1. Introduction
The general trend of urban sprawl generates increasing flows of pollutants discharged into the aquatic environment via urban sewage systems, which include individual wastewaters and rainwater management systems [
1,
2,
3,
4]. The sources of the pollutants discharged by the latter are many: wastewater pollutants, atmospheric pollutants, leached pollutants and particles washed away after accumulation on urban surfaces during dry periods, the erosion of urban materials, the return to suspension of sediments accumulated in sewer networks [
5,
6,
7]. This mixture of urban waters discharged into aquatic environments during wet periods, called Combined Sewer Overflow (CSOs), has been the subject of a large number of studies in recent years [
8,
9,
10,
11]. The input of pollutants linked to these effluents in the natural environment has numerous consequences, such as increased turbidity due to suspended matter, deoxygenation of the environment due to the input of degradable organic matter and nitrous materials, and toxic effects on aquatic organisms via the input of chemical substances [
9,
12,
13,
14,
15].
Up to now, no structured methodology for the predictive assessment of ecotoxicological risks related to these specific discharges has been proposed. The deployment of such a methodology is nonetheless vital when designing new rainwater drainage networks, in order to set up adapted management systems if a risk exists [
16]. After recalling the fundamentals of ecological risk assessment at international level, this article focuses on the formulation of a risk assessment methodology specific to CSOs, and on its application to a discharge of CSOs located in the periphery of the city of Lyon in France. Finally, improvements to the methodology are proposed for optimising the assessment of risks incurred by host aquatic environments.
2. Methodological Approach for the Ecological Risk Assessment
The first Ecological Risk Assessment (ERA) methodologies emerged at the beginning of the 1990s with dawning awareness of the risks liable to impact ecosystems when they are exposed to substances of anthropic origin. In 1992, the United States EPA proposed a framework for the ecological risk assessment of contaminated industrial sites [
17]. Following a certain number of works, especially those of Suter [
18], this guide was improved to become “The Guidelines for Ecological Risk Assessment” [
19] which has now become the reference regarding ERA [
16,
20]. Since then, this guide has been revised by many countries and adapted for the management of their polluted sites [
21,
22,
23].
In addition, methodologies have been formulated to evaluate ecological risks linked to other problems. Mention can be made of the methodology drawn up by the European Union to evaluate risks relating to chemical substances placed on the market [
22,
24], and French works on the assessment of ecotoxicological risks linked to dumping continental dredged sediments [
25], on the assessment of the ecocompatibility of using wastes [
26,
27], and on the assessment of ecotoxicological risks linked to hospital effluents [
28,
29].
Most ERA methods formulated at international level are implemented with four main phases: (1) the formulation of the problem, (2) the characterisation of exposures, (3) the characterisation of effects, and lastly, and (4) the characterisation of the risk itself.
4. Results
The approach described above was applied to CSOs of the discharge site in the Lyon region mentioned previously.
