Ochracenoids A and B, Guaiazulene-Based Analogues from Gorgonian Anthogorgia ochracea Collected from the South China Sea
Abstract
:1. Introduction
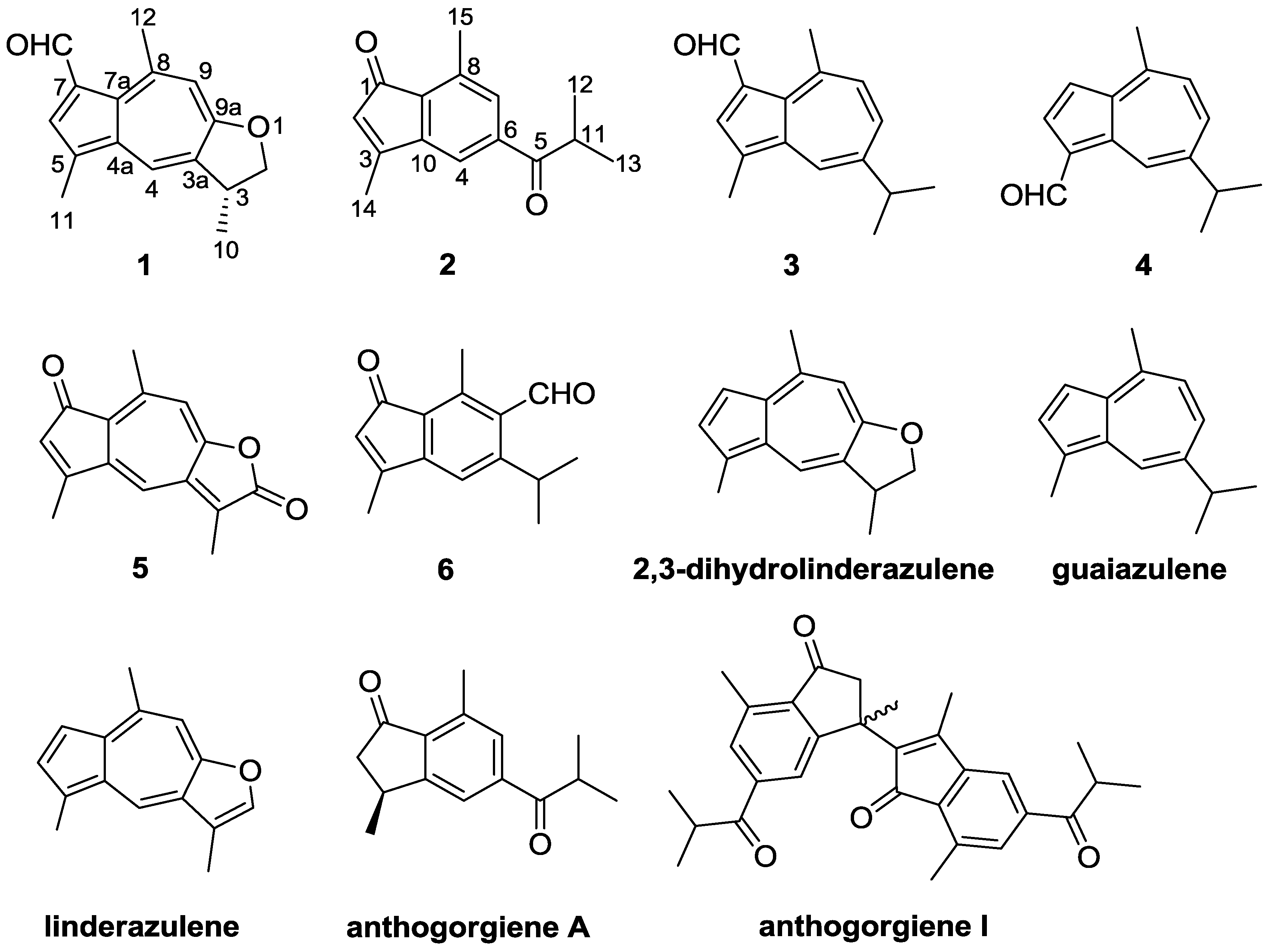
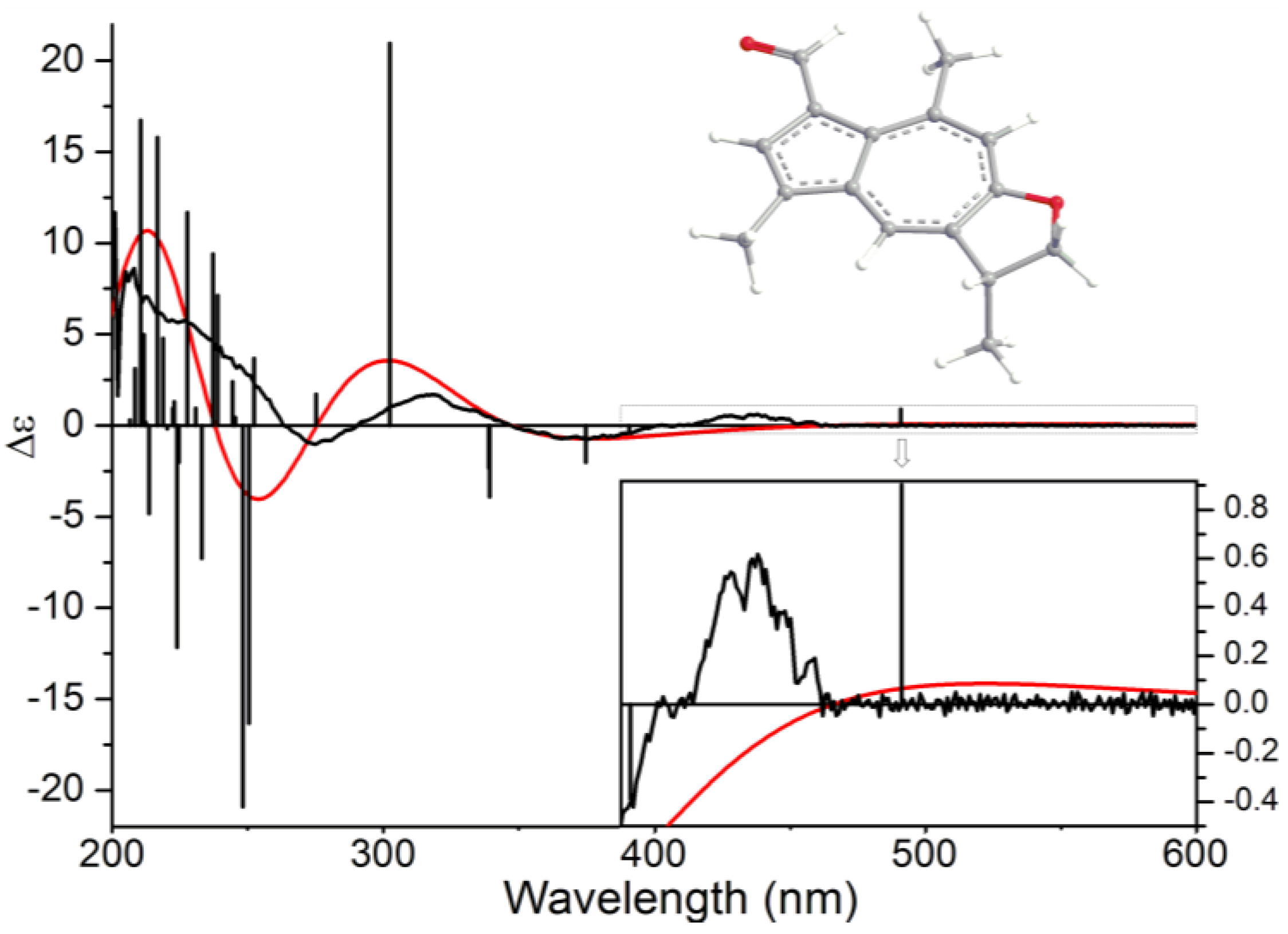
2. Results and Discussion
| Position | δH (J in Hz) | δC Type | HMBC |
|---|---|---|---|
| 2 | 4.82, t (8.4) | 78.1, CH2 | C-10 |
| 4.27, dd (8.4, 6.6) | |||
| 3 | 3.81, m | 39.4, CH | - |
| 3a | - | 130.3, C | - |
| 4 | 8.12, d (1.2) | 131.0, CH | C-3, C-5, C-7a, C-9a |
| 4a | - | 138.5, C | - |
| 5 | - | 127.6, C | - |
| 6 | 7.97, s | 135.6, CH | C-4a, C-7a |
| 7 | - | 129.2, C | - |
| 7a | - | 134.7, C | - |
| 8 | - | 149.0, C | - |
| 9 | 7.09, s | 116.0, CH | C-3a, C-7a, C-9a, C-12 |
| 9a | - | 166.7, C | - |
| 10 | 1.49, d (6.6) | 20.5, CH3 | C-2, C-3, C-3a |
| 11 | 2.55, s | 13.1, CH3 | C-4a, C-5, C-6 |
| 12 | 3.11, s | 30.9, CH3 | C-7a, C-8, C-9 |
| 7-CHO | 10.61, s | 187.0, CH | C-6, C-7 |
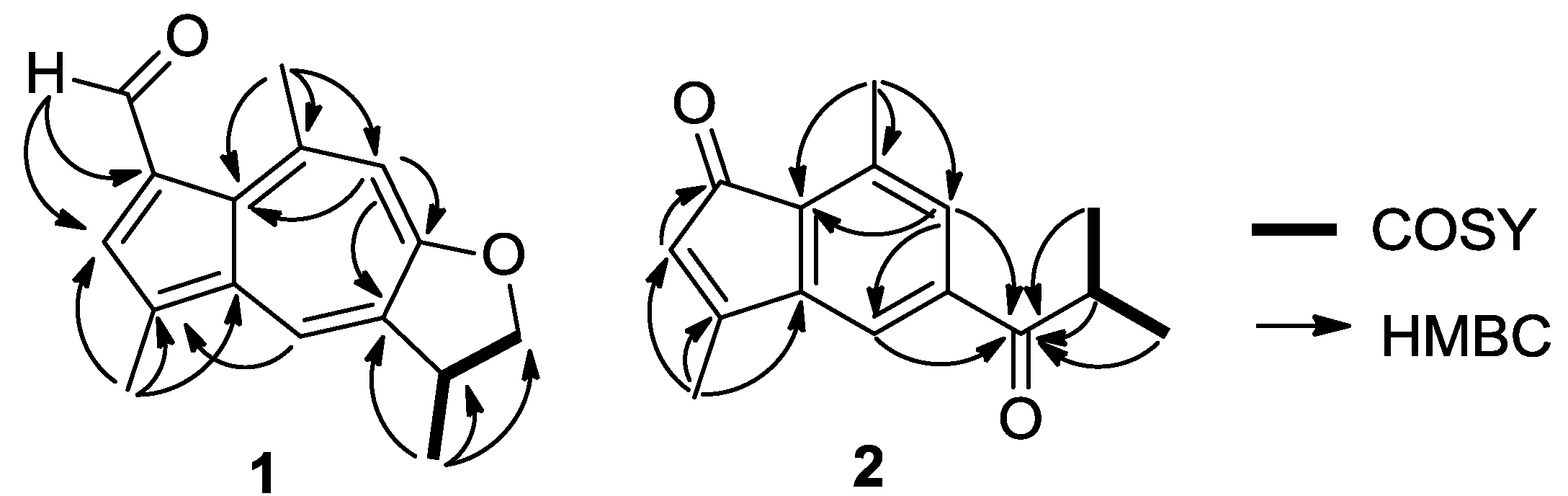
