The Role of Biophysical Parameters in the Antilipopolysaccharide Activities of Antimicrobial Peptides from Marine Fish
Abstract
:1. Introduction
2. Anti-LPS Factors
2.1. StALF and PpALF2
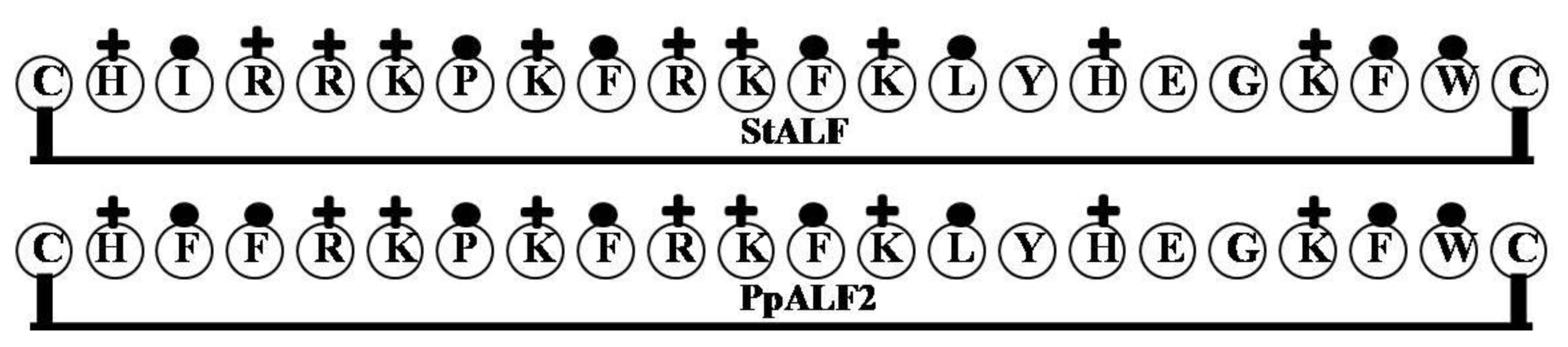
| Name of the Gene or Source | LPS Binding Domain | Net Charge | Hydrophilicity (%) | Hydrophobicity (%) | Molecular Weight |
|---|---|---|---|---|---|
| PpALF2 | CHFFRKPKFRKFKLYHEGKFWC | 8 | 45 | 36 | 2933.56 |
| StALF | CHIRRKPKFRKFKLYHEGKFWC | 9 | 50 | 32 | 2908.55 |
| ALFSp | CHIRRKPKFRKFKLYHEGKFWC | 9 | 50 | 32 | 2908.55 |
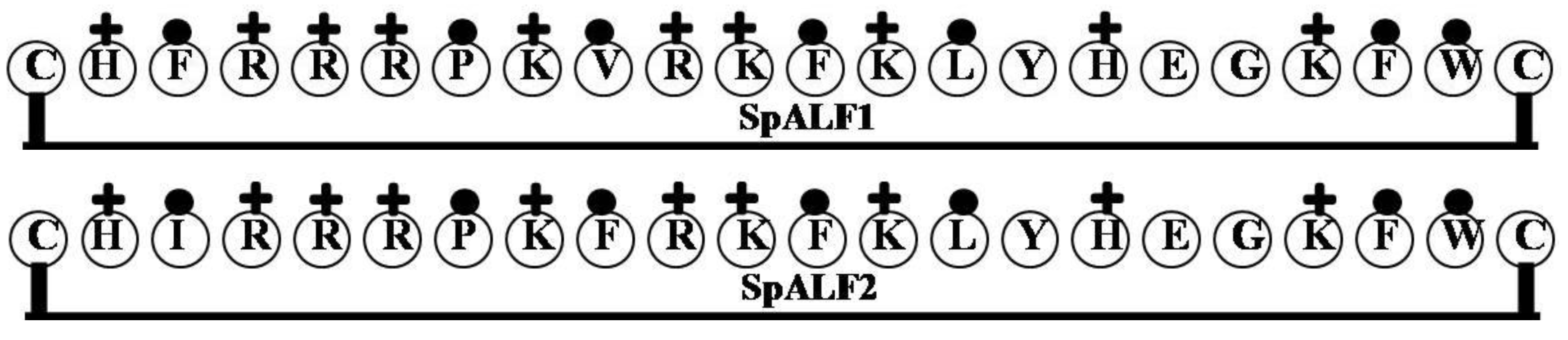
| SpALF1 | CHFRRRPKVRKFKLYHEGKFWC | 9 | 50 | 32 | 2922.54 |
| SpALF2 | CHIRRRPKFRKFKLYHEGKFWC | 9 | 50 | 32 | 2936.57 |
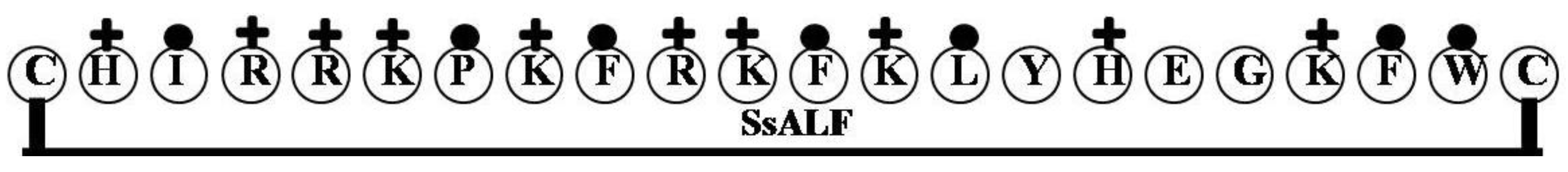
| SsALF | CHIRRKPKFRKFKLYHEGKFWC | 9 | 50 | 32 | 2908.55 |
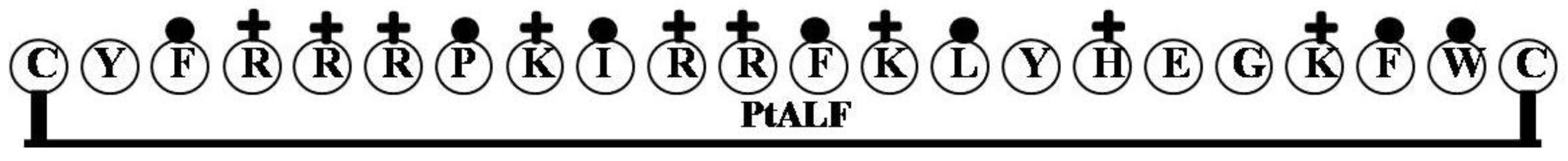
| PtALF | CYFRRRPKIRRFKLYHEGKFWC | 9 | 50 | 32 | 2990.63 |
| PtALF6 | CNFRVMPRLRSWELYFRGDVWC | 2 | 27 | 45 | 2834.37 |
| ALFPm1 | CRYSQRPSFYRWELYFNGRMWC | 3 | 23 | 32 | 2949.43 |
| ALFPm3 | CKFTVKPYLKRFQVYYKGRMWC | 6 | 27 | 36 | 2845.51 |
| ALFPm6 | CSFNVTPKFKRWQLYFRGRMWC | 5 | 23 | 41 | 2854.44 |
| MjALF1 | CNFYVEPKFRNWQLRFKGRMWC | 4 | 27 | 41 | 2909.47 |
| MjALF2 | CRYSQRPTFYRWELYFRGSMWC | 3 | 23 | 32 | 2936.44 |
| ALFFc | CKFTVKPYIKRFQLYYKGRMWC | 6 | 27 | 36 | 2859.54 |
| MrALF5 | CSFQVKPRIKRWELYFRGTMWC | 4 | 27 | 41 | 2835.44 |
| MrALF6 | CIYKRTGYFYKWELHYKAEVRC | 4 | 36 | 27 | 2857.38 |
| MrALF7 | CTYNMRPFFKNWKLYYSASVIC | 3 | 14 | 41 | 2735.27 |
| ALFHa1 | CRFSVKPTVRRFQLYFKGRMWC | 6 | 27 | 41 | 2809.45 |
| ALFHa2 | CNFQVKPKIRRWQLYFVGSMWC | 4 | 18 | 45 | 2790.38 |
| ALFMolf | CQYSVTPRIKKLELWFKGRMWC | 4 | 27 | 41 | 2773.4 |
| EsALF | CNTRVMPTIKKFELYFRGRVWC | 4 | 27 | 41 | 2748.36 |
| LALF | CRKPTFRRLKWKIKFKFKC | 9 | 47 | 37 | 2514.2 |
| CRP19-1 | CRKPTFRRLKWKIKGKFKC | 9 | 47 | 32 | 2424.07 |
| CRP19-2 | CRKPTFRRLKWKYKGKFKC | 9 | 47 | 26 | 2474.09 |
| Pardaxin | GFFALIPKIISSPLFKTLLSAVGSALSSSGGQE | 1 | 9 | 52 | 3323.9 |
| Tachyplesin | KWCFRVCYRGICYRRCR | 6 | 35 | 24 | 2268.82 |
| Sushi 1 | GFKLKGMARISCLPNGQWSNFPPKCIRECAMVSS | 4 | 18 | 44 | 3757.53 |
| Sushi 3 | HAEHKVKIGVEQKYGQFPQGTEVTYTCSGNYFLM | 2 | 24 | 29 | 3891.42 |
| NRC-16 | GWKKWLRKGAKHLGQAAIK | 7 | 37 | 42 | 2176.61 |
2.2. ALFSp

2.3. SpALF1 and SpALF2
2.4. SsALF
2.5. PtALF
2.6. PtALF6

2.7. ALF-Pm3

2.8. MjALF1and MjALF2

2.9. ALFFc

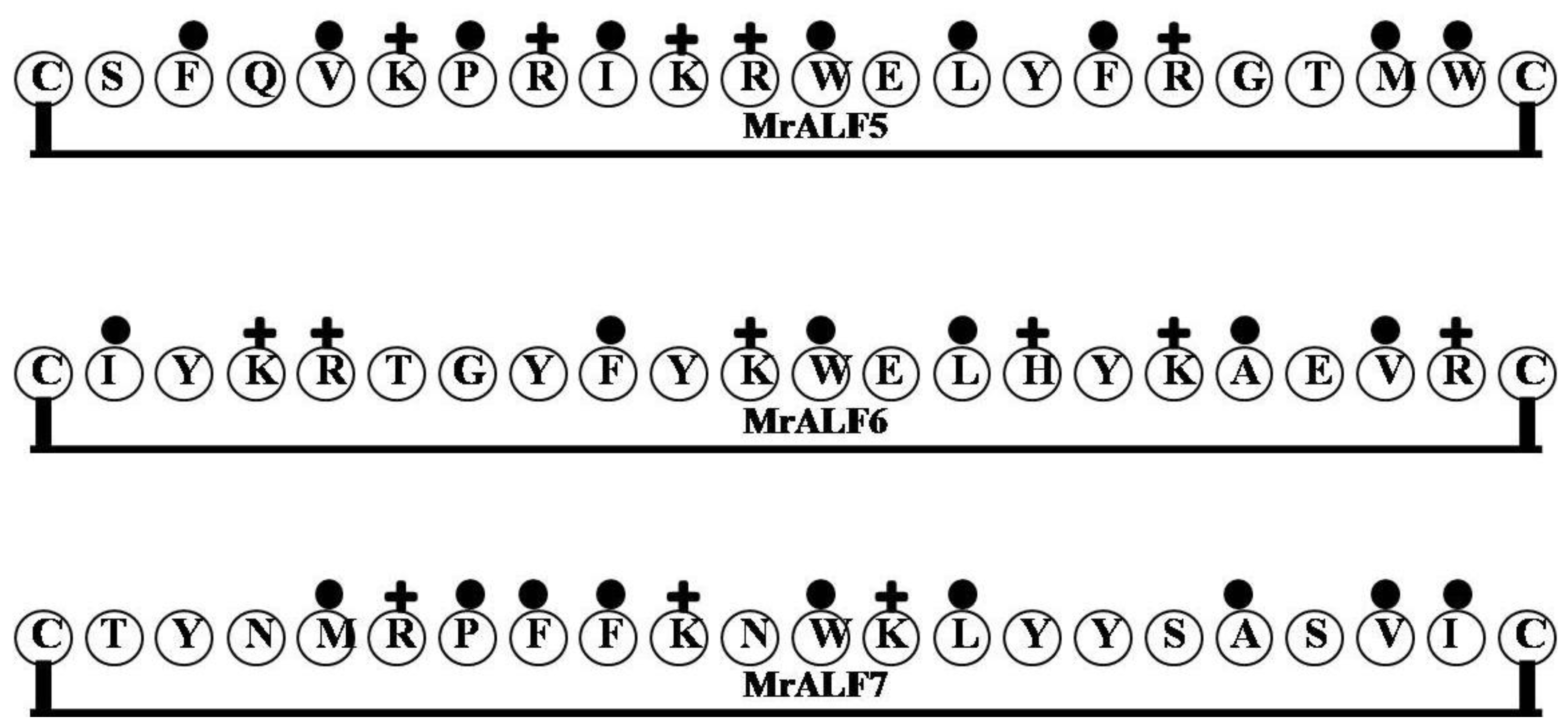
2.10. MrALF5, MrALF6 and MrALF7
2.11. ALFHa1 and ALFHa2

2.12. ALFMolf

2.13. EsALF

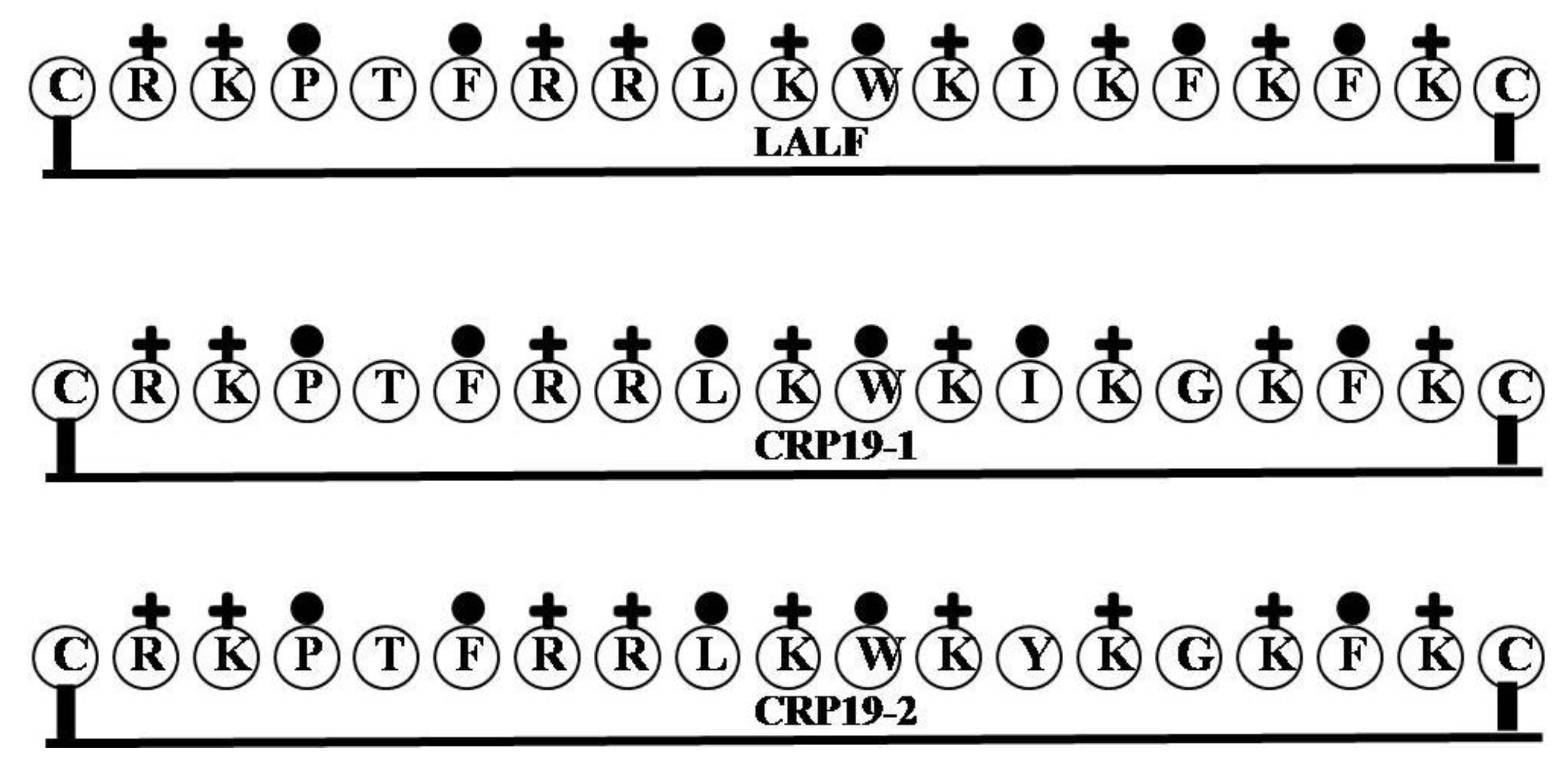
2.14. LALF
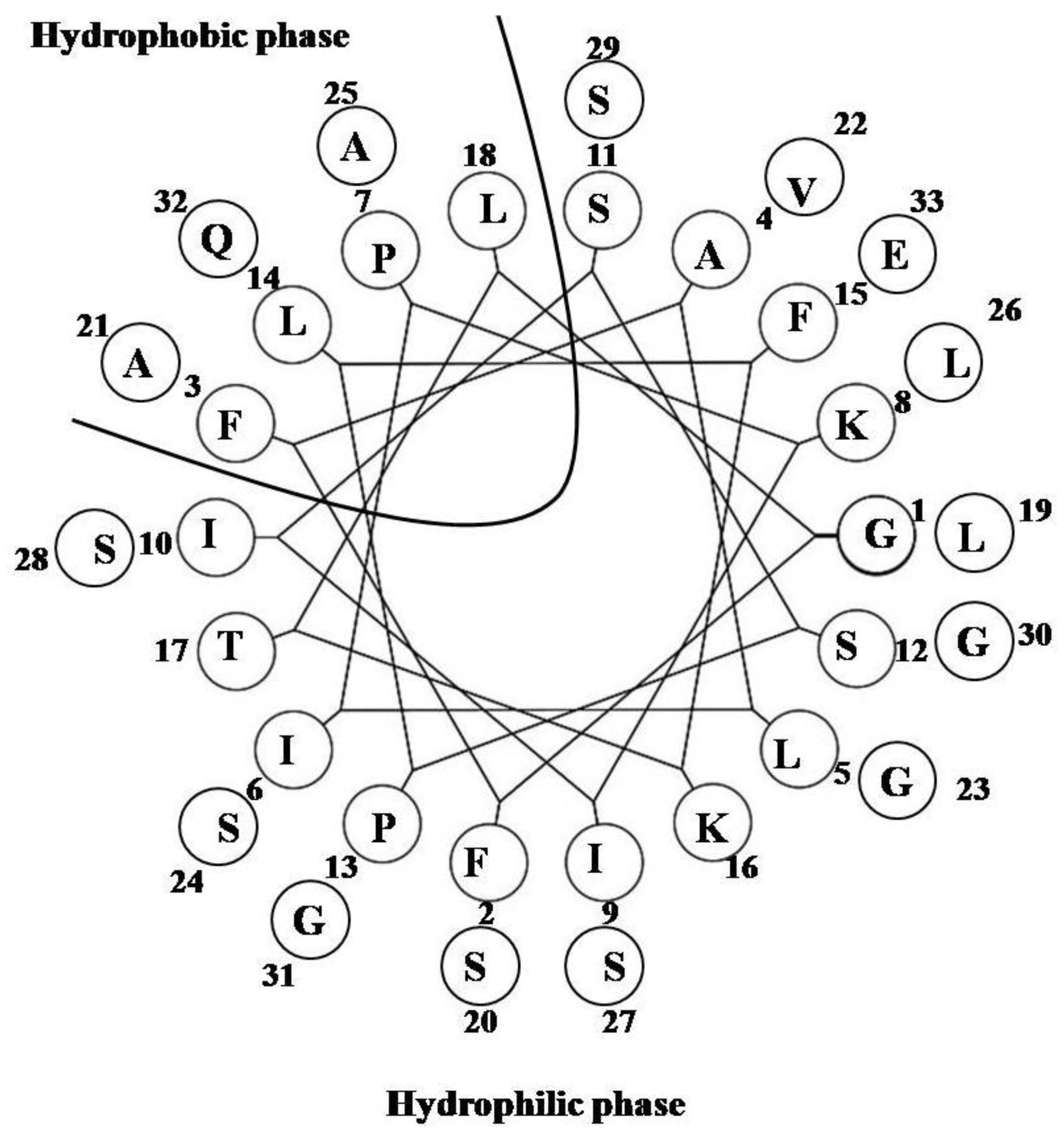
2.15. Pardaxin
2.16. Tachyplesin

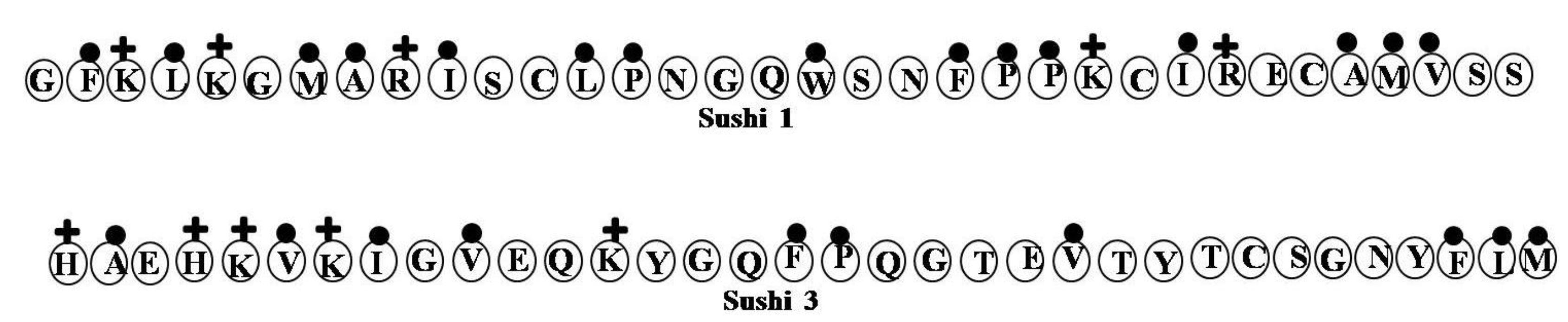
2.17. Sushi Peptides
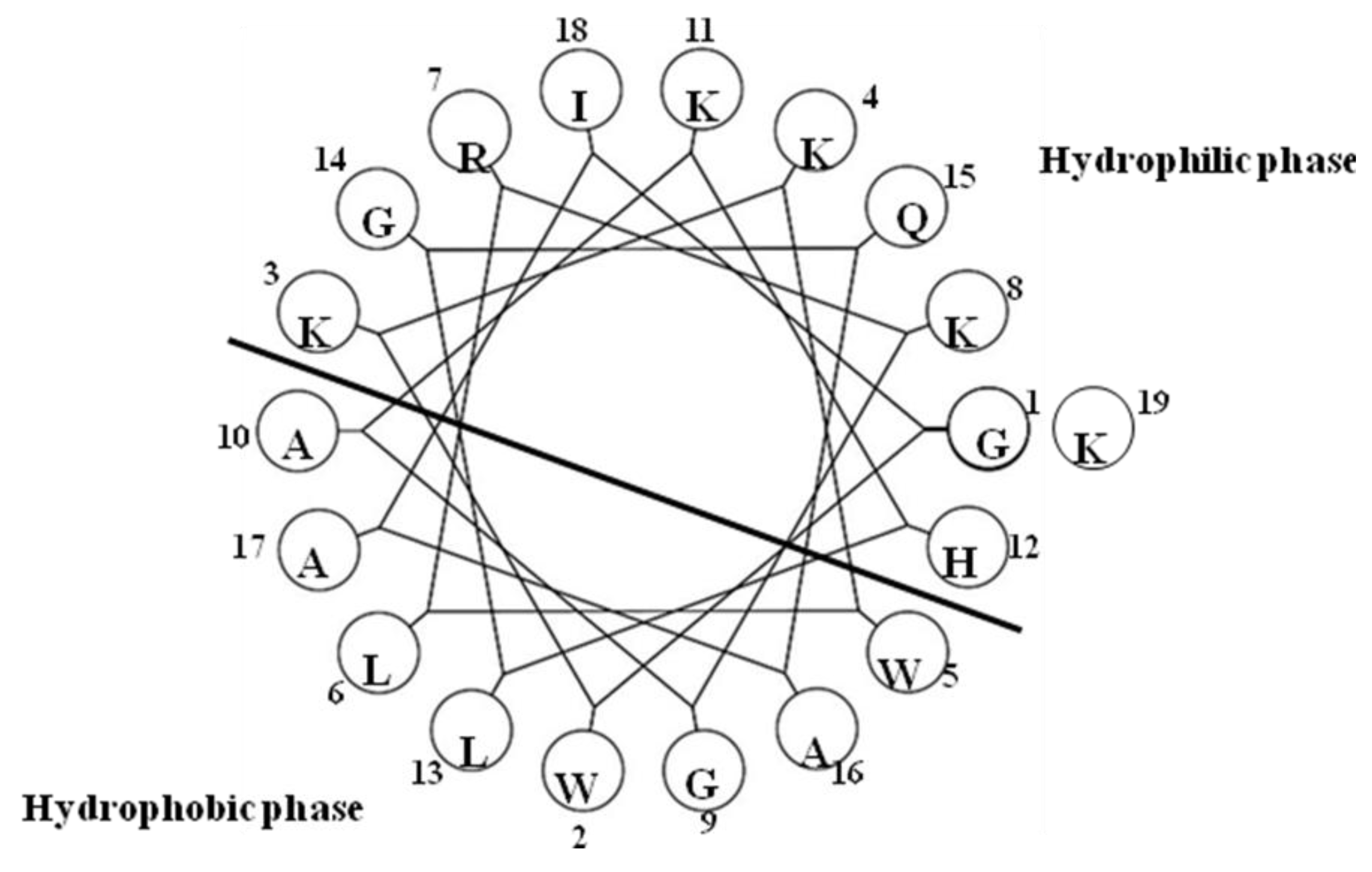
2.18. Pleurocidin-Like Cationic AMP
3. Functions of Cationic ALFs
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Diaz, G.A. Defensins and cystein rich peptides: Two types of antimicrobial peptide in marine molluscs. Invert. Surviv. J. 2010, 7, 157–164. [Google Scholar]
- Rosa, R.D.; Barracco, M.A. Antimicrobial peptides in crustaceans. Invert. Surviv. J. 2010, 7, 262–284. [Google Scholar]
- Matsunaga, S.; Fusetani, N.; Konosu, S. Bioactive marine metabolites, IV. Isolation and the amino acid composition of discodermin A, an antimicrobial peptide, from the marine sponge Discodermia kiiensis. J. Nat. Prod. 1985, 48, 236–241. [Google Scholar] [CrossRef]
- Otero-González, A.J.; Magalhães, B.S.; Garcia-Villarino, M.; López-Abarratequi, C.; Sousa, D.A.; Dias, S.C.; Franco, O.L. Antimicrobial peptides from marine invertebrates as a new frontier for microbial infection control. FASEB J. 2010, 24, 1320–1334. [Google Scholar] [CrossRef]
