The Use of Zebrafish (Danio rerio) Behavioral Responses in Identifying Sublethal Exposures to Deltamethrin
Abstract
:1. Introduction
2. Experimental Section
2.1. Test Species and Test Chemical
2.2. Experimental Setup
2.2.1. Acute Toxicity Test
2.2.2. Behavioral Response Monitoring
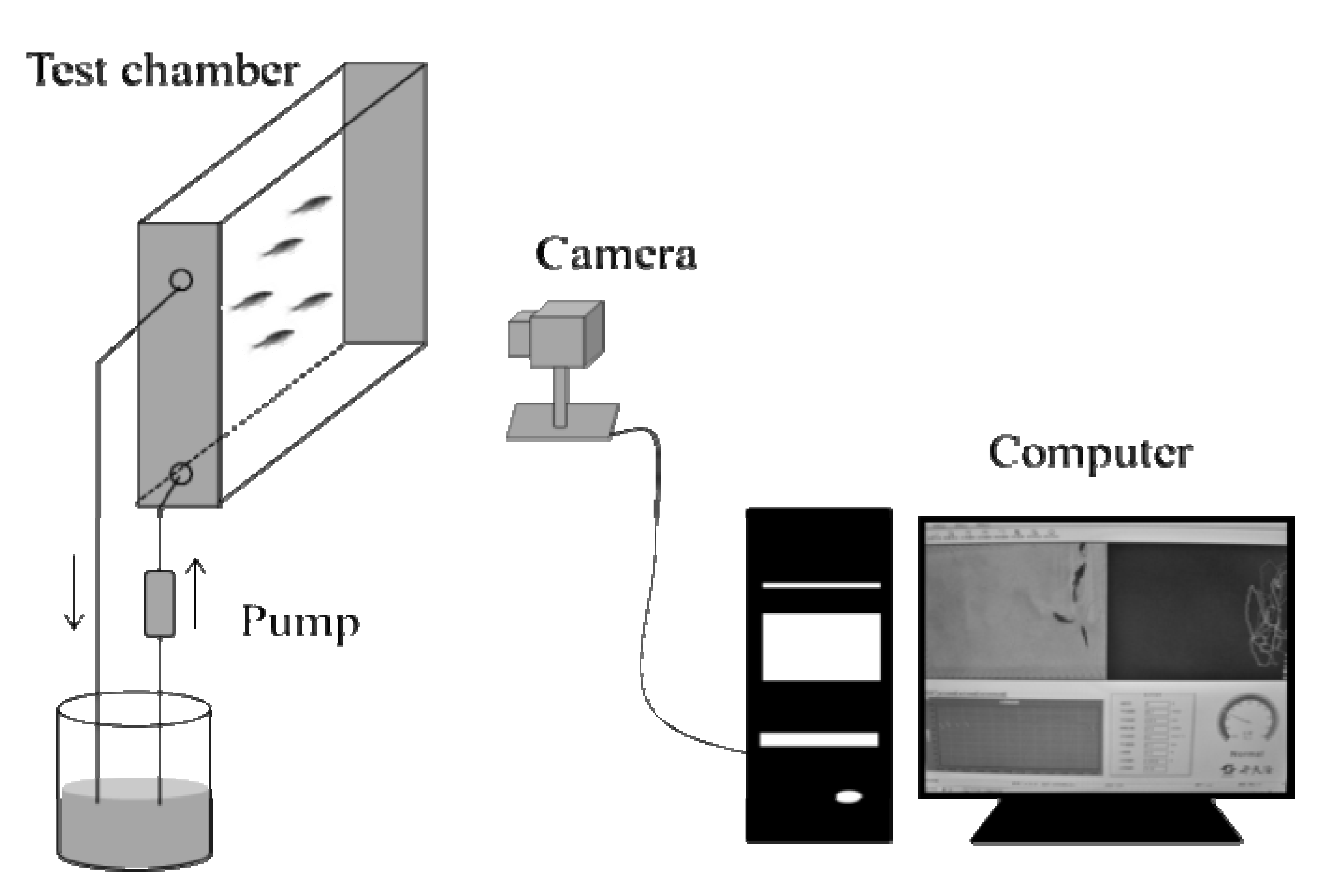
2.3. Data Analysis
3. Results
3.1. Acute Toxicity Test
| Pesticide | Slope | LC50-24h (μg/L) | 95% Confidence Limit | |
|---|---|---|---|---|
| Upper limit | Lower limit | |||
| Deltamethrin | 5.4621 | 14.43 ± 1.031 | 15.36 | 13.56 |
3.2. Behavioral Responses of Zebrafish to DM
| Time Interval (min) | Concentration (μg/L) | |||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 0.15 | 1.5 | 3.75 | 7.5 | 15 | |||
| Speed | Average | 0–60 | 39.6 ± 5.8 | 39.6 ± 1.1 | 42.3 ± 2.5 | 48.7 ± 3.4 | 46.7 ± 3.8 | 43 ± 4.4 |
| 60–120 | 38 ± 4.7 | 49.7 ± 5.9 | 58.6 ± 6.7 | 58 ± 9.9 | 61.2 ± 7.8 * | 54 ± 6.8 * | ||
| 120–180 | 42.6 ± 4.3 | 48.6 ± 1.5 | 57.6 ± 9 | 32.3 ± 9.5 | 46.7 ± 11.2 | 31.3 ± 9.6 | ||
| 180–240 | 42.3 ± 3.5 | 32.8 ± 1.9 | 42.3 ± 9.2 | 22 ± 5.4 * | 38.4 ± 3.2 | 31.1 ± 11.8 | ||
| 240–300 | 44 ± 6.7 | 27.0 ± 5.8 | 41.9 ± 18 | 18.2 ± 16.9 * | 37.3 ± 9.5 | 36.9 ± 12 | ||
| 300–360 | 41.8 ± 10.5 | 28.06 ± 7.18 | 42.5 ± 12.9 | 19.4 ± 16 * | 38.6 ± 7.8 | 38.2 ± 14.1 | ||
| Max | 0–60 | 52 ± 7.3 | 55.7 ± 4.3 | 57.4 ± 4.8 | 61.3 ± 1.4 | 59.9 ± 11.2 | 60.5 ± 0.6 | |
| 60–360 | 60.6 ± 8.3 | 64.26 ± 2.8 | 87.1 ± 13.4 * | 79.8 ± 9.3 * | 84.9 ± 9.4 * | 99.5 ± 5.4 * | ||
| Depth | Average | 0–60 | 156.9 ± 17 | 177.1 ± 25.4 | 173.9 ± 28.7 | 157.5 ± 25.6 | 126.1 ± 21.2 | 131.5 ± 36.3 |
