Screening of Microorganisms Producing Cold-Active Oxidoreductases to Be Applied in Enantioselective Alcohol Oxidation. An Antarctic Survey
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of Microorganisms Producing Oxidoreductases
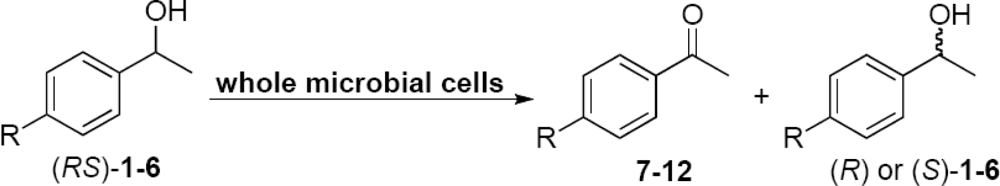
2.2. Evaluation of the Enzyme Activity of Microorganisms
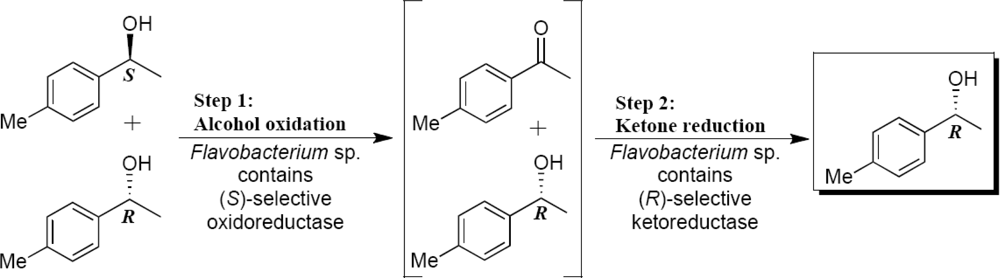
2.3. Optimization of the Enantioselective Oxidation Reaction Using Flavobacterium sp. and Arthrobacter sp
3. Experimental Section
3.1. General Methods
3.2. Preparation of the (RS)-1-(Phenyl)ethanols (1–6) [33]
3.3. Procedures for Bioprospection
3.3.1. Soil and Sediment Sampling Procedure
3.3.2. Culture Media
- -MCFE7 (pH = 5.6) = (RS)-1-(phenyl)ethanol–1 mmol, 0.122 g, 122 μL), KH2PO4 (0.1%; 1.0 g), MgSO4.7H2O (0.04%; 0.4 g), NH4Cl (0.04%; 0.4 g), Dextrose (Oxoid) (0.025%; 0.25 g), Peptone (Oxoid) (0.025%; 0.25 g), agar (Oxoid) (2.0%; 20.0 g; when needed).
- -MCBE7 (pH = 7.0)= (RS)-1-(phenyl)ethanol–1 mmol, 0.122 g, 122 μL), K2HPO4 (0.2%; 2.0 g), KH2PO4 (0.1%; 1.0 g), MgSO4.7H2O (0.04%; 0.4 g), NH4Cl (0.04%; 0.4 g), Meat Extract (Oxoid) (0.025%; 0.25 g), Peptone (Oxoid) (0.025%; 0.25 g), agar (Oxoid) (2.0%; 20.0 g; when needed).
