google Scholar]
- O’Donovan, JT; McClay, AS. Influence of various densities of toadflax on yield of canola. Expert Comm. Weeds Res. Rep. (Western Canada Section) 1987, 191–192. [Google Scholar]
- O’Donovan, JT; Newman, JC. Influence of toadflax on yield of wheat. Expert Comm. Weeds Res. Rep. (Western Canada Section) 1989, 3, 201. [Google Scholar]
- Volenberg, DS; Hopen, HJ; Campobasso, G. Biological control of yellow toadflax (Linaria vulgaris) by Eteobalea serratella in peppermint (Mentha piperita). Weed Sci 1999, 47, 226–232. [Google Scholar]
- McClay, AS; De Clerck-Floate, RA. Linaria vulgaris Miller, yellow toadflax (Scrophulariaceae). In Biological Control Programmes in Canada 1981–2000; Mason, PG, Huber, JT, Eds.; CABI Publishing: New York, NY, USA, 2002; pp. 375–382. [Google Scholar]
- Rist, DL; Lorbeer, JW. Occurrence and overwintering of cucumber mosaic virus and broad bean wilt virus in weeds growing near commercial lettuce fields in New York. Am. Phytopathol. Soc 1989, 79, 65–69. [Google Scholar]
- Francki, RIB; Mossop, DW; Hatta, T. Cucumber mosaic virus. CMI/AAB Descriptions of Plant Viruses, No. 213; Commonwealth Mycological Institute: Kew, UK, 1979. [Google Scholar]
- Olson, BE. Manipulating diet selection to control weeds. In Grazing Behavior of Livestock and Wildlife; Launchbaugh, KL, Sanders, KD, Mosley, JC, Eds.; University of Idaho: Moscow, ID, USA, 1999. [Google Scholar]
- Harbourne, JB; Grayer, RJ. Flavonoids and insects. In The Flavonoids: Advances in Research Since 1986; Harborne, JB, Ed.; Chapman and Hall: London, UK, 1994; pp. 589–618. [Google Scholar]
- Saxena, BP; Tikku, K; Atal, CK; Koul, O. Insect antifertility and antifeedant allelochemics in Adhatoda vasica. Insect Sci. Appl 1986, 7, 489–493. [Google Scholar]
- Kokate, CK; D’Cruz, JL; Kumar, A; Apte, SS. Anti-insect and juvenoidal activity of phytochemicals derived from Adhatoda vasica Nees. Indian J. Nat. Prod 1985, 1, 7–9. [Google Scholar]
- DeClerck-Floate, R; Richards, KW. Pollination Ecology and Biological Control: Developing Release Strategies for Seed-Feeding Insects on Dalmatian Toadflax. In Proceedings of the International Symposium on Pollination, Acta Horticulturae 437; Richards, KW, Ed.; International Society for Horticultural Science: Leuven, Belgium, 1997; pp. 379–381. [Google Scholar]
- Polunin, O. Flowers of Europe, a Field Guide; Oxford University Press: London, UK, 1969. [Google Scholar]
- Provenza, FD. Post-ingestive feedback as an elementary determinant of food preference and intake in ruminants. J. Range Manag 1995, 48, 2–17. [Google Scholar]
- Burrows, GE; Tyrl, RJ. Scrophulariaceae Juss. In Toxic Plants of North America; Iowa State University Press: Ames, IA, USA, 2001; Volume 68, pp. 1092–1103. [Google Scholar]
- Weathers, SA. Field Guide to Plants Poisonous to Livestock: Western U.S; Rosebud Press: Fruitland, UT, USA, 1998. [Google Scholar]
- Karimova, SG; Smirnova, SG; Nasyrov, KM. Chemical composition and pharmacology of the Scrophulariaceae. From Ref. Zh., Farmakol. Khimoter. Sredstva. Toksikol.1967. Chem. Abst 1967, 67, 20384–20385. [Google Scholar]
- Zilke, S; Bell, JM; Coupland, RT. Toxicity Tests of Artificial Dried Leaves and Stems of Toadflax (Linaria Vulgaris Hill). In Res. Rep. National Weed Committee (West. Sec.); Canada Department of Agriculture: Ottawa, ON, Canada, 1954; p. 107. [Google Scholar]
- Atal, CK. Chemistry and Pharmacology of Vasicine—An Oxytocic and Abortifacient; Director, Regional Research Laboratory, Jammu-Tawi: New Delhi, India, 1980; p. 58. [Google Scholar]
- Claeson, UP; Malmfors, T; Wikman, G; Bruhn, JG. Adhatoda vasica: A critical review of ethnopharmacological and toxicological data. J. Ethnopharmacol 2000, 72, 1–20. [Google Scholar]