| 60–120 | 142.1 ± 9.8 | 179.2 ± 11.9 | 187.8 ± 6.5 | 163.1 ± 10.2 | 178 ± 19.6 | 164.2 ± 41.1 | ||
| 120–180 | 135.4 ± 24.2 | 169.5 ± 10.9 | 189.1 ± 31.5 | 193.3 ± 14.2 * | 181.2 ± 36.7 * | 199.4 ± 38.3 * | ||
| 120–240 | 128.7 ± 13.7 | 176.6 ± 15.2 | 198.6 ± 15.2 | 189.3 ± 8.8 * | 212.9 ± 9.7 * | 219.9 ± 21 * | ||
| 240–300 | 144.5 ± 17.5 | 160.2 ± 31 | 209.8 ± 5 * | 168.7 ± 37.6 | 192.2 ± 27.6 * | 244.7 ± 7.6 * | ||
| 300–360 | 137.2 ±19.9 | 171.7 ± 36.7 | 221.9 ± 3.4 * | 121.9 ± 47.1 | 215.2 ± 31.8 * | 228.8 ± 5.4 * | ||
| Max | 0–60 | 232 ± 22.8 | 241.5 ± 28.8 | 237.3 ± 23 | 236.2 ± 2.6 | 191.8 ± 21.2 | 216.7 ± 21.2 | |
| 60–360 | 252.5 ± 15.8 | 258.4 ± 9.1 | 271.8 ± 14.5 | 294.2 ± 10.2 * | 285.2 ± 25 * | 269.9 ± 6.1 * | ||
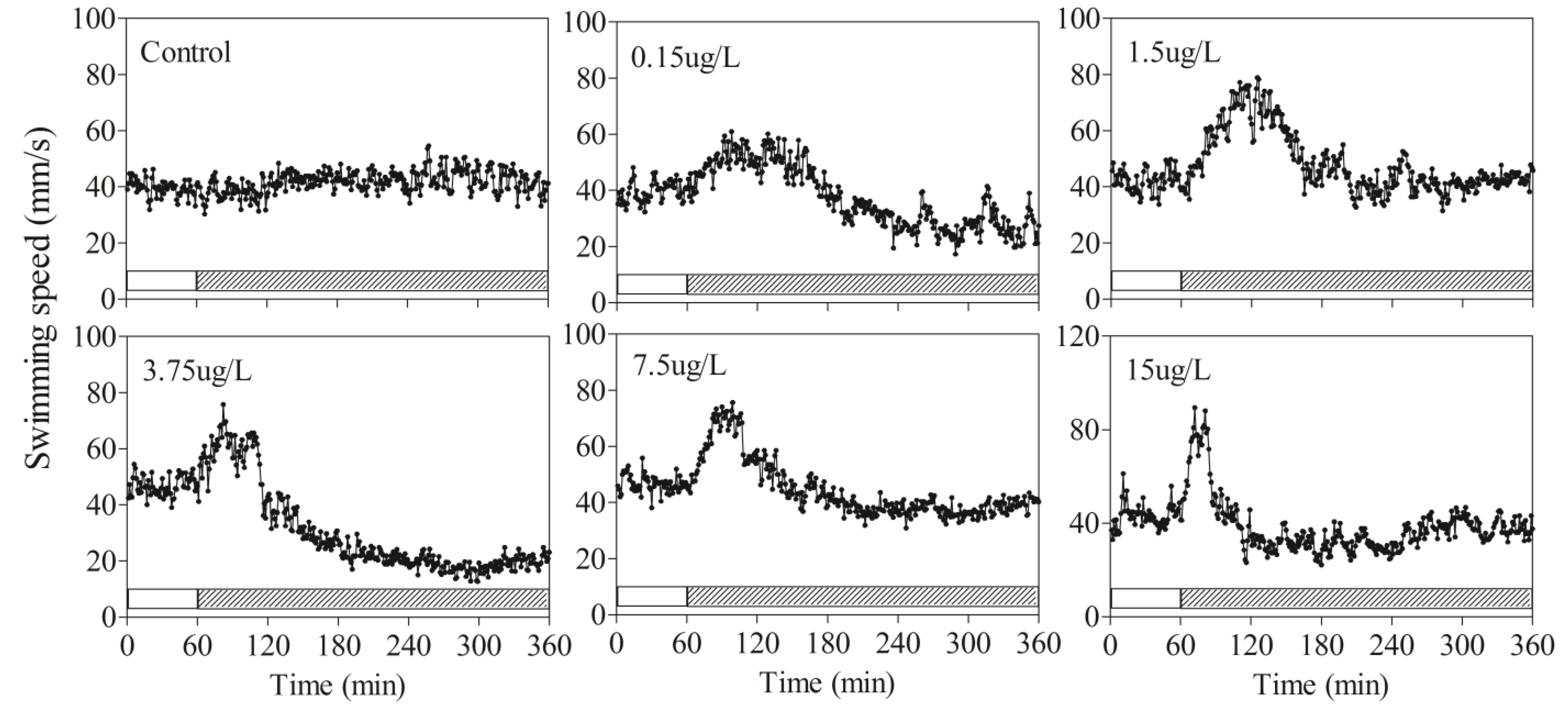
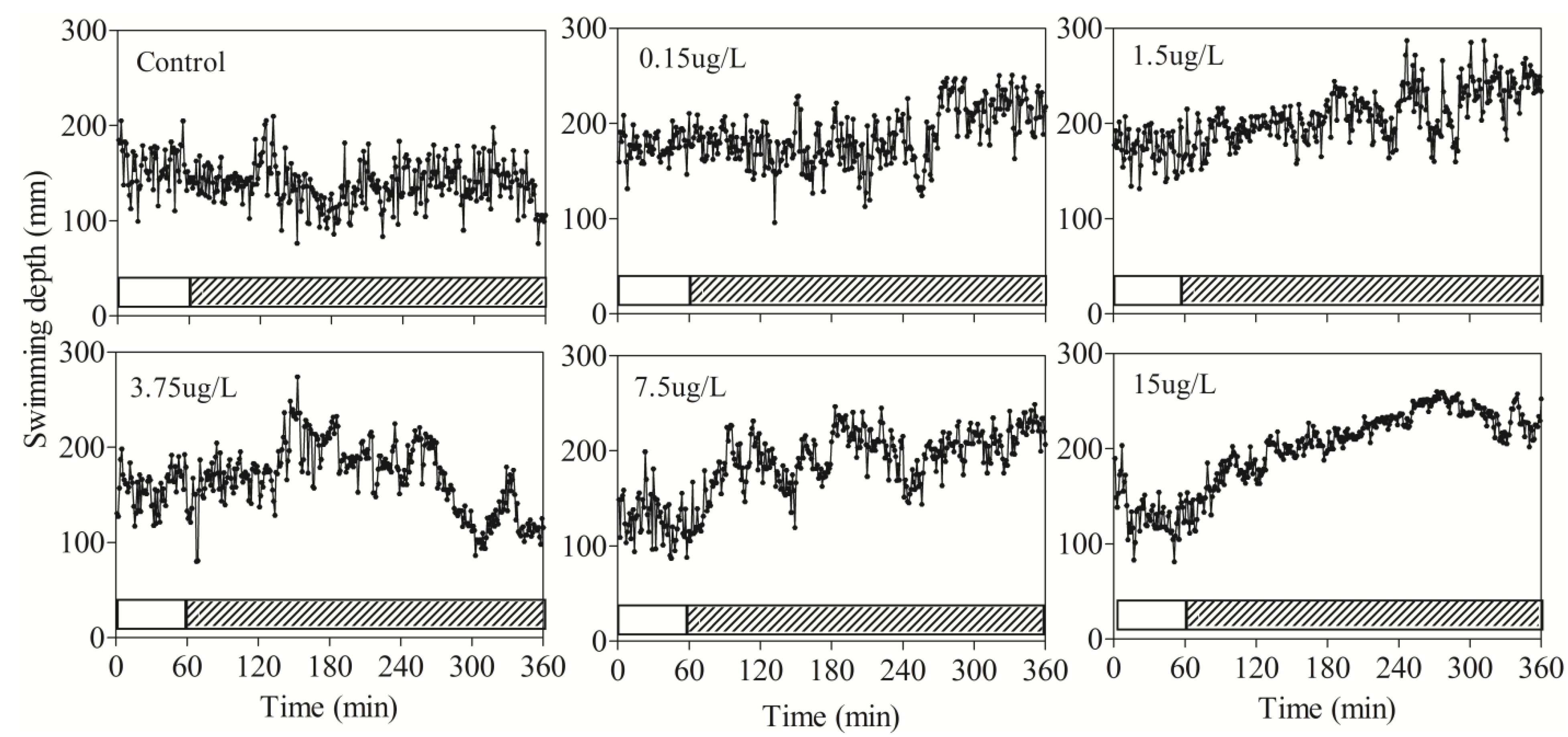

4. Discussion and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shukla, Y.; Yadav, A.; Arora, A. Carcinogenic and cocarcinogenic potential of cypermethrin on mouse skin. Sci. Irel. 2002, 182, 33–41. [Google Scholar]
- Barlow, S.M.; Sullivan, F.M.; Lines, J. Risk assessment of the use of deltamethrin on bednets for the prevention of malaria. Food Chem. Toxicol. 2001, 39, 407–422. [Google Scholar] [CrossRef]
- Azmi, M.A.; Naqvi, S.N.; Akhtar, K.; Moinuddin; Parveen, S.; Parveen, R.; Aslam, M. ffect of pesticide residues on health and blood parameters of farm workers from rural Gadap, Karachi, Pakistan. J. Environ. Biol. 2009, 30, 747–756. [Google Scholar]