- Noga, E.J.; Ullal, A.J.; Corrales, J.; Fernandes, J.M. Application of antimicrobial polypeptide host defenses to aquaculture: Exploitation of downregulation and upregulation responses. Comp. Biochem. Physiol. 2011, 6, 44–54. [Google Scholar]
- Tanaka, S.; Nakamura, T.; Morita, T.; Iwanaga, S. Limulus anti-LPS factor: An anticoagulant which inhibits the endotoxin-mediated activation of Limulus coagulation system. Biochem. Biophys. Res. Comm. 1982, 105, 717–723. [Google Scholar] [CrossRef]
- Muta, T.; Miyata, T.; Tokunaga, F.; Nakamura, T; Iwanaga, S. Primary structure of anti-lipopolysaccharide factor from American horseshoe crab, Limulus polyphemus. J. Biochem. 1987, 101, 1321–1330. [Google Scholar]
- Tassanakajon, A.; Amparyup, P.; Somboonwiwat, K.; Supungul, P. Cationic antimicrobial peptides in penaeid shrimp. Mar. Biotechnol. 2011, 13, 639–657. [Google Scholar] [CrossRef]
- Gross, P.S.; Bartlett, T.C.; Browdy, C.L.; Chapman, R.W.; Warr, G.W. Immune gene discovery by expressed sequence tag analysis of hemocytes and hepatopancreas in the Pacific white shrimp, Litopenaeus vannamei, and the Atlantic white shrimp, L. setiferus. Dev. Comp. Immunol. 2001, 25, 565–577. [Google Scholar] [CrossRef]
- Supungul, P.; Klinbunga, S.; Pichayakura, R.; Jitrapakdee, S.; Hirono, I.; Aoki, T.; Tassanakajon, A. Identification of immune related genes in hemocytes of the black tiger shrimp Penaeus monodon. Mar. Biotechnol. 2002, 4, 487–494. [Google Scholar] [CrossRef]
- Liu, F.S.; Liu, Y.C.; Li, F.H.; Dong, B.; Xiang, J.H. Molecular cloning and expression profile of putative antilipopolysaccharide factor in Chinese shrimp (Fenneropenaeus chinensis). Mar. Biotechnol. 2005, 7, 600–608. [Google Scholar] [CrossRef]
- Nagoshi, H.; Inagawa, H.; Morii, K.; Harada, H.; Kohchi, C.; Nishizawa, T.; Taniguchi, Y.; Uenobe, M.; Honda, T.; Kondoh, M.; et al. Cloning and characterization of a LPS regulatory gene having an LPS binding domain in kuruma prawn Marsupenaeus japonicus. Mol. Immunol. 2006, 43, 2061–2069. [Google Scholar] [CrossRef]
- Mekata, T.; Sudhakaran, R.; Okugawa, S.; Kono, T.; Sakai, M.; Itami, T. Molecular cloning and transcriptional analysis of a newly identified anti-lipopolysaccharide factor gene in kuruma shrimp, Marsupenaeus japonicus. Lett. Appl. Microbiol. 2009, 50, 112–119. [Google Scholar]
- Jiménez-Vega, F; Vargas-Albores, F. Isoforms of Litopenaeus vannamei anti-lipopolysaccharide and its expression by bacterial challenge. J. Shellfish Res. 2007, 26, 1169–1175. [Google Scholar] [CrossRef]
- de-la-Re-Vega, E.; O’Leary, N.A.; Shockey, J.E.; Robalino, J.; Payne, C.; Browdy, C.L.; Warr, G.W.; Gross, P.S. Anti-lipopolysaccharide factor in Litopenaeus vannamei (LvALF): A broad spectrum antimicrobial peptide essential for shrimp immunity against bacterial and fungal infection. Mol. Immunol. 2008, 45, 1916–1925. [Google Scholar] [CrossRef]
- Rosa, R.D.; Stoco, P.H.; Barranco, M.A. Cloning and characterisation of cDNA sequences encoding for anti-lipopolysaccharide factors (ALFs) in Brazilian palaemonid and penaeid shrimps. Fish Shellfish Immunol. 2008, 25, 693–696. [Google Scholar] [CrossRef]
- Tassanakajon, A.; Amparyup, P.; Somboonwiwat, K.; Supungul, P. Cationic antimicrobial peptides in penaeid shrimp. Mar. Biotechnol. 2010, 12, 487–505. [Google Scholar] [CrossRef]