3.3.3. Isolation and Growth of Microorganisms
- One microtiter plate contains MCFE7 culture medium;
- One microtiter plate contains MCBE7 culture medium;
3.4. Enzyme Assays
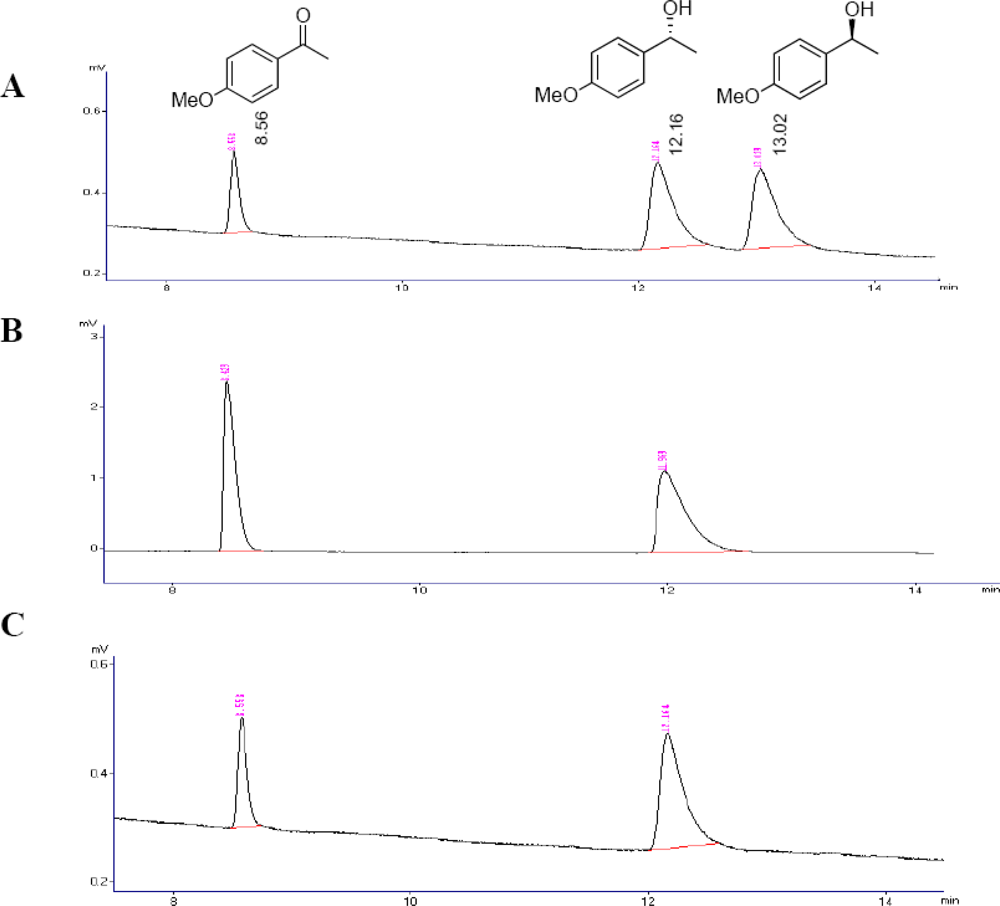
3.5. GC Analysis
3.6. Microorganisms Library and Preservation
3.7. Bacteria Identification
3.8. Effect of Temperature and Reaction Time on the Oxidation Reactions Using Flavobacterium sp. and Arthrobacter sp
3.9. Growth Measurements
3.10. Absolute Configuration
4. Conclusions
Acknowledgments
- Samples Availability: Available from the authors.
