Biological Effects and Safety in Magnetic Resonance Imaging: A Review
Abstract
:1. Introduction
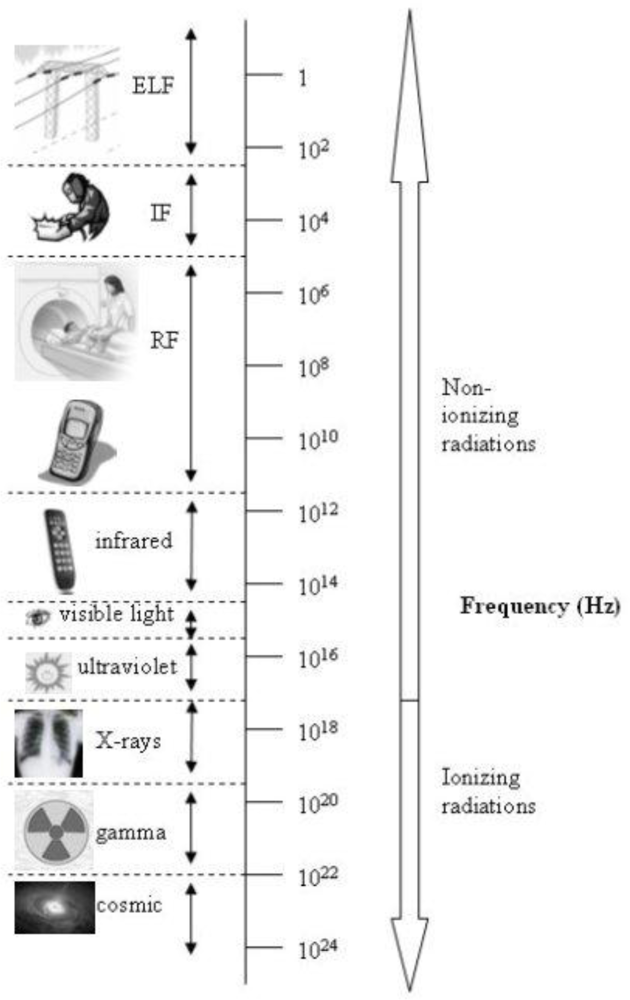
2. Electromagnetic Spectrum
2.1. Static Fields
2.2. Extremely Low Frequency (ELF)
2.3. Intermediate Frequency (IF)
2.4. Radio Frequency (RF)
3. Magnetic Resonance Imaging (MRI)
3.1. MRI Biological Effects
3.1.1. Effects of Static MF
- In vitro effects
- In vivo and ex vivo effects
Mammals
Humans
A document of the World Health Organization (2006, [15]), stated that there are no evidences on the short and long term adverse effects of the MRI static MF on human health. This statement has been confirmed also more recently [16,17]. Considering the increased use of MR scanners with higher static MF values, there is an urgent need to perform studies to provide assurance about their safety.
3.1.2. Effects of Gradient MF
- In vitro effects
- In vivo and ex vivo effects
Mammals
Humans
The use of MRI gradient MF represents a potential health risk beside peripheral nerve and cardiac stimulation to the patient. With the advent of the new generation of MR systems characterized by higher static MF and faster gradient fields, their effects on human health should be the object of further and properly designed studies.
3.1.3. Effects of RF Fields
- - non thermal effects: due to direct interactions between MFs and tissues
- - thermal effects: due to tissue heating caused by the induced electric currents
- In vitro effects
- In vivo and ex vivo effects
Mammals
Humans
Interactions between RF and biological tissues during MR procedures could be unsafe for patients [11]. Most of the reported accidents are burns due to hot spots in presence of conducting materials close to the patient such as the leads of physiological parameters (heart rate, blood pressure, oxygen saturation and temperature) monitoring equipment. This kind of risk can be more serious in case of internal biomedical implants (aneurism clips, stent, etc) especially for implants that have elongated configurations and/or are electronically activated (neurostimulation systems, cardiac pacemakers) [10,99,100].MRI generated RF are unlikely to be genotoxic, but unfortunately, to date no epidemiological studies are available to assess possible long term health effects due to these radiations.