| Position | δH (J in Hz) | δC Type | HMBC |
|---|---|---|---|
| 1 | - | 198.3, C | - |
| 2 | 5.76, s | 125.2, CH | C-1, C-3, C-9, C-10, C-14 |
| 3 | - | 146.7, C | - |
| 4 | 7.48, brs | 116.5, CH | C-5, C-7, C-9, C-10 |
| 5 | - | 204.2, C | - |
| 6 | - | 139.9, C | - |
| 7 | 7.61, brs | 133.4, CH | C-4, C-5, C-9, C-15 |
| 8 | - | 136.6, C | - |
| 9 | - | 131.2, C | - |
| 10 | - | 161.1, C | - |
| 11 | 3.52, septet (8.5) | 35.9, CH | C-5, C-12, C-13 |
| 12 | 1.23, d (8.5) | 19.2, CH3 | C-5, C-11, C-13 |
| 13 | 1.23, d (8.5) | 19.2, CH3 | C-5, C-11, C-12 |
| 14 | 2.27, s | 14.2, CH3 | C-2, C-3, C-10 |
| 15 | 2.58, s | 17.3, CH3 | C-7, C-8, C-9 |
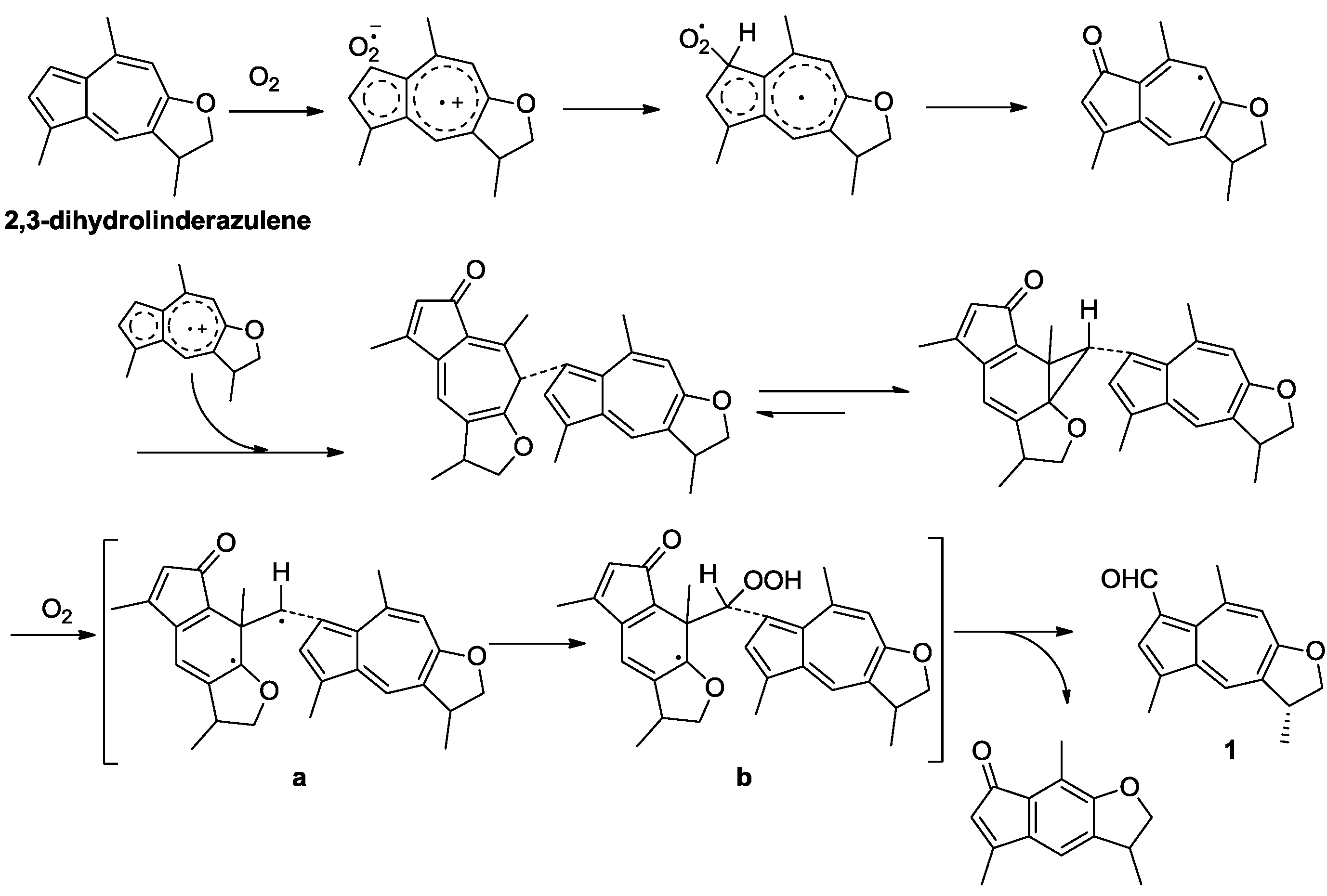
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Materials
3.3. Extraction and Isolation
3.4. Biological Assays
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 2001, 18, 1–49. [Google Scholar] [CrossRef]