- Kim, K.; Anand, S.S.; Kim, H.J.; White, C.A.; Fisher, J.W.; Tornero-Velez, R.; Bruckner, J.V. Age, dose, and time-dependency of plasma and tissue distribution of deltamethrin in immature rats. Toxicol. Sci. 2010, 115, 354–368. [Google Scholar] [CrossRef]
- Hines, R.N. Developmental expression of drug metabolizing enzymes: Impact on disposition in neonates and young children. Int. J. Pharm. 2013, 452, 3–7. [Google Scholar] [CrossRef]
- Ren, Z.M.; Wang, Z.J. Differences in the behavior characteristics between Daphnia magna and Japanese madaka in an on-line biomonitoring system. (in Chinese). J. Environ. Sci. China 2010, 22, 703–708. [Google Scholar] [CrossRef]
- Svecevicius, G. Use of behavioral responses of rainbow trout Oncorhynchus mykiss in identifying sublethal exposure to hexavalent chromium. Bull. Environ. Contam. Toxicol. 2009, 82, 564–568. [Google Scholar] [CrossRef]
- Viran, R.; Erkoc, F.U.; Polat, H.; Kocak, O. Investigation of acute toxicity of deltamethrin on guppies (Poecilia reticulata). Ecotoxicol. Environ. Saf. 2003, 55, 82–85. [Google Scholar] [CrossRef]
- Floyd, E.Y.; Geist, J.P.; Werner, I. Acute, sublethal exposure to a pyrethroid insecticide alters behavior, growth, and predation risk in larvae of the fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 2008, 27, 1780–1787. [Google Scholar] [CrossRef]
- Michalec, F.; Holzner, M.; Menu, D.; Hwang, J.; Souissi, S. Behavioral responses of the estuarine calanoid copepod Eurytemora affinis to sub-lethal concentrations of waterborne pollutants. Aquat. Toxicol. 2013, 138–139, 129–138. [Google Scholar]
- OECD Guidelines for Testing of Chemicals; OECD: Paris, France, 1993.