3.1.4. Effects of combination of Static, Gradient and RF fields during MRI scan
3.2. Occupational Risk
4. Concluding Remarks
References
- Picano, E. Sustainability of medical imaging. BMJ 2004, 328, 578–580. [Google Scholar]
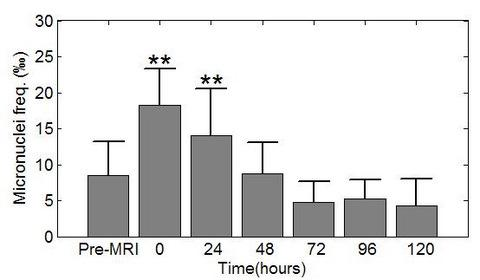
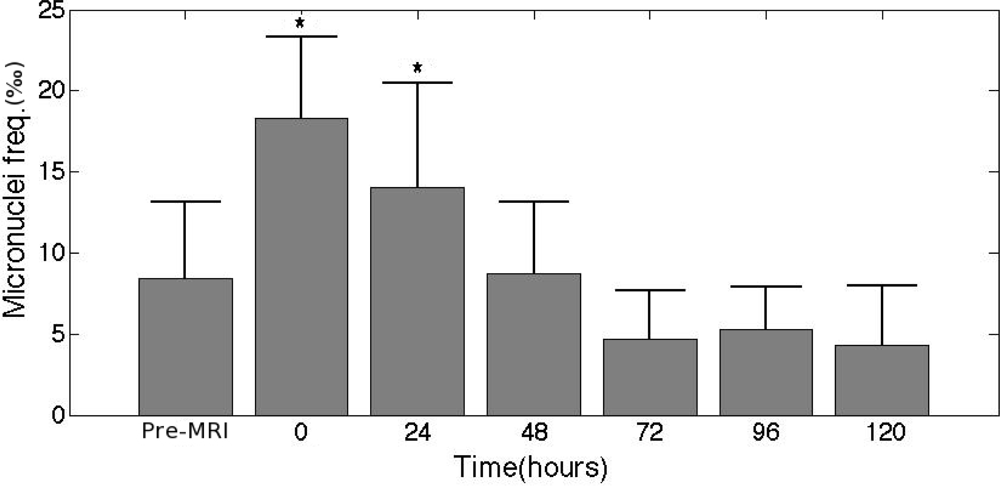
- Simi, S; Ballardin, M; Casella, M; De Marchi, D; Hartwig, V; Giovannetti, G; Vanello, N; Gabbriellini, S; Landini, L; Lombardi, M. Is the genotoxic effect of magnetic resonance negligible? Low persistence of micronucleus frequency in lymphocytes of individuals after cardiac scan. Mutat. Res. Fundam. Mol. Mech. Mutagen 2008, 645, 39–43. [Google Scholar]
- Bonassi, S; Znaor, A; Ceppi, M; Lando, C; Chang, WP; Holland, N; Kirsch-Volders, M; Zeiger, E; Ban, S; Barale, R; Bigatti, MP; Bolognesi, C; Cebulska-Wasilewska, A; Fabianova, E; Fucic, A; Hagmar, L; Joksic, G; Martelli, A; Migliore, L; Mirkova, E; Scarfi, MR; Zijno, A; Norppa, H; Fenech, M. An increased micronucleus frequency in peripheral blood lymphocytes predicts the risk of cancer in humans. Carcinogenesis 2007, 28, 625–631. [Google Scholar]
- McRobbie, DW; Moore, EA; Graves, MJ; Prince, MR. MRI - From Picture to Proton; Cambridge University Press: Cambridge, New York, USA, 2006. [Google Scholar]
- SCENIHR (Scientific Committee on Emerging and Newly Identified Health Risks) Possible effects of Electromagnetic Fields (EMF) on Human Health; European Commission: Brussels, Belgium, March 21 2007.
- International Commission on Non-ionizing Radiation Protection (ICNIRP): Guidelines for limiting exposure to time-varying electric, magnetic, and electromagnetic fields (up to 300GHz). Health Phys 1998, 74, 494–522.
- McNeil, DG. M.R.I.’s Strong Magnets Cited in Accidents. New York Times 2005. [Google Scholar]
- U.S. Food and Drug Administration (FDA) Medical Device Reporting (MDR). Available online: http://www.fda.gov (accessed May 4, 2009).