References
- Soni, P; Banerjee, UC. Enantioselective reduction of acetophenone and its derivatives with a new yeast isolate Candida tropicalis PBR-2MTCC 5158. Biotechnol. J 2006, 1, 80–85. [Google Scholar]
- Matsuda, T; Yamanaka, R; Nakamura, K. Recent progress in biocatalysis for asymmetric oxidation and reduction. Tetrahedron: Asymmetry 2009, 20, 513–557. [Google Scholar]
- Voss, CV; Gruber, CC; Kroutil, W. Deracemisation of secondary alcohols via biocatalytic stereoinversion. Synlett 2010, 7, 991–998. [Google Scholar]
- Ishige, T; Honda, K; Shimizu, S. Whole organism biocatalysis. Curr Opin Chem Biol 2005, 9, 174–180. [Google Scholar]
- Cardenas, F; Alvarez, E; de Castro-Alvarez, MS; Sanchez-Montero, JM; Valmaseda, M; Elson, SW; Sinisterra, JV. Screening and catalytic activity in organic synthesis of novel fungal and yeast lipases. J Mol Catal B: Enzym 2001, 14, 111–123. [Google Scholar]
- Andrade, LH; Comasseto, JV; Rodrigues, DF; Pellizari, VH; Porto, ALM. Enantioselective reduction of ortho-substituted acetophenones by bacterial strains isolated from medium enriched with biphenyl or diesel fuel. J Mol Catal B: Enzym 2005, 33, 73–79. [Google Scholar]
- Yoon, J; Miwa, H; Ahmed, I; Yokota, A; Fujiwara, T. Rhodococcus baikonurensis BTM4c, a boron-tolerant actinobacterial strain isolated from soil. Biosci Biotechnol Biochem 2010, 74, 178–181. [Google Scholar]
- Lo Giudice, A; Bruni, V; Michaud, L. Characterization of Antarctic psychrotrophic bacteria with antibacterial activities against terrestrial microorganisms. J Basic Microb 2007, 47, 496–505. [Google Scholar]
- Marx, JC; Collins, T; D'Amico, S; Feller, G; Gerday, C. Cold-adapted enzymes from marine antarctic microorganisms. Mar Biotechnol 2007, 9, 293–304. [Google Scholar]
- Cavicchioli, R; Siddiqui, KS; Andrews, D; Sowers, KR. Low-temperature extremophiles and their applications. Curr Opin Biotech 2002, 13, 253–261. [Google Scholar]
- Russell, NJ. Toward a molecular understanding of cold activity of enzymes from psychrophiles. Extremophiles 2000, 4, 83–90. [Google Scholar]
- Kazuoka, T; Oikawa, T; Kuroda, IMS; Soda, K. Cold-active and thermostable alcohol dehydrogenase of a psychrotorelant from Antarctic seawater, Flavobacterium frigidimaris KUC-1. Extremophiles 2007, 11, 257–267. [Google Scholar]
- Feller, G; Arpigny, JL; Narinx, E; Gerday, C. Molecular adaptations of enzymes from psychrophilic organisms. Comp Biochem Physiol 1997, 118A, 495–499. [Google Scholar]
- Velonia, K; Tsigos, I; Bouriotis, V; Smonou, I. Stereospecificity of hydrogen transfer by the NAD+-linked alcohol dehydrogenase from the antarctic psychrophile Moraxella sp. TAE123. Bioorg Med Chem Lett 1999, 9, 65–68. [Google Scholar]
