Phylogenetic Identification of Fungi Isolated from the Marine Sponge Tethya aurantium and Identification of Their Secondary Metabolites
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of the Fungal Strains Isolated from T. aurantium
2.2. Secondary Metabolite Analyses
3. Experimental Section
3.1. Sampling Sites
3.2. Sponge Collection
3.3. Isolation, Cultivation and Storage of Fungi
3.4. Morphological Identification of Fungal Isolates
3.5. Scanning Electron Microscopy
3.6. Genetic Identification of Fungal Isolates and Phylogenetic Analysis
3.7. Fermentation and Production of Extracts
3.8. Chemical Analysis
3.9. Bioassays
4. Conclusion
Acknowledgments
- Samples Availability: Available from the authors.
References
- Newman, DJ; Cragg, GM. Natural products as sources of new drugs over the last 25 years. J Nat Prod 2007, 70, 461–477. [Google Scholar]
- Harvey, AL. Natural products in drug discovery. Drug Discov Today 2008, 13, 894–901. [Google Scholar]
- Blunt, JW; Copp, BR; Hu, WP; Munro, MH; Northcote, PT; Prinsep, MR. Marine natural products. Nat Prod Rep 2009, 26, 170–244. [Google Scholar]
- König, GM; Kehraus, S; Seibert, SF; Abdel-Lateff, A; Müller, D. Natural products from marine organisms and their associated microbes. ChemBioChem 2006, 7, 229–238. [Google Scholar]
- Imhoff, JF; Stöhr, R. Sponge-associated bacteria: General overview and special aspects of bacteria associated with Halichondria panicea. Prog Mol Subcell Biol 2003, 37, 35–57. [Google Scholar]
- Gao, Z; Li, B; Zheng, C; Wang, G. Molecular detection of fungal communities in the Hawaiian marine sponges Suberites zeteki and Mycale armata. Appl Environ Microbiol 2008, 74, 6091–6101. [Google Scholar]
- Siepmann, R; Höhnk, W. Über Hefen und einige Pilze (Fungi imp., Hyphales) aus dem Nordatlantik. Veröff Inst Meeresforsch Bremerh 1962, 8, 79–97. [Google Scholar]
- Roth, FJ, Jr; Ahearn, DG; Fell, JW; Meyers, SP; Meyer, SA. Ecology and taxonomy of yeasts isolated from various marine substrates. Limnol Oceanogr 1962, 7, 178–185. [Google Scholar]
- Höhnk, U; Höhnk, W; Ulken, U. Pilze aus marinen Schwämmen. Veröff Inst Meeresforsch Bremerh 1979, 17, 199–204. [Google Scholar]
- Sponga, F; Cavaletti, L; Lazzaroni, A; Borghi, A; Ciciliato, I; Losi, D; Marinelli, F. Biodiversity and potentials of marine-derived microorganisms. J Biotechnol 1999, 70, 65–69. [Google Scholar]
- Höller, U; Wright, AD; Matthée, GF; König, GM; Draeger, S; Aust, HJ; Schulz, B. Fungi from marine sponges: Diversity, biological activity and secondary metabolites. Mycol Res 2000, 104, 1354–1365. [Google Scholar]
- Steffens, S. Prokaryoten und Mikrobielle Eukaryoten aus Marinen Schwämmen. Ph.D. Thesis, University of Bonn, Bonn, Germany. 2003.
- Namikoshi, M; Akano, K; Kobayashi, H; Koike, Y; Kitazawa, A; Rondonuwu, AB; Pratasik, SB. Distribution of marine filamentous fungi associated with marine sponges in coral reefs of Palau and Bunaken Island, Indonesia. J Tokyo Univ Fish 2002, 88, 15–20. [Google Scholar]
- Morrison-Gardiner, S. Dominant fungi from Australian coral reefs. Fung Divers 2002, 9, 105–121. [Google Scholar]
- Pivkin, MV; Aleshko, SA; Krasokhin, VB; Khudyakova, YV. Fungal assemblages associated with sponges of the southern coast of Sakhalin Island. Russ J Mar Biol 2006, 32, 249–254. [Google Scholar]
- Proksch, P; Ebel, R; Edrada, RA; Riebe, F; Liu, H; Diesel, A; Bayer, M; Li, X; Lin, WH; Grebenyuk, V; Müller, WEG; Draeger, S; Zuccaro, A; Schulz, B. Sponge-associated fungi and their bioactive compounds: The Suberites case. Bot Mar 2008, 51, 209–218. [Google Scholar]
- Wang, G; Li, Q; Zhu, P. Phylogenetic diversity of culturable fungi associated with the Hawaiian sponges Suberites zeteki and Gelliodes fibrosa. Antonie Van Leeuwenhoek 2008, 93, 163–174. [Google Scholar]
- Paz, Z; Komon-Zelazowska, M; Druzhinina, IS; Aveskamp, MM; Schnaiderman, A; Aluma, A; Carmeli, S; Ilan, M; Yarden, O. Diversity and potential antifungal properties of fungi associated with a Mediterranean sponge. Fung Divers 2010, 42, 17–26. [Google Scholar]
- Bugni, TS; Ireland, CM. Marine-derived fungi: A chemically and biologically diverse group of microorganisms. Nat Prod Rep 2004, 21, 143–163. [Google Scholar]
- Kohlmeyer, J; Volkmann-Kohlmeyer, B. Koralionastetaceae fam. nov. (Ascomycetes) from coral rock. Mycologia 1987, 79, 764–778. [Google Scholar]
- Amagata, T; Minoura, K; Numata, A. Gymnastatins F–H, cytostatic metabolites from the sponge-derived fungus Gymnascella dankaliensis. J Nat Prod 2006, 69, 1384–1388. [Google Scholar]
- Thiel, V; Neulinger, SC; Staufenberger, T; Schmaljohann, R; Imhoff, JF. Spatial distribution of sponge-associated bacteria in the Mediterranean sponge Tethya aurantium. FEMS Microbiol Ecol 2007, 59, 47–63. [Google Scholar]
- European Marine Life. Available online: http://www.european-marine-life.org/02/tethya-aurantium.php (accessed on 15 December 2010).