- Polat, H.; Erkoc, F.U.; Viran, R.; Kocak, O. Investigation of acute toxicity of beta-cypermethrin on guppies Poecilia reticulata. Chemosphere 2002, 49, 39–44. [Google Scholar] [CrossRef]
- Barry, M.J. Application of a novel open-source program for measuring the effects of toxicants on the swimming behavior of large groups of unmarked fish. Chemosphere 2012, 86, 938–944. [Google Scholar] [CrossRef]
- Beauvais, S.L.; Jones, S.B.; Parris, J.T.; Brewer, S.K.; Little, E.E. Cholinergic and behavioral neurotoxicity of carbaryl and cadmium to larval rainbow trout (Oncorhynchus mykiss). Ecotoxicol. Environ. Saf. 2001, 49, 84–90. [Google Scholar] [CrossRef]
- El-Sayed, Y.S.; Saad, T.T.; El-Bahr, S.M. Acute intoxication of deltamethrin in monosex Nile tilapia, Oreochromis niloticus with special reference to the clinical, biochemical and haematological effects. Environ. Toxicol. Pharmacol. 2007, 24, 212–217. [Google Scholar] [CrossRef]
- Little, E.E.; Archeski, R.D.; Flerov, B.A.; Kozlovskaya, V.I. Behavioral indicators of sublethal toxicity in rainbow trout. Arch. Environ. Contam. Toxicol. 1990, 19, 380–385. [Google Scholar] [CrossRef]
- Robinson, P.D. Behavioural toxicity of organic chemical contaminants in fish: Application to ecological risk assessments (ERAs). Can. J. Fish. Aquat. Sci. 2009, 66, 1179–1188. [Google Scholar] [CrossRef]
- Vellinger, C.; Gismondi, E.; Felten, V.; Rousselle, P.; Mehennaoui, K.; Parant, M.; Usseglio-Polatera, P. Single and combined effects of cadmium and arsenate in Gammarus pulex (Crustacea, Amphipoda). Aquat. Toxicol. 2013, 140–141, 106–116. [Google Scholar]
