Environmental Monitoring and Analysis of Faecal Contamination in an Urban Setting in the City of Bari (Apulia Region, Italy): Health and Hygiene Implications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Territorial Mapping
2.2. Monitoring and Sampling
2.3. Laboratory Analyses
2.3.1. DNA Extraction
2.3.2. Bacteriological Investigations
2.3.3. Virological Investigations
2.3.4. Parasitological Investigations
3. Results and Discussion
3.1. Bacteriological Investigations
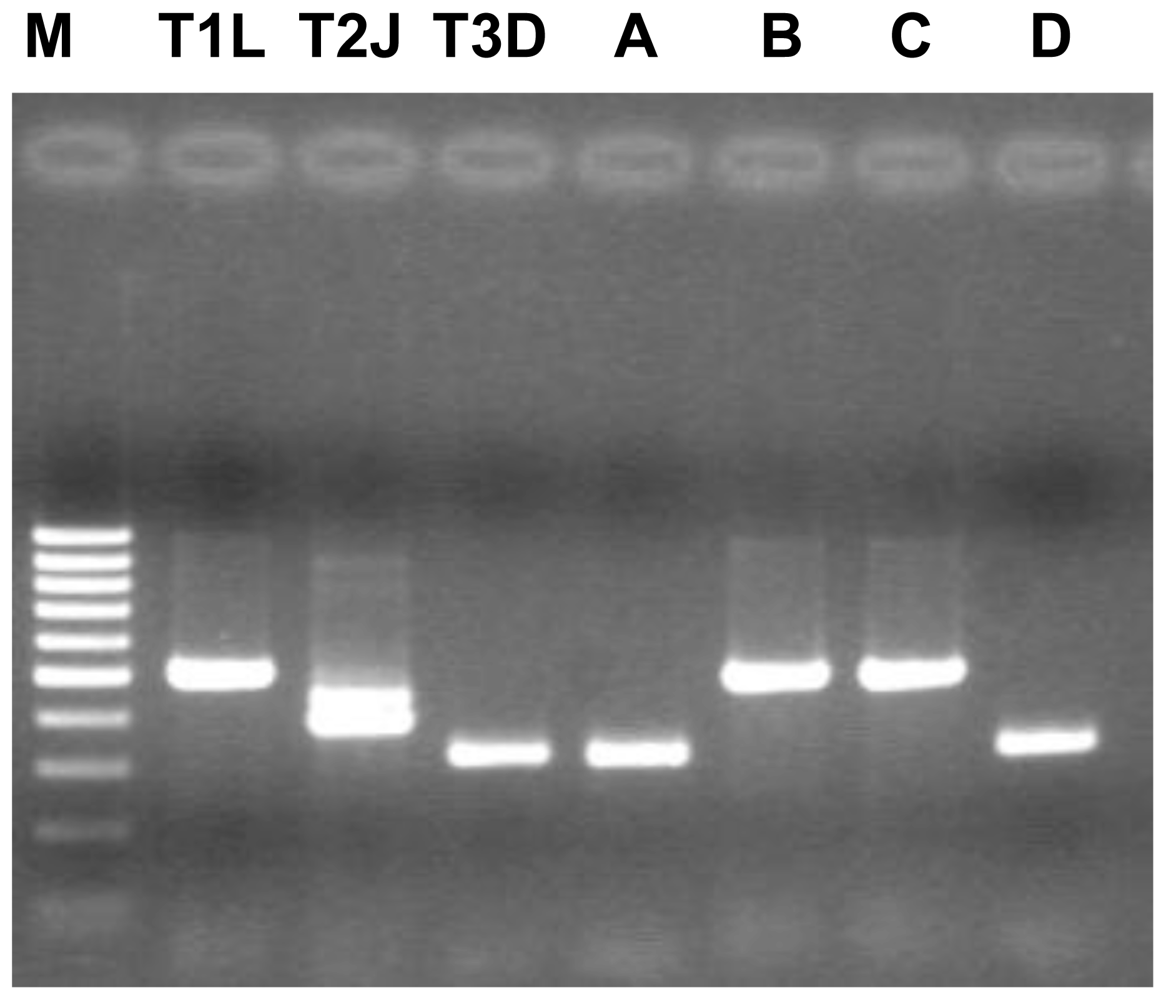
3.2. Virological Investigations
3.3. Parasitological Investigations
4. Conclusions
Acknowledgements
References
- Poglayen, G. Puccini, V, Tarsitano, E, Eds.; Zoonoses by environmental faecalization. In Manual of Urban Parasitology: Cities, Animals and Public Health, 1st ed; Il Sole 24 ORE Edagricole Srl: Bologna, Italy, 2003; pp. 110–117. [Google Scholar]
- Puccini, V; Tarsitano, E. Puccini, V, Tarsitano, E, Eds.; Introduction to Urban Parasitology. In Manual of Urban Parasitology: Cities, Animals and Public Health, 1st ed; Il Sole 24 ORE Edagricole Srl: Bologna, Italy, 2003; pp. 1–13. [Google Scholar]
- Tarsitano, E. Puccini, V, Tarsitano, E, Eds.; Urban Ecosystem. In Manual of Urban Parasitology: Cities, Animals and Public Health, 1st ed; Il Sole 24 ORE Edagricole Srl: Bologna, Italy, 2003; pp. 21–36. [Google Scholar]
- Poglayen, G; Marchesi, B. Urban faecal pollution and parasitic risk: the Italian skill. Parassitologia 2006, 48, 117–119. [Google Scholar]
- Poglayen, G; Giannetto, S; Macrì, B; Grippa, G; Scala, A; Cambosu, C; Giangaspero, A; Paletti, B; Montauti, AE; Traldi, G; Habluetzel, A. Canine zoonoses by environmental faecalization. Parassitologia 2000, 42, 220. [Google Scholar]
- Ferroglio, E; Romano, A; Passera, S; D’Angelo, A; Guiso, P; Ghiggi, E; Bolla, C; Trisciuoglio, A; Biglino, A. Dogs’ parasite and zoonotic risk: from old to new “emergencies” in the North-West of Italy. Parassitologia 2006, 48, 115–116. [Google Scholar]
- Giacometti, A; Cirioni, O; Fortuna, M; Osimani, P; Antonicelli, L; Del Prete, MS; Riva, A; DErrico, MM; Petrelli, E; Scalise, G. Environmental and serological evidence for the presence of toxocariasis in the urban area of Ancona, Italy. Eur. J. Epidemiol 2000, 16, 1023–1026. [Google Scholar]
