Pathophysiological Effects of Synthetic Derivatives of Polymeric Alkylpyridinium Salts from the Marine Sponge, Reniera sarai
Abstract
:1. Introduction
2. Synthetic Analogues of Polymeric Alkylpyridinium Salts
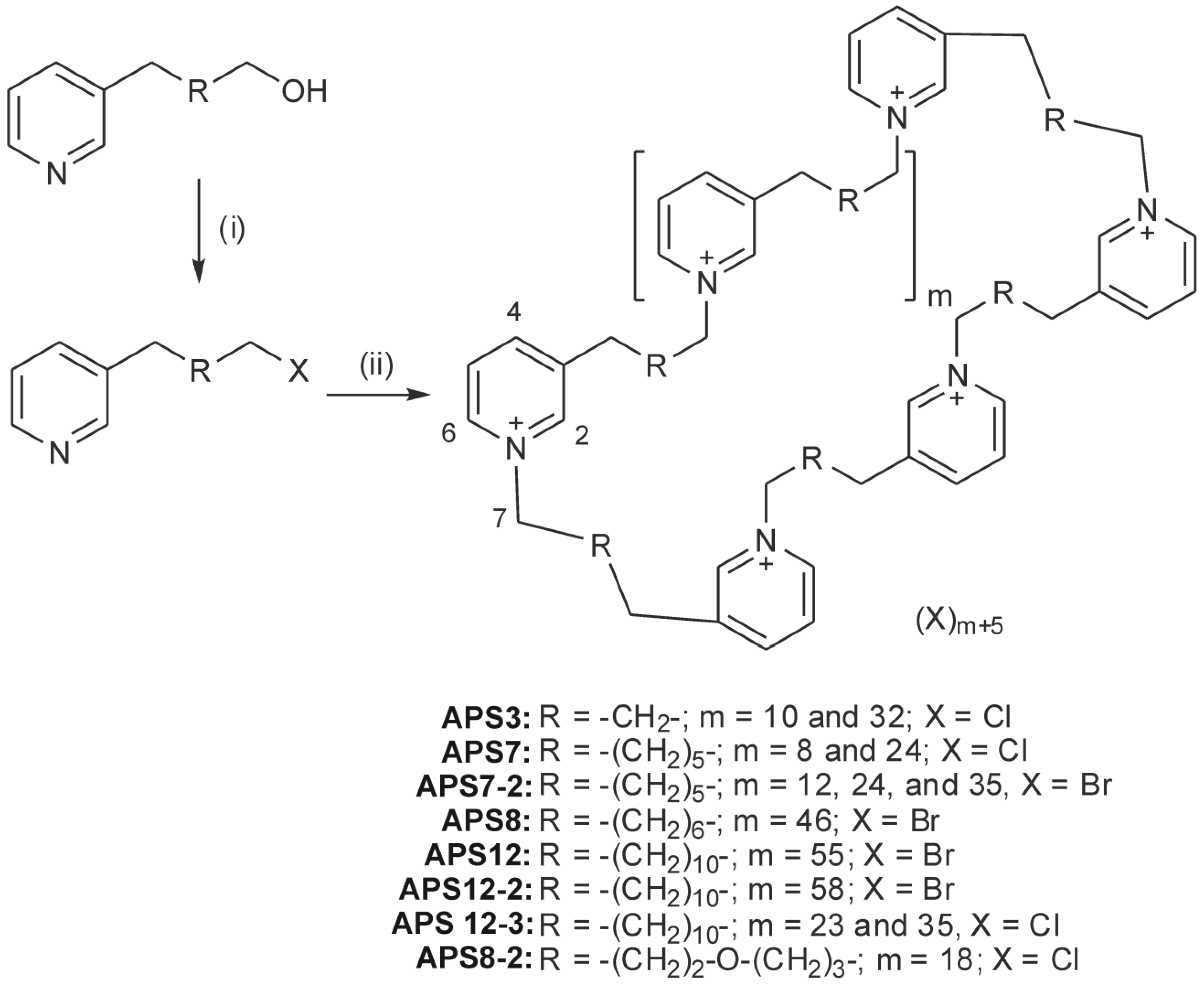
sAPS Synthesis
| Compound | No. of Alky lC-atoms | No. of Polymers and Molar Ratio | Molecular Weight (kDa) | Degree of Polymerization | Counter Ion | Reference |
|---|---|---|---|---|---|---|
| Poly-APS | 8 | 1 | 5.52 | 29 | Cl− | [6] |
| APS3 | 3 | 2 (9:1) | 1.46 (1.2/3.8) | 10 and 32 | Cl− | [29] |
| APS7 | 7 | 2 (2:1) | 2.33 (1.4/4.2) | 8 and 24 | Cl− | [29] |
| APS8 | 8 | 1 | 11.9 | 63 | Br− | [28] |
| APS12 | 12 | 1 | 12.5 | 51 | Br− | [28] |
| APS12-2 | 12 | 1 | 14.7 | 60 | Br− | [28] |
3. Biological Activities of sAPS
3.1. Hemolytic and Antimicrobial Activity
3.2. Effects of sAPS on Acetylcholinesterase
3.3. Antitumor Activity of sAPS
| Compound | AChE Inhibition—Ki (nM) * | Hemolysis (s−1 at 500 nM) ** | IC50 for NSCLC (μM) *** |
|---|---|---|---|
| Poly-APS | irreversible inhibition | 0.05 | 4.41 |
| APS3 | 85 | 0 | 3000 |
| APS7 | 10 | 0.1 | 480 |
| APS8 | 1.875 | 2.6 | 478 |
| APS12-2 | 0.036 | 5.0 | 470 |
4. Toxicity of APS12-2 and APS3
4.1. In Vivo Effects of APS12-2 and APS3
| Measured Parameters | APS12-2 * | APS3 ** |
|---|---|---|
| LD50 (mice) | 11.5 mg/kg | 7.25 mg/kg |
| ECG (rats) |
|
|
| Arterial blood pressure | Steep decrease immediately after application | First a decrease, then an increase above base-line value |
| Breathing | Respiratory arrest soon after application | No effect |
| Biochemical parameters | Statistically significant increase in K+ level (10.44 ± 0.44 mM) | Statistically significant increase in K+ level (5.66 ± 0.37 mM) |
| Muscle contraction | No effect up to 8.6 mg/kg | ID50 = 37.25 mg/kg |
4.2. In Vitro Physiological and Pharmacological Effects of APS12-2 and APS3
| Measured Parameters | APS12-2 * | APS3 ** | |||
|---|---|---|---|---|---|
| Effect | IC50 | Effect | IC50 | ||
| Skeletal muscle contraction | Nerve-evoked stimulation | Inhibition | 0.74 μM | Inhibition | 20.3 μM |
| Direct stimulation | No effect up to 2.72 μM | N/A | No effect up to 20.55 μM | N/A | |
| Pharmacological effect | atropine | No effect up to 80 μM | N/A | No effect up to 80 μM | N/A |
| neostigmine | No effect up to 1 μM | N/A | No effect up to 1 μM | N/A | |
| 3,4-DAP | Stops muscle contraction blockade (300 μM) | N/A | Stops muscle contraction blockade (300 μM) | N/A | |
| Effect on | RP | No effect up to 3.40 μM | N/A | No effect up to 68.49 μM | N/A |
| MEPP | Amplitude decrease, MEPP disappear above 0.68 μM | N/A | Amplitude decrease, MEPP disappear above 6.85 μM | N/A | |
| EPP | Amplitude decrease | 0.36 μM | Amplitude decrease | 7.28 μM | |
| nAChRs inhibition | Inhibition | 0.0005 μM | Inhibition | 0.19 μM | |
| Effect on coronary rings *** | Contraction (4.1–13.6 μM) | N/A | No effect up to 137 μM | N/A | |