4.1. Exposure Characterisation
4.1.1. Proportion of CSOs in the River
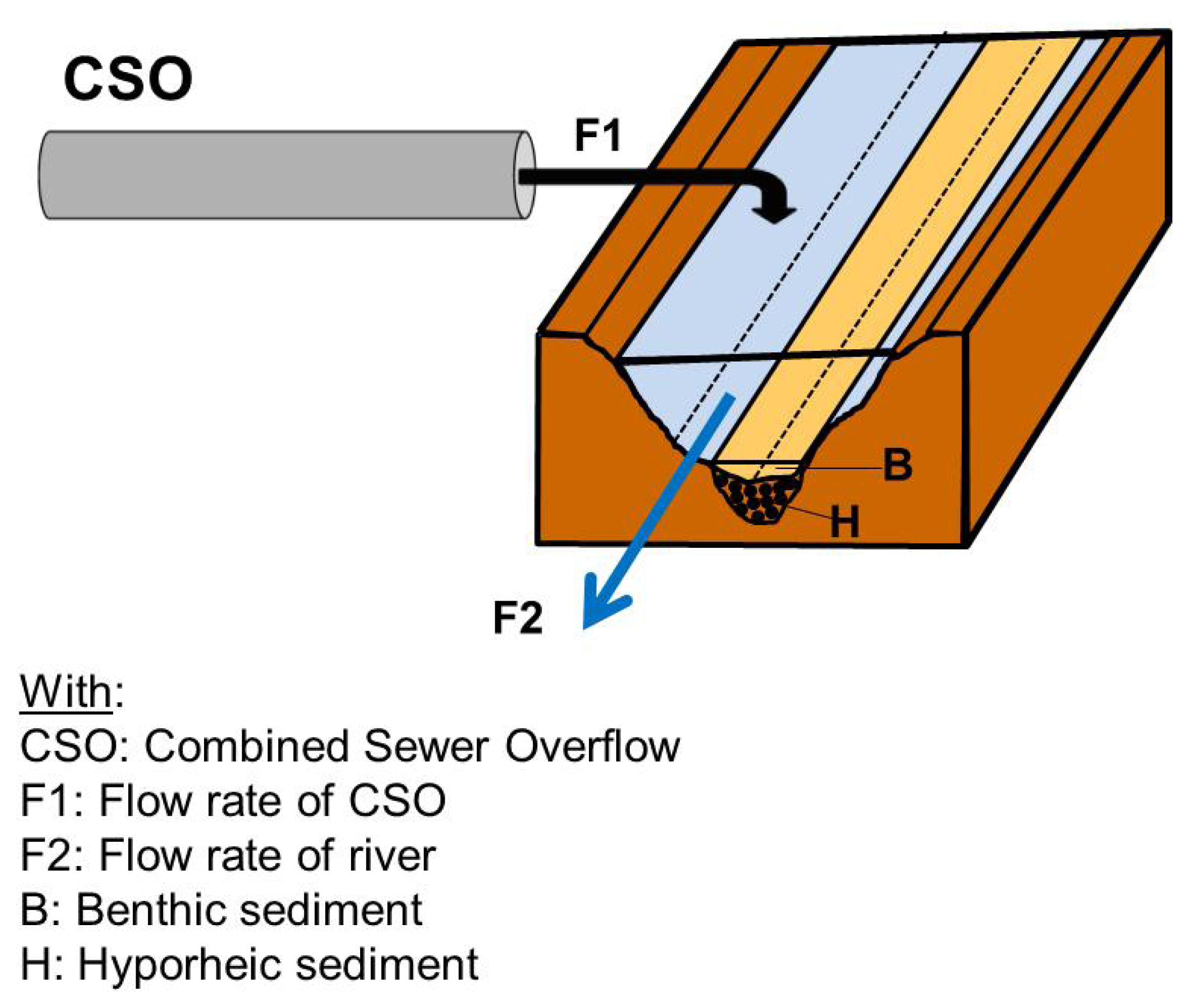
At the critical moment of the scenario, during the overflow of the first discharges of CSOs into the river, the flow of the effluent was evaluated at 1.0 L/s and the flow of the river was evaluated at 16 L/s for the studied site. These values lead the presence of 5.9% effluent in the river at the critical moment of the scenario.
4.1.2. Chemical Analyses
The results of physicochemical analyses of the CSOs filtered at 1.2 μm are given in
Table 3.
Table 3.
Physicochemical analyses of CSOs.
Table 3.
Physicochemical analyses of CSOs.
| Parameters | Units | Concentration * |
|---|
| pH | - | 8.8 |
| Conductivity | µS/cm | 967 |
| DCO | mg/L | 54 |
| NH4+ | mg/L | 17.3 |
| PO43− | mg/L | 8.9 |
| SO42− | mg/L | 27 |
| Cl− | mg/L | 95 |
| Cd | mg/L | <0.0001 |
| Cr | mg/L | 0.0018 |
| Cu | mg/L | 0.0037 |
| Ni | mg/L | 0.0011 |
| Pb | mg/L | 0.0004 |
| Zn | mg/L | 0.0100 |
| Total hydrocarbons | mg/l | <0.1 |
| Naphthalene | µg/L | <0.06 |
| Acenaphthylene | µg/L | <0.02 |
| Acenaphthene | µg/L | <0.02 |
| Fluorine | µg/L | <0.02 |
| Phenanthrene | µg/L | 0.03 |
| Anthracene | µg/L | <0.02 |
| Benzo(a)anthracene | µg/L | <0.02 |
| Dibenzo(ah)anthracene | µg/L | <0.02 |
| Fluoranthene | µg/L | <0.02 |
| Benzo(b)fluoranthene | µg/L | <0.02 |
| Benzo(k)fluoranthene | µg/L | <0.02 |
| Pyrene | µg/L | <0.02 |
| Benzo(a)pyrene | µg/L | <0.02 |
| Indeno(123-cd)pyrene | µg/L | <0.02 |
| Chrysene | µg/L | <0.02 |
| Benzo(ghi)perylene | µg/L | <0.02 |
The pH of the effluent was slightly alkaline (8.8) and its conductivity was high, demonstrating the presence of a strong ionic charge. Also of note in the effluent was a non-negligible concentration of NH4+ (17.3 mg/L) and non-negligible concentrations of heavy metals (particularly copper and zinc).
In contrast, the HAP concentrations are undetectable, except for the phenanthrene (0.03 µg/L) which nevertheless remains low. Globally, these results show pollutant concentrations which are in the range of what is generally observed in the CSOs.
The results of the physicochemical analyses practiced on the particle fraction of the CSOs are given in
Table 4.
Table 4.
Physicochemical analyses CSO particles.
Table 4.
Physicochemical analyses CSO particles.
| Parameters | Units | Concentration * |
|---|
| Heavy metals | | |
| Cd | mg/kg | 0.7 |
| Cr | mg/kg | 24 |
| Cu | mg/kg | 420 |
| Ni | mg/kg | 34 |
| Pb | mg/kg | 92 |
| Zn | mg/kg | 1,190 |
| Loss on ignition (550 °C) | g/kg | 321 |
| Total hydrocarbons | mg/kg | 2,600 |
| PAH | | |
| Naphthalene | µg/kg | <50 |
| Acenaphthylene | µg/kg | <50 |
| Acenaphthene | µg/kg | <50 |
| Fluorine | µg/kg | 58.8 |
| Phénanthrène | µg/kg | 382.3 |
| Anthracene | µg/kg | 117.6 |
| Benzo(a)anthracene | µg/kg | <50 |
| Dibenzo(ah)anthracene | µg/kg | <50 |
| Fluoranthene | µg/kg | 235.3 |
| Benzo(b)fluoranthene | µg/kg | <50 |
| Benzo(k)fluoranthene | µg/kg | <50 |
| Pyrene | µg/kg | 205.9 |
| Benzo(a)pyrene | µg/kg | <50 |
| Indeno(123-cd)pyrene | µg/kg | <50 |
| Chrysene | µg/kg | 88.2 |
| Benzo(ghi)perylene | µg/kg | <50 |
The analyses show an important rate of organic matter in the particles (loss on ignition close to 32%), a high concentration in hydrocarbons, and a high concentration in PAH (especially for phenanthrene, anthracene, fluoranthene and pyrene much higher concentrations of heavy metals than in the filtered water) were also observed.