- ECRI Health Device Alerts (HDA). Available online: http://www.mdsr.ecri.org/ (accessed May 4, 2009).
- Shellock, FG. MR safety update 2002: implants and devices. J. Magn. Reson. Imaging 2002, 16, 485–496. [Google Scholar]
- Formica, D; Silvestri, S. Biological effects of exposure to magnetic resonance imaging: an overview. BioMed. Eng. OnLine 2004, 3, 11. [Google Scholar]
- Drinker, CK; Thomson, RM. Does the magnetic field constitute an industrial hazard? J. Ind. Hyg 1921, 3, 117–129. [Google Scholar]
- Davis, LD; Pappajohn, K; Plavnieks, IM. Bibliography of the biological effects of magnetic fields. Fed. Proc 1962, 21, 1–38. [Google Scholar]
- Schenck, JF. Safety of strong, static magnetic fields. J. Magn. Reson. Imaging 2000, 12, 2–19. [Google Scholar]
- World Health Organization. Environmental Health Criteria 232. Static fields; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Franco, G; Perduri, R; Murolo, A. Effetti biologici da esposizione occupazionale a campi magnetostatici utilizzati in imaging a risonanza magnetica nucleare: una rassegna. Med. Lav 2008, 99, 16–28. [Google Scholar]
- International Commission on Non-ionizing Radiation Protection (ICNIRP): Guidelines on limits of exposure to static magnetic field. Health Phys 2009, 96, 504–514.
- Nakahara, T; Yaguchi, H; Yoshida, M; Miyakoshi, J. Effects of Exposure of CHO-K1 Cells to a 10-T Static Magnetic Field. Radiology 2002, 224, 817–822. [Google Scholar]
- Valiron, O; Peris, L; Rikken, G; Schweitzer, A; Saoudi, Y; Remy, C; Job, D. Cellular disorders induced by high magnetic fields. J. Magn. Reson. Imaging 2005, 22, 334–340. [Google Scholar]
- Yamamoto, T; Nagayama, Y; Tamura, M. A blood oxygenation dependent increase in blood viscosity due to a static magnetic field. Phys. Med. Biol 2004, 49, 3267–3277. [Google Scholar]
- Myakoshi, J. Effects of a static magnetic field at the cellular level. Prog Biophys Mol Biol 2004. [Google Scholar]
- Tenforde, TS; Gaffey, CT; Moyer, BR; Budinger, TF. Cardiovascular alterations in Macaca monkeys exposed to stationary magnetic fields: experimental observations and theoretical analysis. Bioelectromagnetics 1983, 4, 1–9. [Google Scholar]
- Saunders, R. Static magnetic fields: animal studies. Prog. Biophys. Mol. Biol 2005, 87, 225–239. [Google Scholar]
- Wikswo, JP; Barach, JP. An estimate of the steady magnetic field strength required to influence nerve conduction. IEEE Trans. Biomed. Eng 1980, 27, 722–723. [Google Scholar]
- Rosen, AD; Lubowsky, J. Magnetic field influence on central nervous system function. Exp. Neurol 1987, 95, 679–687. [Google Scholar]
- Rosen, AD; Lubowsky, J. Modification of spontaneous unit discharge in the lateral geniculate body by a magnetic field. Exp. Neurol 1990, 108, 261–265. [Google Scholar]
- Rosen, AD. Mechanism of action of moderate-intensity static magnetic fields on biological systems. Cell Biochem. Biophys 2003, 39, 163–173. [Google Scholar]
- Kangarlu, A; Burgess, RE; Zhu, H; Nakayama, T; Hamlin, RL; Abduljalil, AM; Robataille, PML. Cognitive, cardiac, and physiological safety studies in ultra high field magnetic resonance imaging. Magn. Reson. Imaging 1999, 17, 1407–1416. [Google Scholar]
- High, WB; Sikora, J; Ugurbil, K; Garwood, M. Subchronic in vivo effects of a high static magnetic field (9.4 T) in rats. J. Magn. Reson. Imaging 2000, 12, 122–139. [Google Scholar]
- International Commission on Non-ionizing Radiation (ICNIRP). Exposure to static and low frequency electromagnetic fields. In Biological Effects and Health Consequences (0–100 kHz); Matthes, R; McKinlay, AF; Bernhardt, JH; Vecchia, P; Veyret, B (Eds.) ICNIRP: Munich, Germany, 2003.