5. Conclusion
Acknowledgements
Conflicts of Interest
References
- Almeida, A.M.P.; Berlinck, R.G.S. Alcalóides alquilpiridínicos de esponjas marinhas. Quim. Nova 1997, 20, 170–185. [Google Scholar] [CrossRef]
- Sepčić, K. Bioactive alkylpyridinium compounds from marine sponges. Toxin Rev. 2000, 19, 139–160. [Google Scholar] [CrossRef]
- Sepčić, K.; Turk, T. 3-alkylpyridinium compounds as potential non-toxic antifouling agents. In Antifouling Compounds, Progress in Molecular and Subcellular Biology, Subseries Marine Molecular Biotechnology; Fusetani, N., Clare, A.C., Eds.; Springer-Verlag: Berlin, Germany, 2006; pp. 105–124. [Google Scholar]
- Turk, T.; Sepčić, K.; Mancini, I.; Guella, G. 3-Aklypyridinium and 3-alkylpyridine compounds from marine sponges, their synthesis, biological activities, and potential use. In Studies in Natural Products Chemistry; Rahman, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 355–397. [Google Scholar]
- Sepčić, K.; Guella, G.; Mancini, I.; Pietra, F.; Dalla Serra, M.; Menestrina, G.; Tubbs, K.; Maček, P.; Turk, T. Characterization of anticholinesterase-active 3-alkylpyridinium polymers from the marine sponge Reniera sarai in aqueous solutions. J. Nat. Prod. 1997, 60, 991–996. [Google Scholar] [CrossRef]
- Sepčić, K.; Batista, U.; Vacelet, J.; Maček, P.; Turk, T. Biological activities of aqueous extracts from marine sponges and cytotoxic effects of 3-alkylpyridinium polymers from Reniera sarai. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1997, 117, 47–53. [Google Scholar] [CrossRef]
- Sepčić, K.; Muševič, I.; Lahajnar, G.; Turk, T.; Maček, P. AFM imaging of surface adsorbed polymeric 3-alkylpyridinium salts from the marine sponge Reniera sarai. Int. J. Biol. Macromol. 1999, 26, 353–356. [Google Scholar] [CrossRef]
- Grandič, M.; Sepčić, K.; Turk, T.; Juntes, P.; Frangež, R. In vivo toxic and lethal cardiovascular effects of a synthetic polymeric 1,3-dodecylpyridinium salt in rodents. Toxicol. Appl. Pharmacol. 2011, 255, 86–93. [Google Scholar] [CrossRef]
- Faimali, M.; Sepčić, K.; Turk, T.; Geraci, S. Non-toxic antifouling activity of polymeric 3-alkylpyridium salts from the Mediterranean sponge Reniera sarai (Pulitzer Finali). Biofouling 2003, 19, 47–56. [Google Scholar] [CrossRef]
- Garaventa, F.; Faimali, M.; Sepčić, K.; Geraci, S. Laboratory analysis of antimicrofouling activity of Poly-APS extracted from Reniera sarai (Porifera: Demospongiae). Biol. Mar. Mediterr. 2003, 10, 565–567. [Google Scholar]
- Chelossi, E.; Mancini, I.; Sepčić, K.; Turk, T.; Faimali, M. Comparative antibacterial activity of polymeric 3-alkylpyridinium salts isolated from the Mediterranean sponge Reniera sarai and their synthetic analogues. Biomol. Eng. 2006, 23, 317–323. [Google Scholar] [CrossRef]
- Eleršek, T.; Kosi, G.; Turk, T.; Pohleven, F.; Sepčić, K. Influence of polymeric 3-alkylpyridinium salts from the marine sponge Reniera sarai on the growth of algae and wood decay fungi. Biofouling 2008, 24, 137–143. [Google Scholar] [CrossRef]
