Gold Standard Program for Heavy Smokers in a Real-Life Setting
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Outcomes
2.3. Participants
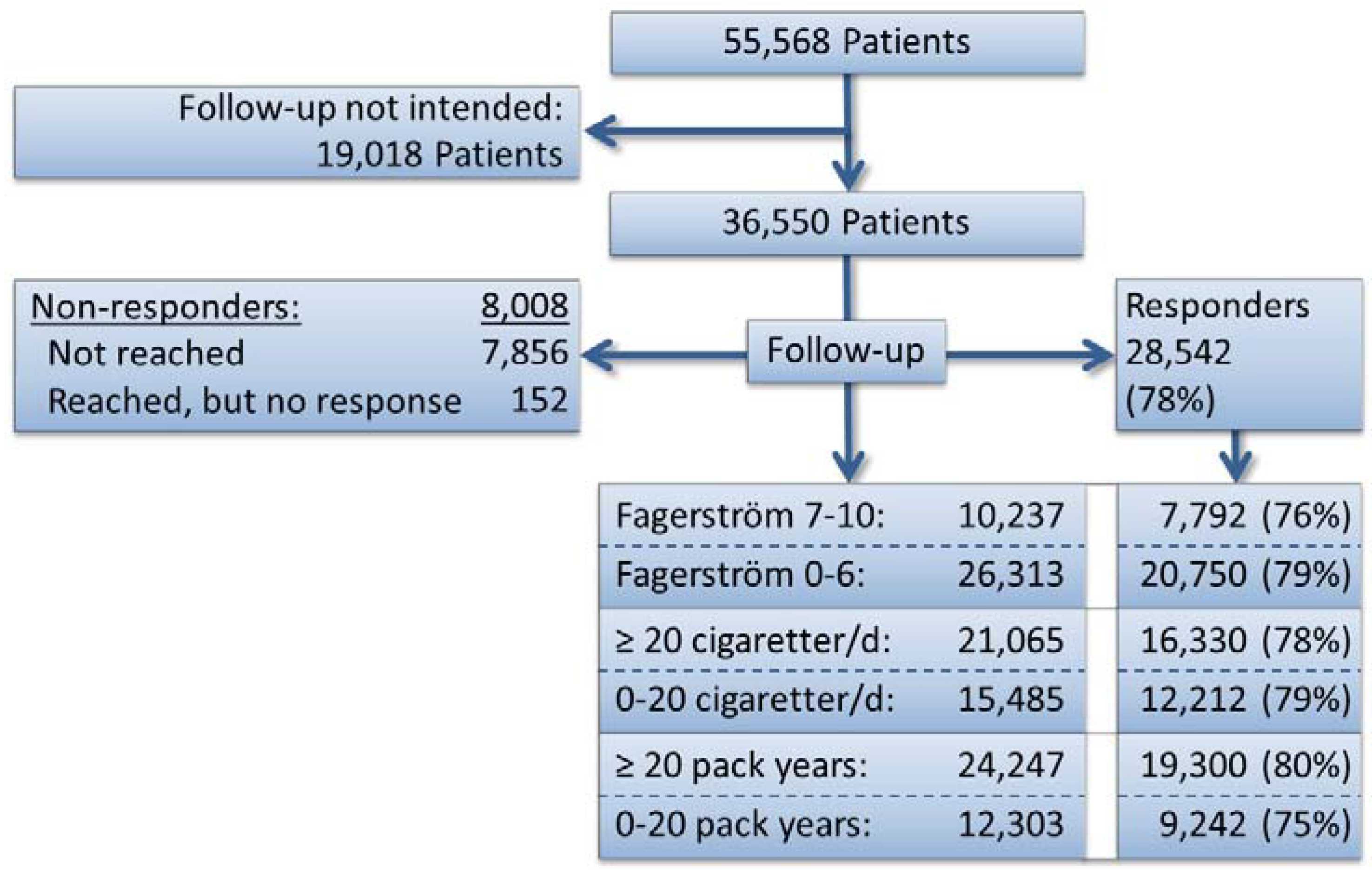
2.4. Setting
2.5. Intervention
2.6. Data
| All | Fagerström score ≥ 7 | Cigarettes/day ≥ 20 | Pack years ≥ 20 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n = | base | (worst-best) | n = | base | (worst-best) | n = | base | (worst-best) | n = | base | (worst-best) | ||
| Total | 36,550 | 33% | (26%–48%) | 10,237 | 27% | (21%–45%) | 21,065 | 30% | (23%–46%) | 24,247 | 32% | (25%–46%) | |
| Capital Region | 12,100 | 32% | (25%–48%) | 3,504 | 26% | (20%–45%) | 7,293 | 30% | (23%–46%) | 8,010 | 31% | (24%–46%) | |
| Central Denmark | 8,216 | 33% | (25%–48%) | 2,183 | 27% | (20%–45%) | 4,419 | 29% | (22%–45%) | 5,200 | 32% | (25%–46%) | |
| North Denmark | 1,477 | 36% | (29%–50%) | 368 | 30% | (24%–46%) | 780 | 33% | (26%–48%) | 982 | 36% | (29%–48%) | |
| Region Zealand | 5,144 | 34% | (28%–47%) | 1,512 | 29% | (23%–43%) | 3,149 | 32% | (26%–44%) | 3,687 | 33% | (28%–44%) | |
| South Denmark | 9,613 | 34% | (27%–48%) | 2,670 | 27% | (21%–45%) | 5,424 | 31% | (24%–47%) | 6,368 | 32% | (26%–46%) | |
| Unknown | 0 | ||||||||||||
| Pharmacy | 9,664 | 33% | (26%–48%) | 2,749 | 27% | (20%–44%) | 5,592 | 29% | (23%–45%) | 6,789 | 31% | (25%–45%) | |
| Hospital | 7,813 | 36% | (28%–50%) | 2,475 | 30% | (22%–47%) | 4,610 | 33% | (25%–49%) | 5,040 | 35% | (27%–49%) | |
| All other settings | 19,073 | 32% | (25%–47%) | 5,013 | 27% | (20%–44%) | 10,863 | 30% | (23%–45%) | 12,418 | 31% | (25%–45%) | |
| Individual format | 4,526 | 37% | (27%–53%) | 1,439 | 34% | (24%–53%) | 2,612 | 34% | (25%–52%) | 2,931 | 35% | (27%–51%) | |
| Group format | 31,918 | 33% | (26%–47%) | 8,773 | 26% | (20%–43%) | 18,401 | 30% | (23%–45%) | 21,260 | 32% | (25%–45%) | |
| All other formats | 106 | 25 | 52 | 56 | |||||||||