- Sanchez Thevenet, P; Nancufil, A; Oyarzo, CM; Torrecillas, C; Raso, S; Mellado, I; Flores, ME; Cordoba, MG; Minvielle, MC; Basualdo, JA. An eco-epidemiological study of contamination of soil with infective forms of intestinal parasites. Eur. J. Epidemiol 2004, 19, 481–489. [Google Scholar]
- Tarsitano, E. Interaction between the environment and animals in urban settings: Integrated and Participatory Planning. Environ. Man 2006, 38, 799–809. [Google Scholar]
- Kleine-Tebbe, J; Heinatz, A; Graser, I; Dautel, H; Hansen, GN; Kespohl, S; Rihs, HP; Raulf-Heimsoth, HP; Vater, MG; Rytter, M; Haustein, UF. Bites of the European pigeon tick (Argas reflexus): Risk of IgE-mediated sensitizations and anaphylactic reactions. J. Allergy Clin. Immunol 2006, 117, 190–195. [Google Scholar]
- Spiewak, R; Lundberg, M; Johansson, G; Buczek, A. Allergy to pigeon tick (Argas reflexus) in Upper Silesia. Poland. Ann. Agric. Environ. Med 2006, 13, 107–112. [Google Scholar]
- Tampieri, MP. Mycetes and urban areas. Parassitologia 2006, 48, 121–124. [Google Scholar]
- Casanovas, L; de Simon, M; Ferrer, D; Arques, J; Monzon, G. Intestinal carriage of campylobacters, salmonellas, yersinias and listerias in pigeons in the city of Barcelona. J. Appl. Bacteriol 1995, 78, 11–13. [Google Scholar]
- Dinetti, M. Urban avifauna: Is it possible to live together? Vet. Res. Comm 2006, 30, 3–7. [Google Scholar]
- Goldshmidt, JN; Fantino, E. Economic context and pigeons’ risk-taking: an integrative approach. Behav. Processes 2004, 65, 133–154. [Google Scholar]
- Haag-Wackernagel, D; Moch, H. Health hazards posed by feral pigeons. J. Infect 2004, 48, 307–313. [Google Scholar]
- Tanaka, C; Miyazawa, T; Watarai, M; Ishiguro, N. Bacteriological survey of feces from feral pigeons in Japan. J. Vet. Med. Sci 2005, 67, 951–953. [Google Scholar]
- Graczyk, TK; Sunderland, D; Rule, AM; da Silva, AJ; Moura, IN; Tamang, L; Girouard, KJ; Schwab, AS; Breysse, PN. Urban feral pigeons (Columba livia) as a source for air- and waterborne contamination with Enterocytozoon bieneusi spores. Appl. Environ. Microbiol 2007, 73, 4357–4358. [Google Scholar]
- Haro, M; Izquierdo, F; Henriques-Gil, N; Andrés, I; Alonso, F; Fenoy, S; del Aguila, C. First detection and genotyping of human-associated microsporidia in pigeons from urban parks. Appl. Environ. Microbiol 2005, 71, 3153–3157. [Google Scholar]