- Indriani, ID. Biodiversity of Marine-Derived Fungi and Identification of their Metabolites. Ph.D. Thesis, University Düsseldorf, Düsseldorf, Germany. 2007. [Google Scholar]
- Yu, Z; Lang, G; Kajahn, I; Schmaljohann, R; Imhoff, JF. Scopularides A and B, cyclodepsipeptides from a marine sponge-derived fungus Scopulariopsis brevicaulis. J Nat Prod 2008, 71, 1052–1054. [Google Scholar]
- Imhoff, JF; Yu, Z; Lang, G; Wiese, J; Kalthoff, H; Klose, S. Production and use of Antitumoral Cyclodepsipeptide. Patent WO/2009/089810, 23 July 2009. [Google Scholar]
- Ebel, R. Secondary Metabolites from Marine-Derived Fungi. In Frontiers in Marine Biotechnology; Proksch, P, Müller, EG, Eds.; Horizon Bioscience: Norfolk, UK, 2006; pp. 73–121. [Google Scholar]
- Taylor, MW; Radax, R; Steger, D; Wagner, M. Sponge-associated microorganisms: Evolution, ecology, and biotechnological potential. Microbiol Mol Biol Rev 2007, 71, 295–347. [Google Scholar]
- Wang, G. Diversity and biotechnological potential of the sponge-associated microbial consortia. J Ind Microbiol Biotechnol 2006, 33, 545–551. [Google Scholar]
- Maldonado, M; Cortadellas, N; Trillas, MI; Rützler, K. Endosymbiotic yeast maternally transmitted in a marine sponge. Biol Bull 2005, 209, 94–106. [Google Scholar]
- Bringmann, G; Lang, G; Bruhn, T; Schäffler, K; Steffens, S; Schmaljohann, R; Wiese, J; Imhoff, JF. Sorbifuranones A–C, sorbicillinoid metabolites from Penicillium strains isolated from Mediterranean sponges. Tetrahedron 2010, 66, 9894–9901. [Google Scholar]
- Bünger, J; Westphal, G; Mönnich, A; Hinnendahl, B; Hallier, E; Müller, M. Cytotoxicity of occupationally and environmentally relevant mycotoxins. Toxicology 2004, 202, 199–211. [Google Scholar]
- SciFinder®. Available online: http://www.cas.org/products/scifindr/index.html (accessed on 13 December 2010).
- Wei, MY; Wang, CY; Liu, QA; Shao, CL; She, ZG; Lin, YC. Five sesquiterpenoids from a marine-derived fungus Aspergillus sp. isolated from a gorgonian Dichotella gemmacea. Mar Drugs 2010, 29, 941–949. [Google Scholar]
- Rochfort, S; Ford, J; Ovenden, S; Wan, SS; George, S; Wildman, H; Tait, RM; Meurer-Grimes, B; Cox, S; Coates, J; Rhodes, D. A novel aspochalasin with HIV-1 integrase inhibitory activity from Aspergillus flavipes. J Antibiot 2005, 58, 279–283. [Google Scholar]
- Zhou, GX; Wijeratne, EM; Bigelow, D; Pierson, LS, III; VanEtten, HD; Gunatilaka, AA. Aspochalasins I, J, and K: Three new cytotoxic cytochalasans of Aspergillus flavipes from the rhizosphere of Ericameria laricifolia of the Sonoran Desert. J Nat Prod 2004, 67, 328–332. [Google Scholar]
- Laakso, JA; Narske, ED; Gloer, JB; Wicklow, DT; Dowd, PF. Isokotanins A–C: New bicoumarins from the sclerotia of Aspergillus alliaceus. J Nat Prod 1994, 57, 128–133. [Google Scholar]
- Junker, B; Walker, A; Connors, N; Seeley, A; Masurekar, P; Hesse, M. Production of indole diterpenes by Aspergillus alliaceus. Biotechnol Bioeng 2006, 95, 919–937. [Google Scholar]
- Slack, GJ; Puniani, E; Frisvad, JC; Samson, RA; Miller, JD. Secondary metabolites from Eurotium species, Aspergillus calidoustus and A. insuetus common in Canadian homes with a review of their chemistry and biological activities. Mycol Res 2009, 113, 480–490. [Google Scholar]
- Wiebe, LA; Bjeldanes, FF. Fusarin C, a mutagen from Fusarium moniliforme grown on corn. J Food Sci 1981, 46, 1424–1426. [Google Scholar]
- Maskey, RP; Grün-Wollny, I; Laatsch, H. Sorbicillin analogues and related dimeric compounds from Penicillium notatum. J Nat Prod 2005, 68, 865–870. [Google Scholar]
