Photodegradation of Selected PCBs in the Presence of Nano-TiO2 as Catalyst and H2O2 as an Oxidant
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Quantum Chemical Calculations
2.3. Photolysis of PCBs
2.4. HPLC Analysis
2.5. Degradation Kinetics
3. Results and Discussions
3.1. Planarity of Selected PCBs Based on Quantum Chemical Calculations
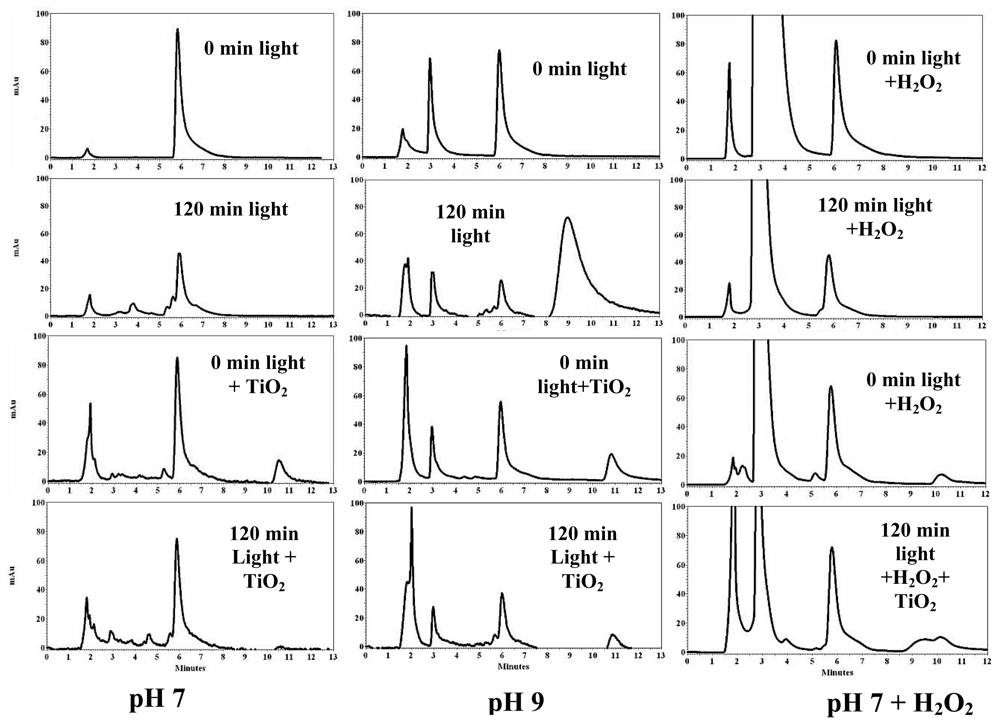
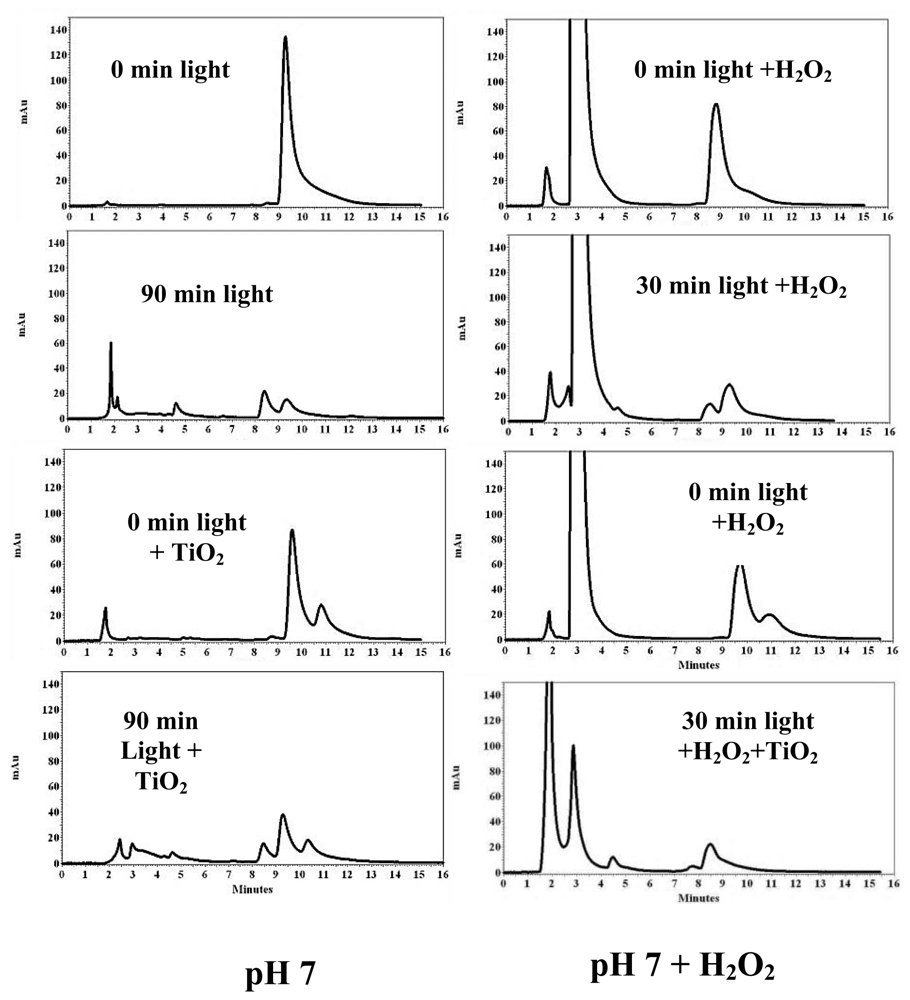
3.2. PCB Photodegradation Monitored by HPLC
3.3. Degradation Rate Constants and Half-Lives
3.4. Effect of PCB Structure on Degradation Rate
3.5. Effect of TiO2, H2O2, and pH on the Photodegradation
4. Conclusions
Acknowledgements
References
- Hopf, NB; Ruder, AM; Succop, P. Background levels of polychlorinated biphenyls in the U.S. population. Sci. Tot. Environ 2009, 407, 6109–6119. [Google Scholar]
- Carpenter, DO. Polychlorinated biphenyls (PCBs): Routes of exposure and effects on human health. Rev. Environ. Health 2006, 21, 1–23. [Google Scholar]
- Drevenkar, V; Fingler, S; Fröbe, Z; Vasilic, Z. Persistent organochlorine compounds in water and soil environments. Environ. Manag. Health 1996, 7, 5–8. [Google Scholar]
- Kimbrough, RD; Krouskas, CA; Leigh Carson, M; Long, TF; Bevan, C; Tardiff, RG. Human uptake of persistent chemicals from contaminated soil: PCDD/Fs and PCBs. Regulatory Toxicol. Pharmacol 2010, 57, 43–54. [Google Scholar]
- Ndountse, LT; Chan, HM. Role of N-methyl-D-aspartate receptors in polychlorinated biphenyl mediated neurotoxicity. Toxicol. Lett 2009, 184, 50–55. [Google Scholar]
- George, CJ; Bennett, GF; Simoneaux, D; George, WJ. Polychlorinated biphenyls: A toxicological review. J. Hazard. Mater 1988, 18, 113–144. [Google Scholar]
- Kopf, PG; Walker, MK. Overview of developmental heart defects by dioxins, PCBs, and pesticides. J. Environ. Sci. Health 2009, 27, 276–285. [Google Scholar]
- Ross, G. The public health implications of polychlorinated biphenyls (PCBs) in the environment. Ecotoxicol. Environ. Safety 2004, 59, 275–291. [Google Scholar]
- Aken, BV; Correa, PA; Schnoor, JL. Phytoremediation of polychlorinated biphenyls: New trends and promises. Environ. Sci. Technol 2010, 44, 2767–2776. [Google Scholar]
- Ohtsubo, Y; Kudo, T; Tsuda, M; Nagata, Y. Strategies for bioremediation of polychlorinated biphenyls. Appl. Microbiol. Biotech 2004, 65, 250–258. [Google Scholar]
- Nobbs, D; Chipman, G. Contaminated site investigation and remediation of chlorinated aromatic compounds. Separat. Purif. Technol 2003, 31, 37–40. [Google Scholar]