- Casanovas, L; de Simón, M; Ferrer, MD; Arqués, J; Monzón, G. Intestinal carriage of campylobacters, salmonellas, yersinias and listerias in pigeons in the city of Barcelona. J. Appl. Bacteriol 1995, 78, 11–13. [Google Scholar]
- Pasmans, F; Van Immerseel, F; Hermans, K; Heyndrickx, M; Collard, JM; Ducatelle, R; Haesebrouck, F. Assessment of virulence of pigeon isolates of Salmonella enterica subsp. enterica serovar typhimurium variant copenhagen for humans. J. Clin. Microbiol 2004, 42, 2000–2002. [Google Scholar]
- Ilieski, V; Ristoski, T; Pendovski, L; Dodovski, A; Mitevski, D. Longbottom, D, Rocchi, M, Eds.; Detection of Chlamydophila psittaci in free-living birds using ELISA and immumohistochemical methods. Proceedings of the 4th Annual Workshop of COST Action 855, Animal Chlamydioses and Zoonotic Implications, Edinburgh UK, 3–5 September, 2006, Moredun Research Institute: Edinburgh, UK, 2006; pp. 86–87. [Google Scholar]
- Residbegovic, E; Kavazovic, A; Satrovic, E; Alibegovic-Zecic, F; Kese, D; Dovc, A. Niemczuk, K, Sachse, K, Sprague, LD, Eds.; Detection of antibodies and isolation of Chlamydophila psittaci in free-living pigeons (Columba livia domestica). Proceedings of the 5th Annual Workshop of Cost Action 855, Animal Chlamydioses and Zoonotic Implication, Pulawy, Poland, 10–11 September, 2007, National Veterinary Research Institute: Pulawy, Poland, 2007; pp. 81–85. [Google Scholar]
- Ceglie, L; Lafisca, S; Guadagno, C; Dalla Pozza, G; Capello, K; Bano, L; Vicari, N; Donati, M; Mion, M; Giurisato, I; Lombardo, D; Pozzato, N; Cevenini, R; Natale, A. Niemczuk, K, Sachse, K, Sprague, LD, Eds.; Serological surveillance in north-eastern Italy for the presence of Chlamydophila spp. from birds and molecular characterization of PCR isolates within the area of Venice. Proceedings of the 5th Annual Workshop of Cost Action 855, Animal Chlamydioses and Zoonotic Implication, Pulawy, Poland, 10–11 September, 2007, National Veterinary Research Institute: Pulawy, Poland, 2007; pp. 62–67. [Google Scholar]
- Magnino, S; Haag-Wackernagel, D; Geigenfeind, I; Helmecke, S; Dovc, A; Prukner-Radovcić, E; Residbegović, E; Ilieski, V; Laroucau, K; Donati, M; Martinov, S; Kaleta, EF. Chlamydial infections in feral pigeons in Europe: Review of data and focus on public health implications. Vet. Microbiol 2009, 135, 54–67. [Google Scholar]