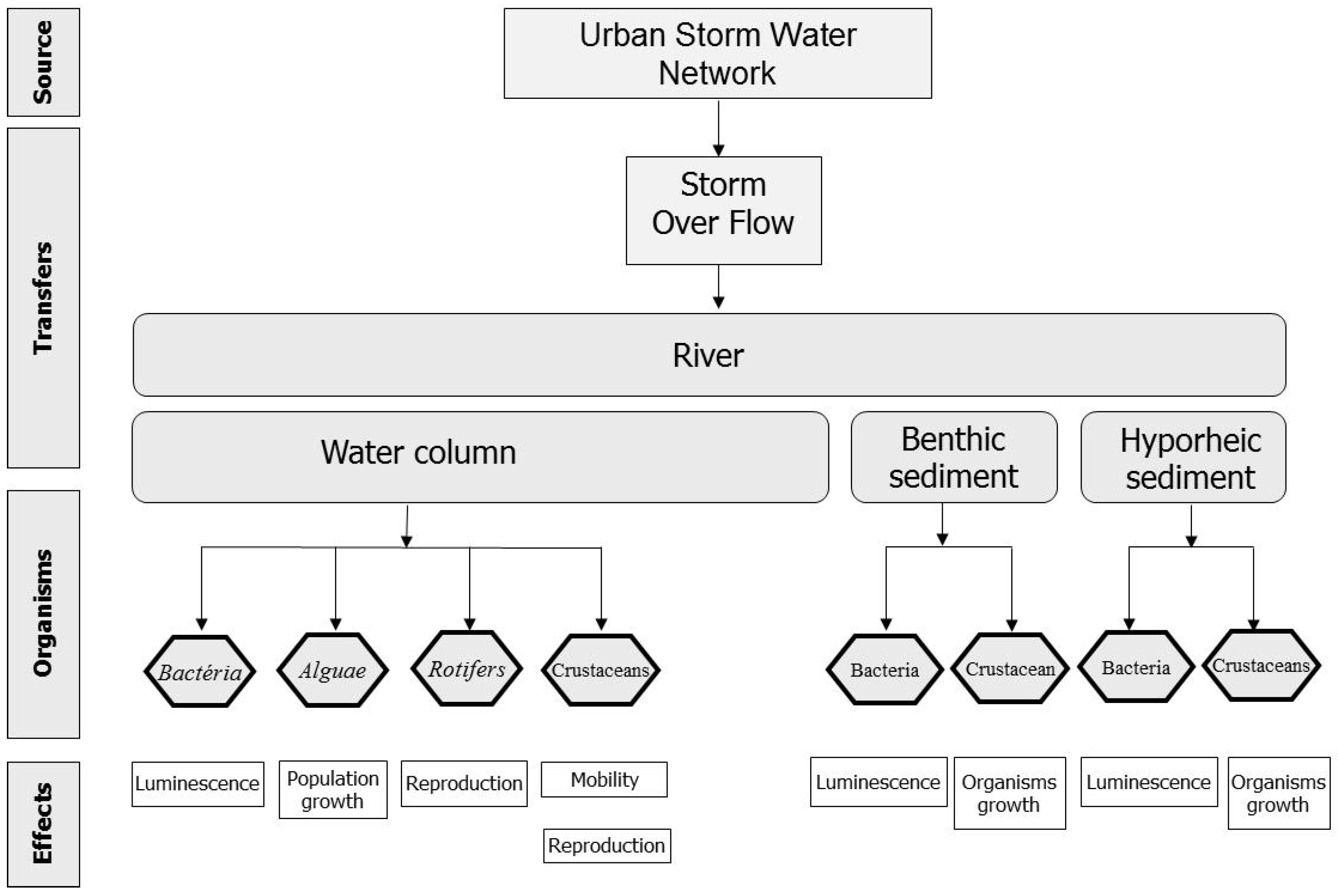
4.2. Effect Characterisation
4.2.1. Bioassays on the Organisms of the Water Column
The results of the bioassays performed with CSOs on the organisms of the water column are presented in
Table 5. The Efficient Concentration for 20% of the organisms (EC
20) is always higher than 80 % of CSOs, which indicates a very low ecotoxicity of theses, for all the tested organisms.
Table 5.
Results of bioassays on the organisms of the water column.
Table 5.