| No free medication/other | 15,112 | 33% | (26%–47%) | 3,894 | 27% | (21%–43%) | 8,400 | 30% | (24%–44%) | 10,008 | 32% | (26%–44%) | |
| Free medication for days | 16,478 | 32% | (25%–48%) | 4,766 | 26% | (20%–45%) | 9,722 | 29% | (22%–46%) | 10,904 | 31% | (25%–46%) | |
| Free for <5 weeks | 4,026 | 36% | (27%–50%) | 1,293 | 31% | (24%–48%) | 2,416 | 34% | (26%–50%) | 2,775 | 35% | (28%–49%) | |
| Free for the total course | 934 | 40% | (32%–53%) | 284 | 35% | (27%–50%) | 527 | 37% | (29%–51%) | 560 | 36% | (30%–48%) | |
| Women | 22,538 | 32% | (25%–46%) | 5,620 | 25% | (19%–42%) | 11,423 | 27% | (21%–43%) | 13,851 | 29% | (24%–43%) | |
| Men | 14,012 | 36% | (28%–50%) | 4,617 | 30% | (23%–48%) | 9,642 | 34% | (26%–49%) | 10,396 | 36% | (28%–49%) | |
| Unknown | 0 | ||||||||||||
| 18-24 years age | 1,407 | 26% | (17%–54%) | 280 | 19% | (11%–53%) | 626 | 22% | (14%–52%) | 7 | – | – | – |
| 25-34 years | 4,855 | 32% | (23%–50%) | 1,164 | 23% | (16%–46%) | 2,341 | 28% | (20%–49%) | 683 | 26% | (19%–46%) | |
| 35-44 years | 8,038 | 33% | (25%–48%) | 2,446 | 26% | (19%–45%) | 4,710 | 29% | (22%–46%) | 4,784 | 29% | (22%–46%) | |
| 45-54 years | 10,135 | 33% | (26%–47%) | 3,277 | 27% | (21%–44%) | 6,316 | 30% | (23%–45%) | 8,207 | 31% | (25%–46%) | |
| 55-64 years | 8,946 | 35% | (29%–47%) | 2,524 | 30% | (24%–44%) | 5,492 | 33% | (27%–45%) | 7,851 | 34% | (28%–46%) | |
| 65+ years | 3,169 | 34% | (28%–46%) | 546 | 32% | (26%–45%) | 1,580 | 33% | (27%–45%) | 2,715 | 33% | (27%–45%) | |
| Unknown | 0 | ||||||||||||
| No previous attempt | 13,168 | 32% | (25%–47%) | 4,323 | 27% | (20%–45%) | 8,351 | 30% | (23%–46%) | 9,371 | 32% | (25%–46%) | |
| Previous attempt | 22,741 | 34% | (27%–48%) | 5,703 | 27% | (21%–44%) | 12,332 | 30% | (24%–46%) | 14,395 | 32% | (26%–46%) | |
| Unknown | 641 | 211 | 382 | 481 | |||||||||
| Living with smoker | 13,291 | 32% | (25%–46%) | 3,912 | 27% | (21%–43%) | 8,059 | 30% | (23%–45%) | 8,762 | 31% | (25%–51%) | |