- Buckingham, J. Dictionary of Natural Products, Version 191; CRC Press: London, UK, 2010. [Google Scholar]
- Blunt, JW; Munro, MH; Laatsch, H. AntiMarin Database; University of Canterbury: Christchurch, New Zealand, 2006. [Google Scholar]
- Niggemann, J; Herrmann, M; Gerth, K; Irschik, H; Reichenbach, H; Höfle, G. Tuscolid and tuscoron A and B: Isolation, structural elucidation and studies on the biosynthesis of novel furan-3(2H)-one-containing metabolites from the myxobacterium Sorangium cellulosum. Eur J Org Chem 2004, 2004, 487–492. [Google Scholar]
- Slaughter, CJ. The naturally occurring furanones: Formation and function from pheromone to food. Biol Rev Camb Philos Soc 1999, 74, 259–276. [Google Scholar]
- White, S; O’Callaghan, J; Dobson, AD. Cloning and molecular characterization of Penicillium expansum genes upregulated under conditions permissive for patulin biosynthesis. FEMS Microbiol Lett 2006, 255, 17–26. [Google Scholar]
- Kanokmedhakul, S; Kanokmedhakul, K; Nasomjai, P; Louangsysouphanh, S; Soytong, K; Isobe, M; Kongsaeree, P; Prabpai, S; Suksamrarn, A. Antifungal azaphilones from the fungus Chaetomium cupreum CC 3003. J. Nat. Prod 2006, 69, 891–895. [Google Scholar]
- Dorner, JW; Cole, RJ; Hill, R; Wicklow, D; Cox, RH. Penicillium rubrum and Penicillium biforme, new sources of rugulovasines A and B. Appl Environ Microbiol 1980, 40, 685–687. [Google Scholar]
- Stierle, AA; Stierle, DB; Patacini, B. The berkeleyamides; amides from the acid lake fungus Penicillum rubrum. J Nat Prod 2008, 71, 856–860. [Google Scholar]
- Shiozawa, H; Takahashi, M; Takatsu, T; Kinoshita, T; Tanzawa, K; Hosoya, T; Furuya, K; Takahashi, S; Furihata, K; Seto, H. Trachyspic acid, a new metabolite produced by Talaromyces trachyspermus, that inhibits tumor cell heparanase: Taxonomy of the producing strain, fermentation, isolation, structural elucidation, and biological activity. J Antibiot 1995, 48, 357–362. [Google Scholar]
- Kuzmanović, N. Preliminarna Israživanja Dinamike Vodenih Masa LIMSKOG Kanala (Zavrsni Izvjestaj); Institut Ruder Boskovic: Rovinj, Croatia, 1985. [Google Scholar]
- Barnett, HL; Hunter, BB. Illustrated Genera of Imperfect Fungi, 4th ed.; APS Press: St. Paul, MN, USA, 1998. [Google Scholar]
- Homepage of MycoBank. Available online: http://www.mycobank.com (accessed on 1 December 2010).
- White, TJ; Bruns, T; Lee, S; Taylor, JW. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, MA, Gelfand, DH, Sninsky, JJ, White, TJ, Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Altschul, SF; Gish, W; Miller, W; Myers, EW; Lipman, DJ. Basic local alignment search tool. J Mol Biol 1990, 215, 403–410. [Google Scholar]
- Larkin, MA; Blackshields, G; Brown, NP; Chenna, R; McGettigan, PA; McWilliam, H; Valentin, F; Wallace, IM; Wilm, A; Lopez, R; Thompson, JD; Gibson, TJ; Higgins, DG. ClustalW and ClustalX version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar]
- Hall, TA. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser. 1999, 41, pp. 95–98. Available online: http://www.mbio.ncsu.edu/BioEdit/bioedit.html (accessed on 15 April 2010).
- Ronquist, F; Huelsenbeck, JP. MRBAYES 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar]
- Huelsenbeck, JP; Ronquist, F. Bayesian analysis of molecular evolution using MrBayes. In Statistical Methods in Molecular Evolution; Nielsen, R, Ed.; Springer-Verlag: New York, NY, USA, 2005; pp. 183–232. [Google Scholar]