- D'Amico, S; Marx, JC; Gerday, C; Feller, G. Activity stability relationships in extremophilic enzymes. J Biol Chem 2003, 278, 7891–7896. [Google Scholar]
- Cripps, RE; Trudgill, PW; Whateley, JG. The metabolism of 1-phenylethanol and acetophenone by Nocardia T5 and an Arthrobacter species. Eur J Biochem 1978, 86, 175–186. [Google Scholar]
- Morita, RY. Psychrophilic bacteria. Bacteriol Rev 1975, 39, 144–167. [Google Scholar]
- Feller, G; D’Amico, D; Gerday, C. Thermodynamic stability of a cold-active α-amylase from the Antarctic bacterium Alteromonas haloplanktis. Biochemistry 1999, 38, 4613–4619. [Google Scholar]
- Rashid, N; Kikuchi, H; Ezaki, S; Atomi, H; Imanaka, T. Isolation and characterization of psychrotrophs from subterranean environments. J Biosci Bioeng 1999, 87, 746–751. [Google Scholar]
- Connaughton, S; Collins, G; O’Flaherty, V. Psychrophilic and mesophilic anaerobic digestion of brewery effluent: A comparative study. Water Res 2006, 40, 2503–2510. [Google Scholar]
- Hoover, RB; Pikuta, EV. Psychrophilic and psychrotolerant microbial extremophiles in polar environments. Polar Microbiol 2010, 115–156. [Google Scholar]
- Irwin, JA. Extremophiles and their application to veterinary medicine. Environ Technol 2010. [Google Scholar]
- Cavicchioli, R; Thomas, T; Curmi, PMG. Cold stress response in Archaea. Extremophiles 2000, 4, 321–331. [Google Scholar]
- Kroutil, W; Mischitz, M; Faber, K. Deracemization of (±)-2,3-disubstituted oxiranes via biocatalytic hydrolysis using bacterial epoxide hydrolases: kinetics of an enantioconvergent process. J Chem Soc Perk Trans 1 1997, 24, 3629–3636. [Google Scholar]
- Allan, GR; Carnell, AJ. Microbial deracemization of 1-aryl and 1-heteroaryl secondary alcohols. J Org Chem 2001, 66, 6495–6497. [Google Scholar]
- Gareth, CJ; Andrew, CJ; Harald, T; Thomas, R. Microbial deracemisation of N-(1-hydroxy-1-phenylethyl)benzamide. Tetrahedron: Asymmetry 2004, 15, 239–243. [Google Scholar]
- Demir, AS; Hamamci, H; Sesenoglu, O; Neslihanoglu, R; Asikoglu, B; Capanoglu, D. Fungal deracemization of benzoin. Tetrahedron Lett 2002, 43, 6447–6449. [Google Scholar]
- Gruber, CC; Lavandera, I; Faber, K; Kroutil, W. From a racemate to a single enantiomer. deracemization by stereoinversion. Adv Synth Catal 2006, 348, 1789–1805. [Google Scholar]
- Orru, RVA; Kroutil, W; Faber, K. Deracemization of (±)-2,2-disubstituted epoxides via enantioconvergent chemoenzymatic hydrolysis using Nocardia EH1 epoxide hydrolase and sulfuric acid. Tetrahedron Lett 1997, 38, 1753–1754. [Google Scholar]