- Sepčić, K.; Marcel, V.; Klaebe, A.; Turk, T.; Šuput, D.; Fournier, D. Inhibition of acetylcholinesterase by an alkylpyridinium polymer from the marine sponge, Reniera sarai. Biochim. Biophys. Acta 1998, 1387, 217–225. [Google Scholar] [CrossRef]
- Sepčić, K.; Poklar, N.; Vesnaver, G.; Fournier, D.; Turk, T.; Maček, P. Interaction of 3-alkylpyridinium polymers from the sea sponge Reniera sarai with insect acetylcholinesterase. J. Protein Chem. 1999, 18, 251–257. [Google Scholar] [CrossRef]
- Garaventa, F.; Piazza, V.; Zovko, A.; Turk, T.; Chelossi, E.; Falugi, C.; Aluigi, M.G.; Angelini, C.; Trombino, S.; Gallus, L.; et al. Multiple functions of the cholinesterase inhibiting polyalkylpyridinium salts extracted from the marine sponge, Haliclona sarai. WSEAS Trans. Biol. Biomed. 2010, 7, 103–113. [Google Scholar]
- McClelland, D.; Evans, R.M.; Abidin, I.; Sharma, S.; Choudhry, F.Z.; Jaspars, M.; Sepčić, K.; Scott, R.H. Irreversible and reversible pore formation by polymeric alkylpyridinium salts (poly-APS) from the sponge Reniera sarai. Br. J. Pharmacol. 2003, 139, 1399–1408. [Google Scholar] [CrossRef]
- Koss, D.; Hindley, K.P.; David, K.C.; Mancini, I.; Guella, G.; Sepčić, K.; Turk, T.; Rebolj, K.; Riedel, G.; Platt, B.; et al. A comparative study of the actions of alkylpyridinium salts from a marine sponge and related synthetic compounds in rat cultured hippocampal neurons. BMC Pharmacol. 2007, 7. [Google Scholar] [CrossRef]
- Tucker, S.J.; McClelland, D.; Jaspars, M.; Sepčić, K.; MacEwan, D.J.; Scott, R.H. The influence of alkyl pyridinium sponge toxins on membrane properties, cytotoxicity, transfection and protein expression in mammalian cells. Biochim. Biophys. Acta 2003, 1614, 171–181. [Google Scholar] [CrossRef]
- Scott, R.H.; Tucker, S.J.; Evans, R.M.; MacEwan, D.J.; Jaspars, M.; Sepčić, K. Cell membrane permeabilisation by alkylpyridinium sponge toxin preparations and their utilisation as novel transfection reagents. In Proceedings of Neurotox’03, Neurotoxicological Targets from Functional Genomics & Proteomics, University of Nottingham, Nottingham, UK, 1–4 September 2003; Beadle, D.J., Mellor, I.R., Usherwood, P.N.R., Eds.; Society of Chemical Industry (SCI): London, UK, 2004; pp. 51–58. [Google Scholar]
- McLaggan, D.; Adjimatera, N.; Sepčić, K.; Jaspars, M.; MacEwan, D.J.; Blagbrough, I.S.; Scott, R.H. Pore forming polyalkylpyridinium salts from marine sponges versus synthetic lipofection systems: Distinct tools for intracellular delivery of cDNA and siRNA. BMC Biotechnol. 2006, 6. [Google Scholar] [CrossRef]
- Catassi, A.; Servent, D.; Paleari, L.; Cesario, A.; Russo, P. Multiple roles of nicotine on cell proliferation and inhibition of apoptosis: implication on lung carcinogenesis. Mutat. Res. 2008, 659, 221–231. [Google Scholar] [CrossRef]