- Kabra, K; Chaudhary, KR; Sawhney, RL. Treatment of hazardous organic and inorganic compounds through aqueous phase photocatalysis: A review. Ind. Eng. Chem. Res 2004, 43, 7683–7696. [Google Scholar]
- Herrmann, JM. Heterogenous photocatalysis: Fundamentals and applications to the removal of various types of aqueous pollutants. Catal. Today 1999, 53, 115–129. [Google Scholar]
- Li, FB; Li, XZ; Ao, CH; Lee, SC; Hou, MF. Enhanced photocatalytic degradation of VOCs using Ln-TiO2 catalysts for indoor air purification. Chemosphere 2005, 59, 787–800. [Google Scholar]
- McCullagh, C; Robertson, JMC; Bahnemann, DW; Robertson, PKJ. The application of TiO2 photocatatlysis for disinfection of water contaminated with pathogenic microorganisms: A Review. Res. Chem. Intermed 2007, 33, 359–375. [Google Scholar]
- Yu, JG; Wang, GH; Chen, B; Zhou, MH. Effects of hydrothermal temperature and time on the photocatalytic activity and microstructures of bimodal mesoporous TiO2 powders. Appl. Catal. B Environ 2007, 69, 171–180. [Google Scholar]
- Sayilkan, H. Improved photocatalytic activity of Sn4+-doped and undoped TiO2 thin film coated stainless steel under UV and Vis-irradiation. Appl. Catal. A Gen 2007, 319, 230–236. [Google Scholar]
- Wang, C; Zhang, X; Liu, H; Li, X; Li, W; Xu, H. Reaction kinetics of photocatalytic degradation of sulfosalicylic acid using TiO2 microspheres. J. Hazard. Mater 2009, 163, 1101–1106. [Google Scholar]
- Wong, KH; Wong, PK. Degradation of polychlorinated biphenyls by UV-catalyzed photolysis. Hum. Ecol. Risk. Assess 2006, 12, 259–269. [Google Scholar]
- Huang, IW; Hong, C-S; Bush, B. Photocatalytic degradation of PCBs in TiO2 aqueous suspensions. Chemosphere 1996, 32, 1869–1881. [Google Scholar]
- Lores, M; Llompart, M; González-García, R; González-Barreiro, C; Cela, R. Photolysis of polychlorinated biphenyls by solid-phase microextraction: “On-fibre” versus aqueous photodegradation. J. Chromatogr. A 2002, 963, 37–47. [Google Scholar]
- Munazano, MA; Perales, JA; Sales, D; Quiroga, JM. Using solar and ultraviolet light to degrade PCBs in sand and transformer oils. Chemosphere 2004, 57, 645–654. [Google Scholar]
- Chu, W; Chan, KH; Kwan, CY; Jafvert, CT. Acceleration of quenching of photolysis of PCB in the presence of surfactant and humic material. Environ. Sci. Technol 2005, 39, 9211–9216. [Google Scholar]
- Miao, XS; Chu, SG; Xu, XB. Degradation pathways of PCBs upon UV irradiation in hexane. Chemosphere 1999, 39, 1639–1650. [Google Scholar]