Results of bioassays on the organisms of the water column.
| Organisms | Measured parameters | Results (% of CSOs) |
|---|
| Vibrio Fischeri | EC 20 (Luminescence) | >80 |
| Daphnia magna | EC 20 (Mobility) | >80 |
| Pseudokirchneriella subcapitata (Ps) | EC 20 (Growth) | >80 |
| Daphnia magna | EC 20 Reproduction | >80 |
| Brachionus calyciflorus | EC 20 Reproduction | >80 |
| Ceriodaphnia dubia | EC 20 Reproduction | 19.6 |
| Brachydanio rerio | Test not performed due to the lack of fish in the studied little river |
4.2.2. Bioassays on CSO Particles
The results of the bioassays performed on CSO particles are given in
Table 6. The result of the first test (
Vibrio Fischeri ) indicates a strong ecotoxicity of the particles. Indeed, it is necessary to dilute fifty times the particles in order to enable the survival of the organisms.
The second test (Heterocypris incongruens) has been made only with undiluted particles. The ecotoxicity observed confirms the ecotoxicity of the solid phase of the CSOs.
Table 6.
Results of bioassays on sedimentary organisms.
Table 6.
Results of bioassays on sedimentary organisms.
| Organisms | Measured parameters | Results |
|---|
| Vibrio fischeri (on solid phase) | EC 20 (Luminescence) | 0.2% of particles |
| Heterocypris incongruens | Mortality of organisms with undiluted particles | 100% mortality with undiluted particles |
4.2.3. Risk Characterisation
The two objectives we chose in the current state of the approach are examined successively:
Risk can be characterised with the quotient method,
i.e., by comparing the PNEC (in % effluent) with the PEC, in this case equal to the proportion of effluent in the river downstream of the discharge (5.9%). To obtain the PNEC, an extrapolation factor is applied to it using experimental laboratory data whose value depends on the data considered [
20,
49]. In the case of our results, which focus on four chronic ecotoxicity assays and two acute ecotoxicity assays. The factor recommended by the Technical Guidance Document of the European Chemicals Bureau responsible for classifying chemical substances is a maximum of 10 with three chronic bioassays. So, we have chosen an extrapolation factor of 5. By applying this factor, we obtain a PNEC of 3.92% (19.6%/5). This leads to a risk quotient of 1.50 (5.9%/3.92%) which indicates a slight risk for the organisms of the water column.
Here again risk can also be characterised by using the quotient method. To obtain the PNEC, we apply a higher extrapolation factor than the previous one; since the experiments concerned are carried out with a battery of fewer bioassays than before [
20,
49]. Under these conditions; and by taking into account the recommendations of the European Chemicals Bureau responsible for classifying chemical substances [
24]; an extrapolation factor of 50 is proposed. By applying this factor to the results obtained for the particle fraction; we obtain a PNEC of 0.004% (0.2%/50).
Previous works [
34] have shown that particles of CSOs can accumulate heavily at certain points of the river downstream of the overspill, notably in hyporheic sediments at downwelling zones. If, by way of example, we assume that this accumulation leads to the presence of 0.01 to 10% CSO particles in the sediment, depending on the sector of the river, this leads to a risk quotient between 2.5 (0.01%/0.004%) and 2,500 (10%/0.004%). These quotients indicate that CSOs present a very high risk for the organisms living in the sediment zones rich in CSO particles.
In brief, with the methodology formulated, the effluent presents a slight risk for the organisms of the water column downstream of the overflow, and a very high one for the sedimentary organisms, especially at downwelling zones in the hyporheic zone.
5. Discussion
5.1. The Results Obtained
The ecological risk estimated for the organisms of benthic and hyporheic sediments is far greater than that calculated for the organisms of the water column. On the one hand, this can be explained by the high concentration in particles estimated at certain points of these sedimentary compartments and, on the other hand, by the very high concentration of pollutants in these particles whose value far exceeds PNEC values for several parameters (
Table 7) [
50].
These conclusions, which emphasise the risk for sedimentary organisms, particularly in the downwelling zones of the river, are consistent with the observations made by other authors for analogous rivers [
34,
51,
52,
53].
Different management recommendations can be made on the basis of these assessments. Regarding the organisms of the water column, the calculated risk first depends on the exposure which itself depends on the concentration of pollutants and the ratio of the CSO and river flow rates. Since it is difficult to control the flow rate of the river, especially at low flow, three improvements for managing the effluent could be considered: (i) the reduction of pollutant emissions at source, (ii), the provisional retention of CSOs in a retention basin and then progressive discharge into the river following the “critical” period, and (iii) setting up a CSO treatment system. In our opinion, the most sustainable solution would be the optimised combination of these three solutions, with a gradual progression to solution 1.
Table 7.
Comparison of concentrations of pollutants with PNECs.
Table 7.
Comparison of concentrations of pollutants with PNECs.
| Pollutants | Concentration in particles | Sediment PNEC (INERIS, 2012) | Concentration in the filtered CSOs | Aquatic PNEC (INERIS, 2012) |
|---|
| Cu | mg/kg | 420 | 0.8 | µg/L | 3.7 | 1.6 |
| Cr | mg/kg | 24 | - | µg/L | 1.8 | 4.1 |
| Pb | mg/kg | 92 | 6.8 | µg/L | 0.4 | 5 |
| Zn | mg/kg | 1,190 | 26 | µg/L | 10 | 8.6 |
| Phenanthrene | µg/kg | 382.3 | 2.3 | µg/L | 0.03 | 1.34 |
| Anthracene | µg/kg | 117.6 | 31.2 | µg/L | <0.02 | 0.063 |
| Fluoranthène | µg/kg | 235.3 | 2.3 | µg/L | <0.02 | 0.1 |
| Pyrene | µg/kg | 205.9 | 23 | µg/L | <0.02 | 0.012 |
Regarding the particle fraction of the CSOs, and the component most responsible for risks to the river, the main, and also inexpensive, improvement for management considered is to install a settling pond downstream of the network intended to collect the CSO particles before their discharge into the river.