- Egleton, R.D.; Brown, K.C.; Dasgupta, P. Nicotinic acetylcholine receptors in cancer: Multiple roles in proliferation and inhibition of apoptosis. Trends Pharmacol. Sci. 2008, 29, 151–158. [Google Scholar] [CrossRef]
- Paleari, L.; Trombino, S.; Falugi, C.; Gallus, L.; Carlone, S.; Angelini, C.; Sepčić, K.; Turk, T.; Faimali, M.; Noonan, D.M.; et al. Marine sponge-derived polymeric alkylpyridinium salts as a novel tumor chemotherapeutic targeting the cholinergic system in lung tumors. Int. J. Oncol. 2006, 29, 1381–1388. [Google Scholar]
- Bunc, M.; Strupi-Šuput, J.; Vodovnik, A.; Šuput, D. Toxic effects of head-to-tail 3-alkylpiridinium polymers isolated from the marine sponge Raniera sarai in rat. Toxicon 2002, 40, 843–849. [Google Scholar]
- Turk, T.; Frangež, R.; Sepčić, K. Mechanisms of toxicity of 3-alkylpyridinium polymers from marine sponge Reniera sarai. Mar. Drugs 2007, 5, 157–167. [Google Scholar] [CrossRef]
- Lunder, M.; Drevenšek, G.; Hawlina, S.; Sepčić, K.; Ziberna, L. Cardiovascular effects induced by polymeric 3-alkylpyridinium salts from the marine sponge Reniera sarai. Toxicon 2012, 60, 1041–1048. [Google Scholar] [CrossRef]
- Mancini, I.; Sicurelli, A.; Guella, G.; Turk, T.; Maček, P.; Sepčić, K. Synthesis and bioactivity of linear oligomers related to polymeric alkylpyridinium metabolites from the Mediterranean sponge Reniera sarai. Org. Biomol. Chem. 2004, 2, 1368–1375. [Google Scholar] [CrossRef]
- Houssen, W.E.; Lu, Z.; Edrada-Ebel, R.A.; Chatzi, C.; Tucker, S.J.; Sepčić, K.; Turk, T.; Zovko, A.; Shen, S.; Mancini, I.; et al. Chemical synthesis and biological activities of 3-alkyl pyridinium polymeric analogues of marine toxins. J. Chem. Biol. 2010, 3, 113–125. [Google Scholar] [CrossRef]
- Zovko, A.; Vaukner Gabrič, M.; Sepčić, K.; Pohleven, F.; Jaklič, D.; Gunde-Cimerman, N.; Lu, Z.; Edrada-Ebel, R.; Houssen, W.E.; Mancini, I.; et al. Antifungal and antibacterial activity of 3-alkylpyridinium polymeric analogues of marine toxins. Int. Biodeterior. Biodegrad. 2012, 68, 71–77. [Google Scholar] [CrossRef]
- Davies-Coleman, M.T.; Faulkner, D.J.; Dubowchik, G.M.; Roth, G.P.; Polson, C.; Fairchild, C. A new EGF-active polymeric pyridinium alkaloid from the sponge Callyspongia fibrosa. J. Org. Chem. 1993, 58, 5925–5930. [Google Scholar] [CrossRef]
- Grandič, M.; Zovko, A.; Frangež, R.; Turk, T.; Sepčić, K. Binding and permeabilization of lipid bilayers by natural and synthetic 3-alkylpyridinium polymers. Bioorg. Med. Chem. 2012, 20, 1659–1664. [Google Scholar] [CrossRef]
- Kappe, C.O. Controlled microwave heating in modern organic synthesis. Angew. Chem. Int. Ed. 2004, 43, 6250–6284. [Google Scholar] [CrossRef]
- Malovrh, P.; Sepčić, K.; Turk, T.; Maček, P. Characterization of hemolytic activity of 3-alkylpyridinium polymers from the marine sponge Reniera sarai. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1999, 124, 221–226. [Google Scholar] [CrossRef]