- Tassanakajon, A.; Klinbunga, S.; Paunglarp, N.; Rimphanitchayakit, V.; Udomkit, A.; Jitrapakdee, S.; Sritunyalucksana, K.; Phongdara, A.; Pongsomboon, S.; Supungul, P.; et al. Penaeus monodon gene discovery project: The generation of an EST collection and establishment of a database. Gene 2006, 384, 104–112. [Google Scholar] [CrossRef]
- Sperstad, S.V.; Haug, T.; Blencke, H.M.; Styrvold, O.B.; Li, C.; Stensvåg, K. Antimicrobial peptides from marine invertebrates: Challenges and perspectives in marine antimicrobial peptide discovery. Biotechnol. Adv. 2011, 29, 519–530. [Google Scholar] [CrossRef]
- Magnadóttir, B. Innate immunity of fish (overview). Fish Shellfish Immunol. 2006, 20, 137–151. [Google Scholar] [CrossRef]
- Liu, Y.; Hou, F.; He, S.; Qian, Z.; Wang, X.; Mao, A.; Sun, C.; Liu, X. Identification, characterization and functional analysis of a serine protease inhibitor (Lvserpin) from the Pacific white shrimp, Litopenaeus vannamei. Dev. Comp. Immunol. 2014, 43, 35–46. [Google Scholar] [CrossRef]
- Li, C.; Ng, M.L.; Zhu, Y.; Ho, B.; Ding, J.L. Tandom repeats of sushi3 peptide with enhanced LPS-binding and-neutralizing activities. Protein Eng. 2003, 16, 629–635. [Google Scholar] [CrossRef]
- Jerala, R.; Porro, M. Endotoxin Neutralizing Peptides. Curr. Top. Med. Chem. 2004, 4, 1173–1184. [Google Scholar] [CrossRef]
- Li, P.; Wohland, T.; Ho, B.; Ding, J.L. Perturbation of Lipopolysaccharide (LPS) Micelles by Sushi 3 (S3) antimicrobial peptide. The importance of an intermolecular disulfide bond in S3 dimer for binding, disruption, and neutralization of LPS. J. Biol. Chem. 2004, 279, 50150–50156. [Google Scholar] [CrossRef]
- Iliev, D.B.; Roach, J.C.; Mackenzie, S.; Planas, J.V.; Goetz, F.W. Endotoxin recognition: in fish or not in fish? FEBS Lett. 2005, 579, 6519–6528. [Google Scholar] [CrossRef]
- Papo, N.; Shai, Y. A molecular mechanism for lipopolysaccharide protection of Gram-negative bacteria from antimicrobial peptides. J. Biol. Chem. 2005, 280, 10378–10387. [Google Scholar] [CrossRef]
- Pristovsek, P.; Fehér, K.; Szilágyi, L.; Kidric, J. Structure of a synthetic fragment of the LALF protein when bound to lipopolysaccharide. J. Med. Chem. 2005, 48, 1666–1670. [Google Scholar] [CrossRef]
- Hancock, R.E.; Brown, K.L.; Mookherjee, N. Host defense peptides from invertebrates- emerging antimicrobial strategies. Immunobiology 2006, 211, 315–322. [Google Scholar] [CrossRef]
- Xian, L.X.; Peng, B.; Dong, W.R.; Yang, Z.F.; Shao, J.Z. Lipopolysaccharide induces apoptosis in Carassius auratus lymphocytes, a possible role in pathogenesis of bacterial infection in fish. Dev. Comp. Immunol. 2008, 32, 992–1001. [Google Scholar] [CrossRef]
- Sepulcre, M.P.; Alcaraz-Pérez, F.; López-Muñoz, A.; Roca, F.J.; Meseguer, J.; Cayuela, M.L.; Mulero, V. Evolution of lipopolysaccharide (LPS) Recognition and signaling: Fish TLR4 does not recognize LPS and negatively regulates NF-kappaB activation. J. Immunol. 2009, 182, 1836–1845. [Google Scholar] [CrossRef]
- Kaisho, T.; Akira, S. Pleiotropic function of Toll-like receptors. Microbes Infect. 2004, 6, 1388–1394. [Google Scholar] [CrossRef]
- Weiss, C.A.; Wasiluk, K.R.; Kellogg, T.A.; Dunn, D.L. Bactericidal and rndotoxin neutralizing activity of a peptide derived from limulus antilipopolysaccharide factor. Surgery 2000, 128, 339–344. [Google Scholar] [CrossRef]
- Afsal, V.V.; Antony, S.P.; Chaithanya, E.R.; Singh, I.S.; Philip, R. Two isoforms of anti lipopolysaccharide factors identified and characterized from the hemocytes of portunid crabs, Portunus pelagicus and Scylla tranquebarica. Mol. Immunol. 2012, 52, 258–263. [Google Scholar] [CrossRef]