- McKinlay, AF; Allen, SG; Cox, R; Dimbylow, PJ; Mann, SM; Muirhead, CR; Saunders, RD; Sienkiewicz, ZJ; Stather, JW; Wainwright, PR. Review of the Scientific Evidence for Limiting Exposure to Electromagnetic Fields (0–300 GHz); NRPB: Chilton, UK, 2003; Volume 15, 3. [Google Scholar]
- Suzuki, Y; Ikehata, M; Nakamura, K; Nishioka, M; Asanuma, K; Koana, T; Shimizu, H. Induction of micronuclei in mice exposed to static magnetic fields. Mutagenesis 2001, 16, 499–501. [Google Scholar]
- International Agency for Research on Cancer (IARC). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Non-Ionising Radiation. Part 1: Static and Extremely Low Frequency (ELF) Electric and Magnetic Fields, vol. 80; IARC: Lyon, France, 2002. [Google Scholar]
- Chakeres, DW; De Vocht, F. Static magnetic field effects on human subjects related to magnetic resonance imaging systems. Prog. Biophys. Mol. Biol 2005, 87, 255–265. [Google Scholar]
- De Vocht, F; van Drooge, H; Engels, H; Kromhout, H. Exposure, health complaints and cognitive performance among employees of an MRI scanners manufacturing department. J. Magn. Reson. Imaging 2006, 23, 197–204. [Google Scholar]
- De Vocht, F; Stevens, T; van Wendel De Joode, B; Engels, H; Kromhout, H. Acute neurobehavioral effects of exposure to static magnetic fields: analyses of exposure-response relations. J. Magn. Reson. Imaging 2006, 23, 291–297. [Google Scholar]
- Evans, JA; Savitz, DA; Kanal, E; Gillen, J. Infertility and pregnancy outcome among magnetic resonance imaging workers. J. Occup. Med 1993, 35, 1191–1195. [Google Scholar]
- De Wilde, JP; Rivers, AW; Proce, DL. A review of the current use of magnetic resonance imaging in pregnancy and safety implications for the fetus. Prog. Biophys. Mol. Biol 2005, 87, 335–353. [Google Scholar]
- Feychting, M. Health effects of static magnetic fields—a review of the epidemiological evidence. Prog. Biophy. Mol. Biol 2005, 87, 241–246. [Google Scholar]
- Greenland, S; Sheppard, AR; Kaune, WT; Poole, C; Kelsh, MA. A pooled analysis of magnetic fields, wire codes, and childhood leukemia. Childhood Leukemia-EMF Study Group. Epidemiology 2000, 11, 624–634. [Google Scholar]
- Foliart, DE; Pollock, BH; Mezei, G; Iriye, R; Silva, JM; Ebi, KL; Kheifets, L; Link, MP; Kavet, R. Magnetic field exposure and long-term survival among children with leukaemia. Br. J. Cancer 2006, 94, 161–164. [Google Scholar]
- Forssen, UM; Rutqvist, LE; Ahlbom, A; Feychting, M. Occupational magnetic fields and female breast cancer: a case-control study using Swedish population registers and new exposure data. Am. J. Epidemiol 2005, 161, 250–259. [Google Scholar]
- Vogt, FM; Ladd, ME; Hunold, P; Mateiescu, S; Hebrank, FX; Zhang, A; Debatin, JF; Göhde, SC. Increased time rate of change of gradient fields: effect on peripheral nerve stimulation at clinical MR imaging. Radiology 2004, 233, 548–554. [Google Scholar]
- Crozier, S; Wang, H; Trakic, A; Liu, F. Exposure of workers to pulsed gradients in MRI. J. Mag. Res. Imaging 2007, 26, 1236–1254. [Google Scholar]
- Lai, H; Singh, NP. Magnetic-field-induced DNA strand breaks in brain cells of the rat. Environ. Health Perspect 2004b, 112, 687–694. [Google Scholar]
- Fedrowitz, M; Westermann, J; Löscher, W. Magnetic field exposure increases cell proliferation but does not affect melatonin levels in the mammary gland of female Sprague Dawley rats. Cancer Res 2002, 62, 1356–1363. [Google Scholar]
- Scarfí, MR; Sannino, A; Perrotta, A; Sarti, M; Mesirca, P; Bersani, F. Evaluation of genotoxic effects in human fibroblasts after intermittent exposure to 50 Hz electromagnetic fields: a confirmatory study. Radiat. Res 2005, 164, 270–276. [Google Scholar]