- Miao, XS; Chu, W; Xu, XB. Photolysis of 2,2′,5,5′-tetrachlorobiphenyl in hexane. Bull. Environ. Contam. Toxicol 1996, 56, 571–574. [Google Scholar]
- Chang, FC; Chiu, TC; Yen, JH; Wang, YS. Dechlorination pathways of ortho-substituted PCBs by UV irradiation in n-hexane and their correlation to the charge distribution on carbon atom. Chemosphere 2003, 51, 775–784. [Google Scholar]
- Ruzo, LO; Zabik, MJ; Schuetz, RD. Photochemistry of bioactive compounds. Photochemical processes of polychlorinated biphenyls. J. Am. Chem. Soc 1974, 96, 3809–3813. [Google Scholar]
- Hawari, J; Demeter, A; Greer, C; Samson, R. Acetone-induced photodechloriantion of Aroclor 1254 in alkaline 2-propanol: Probinng the mechanism by thermolysis in the presence of di-t-butyl peroxide. Chemosphere 1991, 22, 1161–1174. [Google Scholar]
- Grittini, C; Malcomson, M; Fernando, Q; Korte, N. Rapid dechlorination of polychlorinated biphenyls on the surface of a Pd/Fe bimetallic system. Environ. Sci. Technol 1995, 29, 2898–2900. [Google Scholar]
- Yao, Y; Kakimoto, K; Ogawa, HI; Kato, Y; Kadokami, K; Shinohara, R. Further study on the photochemistry of non-ortho substituted PCBs by UV irradiation in alkaline 2-propanol. Chemosphere 2000, 40, 951–956. [Google Scholar]
- Oida, T; Barr, JR; Kimata, K; McClure, PC; Lapeza, CR; Hosoya, K; Ikegami, T; Smith, CJ; Patterson, DG; Tanaka, N. Photolysis of polychlorinated biphenyls on octadecylsilylated silica particles. Chemosphere 1999, 39, 1795–1807. [Google Scholar]
- Kuo, C; Liu, S; Liu, C. Biodegradation of coplanar polychlorinated biphenyls by anaerobic microorganisms from estuarine sediments. Chemosphere 1999, 39, 1445–1458. [Google Scholar]
- Lin, YJ; Lin, C; Yeh, KJ; Lee, A. Photodegradation of the herbicides butachlor and ronstar using natural sunlight and diethylamine. Bull. Environ. Contam. Toxicol 2000, 64, 780–785. [Google Scholar]
- García-Martínez, MJ; Da Riva, I; Canoira, L; Llamas, JF; Alcántara, R; Gallego, JLR. Photodegradation of polycyclic aromatic hydrocarbons in fossil fuels catalysed by supported TiO2. Appl. Catal. B Environ 2006, 67, 279–289. [Google Scholar]
- Swarnalatha, B; Anjaneyulu, Y. Studies on the heterogeneous photocatalytic oxidation of 2,6-dinitrophenol in aqueous TiO2 suspension. J. Mol. Catal. A Chem 2004, 223, 161–165. [Google Scholar]
- Ollis, DF; Al-Ekabi, H (Eds.) Photocatalytic Purification and Treatment of Water and Air; Elsevier: Amsterdam, The Netherlands, 1993.
- Hoffman, MR; Martin, ST; Choi, W; Bahnemann, DW. Environmental applications of semiconductor photocatalysis. Chem. Rev 1995, 95, 69–96. [Google Scholar]