- Nagaoka, I.; Hirota, S.; Niyonsaba, F.; Hirata, M.; Adachi, Y.; Tamura, H.; Tanaka, S.; Heumann, D. Augmentation of the lipopolysaccharide-neutralizing activities of human cathelicidin CAP18/LL-37-derived antimicrobial peptides by replacement with hydrophobic and cationic amino acid residues. Clin. Diagn. Lab. Immunol. 2002, 9, 972–982. [Google Scholar]
- Gopal, R.; Seo, C.H.; Song, P.I.; Park, Y. Effect of repetitive lysine-tryptophan motifs on the bactericidal activity of antimicrobial peptides. Amino Acids 2013, 44, 645–660. [Google Scholar] [CrossRef]
- Frecer, V.; Ho, B.; Ding, J.L. De novo design of potent antimicrobial peptides. Antimicrob. Agents Chemother. 2004, 48, 3349–3357. [Google Scholar] [CrossRef]
- Wasiluk, K.R.; Leslie, D.B.; Vietzen, P.S.; Mayo, K.H.; Dunn, D.L. Structure/function studies of an endotoxin-neutralizing peptide derived from bactericidal/permeability-increasing protein. Surgery 2004, 136, 253–260. [Google Scholar] [CrossRef]
- Imjongjirak, C.; Amparyup, P.; Tassanakajon, A.; Sittipraneed, S. Antilipopolysaccharides factor (ALF) of mud crab scylla paramamosain: molecular cloning, genomic organization and the antimicrobial activity of its synthetic LPS binding domain. Mol. Immunol. 2007, 44, 3195–3203. [Google Scholar] [CrossRef]
- Afsal, V.V.; Antony, S.P.; Sanjeevan, V.N.; Anil Kumar, P.R.; Bright Singh, I.S.; Philip, R. A new isoform of anti-lipopolysaccharide factor identified from the blue swimmer crab, Portunus pelagicus: Molecular characteristics and phylogeny. Aquaculture 2012, 356–357, 119–122. [Google Scholar]
- Zhu, L.; Lan, J.F.; Huang, Y.Q.; Zhang, C.; Zhou, J.F.; Fang, W.H.; Yao, X.J.; Wang, H.; Li, X.C. SpALF4: A newly identified anti-lipopolysaccharide factor from the mud crab Scylla paramamosain with broad spectrum antimicrobial activity. Fish Shellfish Immunol. 2014, 36, 172–180. [Google Scholar] [CrossRef]
- Liu, H.P.; Chen, R.Y.; Zhang, Q.X.; Wang, Q.Y.; Li, C.R.; Peng, H.; Cai, L.; Zheng, C.Q.; Wang, K.J. Characterization of two isoforms of antiliopolysacchride factors (Sp-ALFs) from the mud crab Scylla paramamosain. Fish Shellfish Immunol. 2012, 33, 1–10. [Google Scholar] [CrossRef]
- Yedery, R.D.; Reddy, K.V. Identification, cloning, characterization and recombinant expression of an antilipopolysaccharide factor from the hemocytes of Indian mud crab, Scylla serrata. Fish Shellfish Immunol. 2009, 27, 275–284. [Google Scholar] [CrossRef]
- Yue, F.; Pan, L.; Miao, J.; Zhang, L.; Li, J. Molecular cloning, characterization and mRNA expression of two antibacterial peptides: Crustin and Anti-lipopolysaccharide factor in swimming crab Portunus trituberculatus. Comp. Biochem. Physiol. 2010, 156, 77–85. [Google Scholar] [CrossRef]
- Warren, H.S.; Glennon, M.L.; Wainwright, N.; Amato, S.F.; Black, K.M.; Kirsch, S.J.; Riveau, G.R.; Whyte, R.I.; Zapol, W.M.; Novitsky, T.J. Binding and neutralization of endotoxin by Limulus antilipopolysaccharide factor. Infect. Immun. 1992, 60, 2506–2513. [Google Scholar]
- Liu, Y.; Cui, Z.; Li, X.; Song, C.; Shi, G. A newly identified anti-lipopolysaccharide factor from the swimming crab Portunus trituberculatus with broad spectrum antimicrobial activity. Fish Shellfish Immunol. 2013, 34, 463–470. [Google Scholar] [CrossRef]
- Tharntada, S.; Somboonwiwat, K.; Rimphanitchayakit, V.; Tassanakajon, A. Anti-lipopolysaccharide factors from the black tiger shrimp, Penaeus monodon, are encoded by two genomic loci. Fish Shellfish Immunol. 2008, 24, 46–54. [Google Scholar] [CrossRef]
