Real World Study to Evaluate the Effectiveness of Varenicline and Cognitive-Behavioural Interventions for Smoking Cessation
Abstract
:1. Introduction
2. Methods
2.1. Study Design
2.2. Participants
2.3. Intervention
2.4. Assessments
2.5. Statistical Analysis
3. Results
4. Discussion
References
- Aveyard, P; West, R. Managing smoking cessation. BMJ 2007, 335, 37–41. [Google Scholar]
- Hyland, A; Borland, R; Li, Q; Yong, HH; McNeil, A; Fong, GT; Connor, RJO; Cummings, KM. Individual level predictors of cessation behaviors among participants in the international tobacco control four country survey. Tob. Control 2006, 15, 83–94. [Google Scholar]
- Zhu, S; Melcer, T; Sun, J; Rosbrook, B; Pierce, JP. Smoking cessation with and without assistance: a population-baed analysis. Am. J. Prev. Med 2000, 18, 305–311. [Google Scholar]
- Henningfield, JE; Keenan, RM. Nicotine dilevery kinetics and abuse liability. J. Consult. Clin. Psychol 1993, 61, 743–750. [Google Scholar]
- Siu, CK; Tyndale, RF. Non-Nicotinic therapies for smoking cessatio. Annu. Rev. Pharmacol. Toxicol 2007, 47, 541–564. [Google Scholar]
- Benowitz, NL. Clinical pharmacology of nicotine: implications for understanding, preventing, and treating tobacco addiction. Clin. Pharma. Ther 2008, 83, 531–541. [Google Scholar]
- Learned, SM; Bergstrom, M; Savitvheva, I; Ascher, J; Schimd, VD; Langstrom, B. In vivo activity of bupropion at the human dopamine transporter as measured by positron emission tomography. Biol. Psychiatry 2003, 54, 800–805. [Google Scholar]
- Coe, JW; Brooks, PR; Vetelino, MG; Wirtz, MC; Arnold, EP; Lebel, LA; Fox, CB; Sands, SB; Davis, TI; Schulz, DW; et al. An alpha4-beta2 nicotine receptor partial agonist for smoking cessation. J. Med. Chem 2005, 48, 3474–3477. [Google Scholar]
- Rollema, H; Chmabers, LK; Coe, JW; Glowa, J; Hurst, RS; Lebel, LA; Lu, Y; Mansbach, RS; Mather, RJ; Rovetti, CC; et al. Pharmacological profile of the alpha4-beta2 nicotine acetylcholine receptor partial agonist varenicline, an effetive smoking cessation aid. Neuropharmacology 2007, 52, 985–994. [Google Scholar]
- Williams, KE; Reeves, KR; Billing, CB; Pennington, GJ. A double-blind study evaluating the long-term safety of vareniclie for smoking cessation. Curr. Med. Res. Opin 2007, 23, 793–801. [Google Scholar]