- Linsebigler, AL; Lu, GQ; Yates, JT. Photocatalysis on TiO2 surfaces: Principles, mechanisms, and selected results. Chem. Rev 1995, 95, 735–758. [Google Scholar]
- Serpone, N; Khairutdinov, RF. Application of nanoparticles in the photocatalytic degradation of water pollutants. Stud. Surf. Sci. Catal 1997, 103, 417–444. [Google Scholar]
- Wold, A. Photocatalytic properties of titanium dioxide (TiO2). Chem. Mater 1993, 5, 280–283. [Google Scholar]
- Saritha, P; Dasary, SSR; Aparna, C; Nalini Vijaya Laxmi, P; Himabindu, V; Anjaneyulu, Y. Degradative Oxidation of 2,4,6 Trichlorophenol using advanced oxidation processes—A comparative study. Water Air Soil Pollut 2009, 200, 1–4. [Google Scholar]
- Wang, Y; Hong, CS. Effect of hydrogen peroxide, periodate and persulfate on photocatalysis of 2-chlorobiphenyl in aqueous TiO2 suspensions. Water Res 1999, 33, 2031–2036. [Google Scholar]
- Nobbs, D; Chipmann, G. Contaminated site investigation and remediation of chlorinated aromatic compounds. Sep. Purif. Technol 2003, 31, 37–40. [Google Scholar]
- Zhang, JZ. Photocatalysis in aqueous solution with small TiO2 particles and the dependence of the quantum yield on particle size and light intensity. J. Phys. Chem. B 2000, 104, 7239–7245. [Google Scholar]
- Gerischer, H. Photocatalysis in aqueous solution with small TiO2 particles and the dependence of the quantum yield on particle size and light intensity. Electrochim. Acta 1995, 40, 1277–1281. [Google Scholar]
- Madden, TH; Datye, AK; Fulton, M; Prairie, MR; Majumdar, SA; Stange, BM. Oxidation of metal-EDTA complexes by TiO2 photocatalysis. Environ. Sci. Technol 1997, 31, 3475–3481. [Google Scholar]
- Malato, S; Blanco, J; Richter, C; Braun, B; Maldonado, MI. Enhancement of the rate of solar photocatalytic mineralization of organic pollutants by inorganic oxidizing species. Appl. Catal. B Environ 1998, 17, 347–356. [Google Scholar]
- Doong, RA; Chen, CH; Maithreepala, RA; Chang, SM. The influence of pH and cadmium sulphide on photocatalytic degradation of 2-chlrophenol in titanium dioxide suspensions. Water Res 2001, 35, 2873–2880. [Google Scholar]
- Wong, CC; Chu, W. The hydrogen peroxide-assisted photocatalytic degradation of alachlor in TiO2 suspensions. Environ. Sci. Technol 2003, 37, 2310–2316. [Google Scholar]
- Saritha, P; Aparna, C; Himabindu, V; Anjaneyulu, Y. Comparison of various advanced oxidation processes for the degradation of 4-chloro-2-nitrophenol. J. Hazard. Mater 2007, 149, 609–614. [Google Scholar]