- Chen, D.W.; Yu, S.J.; Ofwegen, L.V.; Proksch, P.; Lin, W.H. Anthogorgienes A–O, new guaiazulene-derived terpenoids from a Chinese gorgonian Anthogorgia species, and their antifouling and antibiotic activities. J. Agric. Food Chem. 2012, 60, 112–123. [Google Scholar] [CrossRef]
- Matsubara, Y.; Takekuma, S.; Yokoi, K.; Yamamoto, H.; Nozoe, T. Autoxidation of guaiazulene and 4,6,8-trimethylazulene in polar aprotic solvent: Structural proof for products. Bull. Chem. Soc. Jpn. 1987, 60, 1415–1428. [Google Scholar] [CrossRef]
- Sakemi, S.; Higa, T. 2,3-Dihydrolinderazulene, a new bioactive azulene pigment from the gorgonian Acalycigorgia sp. Experientia 1987, 43, 624–625. [Google Scholar] [CrossRef]
- Tanaka, J.; Miki, H.; Higa, T. Echinofuran, a new furanosesquiterpene from the gorgonian Echinogorgia praelonga. J. Nat. Prod. 1992, 55, 1522–1524. [Google Scholar] [CrossRef]
- Chen, D.W.; Liu, D.; Shen, S.; Cheng, W.; Lin, W.H. Terpenoids from a Chinese gorgonian Anthogorgia sp. and their antifouling activities. Chin. J. Chem. 2012, 30, 1459–1463. [Google Scholar]