- Rancha, B; Sujata, P; Mamta, KM; Priyanka Sonam, S. Review and future perspectives of using vasicine, and related compounds. Indo-Glob. J. Pharm. Sci. 2011, 1, 85–98. [Google Scholar]
- Engelhorn, R; Puschmann, S. Pharmakologische untersuchungen u ber eine substanz mit sekretolytischer wirkung. Arzneim-Forsch 1963, 13, 474–480. [Google Scholar]
- Burgos, R; Forcelledo, M; Wagner, H; Muller, A; Hancke, J; Wikman, G; Croxatto, H. Non-abortive effect of Adhatoda vasica spissum leaf extract by oral administration in rats. Phytomedicine 1997, 4, 145–149. [Google Scholar]
- Fernandez, SP; Wasowski, C; Loscalzo, LM; Granger, RE; Johnston, GAR; Paladini, AC; Marder, M. Central nervous system depressant action of flavonoid glycosides. Eur. J. Pharmacol 2006, 539, 168–176. [Google Scholar]
- Sigma-Aldrich, Quercetin: MSDS No. Q0125; Sigma-Aldrich: Saint Louis, MO, USA, January 31 2006.
- Sigma-Aldrich, Acacetin: MSDS No. 00017; Sigma-Aldrich: Saint Louis, MO, USA, February 6 2006.
- Hiremath, SP; Rao, SH. Antifertility efficacy of the plant Striga lutea (Scrophulariaceae) on rats. Contraception 1990, 42, 466–477. [Google Scholar]
- Pammel, LH. Manual of Poisonous Plants; Torch Press: Cedar Rapids, MI, USA, 1911. [Google Scholar]
- Hruska, K. Assessment of urban allergophytes using an allergen index. Aerobiologia 2003, 19, 107–111. [Google Scholar]
- JCNM, A dictionary of the traditional Chinese medicines; Jiangsu College of New Medicine: Beijing, China, 1977.
- Zabeer, A; Bhagat, A; Gupta, OP; Singh, GD; Youssouf, MS; Dhar, KL; Suri, OP; Suri, KA; Satti, NK; Gupta, BD; et al. Synthesis and bronchodilator activity of new quinazolin derivative. ChemInform 2006, 37, 429–434. [Google Scholar]
- Gupta, OP; Sharma, ML; Ghatak, BJR; Atal, CK. Pharmacological investigations of vasicine and vasicinone the alkaloids of Adhatoda vasica. Indian J. Med. Res 1977, 66, 680–691. [Google Scholar]
- Amin, AH; Mehta, DR. Bronchodilator alkaloid (vasicinone) from Adhatoda vasica. Nature 1959, 184, 1317–1317. [Google Scholar]
- Gupta, OP; Sharma, ML; Rayghatak, BJ; Atal, CK. Potent uterine activity of the alkaloid vasicine. Indian J. Med. Res 1977, 66, 865–871. [Google Scholar]
- Gupta, OP; Anand, KK; Ghatak, BJR; Atal, CK. Vasicine alkaloid of Adhatoda vasica, a promising utertonic abortifacient. Indian J. Exp. Biol 1978, 16, 1075–1077. [Google Scholar]
- Patel, PK; Bhatt, PH. In vitro study of antimicrobial activity of Adhatoda vasica (leaf extract) on gingival inflammation. Ind. J. Med. Sci 1984, 38, 70–72. [Google Scholar]
- Mathew, AS; Patel, KN. Investigation on antifeedant and anthelmientic potential of Adhatoda vasica Nees. Indian J. Nat. Prod 1998, 14, 11–16. [Google Scholar]
- Fernandez, S; Wasowski, C; Paladini, AC; Marder, M. Sedative and sleep-enhancing properties of linarin, a flavonoid-isolated from Valeriana officinalis. Pharmacol. Biochem. Behav 2004, 77, 399–404. [Google Scholar]
- Oinonen, PP; Jokela, JK; Hatakka, AI; Vuorela, PM. Linarin, a selective acetylcholinesterase inhibitor from Mentha arvensis. Fitoterapia 2006, 77, 429–434. [Google Scholar]
- Peterson, J; Dwyer, J. Flavonoids: dietary occurrence and biochemical activity. Nutr. Res 1998, 18, 1995–2018. [Google Scholar]
- Jacobs, JS; Sheley, RL. Prescribed fire effects on Dalmation toadflax. J. Range Manag 2003, 56, 193–197. [Google Scholar]
- Zouhar, K. Bromus Tectorum; Fire Effects Information System; U.S. Department of Agriculture Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory: Missoula, MT, USA, 2003. Available online: http://www.fs.fed.us/database/feis/plants/graminoid/brotec/all.html accessed on 19 June 2011.