6. Conclusions
This study showed that it is possible to assess risks for the different compartments of a river generated by the discharge of CSOs, using relatively accessible and standardised ecotoxicological investigation resources. Other ecological risk assessment approaches exist around the world. The purpose of our approach, other than taking into account the characteristics of the host environment, and specifying the impact of the effluent on its different compartments, is to define the compartments impacted and improve the realism of the assessment, in view to optimising management decisions. In this respect it is similar to the “Waste ecocompatibility” assessment approach established in France by the Environment and Energy Management Agency to assess the impact on ecosystems of storage scenarios and the reutilisation of mineral wastes for construction [
27].
For all that, the methodology presented can, and must, be improved still further in several directions so it can be used operationally by organisations responsible for managing rivers. It is advisable to check whether the battery of bioassays selected for the sedimentary organisms should be completed, and whether organisms representing fixed epibenthic organisms should be included. When considering these points, it should not be forgotten that a limited number of biological responses leads to increasing the uncertainty on the assessment of effects, and thus increasing the value of the final risk quotient through the necessary augmentation of applicable extrapolation factors.
Lastly, works aimed at improving the final phase of risk characterisation, which is currently based solely on the quotient method, and at ensuring the clarity of the results on the associated uncertainty are required to permit optimal use of the tool formulated [
16,
67,
68].
Acknowledgements
This work was funded by the French Ministry of Ecology and Sustainable Development. The authors would like to thank the technical teams of the laboratories LEHNA, LAQUE and POLDEN for their contribution to this work. They are also grateful to IRSTEA-Lyon which carried out the sampling of CSOs in the framework of the activities of OTHU (Observatoire de Terrain en Hydrologie Urbaine) (Rhône-Alpes Region, France) and the Grand Lyon for its financial support of OTHU activities.
Conflict of Interest
The authors declare no conflict of interest.
References
- Le Coustumer, S.-M. Colmatage et rétention des éléments traces métalliques dans les systèmes d’infiltration des eaux pluviales. Ph.D. Thesis, INSA de Lyon, Lyon, France, 2008. [Google Scholar]
- Winiarski, T.; Bedell, J.; Delolme, C.; Ghidini, M.; Perrodin, Y. Study of spatial distribution of physico-chemical and biological parameters along a soil vertical profile in stormwater infiltration basin in the Lyon area. Hydrogeol. J. 2006, 14, 1244–1251. [Google Scholar]
- Barraud, S.; Gibert, J.; Winiarski, T.; Bertrand-Krajewski, J.-L. Implementation of the monitoring system to measure impact of stormwater runoff infiltration. Water Sci. Technol. 2002, 45, 203–210. [Google Scholar]
- Bedell, J.-P.; Briant, A.; Delolme, C.; Perrodin, Y. Evaluation of the phytotoxicity of contaminated sediments deposited “on soil”: I. Impact of water draining from the deposit on the germination of neighbouring plants. Chemosphere 2002, 50, 393–402. [Google Scholar]
- Davies, D.G. Physiological Events in Biofilm Formation. In Community Structure and Cooperation in Biofilms; Allison, D., Gilbert, P., Lappin-Scott, H., Wilson, M., Eds.; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Mason, Y.; Amman, A.A.; Ulrich, A.; Sigg, L. Behavior of heavy metals, nutrients and major components during roof runoff infiltration. Environ. Sci. Technol. 1999, 33, 1588–1597. [Google Scholar] [CrossRef]
- Davis, A.P.; Shokouhian, M.; Ni, S. Loading estimates of lead, copper, cadmium and zinc in urban runoff from specific sources. Chemosphere 2001, 44, 997–1009. [Google Scholar] [CrossRef]
- Gromaire-Mertz, M.C. La pollution des eaux pluviales urbaines en réseau d'assainissement unitaire: Caractéristiques et origines. Ph.D. Thesis, Ecole Nationale des Ponts et Chaussée, Paris, France, 1998. [Google Scholar]
- Burton, G.; Pitt, R. Stormwater Effects Handbook, a Toolbox for Watershed Managers Scientists and Engineers; CRC Press Lewis: New York, NY, USA, 2002. [Google Scholar]
- Lafont, M.; Grapentine, L.; Rochfort, Q.; Marsalek, J.; Tixier, G.; Breil, P. Bioassessment of wetweather pollution impacts on fine sediments in urban waters by benthic indices and the sediment quality triad. Water Sci. Technol. 2007, 56, 13–20. [Google Scholar] [CrossRef]
- Parent-Raoult, C.; Boisson, J.C. The impact of urban wet weather flows on aquatic environments: The current state of knowledge. Revue des Sciences de l’Eau 2006, 20, 229–239. [Google Scholar]
- Novotny, V.; Witte, J.W. Ascertaining aquatic ecological risks of urban stormwater discharges. Water Res. 1997, 31, 2573–2585. [Google Scholar]
- Buffleben, M.S.; Zayeed, K.; Kimbrough, D.; Stenstrom, M.K.; Suffet, I.H. Evaluation of urban non-point source runoff of hazardous metals entering Santa Monica bay, California. Water Sci. Technol. 2002, 45, 263–268. [Google Scholar]
- Marsalek, J.; Rochfort, Q.; Mayer, T.; Servos, M.; Dutka, B.; Brownlee, B. Toxicity testing for controlling urban wet-weather pollution: Advantages and limitations. Urban Water 1999, 1, 91–103. [Google Scholar] [CrossRef]
- Angerville, R.; Saulais, M.; Emmanuel, E.; Perrodin, Y. Physicochemical and Ecotoxicological Characteristics of Urban Wet Weather Effluents (UWWE) from a French Site. In Proceedings of 20th Annual Conference of Association for Environmental Health and Sciences (AEHS), San Diego, CA, USA, 15–18 March 2010.