- Page, RDM. Treeview: An application to display phylogenetic trees on personal computers. Comput Appl Biosci 1996, 12, 357–358. [Google Scholar]
- Wickerham, LJ. Taxonomy of yeasts. US Dep Agr Tech Bull 1951, 29, 1–6. [Google Scholar]
- Schneemann, I; Kajahn, I; Ohlendorf, B; Zinecker, H; Erhard, A; Nagel, K; Wiese, J; Imhoff, JF. Mayamycin, a cytotoxic polyketide from a marine Streptomyces strain isolated from the marine sponge Halichondria panicea. J Nat Prod 2010, 73, 1309–1312. [Google Scholar]
- Helaly, S; Schneider, K; Nachtigall, J; Vikineswary, S; Tan, GY; Zinecker, H; Imhoff, JF; Süssmuth, RD; Fiedler, HP. Gombapyrones, new alpha-pyrone metabolites produced by Streptomyces griseoruber Acta 3662. J. Antibiot 2009, 62, 445–452. [Google Scholar]



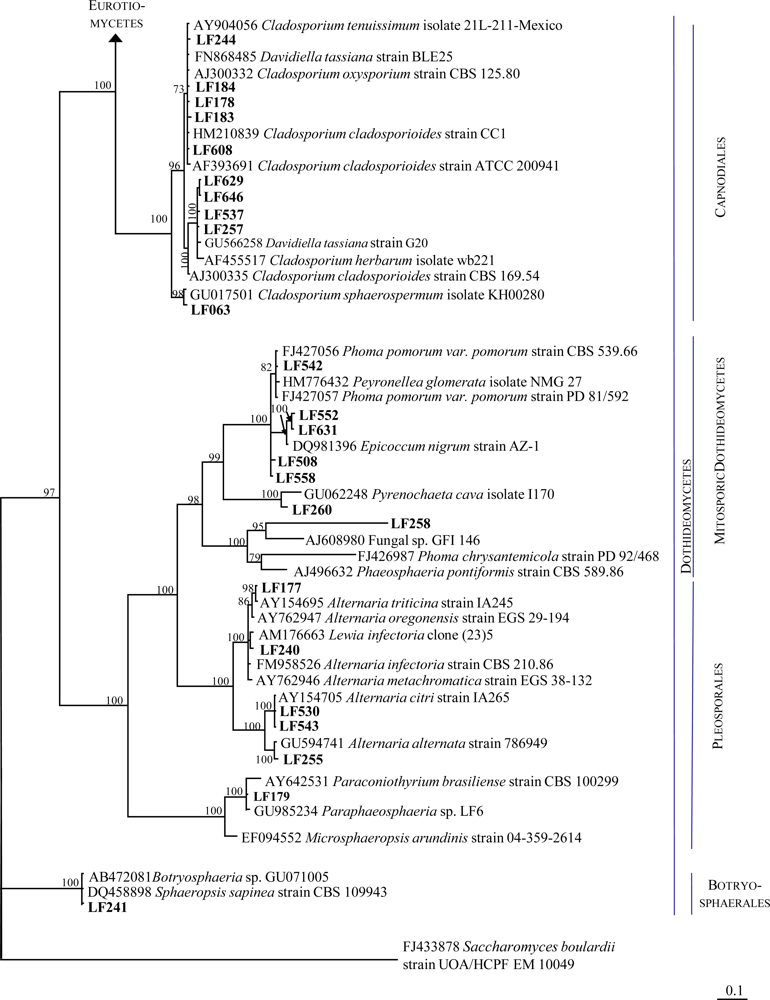
| Strain | Morphological identification | Seq. length (nt) | Next related cultivated strain (BLAST) | Acc. No. | Similarity (%) | Overlap (nt) |
|---|---|---|---|---|---|---|
| LF063 | Cladosporium sp. | 483 | Fungal sp. ARIZ AZ0920 Cladosporium sphaerospermum isolate KH00280 | HM123596.1 GU017501.1 | 99 99 | 482 482 |
| LF064 | Scopulariopsis murina | 473 | Ascomycota sp. 840 Phialemonium obovatum strain CBS 279.76 | GU934604.1 AB278187.1 | 92 89 | 360 340 |
| LF065 | Penicillium sp. | 546 | Penicillium glabrum strain 4AC2K | GU372904.1 | 99 | 545 |
| LF066 | Penicillium sp. | 551 | Penicillium chrysogenum strain JCM 22826 | AB479305.1 | 99 | 549 |
| LF073 | Aspergillus sp. | 533 | Aspergillus versicolor isolate UOA/HCPF 8709 | FJ878627.1 | 100 | 532 |
| LF177 | Alternaria sp. | 568 | Lewia infectoria strain IA310 | AY154718 | 99 | 561 |
| LF178 | Cladosporium sp. | 479 | Fungal endophyte sp. g6 Cladosporium cladosporioides strain CC1 | HM537022.1 HM210839.1 | 100 100 | 479 479 |
| LF179 | Mycelia sterilia | 559 | Fungal endophyte isolate 9137 Paraphaeosphaeria sp. LF6 | EF419991.1 GU985234.1 | 100 99 | 555 557 |
| LF183 | Cladosporium sp. | 523 | Dothideomycetes sp. 11366 Cladosporium cladosporioides isolate SLP001 | GQ153254.1 FJ932747.1 | 99 99 | 522 521 |
| LF184 | Cladosporium sp. | 475 | Fungal endophyte sp. g6 Cladosporium cladosporioides strain CC1 | HM537022.1 HM210839.1 | 100 100 | 475 475 |
| LF236 | Fusarium sp. | 512 | Fusarium sp. CPK3469 Gibberella intricans strain ATCC MYA-3861 | FJ827615.1 GU291255.1 | 99 99 | 511 511 |
| LF237 | Fusarium sp. | 503 | Fusarium sp. CPK3337 Fusarium equiseti strain NRRL 36478 | FJ827616.1 GQ505743.1 | 100 100 | 503 503 |
| LF238 | Fusarium sp. | 513 | Fusarium sp. CPK3469 Fusarium equiseti strain NRRL 36478 | FJ827615.1 GQ505743.1 | 100 100 | 513 513 |
| LF239 | Fusarium sp. | 509 | Fusarium sp. NRRL 45997 Fusarium equiseti strain NRRL 36478 | GQ505761.1 GQ505743.1 | 99 99 | 503 503 |
| LF240 | Mycelia sterilia | 528 | Lewia sp. B32C Lewia infectoria strain IA241 | EF432279.1 AY154692.1 | 99 99 | 525 525 |
| LF241 | Mycelia sterilia | 513 | Botryosphaeria sp. GU071005 Sphaeropsis sapinea strain CBS109943 | AB472081.1 DQ458898.1 | 100 100 | 512 512 |
| LF242 | Penicillium sp. | 538 | Penicillium brevicompactum isolate H66s1 | EF634441.1 | 99 | 537 |
| LF243 | Penicillium sp. | 532 | Penicillium virgatum strain IHB F 536 | HM461858.1 | 100 | 530 |
| LF244 | Cladosporium sp. | 516 | Fungal endophyte sp. g2 Davidiella tassiana strain BLE25 | HM537019.1 FN868485.1 | 99 99 | 516 516 |
| LF245 | Fusarium sp. | 494 | Fusarium sp. CPK3514 Fusarium equiseti strain NRRL 13402 | FJ840530.1 GQ505681.1 | 100 100 | 494 494 |