- Geard, CR; Osmak, RS; Hall, EJ; Simon, HE; Maudsley, AA; Hilal, SK. Magnetic resonance and ionizing radiation: A comparative evaluation in vivo of oncogenic and genotoxic potential. Radiology 1984, 152, 199–202. [Google Scholar]
- Cohen, MM; Kunska, A; Astemborski, JA; McCulloch, D; Paskewitz, DA. Effect of low-level, 60-Hz electromagnetic fields on human lymphoid cells: I. Mitotic rate and chromosome breakage in human peripheral lymphocytes. Bioelectromagnetics 1986, 7, 415–423. [Google Scholar]
- Cohen, MM; Kunska, A; Astemborski, JA; McCulloch, D. Effect of low-level, 60-Hz electromagnetic fields on human lymphoid cells: II. Sister-chromatid exchanges in peripheral lymphocytes and lymphoblastoid cell lines. Mutat. Res 1986, 172, 177–184. [Google Scholar]
- Liboff, AR; Williams, T, Jr; Strong, DM; Wistar, R, Jr. Time-varying magnetic fields: effect on DNA synthesis. Science 1984, 223, 818–820. [Google Scholar]
- Rodegerdts, EA; Gronewaller, EF; Kehlbach, R; Roth, P; Wiskirchen, J; Gebert, R; Claussen, CD; Duda, S. In vitro evaluation of teratogenic effects by time-varying MR gradient fields on fetal human fibroblast. J. Magn. Reson. Imaging 2000, 12, 150–156. [Google Scholar]
- Juutilainen, J; Kumlin, T; Naarala, J. Do extremely low frequency magnetic fields enhance the effects of environmental carcinogens? A meta-analysis of experimental studies. Int. J. Radiat. Biol 2006, 82, 1–12. [Google Scholar]
- Nyenhuis, JA; Bourland, JD; Mouchawar, GA; Elabbady, TZ; Geddes, LA; Schaefer, DJ; Riehl, ME. Comparison of stimulation effects of longitudinal and transverse MRI gradient coils. Proceedings of the Society for Magnetic Resonance in Medicine Annual Meeting, San Francisco, CA, USA, 1990; p. 1275.
- Bourland, JD; Nyenhuis, JA; Mouchawar, GA; Geddes, LA; Schaefer, DJ. Human peripheral nerve stimulation from z-gradients. Proceedings of the Society for Magnetic Resonance in Medicine Annual Meeting, San Francisco, CA, USA, 1990; p. 1157.
- Bourland, JD; Nyenhuis, JA; Schaefer, DJ. Physiologic effects of intense MR Imaging gradient fields. Neuroimaging Clin. N. Am 1999, 9, 363–377. [Google Scholar]
- Juutilainen, J. Developmental effects of electromagnetic fields. Bioelectromagnetics 2005, 7, S107–115. [Google Scholar]
- Schaefer, DJ; Bourland, JD; Nyenhuis, JA. Review of patient safety in time-varying gradient fields. J. Magn. Reson. Imaging 2000, 12, 20–29. [Google Scholar]
- International Electrotechnical Commission. IEC 60601-2-33 Particular requirements for basic safety and essential performance of magnetic resonance equipment for medical diagnosis, 2nd ed; International Electrotechnical Commission: Geneva, Switzerland, 2002. [Google Scholar]
- Reilly, JP. Applied bioelectricity: From electrical stimulation to electropathology; Springer-Verlag: New York, NY, USA, 1998. [Google Scholar]
- Krasin, F; Wagner, H. Biological effects of nonionizing electromagnetic radiation. In Encyclopedia of Medical Devices and Instrumentation; Webster, JG, Ed.; Wiley: Hoboken, NJ, USA, 1988. [Google Scholar]
- Polk, C. Biological effects of nonionizing electromagnetic fields. In Handbook of biomedical engineering; Bronzino, JD, Ed.; J. CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Shellock, FG. Radiofrequency energy-induced heating during MR procedures: a review. J. Magn. Reson. Imaging 2000, 12, 30–36. [Google Scholar]
- Shellock, FG; Rothman, B; Sarti, D. Heating of the scrotum by highfield strength MR imaging. Am. J. Roentgenol 1990, 154, 1229–1232. [Google Scholar]