- Binn, LN; Marchwicki, RH; Keenan, KP; Strano, AJ; Engler, WR. Recovery of reovirus type 2 from an immature dog with respiratory tract disease. Am. J. Vet. Res 1977, 38, 927–929. [Google Scholar]
- Tyler, KL. Knipe, DM, Howley, PM, Eds.; Mammalian reoviruses. In Fields Virology, 4th ed; LWW: Philadelphia, PA, USA, 2001; pp. 1729–1745. [Google Scholar]
- Laroukau, K; Souriau, A; Rodolakis, A. Improved sensivity of PCR for Chlamydophila using pmp genes. Vet. Microbiol 2001, 82, 155–164. [Google Scholar]
- Andreani, E; Poli, A; Tolari, F; Cerri, D; Farina, R; Bandecchi, P. Experimental infection of sheep with Chlamydia psittaci. Br. Vet. J 1987, 143, 221–225. [Google Scholar]
- Khan, AA; Nawaz, MS; Khan, SA; Cerniglia, CE. Detection of multidrug-resistant Salmonella typhimurium DT104 by multiplex polymerase chain reaction. FEMS Microbiol. Lett 2000, 182, 355–360. [Google Scholar]
- Gentsch, JR; Glass, RI; Woods, P; Gouvea, V; Gorziglia, M; Flores, J; Das, BK; Bhan, MK. Identification of group A rotavirus gene 4 types by polymerase chain reaction. J. Clin. Microbiol 1992, 30, 1365–1373. [Google Scholar]
- Leary, PL; Erker, JC; Chalmers, ML; Cruz, AT; Wetzel, JD; Desai, SM; Mushahwar, IK; Dermody, TS. Detection of mammalian reovirus RNA by using reverse transcription-PCR: sequence diversity within the λ3-encoding L1 gene. J. Clin. Microbiol 2002, 40, 1368–1375. [Google Scholar]
- Gouvea, V; Santos, N; Timenetsky, M; do, C. Identification of bovine and porcine rotavirus G types by PCR. J. Clin. Microbiol 1994, 32, 1338–1340. [Google Scholar]
- Gordon, H Mcl; Whitlock, HV. A new technique for counting nematode eggs in sheep faeces. J. Counc. Sci. Ind. Res 1939, 12, 50–52. [Google Scholar]
- Arizmendi, F; Grimes, JE; Relford, RL. Isolation of Chlamydia psittaci from pleural effusion in a dog. J. Vet. Diagn. Invest 1992, 4, 460–463. [Google Scholar]
- Fraser, G; Norwall, J; Withers, AR; Gregor, WW. A case history of psittacosis in the dog. Vet. Rec 1969, 85, 54–58. [Google Scholar]
- Cleaveland, S; Haydon, DT; Taylor, D. Overviews of pathogen emergence: which pathogens emerge, when and why? Curr. Top. Microbiol. Immunol 2007, 315, 85–111. [Google Scholar]
- Elia, G; Lucente, MS; Bellacicco, AL; Camero, M; Cavaliere, N; Decaro, N; Martella, V. Assessment of reovirus epidemiology in dogs. Proceedings of 16th European Congress of Clinical Microbiology and Infectious Diseases, Nice, France, 1–4 April, 2006; p. 1674.