- Ochi, M.; Kataoka, K.; Tatsukawa, A.; Kotsuki, H.; Shibata, K. Gorgiabisazulene and gorgiagallylazulene, two new guaiazulenoid pigments from a gorgonian Acalycigorgia sp. Chem. Lett. 1993, 22, 2003–2006. [Google Scholar]
- Wang, C.Y.; Zhao, J.; Liu, H.Y.; Shao, C.L.; Liu, Q.A.; Liu, Y.; Gu, Y.C. Two new eicosanoids with a unique isovalerianic acid ester moiety from the south China sea gorgonian Dichotella gemmacea. Lipids 2011, 46, 81–85. [Google Scholar] [CrossRef]
- Li, L.; Sheng, L.; Wang, C.Y.; Zhou, Y.M.; Huang, H.; Li, X.B.; Li, J.; Mollo, E.; Gavagnin, M.; Guo, Y.W. Diterpenes from the Hainan soft coral Lobophytum cristatum Tixier-Durivault. J. Nat. Prod. 2011, 74, 2089–2094. [Google Scholar] [CrossRef]
- Shao, C.L.; Wu, H.X.; Wang, C.Y.; Liu, Q.A.; Xu, Y.; Wei, M.Y.; Qian, P.Y.; Gu, Y.C.; Zheng, C.J.; She, Z.G.; et al. Potent antifouling resorcylic acid lactones from the gorgonian-derived fungus Cochliobolus lunatus. J. Nat. Prod. 2011, 74, 629–633. [Google Scholar] [CrossRef]
- Struwe, M.; Csato, M.; Singer, T.; Gocke, E. Comprehensive assessment of the photomutagenicity, photogenotoxicity and photo(cyto)toxicity of azulene. Mutat. Res. 2011, 723, 129–133. [Google Scholar] [CrossRef]
- Manzo, E.; Ciavatta, M.L.; Lopez Gresa, M.P.; Gavagnin, M.; Villani, G.; Naik, C.G.; Cimino, G. New bioactive hydrogenated linderazulene-derivatives from the gorgonian Echinogorgia complexa. Tetrahedron Lett. 2007, 48, 2569–2571. [Google Scholar]
- Imre, S.; Thomson, R.H.; Yalhi, B. Linderazulene, a new naturally occurring pigment from the gorgonian Paramuricea chamaeleon. Experientia 1981, 37, 442–443. [Google Scholar] [CrossRef]
- Spartan 08. Available online: http://www.wavefun.com/products/macintosh/Essential/mac_essential.html (accessed on 2 August 2009).
- Gaussian 09. Available online: http://www.gaussian.com/g_prod/g09.htm (accessed on 23 April 2013).