- Hoess, A.; Watson, S.; Siber, G.R.; Liddington, R. Crystal structure of an endotoxin-neutralizing protein from the horseshoe crab, Limulus anti-LPS factor, at 1.5 A resolution. EMBO J. 1993, 12, 3351–3356. [Google Scholar]
- Somboonwiwat, K.; Marcos, M.; Tassanakajon, A.; Klinbunga, S.; Aumelas, A.; Romestand, B.; Gueguen, Y.; Boze, H.; Moulin, G.; Bachère, E. Recombinant expression and anti-microbial activity of anti-lipopolysaccharide factor (ALF) from the black tiger shrimp Penaeus monodon. Dev. Comp. Immunol. 2005, 29, 841–851. [Google Scholar] [CrossRef]
- Somboonwiwat, K.; Bachère, E.; Rimphanitchayakit, V.; Tassanakajon, A. Localization of anti-lipopolysaccharide factor (ALFPm3) in tissues of the black tiger shrimp, Penaeus monodon, and characterization of its binding properties. Dev. Comp. Immunol. 2008, 32, 1170–1176. [Google Scholar] [CrossRef]
- Jaree, P.; Tassanakajon, A.; Somboonwiwat, K. Effect of the anti-lipopolysaccharide factor isoform 3 (ALFPm3) from Penaeus monodon on Vibrio harveyi cells. Dev. Comp. Immunol. 2012, 38, 554–560. [Google Scholar] [CrossRef]
- Tharntada, S.; Ponprateep, S.; Somboonwiwat, K.; Liu, H.; Söderhäll, I.; Söderhäll, K.; Tassanakajon, A. Role of anti-lipopolysaccharide factor from the black tiger shrimp, Penaeus monodon, in protection from white spot syndrome virus infection. J. Gen. Virol. 2009, 90, 1491–1498. [Google Scholar] [CrossRef]
- Carriel-Gomes, M.C.; Kratz, J.M.; Barracco, M.A.; Bachére, E.; Barardi, C.R.; Simões, C.M. In vitro antiviral activity of antimicrobial peptides against herpes simplex virus 1, adenovirus, and rotavirus. Mem. Inst. Oswaldo Cruz. 2007, 102, 469–472. [Google Scholar] [CrossRef]
- Hikima, S.; Hikima, J.; Rojtinnakorn, J.; Hirono, I.; Aoki, T. Characterization and function of kuruma shrimp lysozyme possessing lytic activity against Vibrio species. Gene 2003, 316, 187–195. [Google Scholar] [CrossRef]
- Yang, Y.; Boze, H.; Chemardin, P.; Padilla, A.; Moulin, G.; Tassanakajon, A.; Pugnière, M.; Roquet, F.; Destoumieux-Garzón, D.; Gueguen, Y.; et al. NMR structure of rALF-Pm3, an anti-lipopolysaccharide factor from shrimp: Model of the possible lipid A-binding site. Biopolymers 2009, 91, 207–220. [Google Scholar] [CrossRef]
- Ried, C.; Wahl, C.; Miethke, T.; Wellnhofer, G.; Landgraf, C.; Schneider-Mergener, J.; Hoess, A. High affinity endotoxin-binding and neutralizing peptides based on the crystal structure of recombinant Limulus anti-lipopolysaccharide factor. J. Biol. Chem. 1996, 271, 28120–28127. [Google Scholar]
- Andrä, J.; Garidel, P.; Majerle, A.; Jerala, R.; Ridge, R.; Paus, E.; Novitsky, T.; Koch, M.H.; Brandenburg, K. Biophysical characterization of the interaction of Limulus polyphemus endotoxin neutralizing protein with lipopolysaccharide. Eur. J. Biochem. 2004, 27, 2037–2046. [Google Scholar]
- Andrä, J.; Lamata, M.; Martinez de Tejada, G.; Bartels, R.; Koch, M.H.; Brandenburg, K. Cyclic antimicrobial peptides based on Limulus anti-lipopolysaccharide factor for neutralization of lipopolysaccharide. Biochem. Pharmacol. 2004, 68, 1297–1307. [Google Scholar] [CrossRef]
- Leslie, D.B.; Vietzen, P.S.; Lazaron, V.; Wasiluk, K.R.; Dunn, D.L. Comparison of endotoxin antagonism of linear and cyclized peptides derived from limulus anti-lipopolysaccharide factor. Surg. Infect. 2006, 7, 45–52. [Google Scholar] [CrossRef]
- Vallespi, M.G.; Alvarez-Obregón, J.C.; Rodriguez-Alonso, I.; Montero, T.; Garay, H.; Reyes, O.; Araña, M.J. A Limulus anti-LPS factor-derived peptide modulates cytokine gene expression and promotes resolution of bacterial acute infection in mice. Int. Immunopharmacol. 2003, 3, 247–256. [Google Scholar] [CrossRef]