- Childs, JE; Richt, JA; Mackenzie, JS. Introduction: conceptualizing and partitioning the emergence process of zoonotic viruses from wildlife to humans. Curr. Top. Microbiol. Immunol 2007, 315, 1–31. [Google Scholar]
- Hochedez, P; Caumes, E. Hookworm-related cutaneous larva migrans. J. Travel. Med 2007, 14, 326–333. [Google Scholar]
- Lia, RP. Puccini, V, Tarsitano, E, Eds.; Visceral and cutaneous Larva Migrans. In Manual of Urban Parasitology: Cities, Animals and Public Health, 1st ed; Il Sole 24 ORE Edagricole Srl: Bologna, Italy, 2003; pp. 117–128. [Google Scholar]
- Gawor, J; Borecka, A. The contamination of the environment with Toxocara eggs in Mazowieckie voivodship as a risk of toxocarosis in children. Wiad. Parazytol 2004, 50, 237–241. [Google Scholar]
- Lim, JH. Toxocariasis of the liver: visceral larva migrans. Abdom. Imaging 2008, 33, 151–156. [Google Scholar]
- Hochedez, P; Caumes, E. Hookworm-related cutaneous larva migrans. J. Travel. Med 2007, 14, 326–333. [Google Scholar]
- Habluetzel, A; Traldi, G; Ruggieri, S; Attili, AR; Scuppa, P; Marchetti, R; Menghini, G; Esposito, F. An estimation of Toxocara canis prevalence in dogs, environmental egg contamination and risk of human infection in the Marche region of Italy. Vet. Parasitol 2003, 113, 243–252. [Google Scholar]
- Legrottaglie, R; Papini, R; Capasso, R; Cardini, G. Prevalence of Toxacara canis eggs in dog faecal deposits from urban areas of Pisa, Italy. Helminthologia 2003, 40, 173–175. [Google Scholar]
- Veneziano, V; Rinaldi, L; Carbone, S; Biggeri, A; Cringoli, G. Geographical Information Systems and canine faecal contamination: the experience in the city of Naples (southern Italy). Parassitologia 2006, 48, 125–128. [Google Scholar]
- Sanchez Thevenet, P; Jensen, O; Mellado, I; Torrecillas, C; Raso, S; Flores, ME; Minvielle, MC; Basualdo, JA. Presence and persistence of intestinal parasites in canine fecal material collected from the environment in the Province of Chubut, Argentine Patagonia. Vet. Parasitol 2003, 28, 263–269. [Google Scholar]
- May, PH; Dabbs, A; Fernandez-Davila, WP; Da Vinha, V; Zaidenweber, N. A corporate approach to social monitoring and assessment for development in a fragile environment. Environ. Monit. Assess 2002, 76, 125–134. [Google Scholar]
- Antolova, D; Reiterova, K; Miterpakova, M; Stanko, M; Dubinsky, P. Circulation of Toxocara spp. in suburban and rural ecosystems in the Slovak Republic. Vet. Parasitol 2004, 126, 317–324. [Google Scholar]
- Fok, E; Szatmari, V; Busak, K; Rozgonyi, K. Prevalence of intestinal parasites in dogs in some urban and rural areas of Hungary. Vet. Quart 2001, 23, 96–98. [Google Scholar]
- Pullola, T; Vierimaa, J; Saari, S; Virtala, AM; Nikander, S; Sukura, A. Canine intestinal helminths in Finland: prevalence, risk factors and endoparasite control practices. Vet. Parasitol 2006, 140, 321–326. [Google Scholar]
- Bradley, CA; Altizer, S. Urbanization and the ecology of wildlife diseases. Trends. Ecol. Evol 2007, 22, 95–102. [Google Scholar]
- Checkoway, B. Political Strategy for Social Planning. In Strategic Perspectives on Planning Practice; Lexington Books: Lexington, KY, USA, 1986. [Google Scholar]
- May, PH; Dabbs, A; Fernandez-Davila, WP; Da Vinha, V; Zaidenweber, N. A corporate approach to social monitoring and assessment for development in a fragile environment. Environ. Monit. Assess 2002, 76, 125–134. [Google Scholar]
- Jardine, C; Hrudey, S; Shortreed, J; Craig, L; Krewski, D; Furgal, C; McColl, S. Risk management frameworks for human health and environmental risks. J. Toxicol. Environ. Health B Crit. Rev 2003, 6, 569–720. [Google Scholar]