- Stephens, P.J.; Harada, N. ECD Cotton effect approximated by the Gaussian curve and other methods. Chirality 2010, 22, 229–233. [Google Scholar]
- Sato, K.; Ozu, T.; Takenaga, N. Solvent-free synthesis of azulene derivatives via Passerini reaction by grinding. Tetrahedron Lett. 2013, 54, 661–664. [Google Scholar] [CrossRef]
- Koul, S.K.; Taneja, S.C.; Ibraham, S.P.; Dhar, K.L.; Atal, C.K. A C-formylated azulene from Lactarius deterrimus. Phytochemistry 1985, 24, 181–182. [Google Scholar] [CrossRef]
- Talzhanov, N.A.; Raldugin, V.A.; Shakirova, G.A.; Adekenov, S.M. Formation of an unusual guaiane ketopentaenolide from dehydroisoleucomisin by the action of anhydrous DMF and HCl. Chem. Nat. Compd. 2005, 41, 420–422. [Google Scholar] [CrossRef]
- Nozoe, T.; Takekuma, S.; Doi, M.; Matsubara, Y.; Yamamoto, H. Oxidation of azulene derivatives. Autoxidation of guaiazulene in a polar aprotic solvent. Chem. Lett. 1984, 13, 627–630. [Google Scholar]
- Matsubara, Y.; Takekuma, S.; Yamamoto, H.; Nozoe, T. 6-(3-Guaiazulenyl)-5-isopropyl-3,8-dimethyl-1(6H)-azulenone and its norcaradiene-isomer. Key intermediates for novel intermolecular one-carbon transfer in autoxidation of guaiazulene. Chem. Lett. 1987, 16, 455–458. [Google Scholar]
- Alpertunga, B.; Imre, S.; Cowe, H.J.; Cox, P.J.; Thomson, R.H. A photo artefact from linderazulene. Tetrahedron Lett. 1983, 24, 4461–4462. [Google Scholar] [CrossRef]
- Takekuma, S.; Matsubara, Y.; Yamamoto, H.; Nozoe, T. Autoxidation of solid guaiazulene and of the solution in DMF in the presence of base or acid: A comparative study of the product distribution. Bull. Chem. Soc. Jpn. 1988, 61, 475–481. [Google Scholar] [CrossRef]
- Stoletov, K.; Klemke, R. Catch of the day: Zebrafish as a human cancer model. Oncogene 2008, 27, 4509–4520. [Google Scholar] [CrossRef]
- Parng, C.; Seng, W.L.; Semino, C.; McGrath, P.A. Zebrafish: A preclinical model for drug screening. Assay Drug Dev. Technol. 2002, 1, 41–48. [Google Scholar] [CrossRef]
- Skehan, P.A.; Storeng, R.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer-drug screening. J. Natl. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar]
- Pierce, C.G.; Uppuluri, P.; Teistan, A.R.; Wormley, J.F.L.; Mowat, E.; Ramage, G.; Lopez-ribot, J.L. A simple and reproducible 96-well plate-based method for the formation of fungal biofilms and its application to antifungal susceptibility testing. Nat. Protoc. 2008, 3, 1494–1500. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zheng, J.-J.; Shao, C.-L.; Chen, M.; Gan, L.-S.; Fang, Y.-C.; Wang, X.-H.; Wang, C.-Y. Ochracenoids A and B, Guaiazulene-Based Analogues from Gorgonian Anthogorgia ochracea Collected from the South China Sea. Mar. Drugs 2014, 12, 1569-1579. https://doi.org/10.3390/md12031569
Zheng J-J, Shao C-L, Chen M, Gan L-S, Fang Y-C, Wang X-H, Wang C-Y. Ochracenoids A and B, Guaiazulene-Based Analogues from Gorgonian Anthogorgia ochracea Collected from the South China Sea. Marine Drugs. 2014; 12(3):1569-1579. https://doi.org/10.3390/md12031569
Chicago/Turabian StyleZheng, Juan-Juan, Chang-Lun Shao, Min Chen, Li-She Gan, Yu-Chun Fang, Xu-Hui Wang, and Chang-Yun Wang. 2014. "Ochracenoids A and B, Guaiazulene-Based Analogues from Gorgonian Anthogorgia ochracea Collected from the South China Sea" Marine Drugs 12, no. 3: 1569-1579. https://doi.org/10.3390/md12031569
APA StyleZheng, J.-J., Shao, C.-L., Chen, M., Gan, L.-S., Fang, Y.-C., Wang, X.-H., & Wang, C.-Y. (2014). Ochracenoids A and B, Guaiazulene-Based Analogues from Gorgonian Anthogorgia ochracea Collected from the South China Sea. Marine Drugs, 12(3), 1569-1579. https://doi.org/10.3390/md12031569