- Ren, Q.; Zhang, Z.; Li, X.C.; Du, J.; Hui, K.M.; Zhang, C.Y.; Wang, W. Three different anti-lipopolysaccharide factors identified from giant freshwater prawn, Macrobrachium rosenbergii. Fish Shellfish Immunol. 2012, 33, 766–774. [Google Scholar] [CrossRef]
- Beale, K.M.; Towle, D.W.; Jayasundara, N.; Smith, C.M.; Shields, J.D.; Small, H.J.; Greenwood, S.J. Anti-lipopolysaccharide factors in the American lobster Homarus americanus: Molecular characterization and transcriptional response to Vibrio fluvialis challenge. Comp. Biochem. Physiol. 2008, 3, 263–269. [Google Scholar]
- Li, C.; Zhao, J.; Song, L.; Mu, C.; Zhang, H.; Gai, Y.; Qiu, L.; Yu, Y.; Ni, D.; Xing, K. Molecular cloning, genomic organization and functional analysis of an anti-lipopolysaccharide factor from Chinese mitten crab Eriocheir sinensis. Dev. Comp. Immunol. 2008, 32, 784–794. [Google Scholar] [CrossRef]
- Wang, D.N.; Chen, L.; Liu, J.W.; He, Z.Y.; Wu, X.F. The native gene of anti-LPS factor Tachypleus tridentatus: Cloning, expression and its bacteriostatic activity in vitro. Protein Pept. Lett. 2001, 8, 273–280. [Google Scholar] [CrossRef]
- Morita, T.; Ohtsubo, S.; Nakamura, T.; Tanaka, S.; Iwanaga, S.; Ohashi, K.; Niwa, M. Isolation and biological activities of limulus anticoagulant (anti-LPS factor) which interacts with lipopolysaccharide (LPS). J. Biochem. 1985, 97, 1611–1620. [Google Scholar]
- Liu, Y.; Ni, B.; Ren, J.D.; Chen, J.H.; Tian, Z.Q.; Tang, M.; Li, D.; Xia, P. Cyclic Limulus anti-lipopolysaccharide (LPS) factor-derived peptide CLP-19 antagonizes LPS function by blocking binding to LPS binding protein. Biol. Pharm. Bull. 2011, 34, 1678–1683. [Google Scholar] [CrossRef]
- Ren, J.; Gao, H.; Tang, M.; Gu, J.; Xia, P.; Xiao, G. Lipopolysaccharide (LPS) detoxification of analogue peptides derived from limulus anti-LPS factor. Peptides 2010, 31, 1853–1859. [Google Scholar] [CrossRef]
- Thompson, S.A.; Tachibana, K.; Nakanishi, K.; Kubota, I. Melittin-like peptides from the shark-repelling defense secretion of the sole Pardachirus pavoninus. Science 1986, 233, 341–343. [Google Scholar]
- Shai, Y.; Fox, J.; Caratsch, C.; Shih, Y.L.; Edwards, C.; Lazarovici, P. Sequencing and synthesis of pardaxin, a polypeptide from the Red Sea Moses sole with ionophore activity. FEBS Lett. 1988, 242, 161–166. [Google Scholar] [CrossRef]
- Bhunia, A.; Domadia, P.N.; Torres, J.; Hallock, K.J.; Ramamoorthy, A.; Bhattacharjya, S. NMR structure of pardaxin, a pore-forming antimicrobial peptide, in lipopolysaccharide micelles: Mechanism of outer membrane permeabilization. J. Biol. Chem. 2010, 285, 3883–3895. [Google Scholar]
- Shai, Y.; Bach, D.; Yanovsky, A. Channel formation properties of synthetic pardaxin and analogues. J. Biol. Chem. 1990, 265, 20202–20209. [Google Scholar]
- Saberwal, G.; Nagaraj, R. Conformations of peptide fragments comprising the amino-terminal, central, and carboxyl-terminal regions of a membrane-active polypeptide. Build-up of secondary structure in pardaxin. J. Biol. Chem. 1993, 268, 14081–14089. [Google Scholar]
- Huang, H.N.; Pan, C.Y.; Chan, Y.L.; Chen, J.Y.; Wu, C.J. Use of the antimicrobial peptide pardaxin (GE33) to protect against MRSA infection in mice with skin injuries. Antimicrob. Agents Chemother. 2014, 58, 1538–1545. [Google Scholar] [CrossRef]
- Desch, C.E.; O’Hara, P.; Harlan, J.M. Antilipopolysaccharide factor from horseshoe crab, Tachypleus tridentatus, inhibits lipopolysaccharide activation of cultured human endothelial cells. Infect Immun. 1989, 57, 1612–1614. [Google Scholar]