| Not living with smoker | 22,975 | 34% | (26%–49%) | 6,233 | 28% | (21%–45%) | 12,825 | 31% | (23%–47%) | 15,293 | 32% | (25%–47%) | |
| Unknown | 284 | 92 | 181 | 192 | |||||||||
| Compliant | 23,400 | 42% | (34%–52%) | 6,144 | 37% | (30%–49%) | 12,990 | 40% | (32%–51%) | 15,919 | 40% | (34%–50%) | |
| Not compliant | 12,677 | 15% | (11%–40%) | 3,968 | 10% | (7%–39%) | 7,805 | 12% | (9%–38%) | 8,032 | 13% | (9%–37%) | |
| Unknown | 473 | 125 | 270 | 296 | |||||||||
| Employed | 24,677 | 34% | (27%–49% | 6,694 | 29% | (22%–45%) | 14,040 | 31% | (24%–47%) | 15,477 | 33% | (26%–47%) | |
| Not employed | 10,941 | 31% | (24%–46%) | 3,285 | 24% | (18%–44%) | 7,025 | 28% | (22%–44%) | 8,770 | 30% | (24%–45%) | |
| Unknown | 698 | 195 | 395 | 465 | |||||||||
| 2006-2010 | 21,516 | 33% | (26%–46%) | 5,577 | 27% | (21%–43%) | 12,172 | 29% | (23%–44%) | 14,011 | 31% | (25%–44%) | |
| 2001-2005 | 15,034 | 34% | (26%–50%) | 4,660 | 28% | (21%–47%) | 8,893 | 31% | (24%–48%) | 10,236 | 33% | (26%–48%) | |
| Unknown | 0 | ||||||||||||
2.7. Statistical Methods
 ) and 95% CI (
) and 95% CI (  ) of continuous abstinence as the dependent variable for Fagerström points (FTND), cigarettes per day (CPD) and pack-year after adjusting (final model) for setting, region, format, payment modality, age, gender, employment, attempts to quit, living with a smoker, compliance and calendar period.
) of continuous abstinence as the dependent variable for Fagerström points (FTND), cigarettes per day (CPD) and pack-year after adjusting (final model) for setting, region, format, payment modality, age, gender, employment, attempts to quit, living with a smoker, compliance and calendar period.
 ) and 95% CI (
) and 95% CI (  ) of continuous abstinence as the dependent variable for Fagerström points (FTND), cigarettes per day (CPD) and pack-year after adjusting (final model) for setting, region, format, payment modality, age, gender, employment, attempts to quit, living with a smoker, compliance and calendar period.