- Kroutil, W; Faber, K. Deracemization of compounds possessing a sec-alcohol or -amino group through a cyclic oxidation-reduction sequence: a kinetic treatment. Tetrahedron: Asymmetry 1998, 9, 2901–2913. [Google Scholar]
- Russell, NJ. Cold adaptation of microorganisms. Phil Trans R Soc B 1990, 326, 595–611. [Google Scholar]
- Feller, G; Arpigny, JL; Narinx, E; Gerday, C. Molecular adaptations of enzymes from psychrophilic organisms. Comp Biochem Physiol 1997, 118, 495–499. [Google Scholar]
- Andrade, LH; Polak, R; Porto, ALM; Schoenlein-Crusius, IH; Comasseto, JV. Application of bioreduction by microorganisms in the enantioselective synthesis of alpha-substituted-1-phenylethanols. Lett Org Chem 2006, 3, 613–618. [Google Scholar]
- Nakamura, K; Matsuda, T. Asymmetric reduction of ketones by the acetone powder of Geotrichum candidum. J Org Chem 1998, 63, 8957–8964. [Google Scholar]
- Salvi, NA; Chattopadhyay, S. Studies on Rhizopus arrhizus mediated enantioselective reduction of arylalkanones. Tetrahedron 2001, 57, 2833–2839. [Google Scholar]
- Nogales, B; Moore, ERB; Brossa, EL; Mora, RR; Amann, R; Timmis, KN. Combined use of 16S ribosomal DNA and 16S rRNA to study the bacterial community of polychlorinated biphenyl-polluted soil. Appl Environ Microbiol 2001, 67, 1874–1884. [Google Scholar]
- Naemura, K; Murata, M; Tanaka, R; Yano, M; Hirose, K; Tobe, Y. Enantioselective acylation of primary and secondary alcohols catalyzed by lipase QL from Alcaligenes sp.: A predictive active site model for lipase QL to identify which enantiomer of an alcohol reacts faster in this acylation. Tetrahedron: Asymmetry 1996, 7, 3285–3294. [Google Scholar]
- Yadav, JS; Nanda, S; Reddy, TP; Rao, AB. Efficient enantioselective reduction of ketones with Daucus carota Root. J Org Chem 2002, 67, 3900–3903. [Google Scholar]
- Kim, MJ; Kim, HM; Kim, D; Ahn, Y; Park, J. Dynamic kinetic resolution of secondary alcohols by enzyme-metal combinations in ionic liquid. Green Chem 2004, 6, 471–474. [Google Scholar]


| Culture medium | Microorganisms (soil) | Microorganisms (sediment) |
|---|---|---|
| MCBE7a | 103 | 17 |
| MCFE7b | 96 | 16 |
| Entry | Microorganism | R | Ketone/Alcohola | Alcohol e.e. (%)a |
|---|---|---|---|---|
| 1 | MPS8D3 | H | 11/89 | 25 (R) |
| 2 | Me | 22/78 | 92 (R) | |
| 3 | MeO | 46/54 | >99 (R) | |
| 4 | Cl | 21/79 | 89 (R) | |
| 5 | Br | 23/77 | 96 (R) | |
| 6 | NO2 | 43/57 | 57 (R) | |
| 7 | HK1D9 | H | 16/84 | 22 (R) |
| 8 | Me | 27/73 | 97 (R) | |
| 9 | MeO | 47/53 | >99 (R) | |
| 10 | Cl | 25/75 | 95 (R) | |
| 11 | Br | 4/96 | 17 (R) | |
| 12 | NO2 | 34/66 | 33 (R) | |
| 13 | HK2D2 | H | 6/94 | 10 (R) |
| 14 | Me | 17/83 | 57 (R) | |
| 15 | MeO | 25/75 | 45 (R) | |
| 16 | Cl | 16/84 | 52 (R) | |
| 17 | Br | 21/79 | 61 (R) | |