- Perrodin, Y.; Boillot, C.; Angerville, R.; Donguy, G.; Emmanuel, E. Ecological risk assessment of urban and industrial systems: A review. Sci. Total Environ. 2011, 409, 5162–5176. [Google Scholar] [CrossRef]
- US-EPA. Framework for Ecological Risk Assessment. In Proceedings of Risk Assessment Forum, Washington, DC, USA, May 1992; p. 161.
- Suter, G.W. Environmental Risk Assessment; Lewis Publishers: Chelsea, UK, 1993; p. 538. [Google Scholar]
- United States Environmental Protection Agency, Guidelines for Ecological Risk Assessment; US-EPA: Washington, DC, USA, 1998; p. 188.
- Rivière, J.L. Évaluation du risque écologique des sols pollués; Tec & Doc Lavoisier: Paris, France, 1998; p. 228. [Google Scholar]
- CEAEQ, Procedure for Ecotoxicological Risk Assessment of Contaminated Land (Procédure d’évaluation des risques écotoxicologiques pour la réhabilitation des terrains contaminés); Centre d’Expertise en Analyse Environnementale du Québec, Ministère de l'Environnement et de la Faune, Gouvernement du Québec: Quebec, Canada, 1998; p. 139.
- EAUK, Ecological Risk Assessment, a Public Consultation on a Frame Work and Methods for Assessing Harm to Ecosystems from Contaminants Soil; Environment Agency of United Kingdoms: Bristol, UK, 2003; p. 104.
- Liliburne, L.; Phillips, C. Risk Assessment for Contaminated Sites in New Zealand. Available online: http://contamsites.landcareresearch.co.nz/ (accessed on 15 April 2013).
- ECB, Technical Guidance Document in Support of Commission Directive 93/67/EEC on Risk Assessment for New Notified Substances, Commission Regulation (EC) No 1488/94 on Risk Assessment for Existing Substances And Directive 98/8/EC of The European Parliament and of the Council Concerning the Placing of Biocidal Products on the Market; Part I, II, III and IV; European Chemical Bureau: Ispra, Italy, 2003; p. 1044.
- CETMEF, Ecotoxicological Risk Assessment of Contaminated Sediments or Dredging Materials. Evaluation (écotoxicologique de sédiments contaminés ou de matériaux de dragage); Centre d’Etudes Techniques Maritimes et Fluviales: Compiègne, France, 2001; p. 12.
- Perrodin, Y.; Grelier-Volatier, L.; Barna, R.; Gobbey, A. Assessment of the ecocompatibility of waste disposal or waste use scenarios: Towards the elaboration and implementation of a comprehensive methodology. Waste Manag. Series 2000, 1, 504–512. [Google Scholar] [CrossRef]
- ADEME, Assessment of the Ecocompatibility of Waste Disposal or Waste Use Scenarios. Evaluation (de l’écocompatibilité des scénarios de stockage et de valorisation des déchets); National Environment and Energy Management Agency. Agence De l’Environnement et de la Maîtrise de l’Énergie: Angers, France, 2002; Rapport No. 4445; p. 148.
- Emmanuel, E.; Perrodin, Y.; Keck, G.; Blanchard, J.M.; Vermande, P. Ecotoxicological risk assessment of hospital wastewater: A proposed framework for raw effluents discharging into urban sewer network. J. Hazard. Mater. 2005, 117, 1–11. [Google Scholar] [CrossRef]
- Boillot, C.; Angerville, R.; Panouillères, M.; Perrodin, Y. Ecotoxicological risk assessment of hospital wastewater discharge: Characterization of combined effect due to binary mixtures of three surfactants and three disinfectants towards daphnia magna (poster). In Proceedings of 17th SETAC Europe, Porto, Portugal, 20–24 May 2007.