) of continuous abstinence as the dependent variable for Fagerström points (FTND), cigarettes per day (CPD) and pack-year after adjusting (final model) for setting, region, format, payment modality, age, gender, employment, attempts to quit, living with a smoker, compliance and calendar period.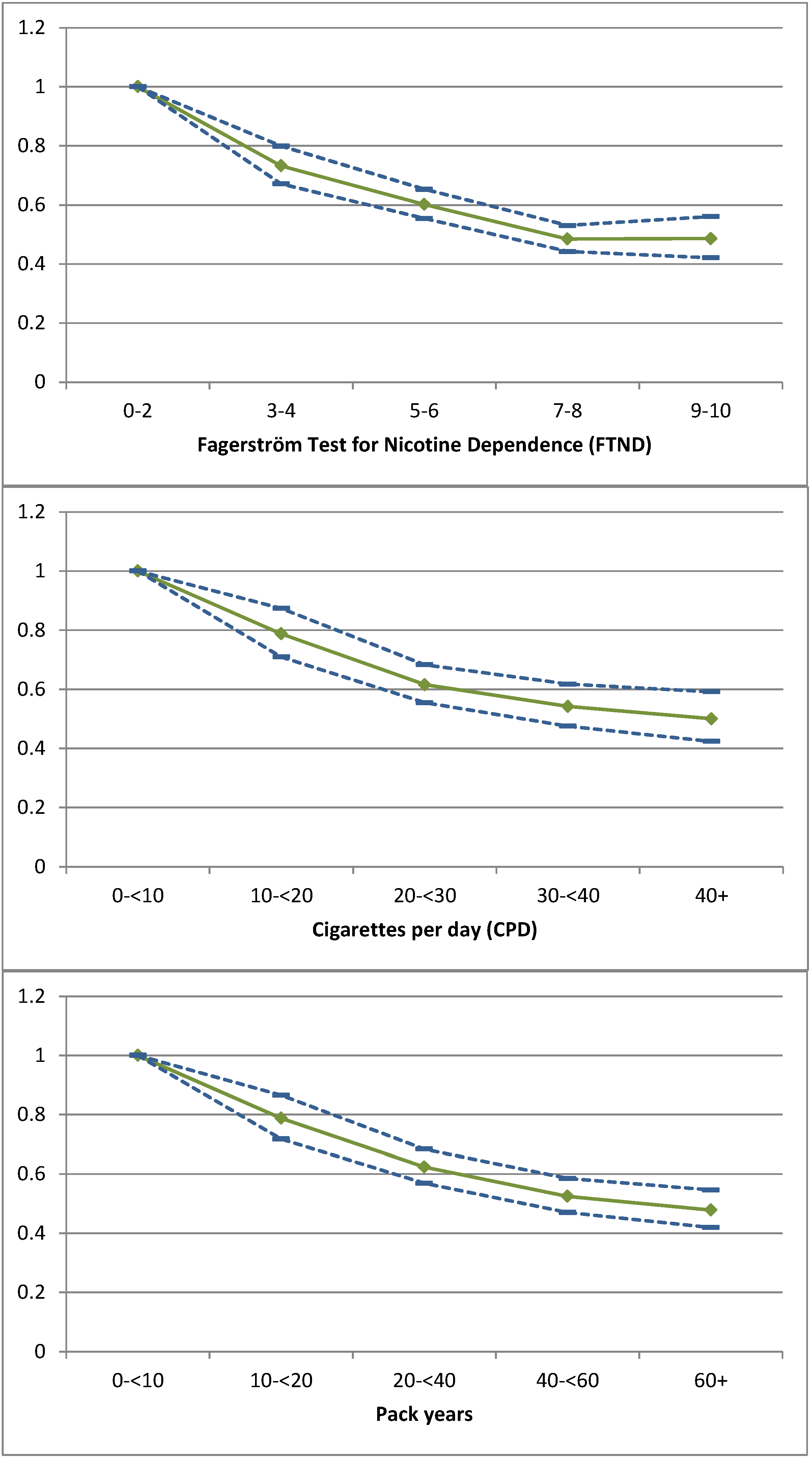
3. Results
| Fagerström score ≥ 7 | Cigarettes/day ≥ 20 | Pack years ≥ 20 | |
|---|---|---|---|
| ORs (95% CI) | Exp(B) (95% CI) | Exp(B) (95% CI) | |
| MULTIVARIATE | |||
| Capital (vs. other regions) | 0.99 (0.87–1.13) | 1.00 (0.92–1.08) | 0.94 (0.87–1.01) |
| (Setting) | * | * | |
| Pharmacy (vs. other settings) | 0.89 (0.78–1.02) | 0.91 (0.83–0.99) * | 0.90 (0.83–0.98) * |
| Hospital (vs. other settings) | 1.05 (0.91–1.22) | 1.11 (1.01–1.23) * | 1.10 (1.00–1.20) * |
| Pharmacy (vs. hospitals) | 0.85 (0.72–1.00) | 0.82 (0.73–0.91) * | 0.82 (0.74–0.91) * |
| Individual (vs. other formats) | 1.44 (1.23–1.68) * | 1.29 (1.15–1.44) * | 1.22 (1.10–1.35) * |
| Payment for weeks (vs. non/shorter payments) | 1.06 (0.78–1.43) | 1.08 (0.87–1.34) | 0.98 (0.80–1.20) |
| Men (vs. women) | 1.25 (1.12–1.39) * | 1.31 (1.22–1.41) * | 1.29 (1.21–1.38) * |
| Age (every 10 years) | 1.07 (1.02–1.13) * | 1.05 (1.02–1.09) * | 1.10 (1.06–1.14) * |
| Compliant (vs. other) | 4.90 (4.28–5.61) * | 4.38 (4.01–4.78) * | 4.35 (4.01–4.73) * |
| Living with a smoker (vs. not) | 0.94 (0.84–1.05) | 0.95 (0.89–1.03) | 0.95 (0.89–1.01) |
| No earlier attempts (vs. earlier attempts) | 1.03 (0.93–1.15) | 0.99 (0.92–1.07) | 1.03 (0.97–1.10) |