- Guo, R.X.; Ren, X.K.; Ren, H.Q. Assessment the toxic effects of dimethoate to rotifer using swimming behavior. Environ. Contam. Toxicol. 2012, 89, 568–571. [Google Scholar] [CrossRef]
- Brewer, S.K.; Little, E.E.; Delonay, A.J.; Beauvais, S.L.; Jones, S.B.; Ellersieck, M.R. Behavioral dysfunctions correlate to altered physiology in rainbow trout (Oncorynchus mykiss) exposed to cholinesterase-inhibiting chemicals. Arch. Environ. Contam. Toxicol. 2001, 40, 70–76. [Google Scholar] [CrossRef]
- Sandahl, J.F.; Baldwin, D.H.; Jenkins, J.J.; Scholz, N.L. Comparative thresholds for acetylcholinesterase inhibition and behavioral impairment in coho salmon exposed to chlorpyrifos. Environ. Toxicol. Chem. 2005, 24, 136–145. [Google Scholar] [CrossRef]
- Gruber, S.J.; Munn, M.D. Organophosphate and carbamate insecticides in agricultural waters and cholinesterase (ChE) inhibition in common carp (Cyprinus carpio). Arch. Environ. Contam. Toxicol. 1998, 35, 391–396. [Google Scholar] [CrossRef]
- De la Torre, F.R.; Ferrari, L.; Salibian, A. Freshwater pollution biomarker: Response of brain acetylcholinesterase activity in two fish species. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2002, 131, 271–280. [Google Scholar] [CrossRef]
- Sayeed, I.; Parvez, S.; Pandey, S.; Bin-Hafeez, B.; Haque, R.; Raisuddin, S. Oxidative stress biomarkers of exposure to deltamethrin in freshwater fish, Channa punctatus Bloch. Ecotoxicol. Environ. Saf. 2003, 56, 295–301. [Google Scholar] [CrossRef]