| LF246 | Volutella sp. | 548 | Volutella ciliata strain BBA 70047 | AJ301966.1 | 99 | 547 |
| LF247 | Fusarium sp. | 520 | Fusarium sp. LD-135 Fusarium equiseti strain NRRL 13402 | EU336989.1 GQ505681.1 | 99 99 | 509 504 |
| LF248 | Botrytis sp. | 504 | Fungal endophyte sp. g18 Botryotinia fuckeliana strain OnionBC-1 | HM537028.1 FJ169667.2 | 100 100 | 504 504 |
| LF249 | Penicillium sp. | 552 | Penicillium sp. BM Penicillium commune isolate HF1 | GU566211.1 GU183165.1 | 99 99 | 551 551 |
| LF250 | Penicillium sp. | 564 | Penicillium chrysogenum strain ACBF 003-2 | GQ241341.1 | 97 | 547 |
| LF251 | Penicillium sp | 550 | Penicillium sp. F6 Penicillium chrysogenum strain ACBF 003-2 | GU566250.1 GQ241341.1 | 100 100 | 550 550 |
| LF252 | Fusarium sp | 513 | Fusarium sp. NRRL 45997 Fusarium equiseti strain NRRL 36478 | GQ505761.1 GQ505743.1 | 99 99 | 511 511 |
| LF253 | Trichoderma sp. | 543 | Hypocrea lixii strain OY3207 | FJ571487.1 | 100 | 540 |
| LF254 | Clonostachys sp | 525 | Bionectria ochroleuca strain G11 | GU566253.1 | 100 | 524 |
| LF255 | Alternaria sp | 518 | Fungal endophyte sp. g76 Alternaria alternata strain 786949 | HM537053.1 GU594741.1 | 100 100 | 518 518 |
| LF256 | Botrytis sp. | 535 | Beauveria bassiana strain G61 | GU566276.1 | 99 | 533 |
| LF257 | Cladosporium sp. | 495 | Davidiella tassiana strain G20 | GU566258.1 | 100 | 495 |
| LF258 | Phoma sp. nov. | 538 | Fungal sp. GFI 146 Septoria arundinacea isolate BJDC06 | AJ608980.1 GU361965.1 | 93 91 | 470 460 |
| LF259 | Penicillium sp. | 522 | Penicillium brevicompactum strain: JCM 22849 | AB479306.1 | 100 | 522 |
| LF260 | Sphaeropsidales | 508 | Pyrenochaeta cava isolate olrim63 | AY354263.1 | 100 | 508 |
| LF491 | Aspergillus sp. | 555 | Petromyces alliaceus isolate NRRL 4181 | EF661556.1 | 99 | 555 |
| LF494 | Fusarium sp. | 469 | Fusarium sp. CB-3 | GU932675.1 | 100 | 469 |
| LF496 | Mycelia sterilia | 531 | Verticillium sp. TF17TTW | FJ948142.1 | 99 | 529 |
| LF501 | Aspergillus sp. | 514 | Aspergillus granulosus isolate NRRL 1932 | EF652430.1 | 100 | 514 |
| LF508 | Not identified | 501 | Phoma sp. W21 | GU045305.1 | 99 | 497 |
| LF509 | Fusarium sp. | 504 | Fusarium sp. CPK3514 | FJ840530.1 | 99 | 503 |
| LF510 | Fusarium sp. | 516 | Fusarium sp. FL-2010c isolate UASWS0396 | HQ166535.1 | 99 | 514 |
| LF514 | Trichoderma sp. | 546 | Trichoderma sp. TM9 | AB369508.1 | 100 | 546 |
| LF526 | Eurotium sp. | 486 | Eurotium sp. FZ Eurotium chevalieri isolate UPM A11 | HQ148160.1 HM152566.1 | 100 100 | 486 486 |
| LF530 | Alternaria sp. | 522 | Alternaria sp. 7 HF-2010 | HQ380788.1 | 100 | 522 |
| LF534 | Penicillium sp. | 540 | Penicillium roseopurpureum strain E2 | GU566239.1 | 99 | 536 |
| LF535 | Acremonium sp. | 525 | Acremonium sp. FSU2858 Lecanicillium lecanii strain V56 | AY633563.1 DQ007047.1 | 99 99 | 523 510 |
| LF537 | Cladosporium sp. | 503 | Cladosporium cladosporioides strain F12 | HQ380766.1 | 100 | 503 |
| LF538 | Mucor hiernalis | 598 | Mucor hiemalis isolate UASWS0442 | HQ166553.1 | 100 | 598 |
| LF540 | Not identified | 578 | Hypocrea lixii isolate FZ1302 | HQ259308.1 | 99 | 575 |
| LF542 | Mycelia sterilia | 458 | Peyronellaea glomerata isolate NMG_27 Phoma pomorum var. pomorum strain CBS 539.66 | HM776432 FJ427056.1 | 99 99 | 457 457 |
| LF543 | Alternaria sp. | 522 | Alternaria citri strain IA265 | AY154705.1 | 100 | 522 |
| LF547 | Aspergillus sp. | 539 | Aspergillus minutus isolate NRRL 4876 | EF652481.1 | 98 | 529 |