| Employed (vs. other) | 1.28 (1.13–1.45) * | 1.22 (1.12–1.33) * | 1.25 (1.16–1.36) * |
| 2001–2005 (vs. 2006–2010) | 0.85 (0.76–0.96) * | 0.87 (0.80–0.94) * | 0.88 (0.82–0.95) * |
| Constant | 0.08 * | 0.10 * | 0.09 * |
| FINAL MODEL | |||
| (Setting ) | * | * | |
| Pharmacy (vs. other settings) | 0.90 (0.78–1.02) | 0.91 (0.83–0.99) * | 0.92 (0.85–0.99) * |
| Hospital (vs. other settings) | 1.05 (0.91–1.21) | 1.11 (1.01–1.23) * | 1.07 (0.98–1.17) |
| Pharmacy (vs. hospitals) | 0.85 (0.73–0.99) | 0.81 (0.73–0.90) * | 0.86 (0.78–0.94) * |
| Individual (vs. other formats) | 1.44 (1.23–1.68) * | 1.30 (1.16–1.45) * | 1.23 (1.11–1.36) * |
| Men (vs. Women) | 1.25 (1.12–1.39) * | 1.31 (1.22–1.41) * | 1.29 (1.21–1.38) * |
| Age (every 10 years) | 1.07 (1.02–1.13) * | 1.05 (1.02–1.09) * | 1.10 (1.06–1.14) * |
| Compliant (vs. other) | 4.89 (4.27–5.60) * | 4.38 (4.01–4.78) * | 4.36 (4.01–4.73) * |
| Employed (vs. Other) | 1.28 (1.13–1.44) * | 1.22 (1.12–1.33) * | 1.25 (1.16–1.35) * |
| 2001–2005 (vs. 2006–2010) | 0.85 (0.76–0.95) * | 0.87 (0.80–0.94) * | 0.89 (0.83–0.95) * |
| Constant | 0.07 * | 0.10 * | 0.09 * |
3.1. Quit Rate and Severity of Smoking
3.2. Point Prevalence
3.3. Sensitivity Analyses
4. Discussion
5. Conclusions
Acknowledgements
Conflicts of Interest
References
- World Health Organization, WHO Report on the Global Tobacco Epidemic, 2011 Warning about the Dangers of Tobacco; World Health Organization: Geneva, Switzerland, 2011.
- Tobacco Use and Dependence Guideline Panel, Treating Tobacco Use and Dependence: 2008 Update; US Department of Health and Human Services: Washington, DC, USA, 2008.