- Tarsitano, E. Puccini, V, Tarsitano, E, Eds.; Management, prevention and integrated control of urban parassitosis. In Manual of Urban Parasitology: Cities, Animals and Public Health, 1st ed; Il Sole 24 ORE Edagricole Srl: Bologna, Italy, 2003; pp. 331–341. [Google Scholar]
- Ballarini, G. Puccini, V, Tarsitano, E, Eds.; Urban fauna plan. In Manual of Urban Parasitology: Cities, Animals and Public Health, 1st ed; Il Sole 24 ORE Edagricole Srl: Bologna, Italy, 2003; pp. 319–330. [Google Scholar]
- Dinetti, M. Dinetti, M, Ed.; Manual of Urban Biodiversity; Il Sole 24 ORE Edagricole Srl: Livorno, Italy, 2009; pp. 1–293. [Google Scholar]
- Burger, J; Myers, O; Boring, CS; Dixon, C; Jeitner, JC; Leonard, J; Lord, C; McMahon, M; Ramos, R; Shukla, S; Gochfeld, M. Perceptual indicators of environmental health, future land use, and stewardship. Environ. Monit. Assess 2003, 89, 285–303. [Google Scholar]
- Eyles, J; Furgal, C. Indicators in environmental health: identifying and selecting common sets. Can. J. Public Health 2002, 93, 62–67. [Google Scholar]




| SITES | AREA - GPS POSITION | ISOLATED PATHOGENS |
|---|---|---|
| 1. LIBERTÀ | B - E 16°51′25″, N 41°07′24″ | C. psittaci |
| C1 - E 16°50′52″, N 41°07′13″ | C. psittaci | |
| C2 - E 16°50′47″, N 41°07′17″ | C. psittaci (Guano) | |
| 2. SAN GIROLAMO | B - E 16°49′52″, N 41°07′57″ | Ancylostoma |
| C - E 16°48′54″, N 41°08′06″ | C. psittaci | |
| 3. MARCONI | B - E 16°50′28″, N 41°08′16″ | Ascarida C. psittaci |
| 4. MURAT | A - E 16°51′54″, N 41°07′32″ | Ascarida Ancylostoma |
| C - E 16°52′02″, N 41°07′13″ | Ancylostoma | |
| 5. SAN NICOLA | B - E 16°52′14″, N 41°07′48″ | Ascarida C. psittaci (Guano) |
| 6. PALESE | B –E 16°45′19″ N 41°09′26″ | Ascarida |
| 8. CATINO - ENZITETO | A - E 16°44′17″, N 41°09′27″ | Reoviridae |
| B - E 16°44′23″, N 41°09′06″ | Ascarida Trichurida | |
| 10. TORRE A MARE | C - E 16°59′42″, N 41°04′56″ | Trichurida |
| 12. SAN PAOLO | A - E 16°48′08″, N 41°07′23″ | Reoviridae Coccidia |
| 17. POGGIOFRANCO | A - E 16°51′34″, N 41°06′13″ | Ascarida Ancylostoma |
| B - E 16°51′43″, N 41°05′55″ | Reoviridae | |
| 18. PICONE | B - E 16°51′18″, N 41°06′32″ | Ascarida |
| 19. CARRASSI | A - E 16°52′30″, N 41°06′03″ | Ascarida |
| C - E 16°52′02″, N 41°06′44″ | Ascarida | |
| 20. CARBONARA | C - E 16°52′13″, N 41°04′28″ | C. psittaci |
| 21. CEGLIE | A - E 16°51′56″, N 41°03′52″ | C. psittaci |
| B - E 16°52′04″, N 41°03′59″ | Ancylostoma | |
| 22. LOSETO | A - E 16°51′13″, N 41°02′28″ | C. psittaci Reoviridae |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tarsitano, E.; Greco, G.; Decaro, N.; Nicassio, F.; Lucente, M.S.; Buonavoglia, C.; Tempesta, M. Environmental Monitoring and Analysis of Faecal Contamination in an Urban Setting in the City of Bari (Apulia Region, Italy): Health and Hygiene Implications. Int. J. Environ. Res. Public Health 2010, 7, 3972-3986. https://doi.org/10.3390/ijerph7113972
Tarsitano E, Greco G, Decaro N, Nicassio F, Lucente MS, Buonavoglia C, Tempesta M. Environmental Monitoring and Analysis of Faecal Contamination in an Urban Setting in the City of Bari (Apulia Region, Italy): Health and Hygiene Implications. International Journal of Environmental Research and Public Health. 2010; 7(11):3972-3986. https://doi.org/10.3390/ijerph7113972
Chicago/Turabian StyleTarsitano, Elvira, Grazia Greco, Nicola Decaro, Francesco Nicassio, Maria Stella Lucente, Canio Buonavoglia, and Maria Tempesta. 2010. "Environmental Monitoring and Analysis of Faecal Contamination in an Urban Setting in the City of Bari (Apulia Region, Italy): Health and Hygiene Implications" International Journal of Environmental Research and Public Health 7, no. 11: 3972-3986. https://doi.org/10.3390/ijerph7113972