| LF550 | Mycelia sterilia | 524 | Bartalinia robillardoides CBS:122686 Ellurema sp. 42-3 | EU552102.1 AY148442.1 | 99 99 | 522 514 |
| LF552 | Epicoccum nigrum | 506 | Epicoccum nigrum strain GrS7 | FJ904918.1 | 99 | 503 |
| LF553 | Aspergillus sp. | 528 | Aspergillus sp. Da91 | HM991178.1 | 100 | 528 |
| LF554 | Aspergillus sp. | 525 | Aspergillus sp. Da91 | HM991178.1 | 100 | 525 |
| LF557 | Mycelia sterilia | 528 | Fusarium sp. FL-2010f Fusarium oxysporum strain TS08-137-1-1 | HQ166539.1 AB470850.1 | 99 | 522 |
| LF558 | Not identified | 498 | Phoma sp. W21 | GU045305.1 | 100 | 498 |
| LF562 | Tritirachium sp. | 504 | Tritirachium sp. F13 | EU497949.1 | 99 | 498 |
| LF563 | Trichoderma sp. | 537 | Hypocrea lixii isolate DLEN2008014 | HQ149778.1 | 100 | 537 |
| LF576 | Clonostachys sp. | 504 | Bionectria cf. ochroleuca CBS 113336 | EU552110.1 | 99 | 503 |
| LF577 | Penicillium sp. | 538 | Penicillium brevicompactum isolate NMG_25 | HM776430.1 | 99 | 534 |
| LF580 * | Scopulariopsis brevicaulis | 916 | Scopulariopsis brevicaulis strain NCPF 2177 | AY083220.1 | 99 | 686 |
| LF581 | Fusarium sp. | 504 | Fusarium sp. NRRL 45996 | GQ505760.1 | 99 | 502 |
| LF584 | Aspergillus sp. | 543 | Aspergillus sp. N13 | GQ169453.1 | 99 | 542 |
| LF590 | Penicillium sp. | 522 | Penicillium citreonigrum strain Gr155 | FJ904848.1 | 100 | 522 |
| LF592 | Paecilomyces sp. | 544 | Fungal endophyte sp. P1201A Paecilomyces lilacinus strain CG 271 | EU977225.1 EU553303.1 | 99 98 | 541 516 |
| LF594 | Mycelia sterilia | 531 | Fusarium sp. FL-2010c Fusarium acuminatum strain NRRL 54217 | HQ166535.1 HM068325.1 | 99 99 | 527 527 |
| LF596 | Penicillium sp. | 529 | Penicillium sp. FF24 Penicillium canescens strain QLF83 | FJ379805.1 FJ025212.1 | 100 100 | 529 529 |
| LF607 | Penicillium sp. | 532 | Penicillium sp. 17-M-1 Penicillium sclerotiorum strain SK6RN3M | EU076929.1 EU807940.1 | 99 99 | 523 520 |
| LF608 | Cladosporium sp. | 495 | Fungal sp. mh2981.6 Cladosporium cladosporioides strain CC1 | GQ996077.1 HM210839.1 | 100 100 | 495 495 |
| LF610 | Clonostachys sp. | 528 | Fungal sp. mh2053.3 Bionectria ochroleuca isolate Rd0801 | GQ996069.1 HQ115728.1 | 99 99 | 524 524 |
| LF626 | Mycelia sterilia | 588 | Trichoderma cerinum isolate C.P.K. 3619 | GU111565.1 | 99 | 586 |
| LF627 | Aspergillus sp. | 509 | Aspergillus sp. 4-1 | HQ316558.1 | 100 | 509 |
| LF629 | Mycelia sterilia | 510 | Cladosporium cladosporioides strain F12 | HQ380766.1 | 99 | 509 |
| LF630 | Penicillium sp. | 518 | Penicillium brevicompactum isolate H66s1 | EF634441.1 | 100 | 518 |
| LF631 | Epicoccum nigrum | 514 | Epicoccum nigrum strain AZ-1 | DQ981396.1 | 99 | 512 |
| LF634 | Aspergillus sp. | 581 | Aspergillus terreus isolate UOA/HCPF 10213 | GQ461911.1 | 99 | 579 |
| LF644 | Clonostachys sp | 528 | Bionectria rossmaniae strain CBS 211.93 | AF210665.1 | 99 | 521 |
| LF646 | Mycelia sterilia | 510 | Cladosporium cladosporioides strain F12 | HQ380766.1 | 99 | 509 |
| Genus | Strain | Compound | Reported from a | Bioactivity a,b | Method of dereplication |
|---|---|---|---|---|---|
| Alternaria | LF177 | infectopyrone | Alternaria infectoria, Leptosphaeria maculans/Phoma lingum | UV, MS, NMR | |
| phomenin A & B | Phoma tracheiphila, Leptosphaeria maculans/Phoma lingum, Ercolaria funera | phytotoxin | UV, MS, NMR | ||
| Aspergillus | LF627 | sterigmatocystin | Aspergillus versicolor, Chaetomium | mycotoxin [32] | UV, MS |