- Kamholz, S.L. Pulmonary and cardiovascular consequences of smoking. Med. Clin. N. Am. 2004, 88, 1415–1430. [Google Scholar] [CrossRef]
- Heatherton, T.F.; Kozlowski, L.T.; Frecker, R.C.; Fagerström, K.O. The fagerström test for nicotine dependence: A revision of the fagerström tolerance questionnaire. Br. J. Addict. 1991, 86, 1119–1127. [Google Scholar] [CrossRef]
- Bierut, L.J. Nicotine dependence and genetic variation in the nicotinic receptors. Drug Alcohol Depend. 2009, 104, S64–S69. [Google Scholar] [CrossRef]
- Olsen, K.R.; Bilde, L.; Juhl, H.H.; Kjaer, N.T.; Mosbech, H.; Evald, T.; Rasmussen, M.; Hiladakis, H. Cost-effectiveness of the Danish smoking cessation interventions: Subgroup analysis based on the Danish Smoking Cessation Database. Eur. J. Health Econ. 2006, 7, 255–264. [Google Scholar] [CrossRef]
- Kjaer, N.T.; Evald, T.; Rasmussen, M.; Juhl, H.H.; Mosbech, H.; Olsen, K.R. The effectiveness of nationally implemented smoking interventions in Denmark. Prev. Med. 2007, 45, 12–14. [Google Scholar] [CrossRef]
- Daughton, D.; Susman, J.; Sitorius, M.; Belenky, S.; Millatmal, T.; Nowak, R.; Patil, K.; Rennard, S.I. Transdermal nicotine therapy and primary care. Importance of counseling, demographic, and participant selection factors on 1-year quit rates. The Nebraska Primary Practice Smoking Cessation Trial Group. Arch. Fam. Med. 1998, 7, 425–430. [Google Scholar] [CrossRef]
- Herrera, N.; Franco, R.; Herrera, L.; Partidas, A.; Rolando, R.; Fagerström, K.O. Nicotine gum, 2 and 4 mg, for nicotine dependence. A double-blind placebo-controlled trial within a behavior modification support program. Chest 1995, 108, 447–451. [Google Scholar] [CrossRef]
- Lind, M.; Jaspers, K. Manual for Smoking Cessation in Groups, 2nd ed.; Danish Cancer Association: Copenhagen, Denmark, 2008. [Google Scholar]
- Peng, R.; Wang, S. Nicotine sublingual tablet for smoking cessation in 115 cases: A double-blind randomized placebo-controlled clinical trial. J. Clin. Rehabil. Tissue Eng. Res. 2007, 11, 10443–10446. [Google Scholar]
- Pisinger, C.; Vestbo, J.; Borch-Johnsen, K.; Jørgensen, T. It is possible to help smokers in early motivational stages to quit. The Inter99 study. Prev. Med. 2005, 40, 278–284. [Google Scholar]
- Baker, T.B.; Piper, M.E.; McCarthy, D.E.; Bolt, D.M.; Smith, S.S.; Kim, S.-Y.; Colby, S.; Conti, D.; Giovino, G.A.; Hatsukami, D.; et al. Time to first cigarette in the morning as an index of ability to quit smoking: Implications for nicotine dependence. Nicot. Tob. Res. 2007, 9, S555–S570. [Google Scholar] [CrossRef]
- Kozlowski, L.T.; Porter, C.Q.; Orleans, C.T.; Pope, M.A.; Heatherton, T. Predicting smoking cessation with self-reported measures of nicotine dependence: FTQ, FTND, and HSI. Drug Alcohol Depend. 1994, 34, 211–216. [Google Scholar] [CrossRef]
- Ferguson, J.; Bauld, L.; Chesterman, J.; Judge, K. The English smoking treatment services: One-year outcomes. Addiction 2005, 100, 59–69. [Google Scholar] [CrossRef]
- Stead, L.F.; Perera, R.; Bullen, C.; Mant, D.; Lancaster, T. Nicotine replacement therapy for smoking cessation. Cochrane Database Syst. Rev. 2008, 23. [Google Scholar] [CrossRef]
- Lai, D.T.; Cahill, K.; Qin, Y.; Tang, J.-L. Motivational interviewing for smoking cessation. Cochrane Database Syst. Rev. 2010, 20. [Google Scholar] [CrossRef]
- Lancaster, T.; Stead, L.F. Individual behavioural counselling for smoking cessation. Cochrane Database Syst. Rev. 2005, 18. [Google Scholar] [CrossRef]
- Stead, L.F.; Lancaster, T. Group behaviour therapy programmes for smoking cessation. Cochrane Database Syst. Rev. 2005. [Google Scholar] [CrossRef]
- Thomsen, T.; Villebro, N.; Møller, A.M. Interventions for preoperative smoking cessation. Cochrane Database Syst. Rev. 2010, 20. [Google Scholar] [CrossRef]
- Lancaster, T.; Stead, L.F. Physician advice for smoking cessation. Cochrane Database Syst. Rev. 2004, 18. [Google Scholar] [CrossRef]
- Stead, L.; Lancaster, T. Behavioural interventions as adjuncts to pharmacotherapy for smoking cessation. Cochrane Database Syst. Rev. 2012, 12. [Google Scholar] [CrossRef]
- WHO Collaborating Centre—Smoking Cessation Database. Available online: http://www.scdb.dk/ (accessed on 17 October 2011).