- Kondo, T.; Tomizawa, M. Mechanism of hemolysis by cationic surface-active agents. J. Pharm. Sci. 1969, 58, 1378–1381. [Google Scholar] [CrossRef]
- Zaslavsky, B.Y.; Ossipov, N.N.; Rogozhin, S.V. Action of surface-active substances on biological membranes III. Comparison of hemolytic activity of ionic and nonionic surfactants. Biochim. Biophys. Acta 1978, 510, 151–159. [Google Scholar]
- Faimali, M.; Garaventa, F.; Mancini, I.; Sicurelli, A.; Guella, G.; Piazza, V.; Greco, G. Antisettlement activity of synthetic analogues of polymeric 3-alkylpyridinium salts isolated from the sponge Reniera sarai. Biofouling 2005, 21, 49–57. [Google Scholar] [CrossRef]
- Piazza, V.; Dragić, I.; Sepčić, K.; Faimali, M.; Garaventa, F.; Turk, T.; Berne, S. Antifouling activity of synthetic alkylpyridinium polymers using the barnacle model. Mar. Drugs. 2014, 12, 1959–1976. [Google Scholar] [CrossRef]
- Quinn, D.M. Acetylcholinesterase: Enzyme structure, reaction dynamics, and virtual transitionstates. Chem. Rev. 1987, 87, 955–979. [Google Scholar] [CrossRef]
- Cummings, J.L. Cholinesterase inhibitors: A new class of psychotropic compounds. Am. J. Psychiatry 2000, 157, 4–15. [Google Scholar]
- Zovko, A.; Viktorsson, K.; Lewensohn, R.; Kološa, K.; Filipič, M.; Xing, H.; Kem, W.R.; Paleari, L.; Turk, T. APS8, a polymeric alkylpyridinium salt blocks α7 nAChR and induces apoptosis in non-small cell lung carcinoma. Mar. Drugs 2013, 11, 2574–2594. [Google Scholar] [CrossRef]
- Zovko, A. Personal communication. Department of Biology, Biotechnical Faculty, University of Ljubljana: Ljubljana, Slovenia, 2014. [Google Scholar]
- Grandič, M.; Aráoz, R.; Molgó, J.; Turk, T.; Sepčić, K.; Benoit, E.; Frangež, R. Toxicity of the synthetic polymeric 3-alkylpyridinium salt (APS3) is due to specific block of nicotinic acetylcholine receptors. Toxicology 2013, 303, 25–33. [Google Scholar] [CrossRef]
- Emberson, J.W.; Muir, A.R. Changes in the ultrastructure of rat myocardium induced by hyperkalaemia. J. Anat. 1969, 104, 411–421. [Google Scholar]
- Van der Meer, C.; Valkenburg, P.W.; Snijders, P.M. Studies on hyperkalemia as a cause of death in intestinal ischemia shock in rats. Circ. Shock 1986, 19, 329–345. [Google Scholar]
- Parham, W.A.; Mehdirad, A.A.; Biermann, K.M.; Fredman, C.S. Hyperkalemia revisited. Tex. Heart Inst. J. 2006, 33, 40–47. [Google Scholar]
- McCaffrey, T.V.; Kern, E.B. Laryngeal regulation of airway resistance. II. Pulmonary receptor reflexes. Ann. Otol. Rhinol. Laryngol. 1980, 89, 462–466. [Google Scholar]
- Willette, R.N.; Barcas, P.P.; Krieger, A.J.; Sapru, H.N. Pulmonary resistance and compliance changes evoked by pulmonary opiate receptor stimulation. Eur. J. Pharmacol. 1983, 91, 181–188. [Google Scholar] [CrossRef]
- Paintal, A.S.; Damodaran, V.N.; Guz, A. Mechanism of excitation of type J receptors. Acta Neurobiol. Exp. 1973, 33, 15–19. [Google Scholar]