- Borchardt, D.; Sperling, F. Urban stormwater discharges: Ecological effects on receiving waters and consequences for technical measures. Water Sci. Technol. 1997, 36, 173–178. [Google Scholar]
- Mulliss, R.M.; Revitt, D.M.; Shutes, R.B.E. The impacts of discharges from two combined sewer overflows on the water quality of an urban watercourse. Water Sci. Technol. 1997, 36, 195–199. [Google Scholar] [CrossRef]
- Seidl, M.; Viviane, H.; Mouchel, J.M. Toxicity of combined sewer overflows on phytoplankton: The role of heavy metals. Environ. Pollut. 1998, 101, 107–116. [Google Scholar]
- Rochfort, Q.; Grapentine, L.; Marsalek, J.; Brownlee, B.; Reynoldson, T.; Thompson, S.; Milani, D.; Logan, C. Using benthic assessment techniques to determine combined sewer overflow and stormwater impacts in the aquatic ecosystem. Water Qual. Res. J. Canada 2000, 35, 365–397. [Google Scholar]
- Lafont, M.; Vivier, A.; Nogueira, S.; Namour, P.; Breil, P. Surface and hyporheic oligochaete assemblages in a French suburban stream. Hydrobiologia 2006, 564, 183–193. [Google Scholar] [CrossRef]
- Davoren, M.; Fogarty, A.M. A test battery for the ecotoxicological evaluation of the agri-chemical environ. Ecotox. Environ. Saf. 2004, 59, 116–122. [Google Scholar] [CrossRef]
- Kim, Y.; Choi, K.; Jung, J.; Park, S.; Kim, P.-G.; Park, J. Aquatic toxicity of acetaminophen, carbamazepine, cimetidine, diltiazem and six major sulfonamides, and their potential ecological risks in Korea. Environ. Int. 2007, 33, 370–375. [Google Scholar] [CrossRef]
- Radix, P.; Léonard, M.; Papantoniou, C.; Roman, G.; Saouter, E.; Gallotti-Schmitt, S.; Thiébaud, H.; Vasseur, P. Comparison of four chronic toxicity tests using algae, bacteria, and invertebrates assessed with sixteen chemicals. Ecotox. Environ. Saf. 2000, 47, 186–194. [Google Scholar]
- Andrén, C.; Eklund, B.; Gravenfors, E.; Kukulska, Z.; Tarkpea, M. A multivariate biological and chemical characterization of industrial effluents connected to municipal sewage treatment plants. Environ. Toxicol. Chem. 1998, 17, 228–233. [Google Scholar]
- Naudin, S.; Garric, J.; Vindimian, E.; Bray, M.; Migeon, B.; Vollat, B.; Lenon, G. Influence of the sample preservation mode to assess the chronic toxicity of an industrial effluent. Ecotox. Environ. Saf. 1995, 30, 54–62. [Google Scholar] [CrossRef]
- Persoone, G.; Marsalek, B.; Blinova, I.; Torokne, A.; Zarina, D.; Manusadzianas, L.; Nalecz-Jawecki, G.; Tofan, L.; Stepanova, N.; Tothova, L.; et al. A practical and user-friendly toxicity classification system with microbiotests for natural waters and wastewaters. Environ. Toxicol. 2003, 18, 395–402. [Google Scholar] [CrossRef]
- Ren, S.; Frymier, P.D. Use of multidimensional scaling in the selection of wastewater toxicity test battery components. Water Res. 2003, 37, 1655–1661. [Google Scholar] [CrossRef]
- Davoren, M.; Ni Shúilleabháin, S.; O’Halloran, J.; Hartl, M.G.J.; Sheehan, D.; O’Brien, N.M.; Van Pelt, F.N.A.M.; Mothersill, C. A test battery approach for the ecotoxicological evaluation of estuarine sediments. Ecotoxicology 2005, 14, 741–755. [Google Scholar] [CrossRef]
- Clément, B.; Persoone, G.; Colin, J.; Du-Delepierre, A. Estimation of the hazard of landfills through toxicity testing of leachates. I. Determination of leachate toxicity with a battery of acute tests. Chemosphere 1996, 33, 2303–2320. [Google Scholar] [CrossRef]
- Isidori, M.; Lavorgna, M.; Nardelli, A.; Parrella, A. Toxicity identification evaluation of leachates from municipal solid waste landfills: A multispecies approach. Chemosphere 2003, 52, 85–94. [Google Scholar] [CrossRef]
- Pandard, P.; Devillers, J.; Charissou, A.-M.; Poulsen, V.; Jourdain, M.-J.; Ferard, J.-F.; Grand, C.; Bispo, A. Selecting a battery of bioassays for ecotoxicological characterization of wastes. Sci. Total Environ. 2006, 363, 114–125. [Google Scholar] [CrossRef]
- Rojícková-Padrtová, R.; MarIsálek, B.; Holoubek, I. Evaluation of alternative and standard toxicity assays for screening of environmental samples: Selection of an optimal test battery. Chemosphere 1998, 37, 495–507. [Google Scholar]
- Juvonen, R.; Martikainen, E.; Schultz, E.; Joutti, A.; Ahtiainen, J.; Lehtokari, M. A battery of toxicity tests as indicators of decontamination in composting oily waste. Ecotox. Environ. Saf. 2000, 47, 156–166. [Google Scholar] [CrossRef]
- Schaefer, M. Assessing 2,4,6-trinitrotoluene (TNT)-contaminated soil using three different earthworm test methods. Ecotox. Environ. Saf. 2004, 57, 74–80. [Google Scholar]
- Chapman, P.M.; Fairbrother, A.; Brown, D. A critical assessment of safety (uncertainety) factors for ecological risk assessment. Environ. Toxicol. Chem. 1998, 17, 99–108. [Google Scholar] [CrossRef]
- INERIS. Portail Substances Chimiques. Available online: http://www.Ineris.Fr/fr/rapports-d%c3%a9tude/toxicologie-et-environnement/fiches-de-donn%c3%a9es-toxicologiques-et-environnementales (accessed on 15 April 2013).