- De Felip, E; Ferri, F; Lupi, C; Trieff, NM; Volpi, F; Di Domenico, A. Structure-dependent photocatalytic degradation of polychlorinatedbiphenyls in a TiO2 aqueous system. Chemosphere 1996, 33, 2263–2271. [Google Scholar]
- Al-Sheikhly, M; Silverman, J; Neta, P; Karam, L. Mechanisms of ionizing radiation-induced destruction of 2,6-dichlorobiphenyl in aqueous solutions. Environ. Sci. Technol 1997, 31, 2473–2477. [Google Scholar]
- Lin, YA; Teng, LS; Lee, A; Chen, Y. Effect of photosensitizer diethylamine on the photodegradation of polychlorinated biphenyls. Chemosphere 2004, 55, 879–884. [Google Scholar]
- Al Momani, F; Sans, C; Esplugas, SA. Comparative study of the advanced oxidation of 2,4-dichlorophenol. J. Hazard. Mater 2004, 107, 123–129. [Google Scholar]
- Choi, W; Hoffman, MR. Novel photocatalytic mechanisms for CHCl3, CHBr3, and CCl3CO−2 degradation and the fate of photogenerated trihalomethyl radicals on TiO2. Environ. Sci. Technol 1996, 31, 89–95. [Google Scholar]
- Wang, S; Mosley, C; Stewart, G; Yu, H. Photochemical reaction of a dye precursor 4-chloro-1,2-phenylenediamine and its associated mutagenic effects. J. Photochem. Photobiol. A 2008, 197, 34–39. [Google Scholar]
- Hu, Q; Zhang, C; Wang, Z; Chen, Y; Mao, K; Zhang, X; Xiong, Y; Zhu, M. Photodegradation of methyl tert-butyl ether (MTBE) by UV/H2O2 and UV/TiO2. J. Hazard. Mater 2008, 154, 795–803. [Google Scholar]
- Pignatello, JJ. Dark and photoassisted Fe(III) catalyzed degradation of chlorophenoxy herbicides by hydrogen peroxide. Environ. Sci. Technol 1992, 26, 944–951. [Google Scholar]
- Esplugas, S; Vicente, M; Ibarx, A; Prat, C; Costa, J. Influence of lamp position on available radiation flux in an annular photoreactor. Chem. Eng. J 1987, 34, 111–115. [Google Scholar]
- Lin, YJ; Chen, YL; Huang, CY; Wu, MF. Photocatalysis of 2,2′,3,4,4′,5′-hexachlorobiphenyl and its intermediates using various catalytical preparing methods. J. Hazard. Mater 2006, B136, 902–910. [Google Scholar]
- Bunce, NJ. Photodechlorination of PCBs: Current status. Chemosphere 1982, 11, 701–714. [Google Scholar]
- Chiou, CH; Wu, CY; Juang, RS. Influence of operating parameters on the photocatalytic degradation of phenol in UV/TiO2 process. Chem. Eng. J 2007, 139, 322–329. [Google Scholar]
- Quici, N; Morgada, ME; Gettar, RT; Bolte, M; Litter, MI. Photocatalytic degradation of citric acid under different conditions: TiO2 heterogeneous photocatalysis against homogeneous photolytic processes promoted by Fe(III) and H2O2. Appl. Catal. B Environ 2007, 71, 117–124. [Google Scholar]