- Grandič, M.; Aráoz, R.; Molgó, J.; Turk, T.; Sepčić, K.; Benoit, E.; Frangež, R. The non-competitive acetylcholinesterase inhibitor APS12-2 is a potent antagonist of skeletal muscle nicotinic acetylcholine receptors. Toxicol. Appl. Pharmacol. 2012, 265, 221–228. [Google Scholar] [CrossRef]
- Sherby, S.M.; Eldefrawi, A.T.; Albuquerque, E.X.; Eldefrawi, M.E. Comparison of the actions of carbamate anticholinesterases on the nicotinic acetylcholine receptor. Mol. Pharmacol. 1985, 27, 343–348. [Google Scholar]
- Okonjo, K.O.; Kuhlmann, J.; Maelicke, A. A second pathway of activation of the Torpedo acetylcholine receptor channel. Eur. J. Biochem. 1991, 200, 671–677. [Google Scholar] [CrossRef]
- Ros, E.; Aleu, J.; Gomez de Aranda, I.; Cantí, C.; Pang, Y.P.; Marsal, J.; Solsona, C. Effects of bis(7)-tacrine on spontaneous synaptic activity and on the nicotinic ACh receptor of Torpedo electric organ. J. Neurophysiol. 2001, 86, 183–189. [Google Scholar]
- Olivera-Bravo, S.; Ivorra, I.; Morales, A. The acetylcholinesterase inhibitor BW284c51 is a potent blocker of Torpedo nicotinic AChRs incorporated into the Xenopus oocyte membrane. Br. J. Pharmacol. 2005, 144, 88–97. [Google Scholar] [CrossRef]
- Kasai, H.; Neher, E. Dihydropyridine-sensitive and omega-conotoxin-sensitive calcium channels in a mammalian neuroblastoma-glioma cell line. J. Physiol. 1992, 448, 161–188. [Google Scholar]
- Hogestatt, E.D.; Andersson, K.E. Mechanisms behind the biphasic contractile response to potassium depolarization in isolated rat cerebral arteries. J. Pharmacol. Exp. Ther. 1984, 228, 187–195. [Google Scholar]
- Triggle, D.J. The pharmacology of ion channels: With particular reference to voltage-gated Ca2+ channels. Eur. J. Pharmacol. 1999, 375, 311–325. [Google Scholar] [CrossRef]
- Grandič, M.; Bajuk, B.P.; Sepčić, K.; Košorok, M.D.; Frangež, R. Effects of synthetic analogues of poly-APS on contractile response of porcine coronary arteries. Toxicol. In Vitro 2012, 27, 627–631. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Grandič, M.; Frangež, R. Pathophysiological Effects of Synthetic Derivatives of Polymeric Alkylpyridinium Salts from the Marine Sponge, Reniera sarai. Mar. Drugs 2014, 12, 2408-2421. https://doi.org/10.3390/md12052408
Grandič M, Frangež R. Pathophysiological Effects of Synthetic Derivatives of Polymeric Alkylpyridinium Salts from the Marine Sponge, Reniera sarai. Marine Drugs. 2014; 12(5):2408-2421. https://doi.org/10.3390/md12052408
Chicago/Turabian StyleGrandič, Marjana, and Robert Frangež. 2014. "Pathophysiological Effects of Synthetic Derivatives of Polymeric Alkylpyridinium Salts from the Marine Sponge, Reniera sarai" Marine Drugs 12, no. 5: 2408-2421. https://doi.org/10.3390/md12052408
APA StyleGrandič, M., & Frangež, R. (2014). Pathophysiological Effects of Synthetic Derivatives of Polymeric Alkylpyridinium Salts from the Marine Sponge, Reniera sarai. Marine Drugs, 12(5), 2408-2421. https://doi.org/10.3390/md12052408