- Shellock, FG; Crues, JV. Corneal temperature changes associated with high-field MR imaging using a head coil. Radiology 1988, 167, 809–811. [Google Scholar]
- Vahlensieck, M. Tattoo-related cutaneous inflammation (burn grade I) in a mid-field MR scanner (letter). Eur. Radiol 2000, 10, 197. [Google Scholar]
- Wagle, WA; Smith, M. Tattoo-induced skin burn during MR imaging (letter). Am. J. Roentgenol 2000, 174, 1795. [Google Scholar]
- Kreidstein, ML; Giguere, D; Freiberg, A. MRI interaction with tattoo pigments: case report, pathophysiology, and management. Plast. Reconstr. Surg 1997, 99, 1717–1720. [Google Scholar]
- Bottomley, PA; Redington, RW; Edelstein, WA; Schenck, JF. Estimating radiofrequency power deposition in body NMR imaging. Magn. Reson. Med 1985, 2, 336–349. [Google Scholar]
- Adair, ER; Berglund, LG. On the thermoregulatory consequences of NMR imaging. Magn. Reson. Imaging 1986, 4, 321–333. [Google Scholar]
- Shellock, FG; Schaefer, DJ; Crues, JV. Alterations in body and skin temperatures caused by MR imaging: is the recommended exposure for radiofrequency radiation too conservatice? Brit. J. Radiol 1989, 62, 904–909. [Google Scholar]
- Van den Berg, CAT; van den Bergen, B; van de Kamer, JB; Raaymakers, BW; Kroeze, H; Bartels, LW; Lagendijk, JJW. Simultaneous B+1 Homogenization and Specific Absorption Rate Hotspot Suppression Using a Magnetic Resonance Phased Array Transmit Coil. Mag. Res. Med 2007, 57, 577–586. [Google Scholar]
- Liu, F; Zhao, HW; Crozier, S. Calculation of electric fields induced by body and head motion in high-field MRI. J. Magn. Reson 2003, 161, 99–107. [Google Scholar]
- Meltz, ML. Radiofrequency exposure and mammalian cell toxicity, genotoxicity, and transformation. Biolectrom. Suppl 2003, 6, S196–S213. [Google Scholar]
- Diem, E; Schwarz, C; Adlkofer, F; Jahn, O; Rudiger, H. Non-thermal DANN breakage by mobile phone radiation (1800 MHz) in human fibroblasts and in transformed GFSH-R17 rat granulosa cells in vitro. Mutat. Res 2005, 583, 178–183. [Google Scholar]
- Nikolova, T; Czyz, J; Rolletschek, A; Blyszczuk, P; Fuchs, J; Jovtchev, G. Electromagnetic fields affect transcript levels of apoptosis-related genes in embryonic stem cell-derived neural progenitor cells. FASEB J 2005, 19, 1686–1693. [Google Scholar]
- Speit, G; Schutz, P; Hoffmann, H. Genotoxic effects of exposure to radiofrequency electromagnetic fields (RF-EMF) in cultured mammalian cells are not independently reproducible. Mutat. Res 2007, 626, 42–47. [Google Scholar]
- Lantow, M; Viergutz, T; Weiss, DG; Simkó, M. Comparative study of cell cycle kinetics and induction of apoptosis or necrosis after exposure to radiofrequency radiation in human Mono Mac 6 cells. Radiat. Res 2006c, 166, 539–543. [Google Scholar]
- Caraglia, M; Marra, M; Mancinelli, F; D’Ambrosio, G; Massa, R; Giordano, A; Budillon, A; Abbruzzese, A; Bismuto, E. Electromagnetic fields at mobile phone frequency induce apoptosis and inactivation of the multi-chaperone complex in human epidermoid cancer cells. J. Cell. Physiol 2005, 204, 39–48. [Google Scholar]
- Scarfì, MR; Fresegna, AM; Villani, P; Pinto, R; Marino, C; Sarti, M; Altavista, P; Sannino, A; Lovisolo, GA. Exposure to radiofrequency radiation (900 MHz, GSM signal) does not affect micronucleus frequency and cell proliferation in human peripheral blood lymphocytes: an interlaboratory study. Radiat. Res 2006, 165, 655–663. [Google Scholar]
- Zeni, O; Schiavoni, A; Perrotta, A; Forigo, D; Depilano, M; Scarfi, MR. Evaluation of genotoxic effects in human leukocytes after in vitro exposure to 1950 MHz UMTS radiofrequency field. Bioelectromagnetics 2008, 29, 177–184. [Google Scholar]