- Maxwell, BD; Lehnhoff, EA; Rew, LJ. The rationale for monitoring invasive plant populations as a crucial step for management. Invasive Plant Sci. Manag 2009, 1, 1–9. [Google Scholar]
- Gates, DH; Robocker, WC. Revegetation with adapted grasses in competition with Dalmatian toadflax and St. Johnswort. J. Range Manag 1960, 13, 322–326. [Google Scholar]
- Phillips, BG; Crisp, D. Dalmatian Toadflax, an Invasive Exotic Noxious Weed, Threatens Flagstaff Pennyroyal Community Following Prescribed Fire. Southwestern Rare and Endangered Plants: Proceedings of the Third Conference, Flagstaff, AZ, USA, 25–28 September 2000, Maschinski, J, Holter, L, Eds.; Rocky Mountain Research Station, USDA: Flagstaff, AZ, USA, 2001; pp. 200–205. [Google Scholar]
- Lehnhoff, EA; Rew, LJ; Maxwell, BD; Taper, ML. Quantifying invasiveness: A case study of Linaria vulgaris. Invasive Plant Sci. Manag 2008, 1, 319–325. [Google Scholar]
- Pariera Dinkins, CL; Brumfield, SK; Peterson, RKD; Grey, WE; Sing, SE. Dalmatian toadflax (Linaria dalmatica): New host for cucumber mosaic virus. Weed Technol 2007, 21, 41–44. [Google Scholar]
- USDA-NRCS. National Range and Pasture Handbook; Grazing Lands Technology Institute: Fort Worth, Texas, USA, 2003; Chapter 6. Available online: http://www.glti.nrcs.usda.gov/technical/publications/nrph.html accessed on 19 June 2011.
- Nikolova-Damyanova, B; Ilieva, E; Handjieva, N; Bankova, V. Quantitative thin layer chromatography of iridoid and flavonoid glucosides in species of Linaria. Phytochem. Anal 1994, 5, 38–40. [Google Scholar]
- Singh, RP; Agrawal, P; Yim, D; Agarwal, C; Agarwal, R. Acacetin inhibits cell growth and cell cycle progression, and induces apoptosis in human prostate cancer cells: Structure-activity relationship with linarin and linarin acetate. Carcinogenesis 2005, 26, 845–854. [Google Scholar]
- Hsu, YL; Kuo, PL; Liu, CF; Lin, CC. Acacetin-induced cell cycle arrest and apoptosis in human non-small cell lung cancer A549 cells. Cancer Lett 2004, 212, 53–60. [Google Scholar]
- Miyazawa, M; Hisama, M. Antimutagenic activity of flavonoids from Chrysanthemum morifolium. Biosci. Biotechnol. Biochem 2003, 67, 2091–2099. [Google Scholar]
- Singh, RP; Dhanalakshmi, S; Agarwal, R. Phytochemicals as cell cycle modulators: A less toxic approach in halting human cancer. Cell Cycle 2002, 1, 156–161. [Google Scholar]
- Sample, BE; Arenal, CA. Allometric models for interspecies extrapolation of wildlife toxicity data. Bull. Environ. Contam. Toxicol 1999, 62, 653–663. [Google Scholar]
- Slooff, W; van Oers, JAM; de Zwart, D. Margins of uncertainty in ecotoxicological hazard assessment. Environ. Toxicol. Chem 1986, 5, 841–852. [Google Scholar]
- Matarczyk, JA; Willis, AJ; Vranjic, JA; Ash, JE. Herbicides, weeds and endangered species: Management of bitou bush (Chrysanthemoides monilifera ssp rotundata) with glyphosate and impacts on the endangered shrub, Pimelea spicata. Biol. Conserv 2002, 108, 133–141. [Google Scholar]
- Pywell, RF; Nowakowski, M; Walker, KJ; Barratt, D; Sparks, TH. Preliminary studies of the effects of pre-emergence herbicides on the establishment of injurious weed and wildflower species. In Weed Seedbanks: Determination, Dynamics and Manipulation; Champion, GT, Grundy, AC, Jones, NE, Marshall, EJP, Froud-Williams, RJ, Eds.; St Catherine’s College; Oxford UK, 1998; pp. 173–178. [Google Scholar]