- Kang, I.J.; Moroishi, J.; Yamasuga, M.; Kim, S.; Oshima, Y. Swimming behavioral toxicity in Japanese medaka (Oryzias latipes) exposed to various chemicals for biological monitoring of water quality. Atmos. Biol. Environ. Monit. 2009, 285–293. [Google Scholar]
- Scott, G.R.; Sloman, K.A. The effects of environmental pollutants on complex fish behaviour: Integrating behavioural and physiological indicators of toxicity. Aquat. Toxicol. 2004, 68, 369–392. [Google Scholar] [CrossRef]
- Gerhardt, A. Aquatic behavioral ecotoxicology—Prospects and limitations. Hum. Ecol. Risk Assess. 2007, 13, 481–491. [Google Scholar] [CrossRef]
- Van der Schalie, W.H.; Shedd, T.R.; Knechtges, P.L.; Widder, M.W. Using higher organisms in biological early warning systems for real-time toxicity detection. Biosens. Bioelectron. 2001, 16, 457–465. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, Y.; Zhang, J.; Han, X.; Huang, T. The Use of Zebrafish (Danio rerio) Behavioral Responses in Identifying Sublethal Exposures to Deltamethrin. Int. J. Environ. Res. Public Health 2014, 11, 3650-3660. https://doi.org/10.3390/ijerph110403650
Huang Y, Zhang J, Han X, Huang T. The Use of Zebrafish (Danio rerio) Behavioral Responses in Identifying Sublethal Exposures to Deltamethrin. International Journal of Environmental Research and Public Health. 2014; 11(4):3650-3660. https://doi.org/10.3390/ijerph110403650
Chicago/Turabian StyleHuang, Yi, Jinsong Zhang, Xiaobo Han, and Tinglin Huang. 2014. "The Use of Zebrafish (Danio rerio) Behavioral Responses in Identifying Sublethal Exposures to Deltamethrin" International Journal of Environmental Research and Public Health 11, no. 4: 3650-3660. https://doi.org/10.3390/ijerph110403650