| 18 | NO2 | 7/93 | 6 (R) | |
| 19 | MPS4D2 | H | 8/92 | 22 (R) |
| 20 | Me | 17/83 | 93 (R) | |
| 21 | MeO | 44/56 | >99 (R) | |
| 22 | Cl | 15/85 | 69 (R) | |
| 23 | Br | 19/81 | 87 (R) | |
| 24 | NO2 | 21/79 | 22 (R) | |
| 25 | HK1D8 | H | 2/98 | 3 |
| 26 | Me | 5/95 | 17 (R) | |
| 27 | MeO | 15/85 | 22 (R) | |
| 28 | Cl | 5/95 | 15 (R) | |
| 29 | Br | 5/95 | 15 (R) | |
| 30 | NO2 | 5/95 | 15 (R) | |
| 31 | HK1D10 | H | 11/89 | 17 (R) |
| 32 | Me | 9/91 | 24 (R) | |
| 33 | MeO | 41/59 | 79 (R) | |
| 34 | Cl | 1/99 | 1 | |
| 35 | Br | 1/99 | 2 | |
| 36 | NO2 | 4/96 | 2 | |
| Flavobacterium sp. (HK1D9) | ||||
|---|---|---|---|---|
| Entry | Temp. (°C)a | Time (h) | Ketone/Alcoholb | Alcohol e.e. (%)b |
| 1 | 5 | 24 | nd | nd |
| 2 | 48 | nd | nd | |
| 3 | 72 | nd | nd | |
| 4 | 10 | 24 | 5/95 | 10 (R) |
| 5 | 48 | 14/86 | 21 (R) | |
| 6 | 72 | 14/86 | 24 (R) | |
| 7 | 15 | 24 | 32/68 | 42 (R) |
| 8 | 48 | 46/54 | 78 (R) | |
| 9 | 72 | 48/52 | 91 (R) | |
| 10 | 20 | 24 | 42/58 | 76 (R) |
| 11 | 48 | 41/59 | 83 (R) | |
| 12 | 72 | 49/51 | 96 (R) | |
| 13 | 25 | 24 | 45/55 | 89 (R) |
| 14 | 48 | 40/60 | 93 (R) | |
| 15 | 72 | 33/67 | 95 (R) | |
| 16 | 30 | 24 | nd | nd |
| 17 | 48 | nd | nd | |
| 18 | 72 | nd | nd | |
| Arthrobacter sp. (MPS8D3) | ||||
| 19 | 5 | 24 | - | - |
| 20 | 48 | - | - | |
| 21 | 72 | 1/99 | - | |
| 22 | 10 | 24 | 1/99 | 1 |
| 23 | 48 | 2/98 | 1 | |
| 24 | 72 | 5/95 | 11 (R) | |
| 25 | 15 | 24 | 40/60 | 94 (R) |
| 26 | 48 | 37/63 | 96 (R) | |
| 27 | 72 | 34/66 | 96 (R) | |
| 28 | 20 | 24 | 40/60 | 95 (R) |
| 29 | 48 | 36/64 | 95 (R) | |
| 30 | 72 | 30/70 | 96 (R) | |
| 31 | 25 | 24 | 38/62 | 96 (R) |
| 32 | 48 | 34/66 | 95 (R) | |
| 33 | 72 | 30/70 | 95 (R) | |
| 34 | 30 | 24 | - | - |
| 35 | 48 | 1/99 | - | |
| 36 | 72 | 2/98 | 4 | |
| Flavobacterium sp. (HK1D9) | ||||
|---|---|---|---|---|
| Entry | Temp. (°C)a | Time (h) | Ketone/Alcoholb | Alcohol e.e. (%)b |
| 1 | 24 | 39/61 | 39 (R) | |
| 2 | 5 | 48 | 39/61 | 79 (R) |
| 3 | 72 | 45/55 | 95 (R) | |
| 4 | 24 | 35/65 | 62 (R) | |
| 5 | 10 | 48 | 44/56 | 95 (R) |
| 6 | 72 | 51/49 | >99 (R) | |
| 7 | 24 | 25/75 | 36 (R) | |
| 8 | 15 | 48 | 43/57 | 83 (R) |
| 9 | 72 | 43/57 | 95 (R) | |
| 10 | 24 | 28/72 | 49 (R) | |
| 11 | 20 | 48 | 35/65 | 80 (R) |
| 12 | 72 | 38/62 | 83 (R) | |
| 13 | 24 | 45/55 | 89 (R) | |
| 14 | 25 | 48 | 40/60 | 93 (R) |
| 15 | 72 | 33/67 | 95 (R) | |
| 16 | 24 | 7/93 | 6 (R) | |
| 17 | 30 | 48 | 6/94 | 9 (R) |
| 18 | 72 | 7/93 | 18 (R) | |
| Arthrobacter sp. (MPS8D3) | ||||
| 19 | 24 | 40/60 | 88 (R) | |
| 20 | 5 | 48 | 44/56 | 96 (R) |
| 21 | 72 | 43/57 | 96 (R) | |
| 22 | 24 | 40/60 | 94 (R) | |
| 23 | 10 | 48 | 46/54 | 97 (R) |