| notoamid D | Aspergillus sp. | UV, MS | |||
| stephacidin A | Aspergillus ochraceus | cytotoxic | UV, MS, NMR | ||
| LF547 | cinereain | Botrytis cinerea | plant growth regulator, phytotoxin | UV, MS, NMR | |
| (2′E,4′E,6′E)-6-(1′-carboxyocta-2′,4′,6′-trien)-9-hydroxydrim-7-ene-11,12-olide | Aspergillus ustus | UV, MS, NMR | |||
| (2′E,4′E,6′E)-6-(1′-carboxyocta-2′,4′,6′-trien)-9-hydroxydrim-7-ene-11-al | Aspergillus ustus | UV, MS, NMR | |||
| compound A | hit in Scifinder [33], but no publication available | UV, MS, NMR | |||
| compound B | no hit in database | UV, MS, NMR | |||
| LF553 | sydonic acid | Aspergillus sydowii | weakly antibacterial [34] | UV, MS, NMR | |
| hydroxysydonic acid | Aspergillus sydowii | UV, MS | |||
| LF584 | WIN-6 6306 | Aspergillus flavipes | substance P antagonist, inhibition of HIV-1 integrase [35] | UV, MS | |
| aspochalasines | Aspergillus flavipes and other Aspergillus sp. | antibiotic, moderately cytotoxic [36] | UV, MS, NMR | ||
| Aspergillus/ Petromyces | LF491 | isokotanin A–C | Aspergillus alliaceus and Petromyces alliaceus | moderate antiinsectan activities [37] | UV, MS, NMR |
| 14-(N,N-Dimethyl-l-leucinyloxy)paspalinine | Aspergillus alliaceus | potassium channel antagonist [38] | UV, MS, NMR | ||
| nominine or a similar indoloditerpene | Aspergillus nomius, Aspergillus flavus, Petromyces alliaceus | insecticidal properties | UV, MS, NMR | ||
| Phoma | LF258 | monocerin | Helminthosporium monoceras, Fusarium larvarum, Dreschlera ravenelii, Exserohilum rostratum, Readeriella mirabilis | antifungal, insecticidal and phytotoxic properties | UV, MS |
| intermediate in the bio-synthesis of monocerin | Dreschlera ravenelii | UV, MS, NMR | |||
| evernin- or isoeverninaldehyde | Guignardia laricina | weak phytotoxin | UV, MS, NMR | ||
| Epicoccum | LF552 | epicoccamide | Epicoccum purparescens and other Epicoccum sp., Aurelia aurita | UV, MS | |
| orevactaene | Epicoccum nigrum | binding inhibitor of HIV-1 rev protein to Rev response element (RRE) | UV, MS | ||
| Eurotium | LF526 | echinulin | Eurotium repens, Aspergillus amstelodami, Aspergillus echinulatus, Aspergillus glaucus | experimentally hepatic and pulmonary effects | UV, MS |
| neoechinulines | Aspergillus amstelodami | antioxidative activity | UV, MS | ||
| auroglaucines and flavoglaucine | Aspergillus and Eurotium spp. | mycotoxin, shows antineo-plastic properties [39] | UV, MS | ||
| Fusarium | LF236 | equisetin | Fusarium equiseti and Fusarium heterosporum | antibacterial activity, inhi-bition of HIV-1 integrase | UV, MS, NMR |
| LF238 | equisetin | Fusarium equiseti and Fusarium heterosporum | antibacterial activity, inhi-bition of HIV-1 integrase | UV, MS, NMR | |
| fusarins | Fusarium moniliforme | mutagenic [40] | UV, MS | ||
| LF594 | enniatine | various Fusarium sp. | ionophore, insecticidal, ACAT inhibition, GABA receptor binding | UV, MS | |
| Paecilomyces | LF592 | leucinostatins | Paecilomyces lilacinus and other Paecilomyces sp. | active against Gram-positive bacteria and fungi | UV, MS |
| Penicillium | LF066 | compound C | no hit in database | UV, MS, NMR | |
| meleagrin | Penicillium meleagrinum and Penicillium chrysogenum | structurally similar to tremorgenic mycotoxins | UV, MS, NMR | ||
| roquefortin C | Penicillium roquefortii and other Penicillium sp. | neurotoxin | UV, MS, NMR | ||
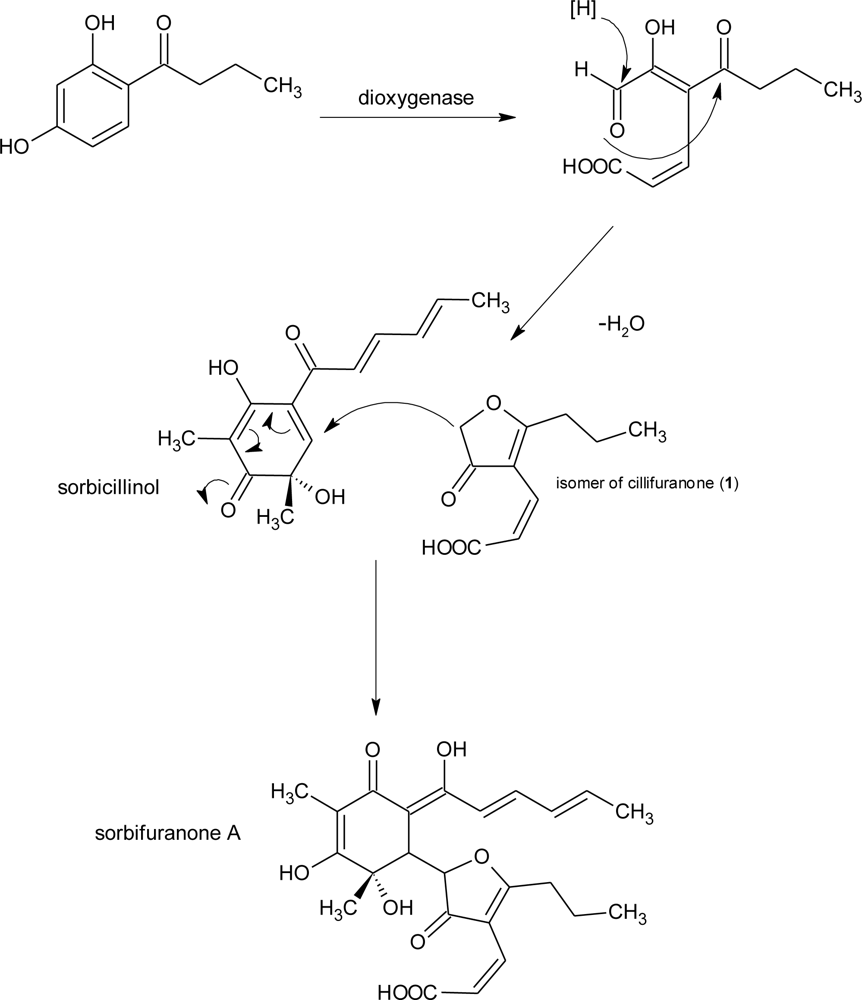
| sorbifuranones A–C | Penicillium chrysogenum [31] | UV, MS, NMR | |||