- Saquib, M; Abu Tariq, M; Haque, MM; Muneer, M. Photocatalytic degradation of dispersed blue 1 using UV/TiO2/H2O2. J. Environ. Manag 2008, 88, 300–306. [Google Scholar]
- Barkat, MA; Tseng, JM; Huang, CP. Hydrogen peroxide-assisted photocatalytic oxidation of phenolic compounds. Appl. Catal. B Environ 2005, 59, 99–104. [Google Scholar]



| Name and Abbreviation | 3-D Structure | Dihedral Angle | Position of Cl |
|---|---|---|---|
| 3,3′,4,4′-Tetrachlorobiphenyl (3,3′,4,4′-TCB) | 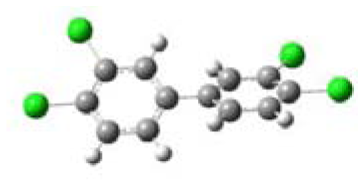 | 37.09 | m,m′,p,p′ |
| 3,4′,5-Trichlorobiphenyl (3,4′,5-TCB) | 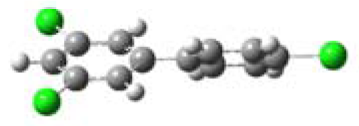 | 37.35 | m,p′,m |
| 2,2′,4,4′-Tetrachlorobiphenyl (2,2′,4,4′-TCB) | 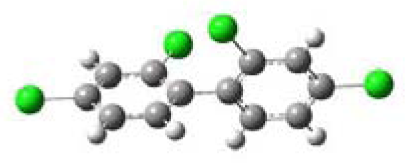 | 68.27 | o,o′,p,p′ |
| 2,6-Dichlorobipheny (2,6-DCB) | 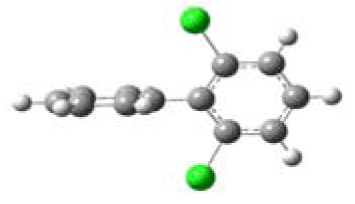 | 76.84 | o,o |
| 2,3,5,6-Tetrachlorobiphenyl (2,3,5,6-TCB) | 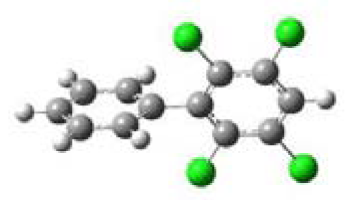 | 90.00 | o,m,m,o |
| PCBs | pH 7 | pH 7+TiO2 | pH 9 | pH 9+TiO2 | H2O2 | H2O2+TiO2 |
|---|---|---|---|---|---|---|
| 2,6-DCB | 139 min 0.0050 | 192 min 0.0036 | 157 min 0.0044 | b | 178 min 0.0039 | 110 min 0.0063 |
| 3,4′,5-TCB | 630 min 0.0011 | 141 min 0.0049 | 533 min 0.0013 | 124 min 0.0056 | 18 min 0.038 | 55 min 0.013 |
| 2,3,5,6-TCB | 107 min 0.0065 | 239 min 0.0029 | a | a | 48 min 0.0143 | 34 min 0.020 |
| 2,2′,4,4′-TCB | 38 min 0.018 | 66 min 0.010 | a | a | 46 min 0.015 | 35 min 0.021 |
| 3,3′,4,4′-TCB | 385 min 0.0018 | 161 min 0.0043 | 217 min 0.0032 | 128 min 0.0054 | 20 min 0.033 | 88 min 0.0079 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dasary, S.S.R.; Saloni, J.; Fletcher, A.; Anjaneyulu, Y.; Yu, H. Photodegradation of Selected PCBs in the Presence of Nano-TiO2 as Catalyst and H2O2 as an Oxidant. Int. J. Environ. Res. Public Health 2010, 7, 3987-4001. https://doi.org/10.3390/ijerph7113987
Dasary SSR, Saloni J, Fletcher A, Anjaneyulu Y, Yu H. Photodegradation of Selected PCBs in the Presence of Nano-TiO2 as Catalyst and H2O2 as an Oxidant. International Journal of Environmental Research and Public Health. 2010; 7(11):3987-4001. https://doi.org/10.3390/ijerph7113987
Chicago/Turabian StyleDasary, Samuel S. R., Julia Saloni, Amanda Fletcher, Yerramilli Anjaneyulu, and Hongtao Yu. 2010. "Photodegradation of Selected PCBs in the Presence of Nano-TiO2 as Catalyst and H2O2 as an Oxidant" International Journal of Environmental Research and Public Health 7, no. 11: 3987-4001. https://doi.org/10.3390/ijerph7113987