- Saravanan, R.; Mohanram, H.; Joshi, M.; Domadia, P.N.; Torres, J.; Ruedl, C.; Bhattacharjya, S. Structure, activity and interactions of the cysteine deleted analog of tachyplesin-1 with lipopolysaccharide micelle: Mechanistic insights into outer-membrane permeabilization and endotoxin neutralization. Biochim. Biophys. Acta 2012, 1818, 1613–1624. [Google Scholar] [CrossRef]
- Kushibiki, T.; Kamiya, M.; Aizawa, T.; Kumaki, Y.; Kikukawa, T.; Mizuguchi, M.; Demura, M.; Kawabata, S.I.; Kawano, K. Interaction between tachyplesin I, an antimicrobial peptide derived from horseshoe crab, and lipopolysaccharide. Biochim. Biophys. Acta 2014, 1844, 527–534. [Google Scholar] [CrossRef]
- Ding, J.L.; Navas, M.A.; Ho, B. Molecular cloning and sequence analysis of factor C cDNA from the Singapore horseshoe crab, Carcinoscorpius rotundicauda. Mol. Mar. Biol. Biotechnol. 1995, 4, 90–103. [Google Scholar]
- Tan, N.S.; Ng, M.L.; Yau, Y.H.; Chong, P.K.; Ho, B.; Ding, J.L. Definition of endotoxin binding sites in horseshoe crab factor C recombinant sushi proteins and neutralization of endotoxin by sushi peptides. FASEB J. 2000, 14, 1801–1813. [Google Scholar] [CrossRef]
- Gopal, R.; Lee, J.H.; Kim, Y.G.; Kim, M.S.; Seo, C.H.; Park, Y. Anti-microbial, anti-biofilm activities and cell selectivity of the NRC-16 peptide derived from witch flounder, Glyptocephalus cynoglossus. Mar. Drugs 2013, 11, 1836–1852. [Google Scholar] [CrossRef]
- Patrzykat, A.; Gallant, J.W.; Seo, J.K.; Pytyck, J.; Douglas, S.E. Novel antimicrobial peptides derived from flatfish genes. Antimicrob. Agents Chemother. 2003, 47, 2464–2470. [Google Scholar] [CrossRef]
- Gopal, R.; Park, J.S.; Seo, C.H.; Park, Y. Applications of circular dichroism for structural analysis of gelatin and antimicrobial peptides. Int. J. Mol. Sci. 2012, 13, 3229–3244. [Google Scholar] [CrossRef]
- Domadia, P.N.; Bhunia, A.; Ramamoorthy, A.; Bhattacharjya, S. Structure, interactions, and antibacterial activities of MSI-594 derived mutant peptide MSI-594F5A in lipopolysaccharide micelles: Role of the helical hairpin conformation in outer-membrane permeabilization. J. Am. Chem. Soc. 2010, 132, 18417–18428. [Google Scholar] [CrossRef]
- Bhunia, A.; Ramamoorthy, A.; Bhattacharjya, S. Helical hairpin structure of a potent antimicrobial peptide MSI-594 in lipopolysaccharide micelles by NMR spectroscopy. Chemistry 2009, 15, 2036–2040. [Google Scholar] [CrossRef]
- Fazio, M.A.; Oliveira, V.X., Jr.; Bulet, P.; Miranda, M.T.; Daffre, S.; Miranda, A. Structure-activity relationship studies of gomesin: Importance of the disulfide bridges for conformation, bioactivities, and serum stability. Biopolymers 2006, 84, 205–218. [Google Scholar] [CrossRef]
- Chandrababu, K.B.; Ho, B.; Yang, D. Structure, dynamics, and activity of an all-cysteine mutated human beta defensin-3 peptide analogue. Biochemistry 2009, 48, 6052–6061. [Google Scholar] [CrossRef]
- Bhunia, A.; Saravanan, R.; Mohanram, H.; Mangoni, M.L.; Bhattacharjya, S. NMR structures and interactions of temporin-1Tl and temporin-1Tb with lipopolysaccharide micelles: Mechanistic insights into outer membrane permeabilization and synergistic activity. J. Biol. Chem. 2011, 286, 24394–24406. [Google Scholar]
- Lee, J.K.; Park, S.C.; Hahm, K.S.; Park, Y. A helix-PXXP-helixpeptide with antibacterial activity without cytotoxicity against MDRPA-infected mice. Biomaterials 2014, 35, 1025–1039. [Google Scholar] [CrossRef]
- Giuliani, A.; Pirri, G.; Rinaldi, A.C. Antimicrobialpeptides: the LPSconnection. Methods Mol. Biol. 2010, 618, 137–154. [Google Scholar] [CrossRef]