| 24 | 72 | 41/59 | 96 (R) | |
| 25 | 24 | 41/59 | >99 (R) | |
| 26 | 15 | 48 | 37/63 | >99 (R) |
| 27 | 72 | 36/64 | >99 (R) | |
| 28 | 24 | 43/57 | 98 (R) | |
| 29 | 20 | 48 | 38/63 | 98 (R) |
| 30 | 72 | 36/64 | >99 (R) | |
| 31 | 24 | 45/55 | 89 (R) | |
| 32 | 25 | 48 | 40/60 | 93 (R) |
| 33 | 72 | 33/67 | 95 (R) | |
| 34 | 24 | 42/58 | 97 (R) | |
| 35 | 30 | 48 | 33/67 | 97 (R) |
| 36 | 72 | 27/73 | 97 (R) | |
| Flavobacterium sp. (HK1D9) | ||||
|---|---|---|---|---|
| Entry | Temp. (°C)a | Time (h) | Ketone/Alcoholb | Alcohol e.e. (%)b |
| 1 | 5 | 24 | 5/95 | 15 (R) |
| 2 | 48 | 16/84 | 30 (R) | |
| 3 | 72 | 21/79 | 43 (R) | |
| 4 | 10 | 24 | 19/81 | 21 (R) |
| 5 | 48 | 34/66 | 65 (R) | |
| 6 | 72 | 42/58 | 89 (R) | |
| 7 | 15 | 24 | 25/75 | 36 (R) |
| 8 | 48 | 43/57 | 83 (R) | |
| 9 | 72 | 43/57 | 95 (R) | |
| 10 | 20 | 24 | 16/84 | 44 (R) |
| 11 | 48 | 34/66 | 73 (R) | |
| 12 | 72 | 41/59 | 96 (R) | |
| 13 | 25 | 24 | 27/73 | 53 (R) |
| 14 | 48 | 33/67 | 77 (R) | |
| 15 | 72 | 34/66 | 87 (R) | |
| 16 | 30 | 24 | 9/91 | 9 (R) |
| 17 | 48 | 9/91 | 10 (R) | |
| 18 | 72 | 6/94 | 12 (R) | |
| Arthrobacter sp. (MPS8D3) | ||||
| 19 | 5 | 24 | 42/58 | 90 (R) |
| 20 | 48 | 42/58 | >99 (R) | |
| 21 | 72 | 45/55 | >99 (R) | |
| 22 | 10 | 24 | 41/59 | 95 (R) |
| 23 | 48 | 41/59 | 97 (R) | |
| 24 | 72 | 43/47 | 97 (R) | |
| 25 | 15 | 24 | 35/65 | 89 (R) |
| 26 | 48 | 34/66 | 97 (R) | |
| 27 | 72 | 31/69 | 98 (R) | |
| 28 | 20 | 24 | 42/58 | 98 (R) |
| 29 | 48 | 36/64 | 98 (R) | |
| 30 | 72 | 33/67 | 98 (R) | |
| 31 | 25 | 24 | 43/57 | >99 (R) |
| 32 | 48 | 36/64 | >99 (R) | |
| 33 | 72 | 32/68 | >99 (R) | |
| 34 | 30 | 24 | 37/63 | 96 (R) |
| 35 | 48 | 33/67 | 97 (R) | |
| 36 | 72 | 25/75 | 97 (R) | |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Araújo, L.S.; Kagohara, E.; Garcia, T.P.; Pellizari, V.H.; Andrade, L.H. Screening of Microorganisms Producing Cold-Active Oxidoreductases to Be Applied in Enantioselective Alcohol Oxidation. An Antarctic Survey. Mar. Drugs 2011, 9, 889-905. https://doi.org/10.3390/md9050889
Araújo LS, Kagohara E, Garcia TP, Pellizari VH, Andrade LH. Screening of Microorganisms Producing Cold-Active Oxidoreductases to Be Applied in Enantioselective Alcohol Oxidation. An Antarctic Survey. Marine Drugs. 2011; 9(5):889-905. https://doi.org/10.3390/md9050889
Chicago/Turabian StyleAraújo, Lidiane S., Edna Kagohara, Thaís P. Garcia, Vivian H. Pellizari, and Leandro H. Andrade. 2011. "Screening of Microorganisms Producing Cold-Active Oxidoreductases to Be Applied in Enantioselective Alcohol Oxidation. An Antarctic Survey" Marine Drugs 9, no. 5: 889-905. https://doi.org/10.3390/md9050889