- Saunders, RD; Kowalczuk, CI; Beechley, CV; Dunford, R. Studies of the induction of dominant lethal and translocations in male mice after chronic exposure to microwave radiation. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med 1988, 53, 983–992. [Google Scholar]
- Lloyd, DC; Saunders, RD; Moquet, JE; Kowalczuk, CI. Absence of chromosomal damage in human lymphocytes exposed to microwave radiation with hyperthermia. Bioelectromagnetics 1986, 7, 235–237. [Google Scholar]
- Muscat, JE; Malkin, MG; Thompson, S; Shore, RE; Stellman, SD; McRee, D; Neugut, AI; Wynder, EL. Handheld cellular telephone use and risk of brain cancer. JAMA 2000, 284, 3001–3007. [Google Scholar]
- Inskip, PD; Hatch, EE; Stewart, PA; Heineman, EF; Ziegler, RG; Dosemeci, M; Parry, D; Rothman, N; Boice, JD; Wilcosky, TC; Watson, DJ; Shapiro, WR; Selker, RG; Fine, HA; Black, P; Loeffler, JS; Linet, MS. Study design for a case-control investigation of cellular telephones and other risk factors for brain tumours in adults. Radiat. Prot. Dosim 1999, 86, 45–52. [Google Scholar]
- Hardell, L; Nasman, A; Pahlson, A; Hallquist, A; Hansson Mild, K. Use of cellular telephones and the risk for brain tumours: a case–control study. Int. J. Oncol 1999, 15, 113–116. [Google Scholar]
- Lonn, S; Ahlbom, A; Hall, P; Feychting, M. Long-term mobile phone use and brain tumour risk. Am. J. Epidemiol 2005, 161, 526–535. [Google Scholar]
- Schüz, J; Böhler, E; Berg, G; Schlehofer, B; Hettinger, I; Schlaefer, K; Wahrendorf, J; Kunna-Grass, K; Blettner, M. Cellular phones, cordless phones, and the risk of glioma and meningioma (Interphone study group, Germany). Am. J. Epidemiol 2006a, 163, 512–520. [Google Scholar]
- Elwood, JM. Epidemiological studies of radio frequency exposures and human cancer. Biolectrom Suppl 2003, 6, S63–S73. [Google Scholar]
- The Possible Harmful Biological Effects of Low Level Electromagnetic Fields of Frequencies up to 300 GHz. 2002. Available online: http://materias.fi.uba.ar/6209/download/IEE2002.pdf (accessed June 5, 2009).
- Christ, A; Kuster, N. Differences in RF energy absorption in the heads of adults and children. Bioelectromagnetics 2005, 7, S31–44. [Google Scholar]
- Regel, SJ; Negovetic, S; Röösli, M; Berdinas, V; Schuderer, J; Huss, A; Lott, U; Kuster, N; Achermann, P. UMTS base station-like exposure, well being and cognitive performance. Environ. Health Perspect 2006, 114, 1270–1275. [Google Scholar]
- Schaefer, DJ; Barber, BJ; Gordon, CJ; Zielonka, J; Hecker, J. Thermal effects of magnetic resonance imaging. In Book of abstracts, Society for magnetic resonance in medicine Volume 2; Society for Magnetic Resonance in Medicine: Berkeley, CA, USA, 1985; p. 925. [Google Scholar]
- Shellock, FG; Schaefer, DJ; Crues, JV. Evaluation of skin blood flow, body and skin temperatures in man during MR imaging at high levels of RF energy. Magn. Reson. Imaging 1989, 7, 335. [Google Scholar]
- Kido, DK; Morris, TW; Erickson, JL; Plewes, DB; Simon, JH. Physiologic changes during high field strength MR imaging. Am. J. Roentgenol 1987, 148, 1215–1218. [Google Scholar]
- Shellock, FG; Schaefer, DJ; Kanal, E. Physiologic responses to MR imaging performed at an SAR level of 6.0 W/Kg. Radiology 1994, 192, 865–868. [Google Scholar]
- Shellock, FG; Schatz, CJ. Increases in corneal temperature caused by MR imaging of the eye with a dedicated local coil. Radiology 1992, 185, 697–699. [Google Scholar]
- International Commission on Non-ionizing Radiation Protection (ICNIRP): medical magnetic resonance (MR) procedures: Protection of patients. Health Phys. Soc 2004, 87, 197–216.