- Bridle, A.; Nosworthy, E.; Polinski, M.; Nowak, B. Evidence of an antimicrobial-immunomodulatory role of atlantic salmon cathelicidins during infection with Yersinia ruckeri. PloS One 2011, 6, e23417. [Google Scholar]
- Beloin, C.; Michaelis, K.; Lindner, K.; Landini, P.; Hacker, J.; Ghigo, J.M.; Dobrindt, U. The transcriptional antiterminator RfaH represses biofilm formation in Escherichia coli. J. Bacteriol. 2006, 188, 1316–1331. [Google Scholar] [CrossRef]
- Hori, K.; Matsumoto, S. Bacterial adhesion: From mechanism to control. Biochem. Eng. J. 2010, 48, 424–443. [Google Scholar] [CrossRef]
- Lau, P.C.; Lindhout, T.; Beveridge, T.J.; Dutcher, J.R.; Lam, J.S. Differential lipopolysaccharide core capping leads to quantitative and correlated modifications of mechanical and structural properties in Pseudomonas aeruginosa biofilms. J. Bacteriol. 2009, 191, 6618–6631. [Google Scholar] [CrossRef]
- Benitez, J.A.; Spelbrink, R.G.; Silva, A.; Phillips, T.E.; Stanley, C.M.; Boesmann Finkelstein, M. Adherence of Vibrio cholerae to cultured differentiated human intestinal cells: An in vitro colonization model. Infect. Immunol. 1997, 65, 3474–3477. [Google Scholar]
- Bandara, H.M.; Lam, O.L.; Watt, R.M.; Jin, L.J.; Samaranayake, L.P. Bacterial lipopolysaccharides variably modulate in vitro biofilm formation of Candida species. J. Med. Microbiol. 2010, 59, 1225–1234. [Google Scholar] [CrossRef]
- Rendueles, O.; Kaplan, J.B.; Ghigo, J.M. Antibiofilm polysaccharides. Environ. Microbiol. 2013, 15, 334–346. [Google Scholar] [CrossRef]
- Hugouvieux, V.; Barber, C.E.; Daniels, M.J. Entry of Xanthomonas campestris pv. campestris into hydathodes of Arabidopsis thaliana leaves: A system for studying early infection events in bacterial pathogenesis. Mol. Plant Microbe Interact. 1998, 11, 537–543. [Google Scholar] [CrossRef]
- Steinstraesser, L.; Koehler, T.; Jacobsen, F.; Daigeler, A.; Goertz, O.; Langer, S.; Kesting, M.; Steinau, H.; Eriksson, E.; Hirsch, T. Host defense peptides in wound healing. Mol. Med. 2008, 14, 528–537. [Google Scholar]
- Duplantier, A.J.; van Hoek, M.L. The human cathelicidin antimicrobial peptide LL-37 as a potential treatment for polymicrobial infected wounds. Front Immunol. 2013, 4, 143–156. [Google Scholar]
- Gutsmann, T.; Razquin-Olazaran, I.; Kowalski, I.; Kaconis, Y.; Howe, J.; Bartels, R.; Hornef, M.; Schurholz, T.; Rossle, M.; Sanchez-Gomez, S.; et al. New antiseptic peptides to protect against endotoxin-mediated shock. Antimicrob. Agents Chemother. 2010, 54, 3817–3824. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gopal, R.; Seo, C.H.; Park, Y. The Role of Biophysical Parameters in the Antilipopolysaccharide Activities of Antimicrobial Peptides from Marine Fish. Mar. Drugs 2014, 12, 1471-1494. https://doi.org/10.3390/md12031471
Gopal R, Seo CH, Park Y. The Role of Biophysical Parameters in the Antilipopolysaccharide Activities of Antimicrobial Peptides from Marine Fish. Marine Drugs. 2014; 12(3):1471-1494. https://doi.org/10.3390/md12031471
Chicago/Turabian StyleGopal, Ramamourthy, Chang Ho Seo, and Yoonkyung Park. 2014. "The Role of Biophysical Parameters in the Antilipopolysaccharide Activities of Antimicrobial Peptides from Marine Fish" Marine Drugs 12, no. 3: 1471-1494. https://doi.org/10.3390/md12031471
APA StyleGopal, R., Seo, C. H., & Park, Y. (2014). The Role of Biophysical Parameters in the Antilipopolysaccharide Activities of Antimicrobial Peptides from Marine Fish. Marine Drugs, 12(3), 1471-1494. https://doi.org/10.3390/md12031471