| 2′,3′-dihydrosorbicillin | Penicillium notatum, Verticillium intertextum | weakly antibacterial [41] | UV, MS | ||
| bisvertonolone | Penicillium chrysogenum, Verticillium intertextum, Acremonium strictum, Trichoderma longibrachiatum | β-1,6glucan biosynthesis inhibitor, antioxidative, inducer of hyphal malformation in fungi | UV, MS | ||
| ergochromes | Aspergillus ochraceus, Claviceps purpurea, Aspergillus aculeatus, Gliocladium sp., Penicillium oxalicum, Phoma terrestris, Pyrenochaeta terrestris | teratogenic effects | UV, MS | ||
| LF259 | mycophenolic acid | Penicillium brevicompactum and other Penicillium sp. | antineoplastig, antiviral immunosuppressant properties, useful in treating psoriasis and leishmaniasis, | UV, MS | |
| LF590 | citreoviridins | Penicillium citreoviride, Penicillium toxicarium, Penicillium ochrosalmoneum, Aspergillus terreus | neurotoxic | UV, MS, NMR | |
| territrem B | Penicillium sp. and Aspergillus terreus | inhibitor of acetylcholinesterase | UV, MS | ||
| LF607 | sclerotiorin | Penicillium sclerotiorum and Penicillium multicolor | inhibits cholesterin ester transfer protein activity | UV, MS | |
| sclerotioramine | UV, MS, NMR | ||||
| compound D | no hit in database | UV, MS, NMR | |||
| Penicillium | LF596 | griseofulvin | Penicillium griseofulvum and other Penicillium sp. | antifungal, possible human carcinogen | UV, MS, NMR |
| tryptoquivalin | Aspergillus clavatus | tremorgenic toxin | UV, MS | ||
| nortryptoquivalin | Aspergillus clavatus and Aspergillus fumigatus | tremorgenic toxin | UV, MS | ||
| fiscalins A and C | Neosartorya fischeri | substance P inhibitor, neurokinin binding inhibitor | UV, MS, NMR | ||
| Scopulariopsis | LF580 | scopularide A and B | Scopulariopsis brevicaulis | antiproliferative [25,26] | UV, MS, NMR |
| Clonostachys | LF254 | T-988B | Tilachlidium sp. | cytotoxic | UV, MS, NMR |
| bionectin B | Bionectria byssicola | antibacterial (MRSA) | UV, MS, NMR | ||
| verticillin C | Verticillium sp. | antibiotic | UV, MS |
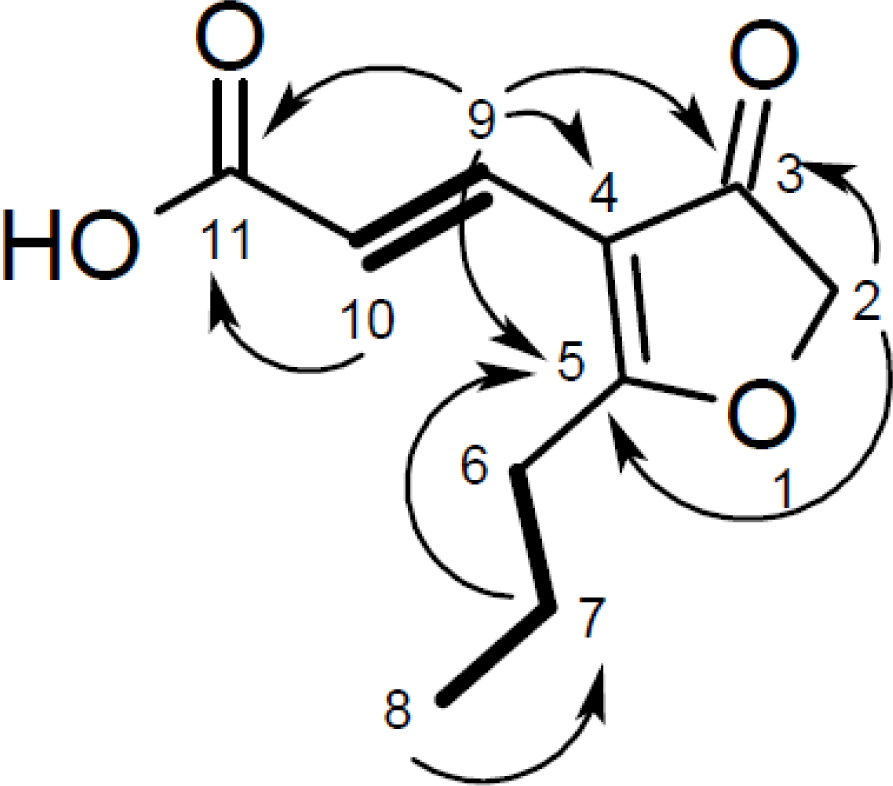
| Cillifuranone (1) | ||||
|---|---|---|---|---|
| Position | δC, mult. | δH, (J in Hz) | COSY | HMBC |
| 1 | ||||
| 2 | 76.3, CH2 | 4.67, s | 6 | 3, 5, 6, 7 |
| 3 | 201.7, C | |||
| 4 | 112.6, C | |||
| 5 | 196.7, C | |||
| 6 | 31.6, CH2 | 2.76, t (7.5) | 2, 7 | 4, 5, 7, 8 |
| 7 | 20.9, CH2 | 1.76, sext. (7.5) | 6, 8 | 5, 6, 8 |
| 8 | 14.0, CH3 | 1.03, t (7.5) | 7 | 6, 7 |
| 9 | 133.0, CH | 7.32, d (16.0) | 10 | 2, 3, 4, 5, 10, 11 |
| 10 | 118.5, CH | 6.83, d (16.0) | 9 | 3, 4, 5, 9, 11 |
| 11 | 170.9, C | |||
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wiese, J.; Ohlendorf, B.; Blümel, M.; Schmaljohann, R.; Imhoff, J.F. Phylogenetic Identification of Fungi Isolated from the Marine Sponge Tethya aurantium and Identification of Their Secondary Metabolites. Mar. Drugs 2011, 9, 561-585. https://doi.org/10.3390/md9040561
Wiese J, Ohlendorf B, Blümel M, Schmaljohann R, Imhoff JF. Phylogenetic Identification of Fungi Isolated from the Marine Sponge Tethya aurantium and Identification of Their Secondary Metabolites. Marine Drugs. 2011; 9(4):561-585. https://doi.org/10.3390/md9040561
Chicago/Turabian StyleWiese, Jutta, Birgit Ohlendorf, Martina Blümel, Rolf Schmaljohann, and Johannes F. Imhoff. 2011. "Phylogenetic Identification of Fungi Isolated from the Marine Sponge Tethya aurantium and Identification of Their Secondary Metabolites" Marine Drugs 9, no. 4: 561-585. https://doi.org/10.3390/md9040561