- Møller, A.M.; Villebro, N.; Pedersen, T.; Tønnesen, H. Effect of preoperative smoking intervention on postoperative complications: A randomised clinical trial. Lancet 2002, 359, 114–117. [Google Scholar] [CrossRef]
- Tønnesen, H. Surgery and smoking at first and second hand. Anesthesiology 2011, 115, 1–3. [Google Scholar] [CrossRef]
- Rasmussen, M.; Ammari, A.B.H.; Pedersen, B.; Tønnesen, H. Smoking cessation intervention activities and outcome before, during and after the national Healthcare Reform in Denmark. Clin. Health Promot. 2012, 2, 26–35. [Google Scholar]
- Neumann, T.; Rasmussen, M.; Ghith, N.; Heitmann, B.L.; Tønnesen, H. The Gold Standard Programme: Smoking cessation interventions for disadvantaged smokers are effective in a real-life setting. Tob. Control 2012. [Google Scholar] [CrossRef] [Green Version]
- West, R.; Hajek, P.; Stead, L.; Stapleton, J. Outcome criteria in smoking cessation trials: Proposal for a common standard. Addiction 2005, 100, 299–303. [Google Scholar] [CrossRef]
- Bauld, L.; Bell, K.; McCullough, L.; Richardson, L.; Greaves, L. The effectiveness of NHS smoking cessation services: A systematic review. J. Public Health 2010, 32, 71–82. [Google Scholar]
- Simon, J.A.; Carmody, T.P.; Hudes, E.S.; Snyder, E.; Murray, J. Intensive smoking cessation counseling vs. minimal counseling among hospitalized smokers treated with transdermal nicotine replacement: A randomized trial. Am. J. Med. 2003, 114, 555–562. [Google Scholar] [CrossRef]
- Hiscock, R.; Judge, K.; Bauld, L. Social inequalities in quitting smoking: What factors mediate the relationship between socioeconomic position and smoking cessation? J. Public Health 2011, 33, 39–47. [Google Scholar] [CrossRef]
- Lemmens, V.; Oenema, A.; Knut, I.K.; Brug, J. Effectiveness of smoking cessation interventions among adults: A systematic review of reviews. Eur. J. Cancer Prev. 2008, 17, 535–544. [Google Scholar] [CrossRef]
- Mottillo, S.; Filion, K.B.; Bélisle, P.; Joseph, L.; Gervais, A.; O’Loughlin, J.; Paradis, G.; Pihl, R.; Pilote, L.; Rinfret, S.; et al. Behavioural interventions for smoking cessation: A meta-analysis of randomized controlled trials. Eur. Heart J. 2009, 30, 718–730. [Google Scholar]
- Bauld, L.; Coleman, T.; Adams, C.; Pound, E.; Ferguson, J. Delivering the English smoking treatment services. Addiction 2005, 100, 19–27. [Google Scholar]
- Christen, A.G.; Christen, J.A. The female smoker: From addiction to recovery. Am. J. Med. Sci. 2003, 326, 231–234. [Google Scholar] [CrossRef]
- Judge, K.; Bauld, L.; Chesterman, J.; Ferguson, J. The English smoking treatment services: Short-term outcomes. Addiction 2005, 100, 46–58. [Google Scholar]
- Schnoll, R.A.; Patterson, F. Sex heterogeneity in pharmacogenetic smoking cessation clinical trials. Drug Alcohol Depend. 2009, 104, S94–S99. [Google Scholar] [CrossRef]
- Lumley, J.; Chamberlain, C.; Dowswell, T.; Oliver, S.; Oakley, L.; Watson, L. Interventions for promoting smoking cessation during pregnancy. Cochrane Database Syst. Rev. 2009, 8. [Google Scholar] [CrossRef]