- Shellock, FG; Crues, JV. MR Procedures: biologic effects, safety, and patient care. Radiology 2004, 232, 635–652. [Google Scholar]
- Shellock, FG; Slimp, G. Severe burn of the finger caused by using a pulse oximeter during MR imaging (letter). Am. J. Roentgenol 1989, 153, 1105. [Google Scholar]
- Shellock, FG; Spinazzi, A. MRI safety update 2008: part 1, MRI contrast agents and nephrogenic systemic fibrosis. Am. J. Roentgenol 2008, 191, 1129–1139. [Google Scholar]
- Schiffer, IB; Schreiber, WG; Graf, R; Schreiber, EM; Jung, D; Rose, DM; Hehn, M; Gebhard, S; Sagemüller, J; Spiess, HW; Oesch, F; Thelen, M; Hengstler, JG. No influence of magnetic fields on cell cycle progression using conditions relevant for patients during MRI. Bioelectromagnetics 2003, 24, 241–250. [Google Scholar]
- Magin, RL; Lee, JK; Klintsova, A; Carnes, KI; Dunn, F. Biological Effects of Long-Duration, High-Field (4 T) MRI on Growth and Development in the Mouse. J. Mag. Res. Imaging 2000, 12, 140–149. [Google Scholar]
- Ali, MA. Magnetic resonance imaging and associated alteration in some biophysical properties of blood. Rom. J. Biophys 2007, 17, 277–286. [Google Scholar]
- Karpowicz, J; Hietanen, M; Gryz, K. Occupational risk from static magnetic fields of MRI scanners. Environmentalist 2007, 27, 533–538. [Google Scholar]
- Available online: http://www.hse.gov.uk/radiation/nonionising/l184emf.pdf (accessed June 21, 2005).
- Zaremba, LA. Guidance for Industry and FDA Staff: Criteria for Significant Risk Investigations of Magnetic Resonance Diagnostic Devices US; Department of Health and Human Services Food and Drug Administration: Silver Spring, MD, USA, 2003. Available online: http://www.fda.gov/cdrh/ode/guidance/793.pdf (accessed December 2003).
- IAPO. Policy Statement on Patient Involvement; IAPO: London, UK, 2005. Available online: http://www.patientsorganizations.org/ (accessed June 2005).
- Welch, HG; Schwartz, L; Woloshin, S. What’s making us sick is an epidemic of diagnoses. New York Times 2007. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hartwig, V.; Giovannetti, G.; Vanello, N.; Lombardi, M.; Landini, L.; Simi, S. Biological Effects and Safety in Magnetic Resonance Imaging: A Review. Int. J. Environ. Res. Public Health 2009, 6, 1778-1798. https://doi.org/10.3390/ijerph6061778
Hartwig V, Giovannetti G, Vanello N, Lombardi M, Landini L, Simi S. Biological Effects and Safety in Magnetic Resonance Imaging: A Review. International Journal of Environmental Research and Public Health. 2009; 6(6):1778-1798. https://doi.org/10.3390/ijerph6061778
Chicago/Turabian StyleHartwig, Valentina, Giulio Giovannetti, Nicola Vanello, Massimo Lombardi, Luigi Landini, and Silvana Simi. 2009. "Biological Effects and Safety in Magnetic Resonance Imaging: A Review" International Journal of Environmental Research and Public Health 6, no. 6: 1778-1798. https://doi.org/10.3390/ijerph6061778