- Jorenby, DE; Hays, JT; Rigotti, NA; Azoulay, S; Watsky, EJ; Williams, KE; Billing, CB; Gong, J; Reeves, KR; Varenicline Phase 3 Study Group. Efficacy of varenicline, an alpha4-beta2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: A randomized controlled trial. JAMA 2006, 296, 56–63. [Google Scholar]
- Gonzales, D; Renard, SI; Nides, M; Oncken, C; Azoulay, S; Billing, CB; Watsky, EJ; Gong, J; Williams, KE; Reeves, KR; Varenicline Phase 3 Study Group. Varenicline, an alpha4-beta2 nicotinic acetylcholine receptor partial agonist, vs sustained-release bupropion and placebo for smoking cessation: A randomized controlled trial. JAMA 2006, 296, 47–55. [Google Scholar]
- Tsai, ST; Cho, HJ; Cheng, HS; Kim, CH; Hsueh, KC; Billing, CB; Williams, KE. A randomized placebo-controlled trial of varenicline, a selective alpha4-beta2 nicotinic acetylcholine receptor partial agonist, as a new therapy for smoking cessation in asian smokers. Clin. Thepareutics 2007, 29, 1027–1039. [Google Scholar]
- Aubin, HJ; Bobak, A; Britton, JR; Oncken, CH; Billing, CB; Gong, J; Williams, KE; Reeves, KR. Varenicline versus transdermal nicotine patch for smoking cessation: Results from a randomized, open-label trial. Thorax 2008, 63, 717–724. [Google Scholar]
- Eisenberg, MJ; Filion, KB; Yavin, D; Bélisle, P; Mottillo, S; Joseph, L; Gervais, A; O'Loughlin, J; Paradis, D; Rinfret, S; Pilote, L. Pharmacotherapies for smoking cessation: a meta-analysis of randomized controlled trials. CMAJ 2008, 179, 135–144. [Google Scholar]
- Caponnetto, P; Polosa, R. Common predictors of smoking cessation in clinical practice. Respir. Med 2008, 102, 1182–1192. [Google Scholar]
- Stapleton, JA; Watson, L; Spirling, L; Smith, R; Milbrandt, A; Ratcliffe, M; Sutherland, G. Verenicline in the routine treatment of tobacco dependence: a pre-post comparison with nicotine replacement therapy and an evaluation in those with mental illness. Addiction 2007, 103, 146–154. [Google Scholar]
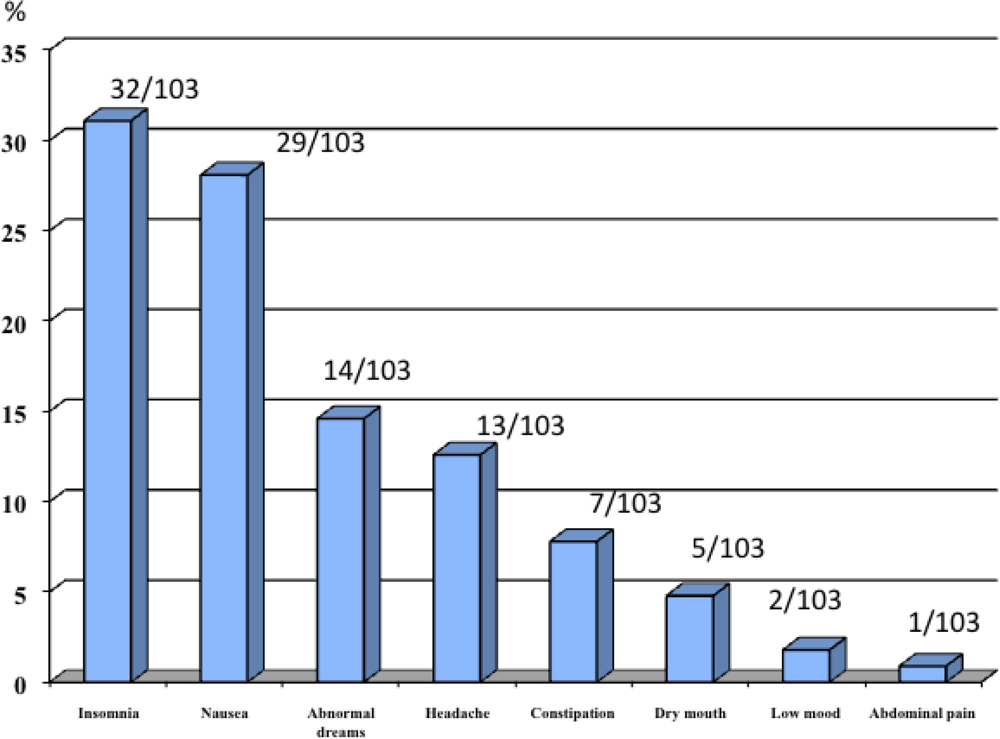
| Male N = 155 | Female N = 109 | Total N = 264 | p value | |
|---|---|---|---|---|
| Age mean (SD) | 45.9 (10.4) | 39.5 (7.9) | 43.7 (10.1) | < 0.001* |
| Cig/day. Mean (Range) | 28 (15–50) | 22 (10–40) | 23 (10–50) | 0.008* |
| N° previous attempts. Mean (SD) | 1.6 (1.0) | 1.4 (1.3) | 1.5 (1.1) | 0.6* |
| Age starting smoking. Mean (SD) | 17.1 (5.0) | 17.5 (3.4) | 17.3 (4.5) | 0.9* |
| Fagerström test score. Mean (SD) | 6.4 (2.5) | 6.0 (2.6) | 6.3 (2.5) | 0.4* |