- Hynes, H.B.N. Groundwater and stream ecology. Hydrobiologia 1983, 100, 93–99. [Google Scholar] [CrossRef]
- Jones, J.B.; Mulholland, P.J. Streams and Ground Waters; Academic Press: San Diego, CA, USA, 2000; p. 425. [Google Scholar]
- Danielopol, D.L. Groundwater fauna associated with riverine aquifers. J. N. Am. Benthol. Soc. 1989, 8, 18–35. [Google Scholar] [CrossRef]
- Charissou, A.M.; Jourdain, M.J.; Pandard, P.; Poulsen, V.; Devillers, J.; Férard, J.F.; Bispo, A. Démarche optimale de sélection de batterie de bioessais pour l’évaluation écotoxicologique des milieux complexes. Synthèse bibliographique. Tech. Sci. Meth. 2006, 5, 101–110. [Google Scholar]
- Clément, B.; Cadier, C. Development of a new laboratory freshwater/sediment microcosm test. Ecotoxicology 1998, 7, 279–290. [Google Scholar] [CrossRef]
- Austin, A.; Deniseger, J. Periphyton community changes along a heavy metals gradient in a long narrow lake. Environ. Exp. Bot. 1985, 25, 41–52. [Google Scholar]
- Bradac, P.; Wagner, B.; Kistler, D.; Traber, J.; Behra, R.; Sigg, L. Cadmium speciation and accumulation in periphyton in a small stream with dynamic concentration variations. Environ. Pollut. 2010, 158, 641–648. [Google Scholar] [CrossRef]
- Ramelow, G.J.; Maples, R.S.; Thompson, R.L.; Mueller, C.S.; Webre, C.; Beck, J.N. Periphyton as monitors for heavy metal pollution in the calcasieu river estuary. Environ. Pollut. 1987, 43, 247–261. [Google Scholar] [CrossRef]
- Serra, A.; Guasch, H. Effects of chronic copper exposure on fluvial systems: Linking structural and physiological changes of fluvial biofilms with the in-stream copper retention. Sci. Total Environ. 2009, 407, 5274–5282. [Google Scholar] [CrossRef]
- Würgler, F.E.; Kramers, P.G.N. Environmental effects of genotoxins (eco-genotoxicology). Mutagenesis 1992, 7, 321–327. [Google Scholar] [CrossRef]
- Devaux, A.; Pesonen, M.; Monod, G. Alkaline comet assay in rainbow trout hepatocytes. Toxicol. In Vitro 1997, 11, 71–79. [Google Scholar] [CrossRef]
- Theodorakis, C.W. Integration of genotoxic and population genetic endpoints in biomonitoring and risk assessment. Ecotoxicology 2001, 10, 245–256. [Google Scholar] [CrossRef]
- Devaux, A.; Flammarion, P.; Bernardon, V.; Garric, J.; Monod, G. Monitoring of the chemical pollution of the river rhône through measurement of DNA damage and cytochrome p4501a induction in chub (Leuciscus cephalus). Mar. Environ. Res. 1998, 46, 257–262. [Google Scholar]
- Anderson, S.L.; Wild, G.C. Linking genotoxic responses and reproductive success in ecotoxicology. Environ. Health Perspect. 1994, 102, 9–12. [Google Scholar] [CrossRef]
- Depledge, M.H. Genetic ecotoxicology: An overview. J. Exp. Mar. Biol. Ecol. 1996, 200, 57–66. [Google Scholar]
- Lacaze, E.; Geffard, O.; Goyet, D.; Bony, S.; Devaux, A. Linking genotoxic responses in Gammarus fossarum germ cells with reproduction impairment, using the comet assay. Environ. Res. 2010, 111, 626–634. [Google Scholar]
- Calow, P. Ecological risk assessment: Risk for what? How do we decide? Ecotoxicol. Environ. Saf. 1998, 40, 15–18. [Google Scholar] [CrossRef]
- Babut, M.; Delmas, H.; Bray, M.; Durrieu, C.; Perrodin, Y.; Garric, J. Characterizing the risks to aquatic ecosystems: A tentative approach in the context of freshwater dredged material disposal. Integr. Environ. Assess. Manag. 2006, 2, 330–343. [Google Scholar] [CrossRef]
- Klaine, S.J.; Cobb, G.P.; Dickerson, R.L.; Dixon, K.R.; Kendall, R.J.; Smith, E.E.; Solomon, K.R. An ecological risk assessment for the use of the biocide, dibromonitrilopropionamide (DBNPA), in industrial cooling systems. Environ. Toxicol. Chem. 1996, 15, 21–30. [Google Scholar]
- Solomon, K.R.; Baker, D.B.; Richards, R.P.; Dixon, K.R.; Klaine, S.J.; La Point, T.W.; Kendall, R.J.; Weisskopf, C.P.; Giddings, J.M.; Giesy, J.P.; et al. Ecological risk assessment of atrazine in north American surface waters. Environ. Toxicol. Chem. 1996, 15, 31–76. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).