- Pisinger, C.; Vestbo, J.; Borch-Johnsen, K.; Thomsen, T.; Jørgensen, T. Acceptance of the smoking cessation intervention in a large population-based study: The Inter99 study. Scand. J. Public Health 2005, 33, 138–145. [Google Scholar] [CrossRef]
- Batra, A. Treatment of tobacco dependence. Deutsches Ärzteblatt Int. 2011, 108, 555–564. [Google Scholar]
- Fidler, J.A.; Shahab, L.; West, R. Strength of urges to smoke as a measure of severity of cigarette dependence: Comparison with the Fagerström Test for Nicotine Dependence and its components. Addiction 2011, 106, 631–638. [Google Scholar] [CrossRef]
- Li, M.D.; Cheng, R.; Ma, J.Z.; Swan, G.E. A meta-analysis of estimated genetic and environmental effects on smoking behavior in male and female adult twins. Addiction 2003, 98, 23–31. [Google Scholar] [CrossRef]
- Tate, J.C.; Pomerleau, C.S.; Pomerleau, O.F. Pharmacological and non-pharmacological smoking motives: A replication and extension. Addiction (Abingdon, England) 1994, 89, 321–330. [Google Scholar] [CrossRef]
- Uhl, G.R.; Drgon, T.; Li, C.-Y.; Johnson, C.; Liu, Q.-R. Smoking and smoking cessation in disadvantaged women: Assessing genetic contributions. Drug Alcohol Depend. 2009, 104, S58–S63. [Google Scholar] [CrossRef]
- Le Strat, Y.; Rehm, J.; Le Foll, B. How generalisable to community samples are clinical trial results for treatment of nicotine dependence: A comparison of common eligibility criteria with respondents of a large representative general population survey. Tob. Control 2011, 20, 338–343. [Google Scholar] [CrossRef] [Green Version]
- Baha, M.Y.; Le Faou, A.-L. Smoking cessation interventions offered to French adult light smokers: A heterogeneous population with specific needs. Eur. Add. Res. 2010, 16, 162–169. [Google Scholar] [CrossRef]
- Schuurmans, M.M.; Diacon, A.H.; van Biljon, X.; Bolliger, C.T. Effect of pre-treatment with nicotine patch on withdrawal symptoms and abstinence rates in smokers subsequently quitting with the nicotine patch: A randomized controlled trial. Addiction 2004, 99, 634–640. [Google Scholar] [CrossRef]
- Carlson, L.E.; Taenzer, P.; Koopmans, J.; Bultz, B.D. Eight-year follow-up of a community-based large group behavioral smoking cessation intervention. Add. Behav. 2000, 25, 725–741. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Neumann, T.; Rasmussen, M.; Heitmann, B.L.; Tønnesen, H. Gold Standard Program for Heavy Smokers in a Real-Life Setting. Int. J. Environ. Res. Public Health 2013, 10, 4186-4199. https://doi.org/10.3390/ijerph10094186
Neumann T, Rasmussen M, Heitmann BL, Tønnesen H. Gold Standard Program for Heavy Smokers in a Real-Life Setting. International Journal of Environmental Research and Public Health. 2013; 10(9):4186-4199. https://doi.org/10.3390/ijerph10094186
Chicago/Turabian StyleNeumann, Tim, Mette Rasmussen, Berit L. Heitmann, and Hanne Tønnesen. 2013. "Gold Standard Program for Heavy Smokers in a Real-Life Setting" International Journal of Environmental Research and Public Health 10, no. 9: 4186-4199. https://doi.org/10.3390/ijerph10094186