| Longest previous abstinence (days) Mean (Range) | 6.2 (0–60) | 7.3 (0–100) | 6.5 (0–100) | 0.7* |
| wks 2–4 CAR (n/N) | wks 2–6 CAR (n/N) | wks 2–12 CAR (n/N) | |
|---|---|---|---|
| Males | 79.3% (123/155) | 67.1% (104/155) | 56.1% (87/155) |
| Females | 77.9% (85/109) | 66.0% (72/109) | page 6 second parag. |
| Total | 78.8% (208/264) | 66.7% (176/264) | 58.3% (154/264) |
| Abstainers at week 12 (Quitters/Total) | Crude OR for abstinence at week 12 (95% CI**) | Adjusted OR* for abstinence at week 12 (95% CI**) | |
|---|---|---|---|
| Associated Disease | |||
| None | 114/185 | 1.0 | |
| Yes
| 40/79
| 1.21 (0.81–1.93)
| |
| Cig/day | |||
| > 30 | 25/68 | 1.0 | 1.0 |
| 21–30 | 41/58 | 0.52 (0.16–0.60) | 0.46 (0.10–0.68) |
| ≤ 20 | 88/138 | 0.58 (0.20–0.78) | 0.50 (0.16–0.80) |
| p for trend 0.02
| |||
| Previous attempts | |||
| 0 | 51/102 | 1.0 | 1.0 |
| >= 1
| 103/162
| 0.79 (0.25–1.00)
| 0.67 (0.22–0.96)
|
| FTND score | |||
| > 6 | 70/136 | 1.0 | 1.0 |
| 3–6 | 49/80 | 0.84 (0.47–1.97) | 0.74 (0.38–2.0) |
| < 3 | 35/48 | 0.71 (0.17–0.90) | 0.80 (0.51–1.8) |
| p for trend 0.4
| |||
| Other smoker in household | |||
| Yes | 43/105 | 1.0 | 1.0 |
| Non | 111/159 | 0.59 (0.25–0.98) | 0.50 (0.21–0.87) |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ramon, J.M.; Bruguera, E. Real World Study to Evaluate the Effectiveness of Varenicline and Cognitive-Behavioural Interventions for Smoking Cessation. Int. J. Environ. Res. Public Health 2009, 6, 1530-1538. https://doi.org/10.3390/ijerph6041530
Ramon JM, Bruguera E. Real World Study to Evaluate the Effectiveness of Varenicline and Cognitive-Behavioural Interventions for Smoking Cessation. International Journal of Environmental Research and Public Health. 2009; 6(4):1530-1538. https://doi.org/10.3390/ijerph6041530
Chicago/Turabian StyleRamon, Josep Mª., and Eugeni Bruguera. 2009. "Real World Study to Evaluate the Effectiveness of Varenicline and Cognitive-Behavioural Interventions for Smoking Cessation" International Journal of Environmental Research and Public Health 6, no. 4: 1530-1538. https://doi.org/10.3390/ijerph6041530
APA StyleRamon, J. M., & Bruguera, E. (2009). Real World Study to Evaluate the Effectiveness of Varenicline and Cognitive-Behavioural Interventions for Smoking Cessation. International Journal of Environmental Research and Public Health, 6(4), 1530-1538. https://doi.org/10.3390/ijerph6041530