- Heagy, MI; Cavers, PB. The Natural Vegetation of Abandoned Gravel Pit Slopes. Proceedings of 5th Annual Meeting Canadian Land Reclamation Association, Timmins, ON, Canada, 18–20 August 1980, Winterhalder, K, Ed.; Canadian Land Reclamation Association: Guelph, ON, Canada, 1981; pp. 169–188. [Google Scholar]
- Long, SW. Differentiation of copper tolerant populations in Linaria vulgaris and Muhlenbergia sobolifera. Bull. New Jersey Acad. Sci 1974, 19, 22. [Google Scholar]
- USDA, NRCS. Montana Grazing Lands Conservation Initiative (GLCI)—2009 GLCI Demonstration Projects: “Cows Eat Weeds”; Montana GLCI Steering Committee: Bozeman, MT, USA, 2009. Available online: http://www.mt.nrcs.usda.gov/technical/glci/proj09cowsmussel.html accessed on 19 June 2011.
- Davison, JC; Smith, E; Wilson, LM. Livestock grazing guidelines for controlling noxious weeds in the western United States; UNR Cooperative Extension Publication No. EB-06-05; University of Nevada Cooperative Extension: Reno, NV, USA, 2006. [Google Scholar]
- Ricciardi, A; Cohen, J. The invasiveness of an introduced species does not predict its impact. Biol. Invasions 2007, 9, 309–315. [Google Scholar]
- Stohlgren, TJ; Barnett, DT; Kartesz, JT. The rich get richer: Patterns of plant invasions in the United States. Frontiers Ecol 2003, 1, 11–14. [Google Scholar]
- Stohlgren, TJ; Otsuki, Y; Villa, CA; Lee, M; Belnap, J. Patterns of plant invasions: A case example in native species hotspots and rare habitats. Biol. Invasions 2001, 3, 37–50. [Google Scholar]
- Reid, AM; Morin, L; Downey, PO; French, K; Virtue, JG. Does invasive plant management aid the restoration of natural ecosystems? Biol. Conserv 2009, 142, 2342–2349. [Google Scholar]
- Sing, SE; Peterson, RKD; Weaver, DK; Hansen, RW; Markin, GP. A retrospective analysis of known and potential risks associated with exotic toadflax-feeding insects. Biol. Control 2005, 35, 276–287. [Google Scholar]
| Effect | Dalmatian Toadflax | Yellow Toadflax | ||
|---|---|---|---|---|
| Effect Uncertainty | Exposure Uncertainty | Effect Uncertainty | Exposure Uncertainty | |
| Competitive Displacement | low | medium | low | medium |
| Reservoir of Plant Disease | medium | medium | low | medium |
| Animal Use | medium | medium | medium | medium |
| Animal Toxicity | high | medium | high | medium |
| Human Toxicity | high | low | high | low |
| Erosion | high | high | high | high |
| Wildfire | high | high | high | high |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sing, S.E.; Peterson, R.K.D. Assessing Environmental Risks for Established Invasive Weeds: Dalmatian (Linaria dalmatica) and Yellow (L. vulgaris) Toadflax in North America. Int. J. Environ. Res. Public Health 2011, 8, 2828-2853. https://doi.org/10.3390/ijerph8072828
Sing SE, Peterson RKD. Assessing Environmental Risks for Established Invasive Weeds: Dalmatian (Linaria dalmatica) and Yellow (L. vulgaris) Toadflax in North America. International Journal of Environmental Research and Public Health. 2011; 8(7):2828-2853. https://doi.org/10.3390/ijerph8072828
Chicago/Turabian StyleSing, Sharlene E., and Robert K. D. Peterson. 2011. "Assessing Environmental Risks for Established Invasive Weeds: Dalmatian (Linaria dalmatica) and Yellow (L. vulgaris) Toadflax in North America" International Journal of Environmental Research and Public Health 8, no. 7: 2828-2853. https://doi.org/10.3390/ijerph8072828




