A Novel Lipid Extraction Method from Wet Microalga Picochlorum sp. at Room Temperature
Abstract
:1. Introduction
2. Results and Discussion
2.1. Examine Ethanol for Lipid Extraction from Wet Biomass
2.2. Investigating the Optimum Procedure of Extracting Lipids Using Ethanol
| Variables | Levels | ||||
|---|---|---|---|---|---|
| −α | −1 | 0 | 1 | α | |
| Extraction time (min) | 2.5 | 10 | 25 | 40 | 47.5 |
| Extraction temperature (°C) | 20 | 25 | 35 | 45 | 50 |
| the ratio of solvent to biomass (mL/g) | 1 | 2 | 4 | 6 | 7 |
| NO. | Extraction time (min) | Extraction temperature (°C) | Solvent to biomass ratio (mL/g) | Extraction yield (of the dry weight) | |
|---|---|---|---|---|---|
| Experimental | Predicted | ||||
| 1 | 10.00 | 25.00 | 2.00 | 22.62 | 22.26 |
| 2 | 40.00 | 25.00 | 2.00 | 25.20 | 25.01 |
| 3 | 10.00 | 45.00 | 2.00 | 22.77 | 22.57 |
| 4 | 40.00 | 45.00 | 2.00 | 26.26 | 25.87 |
| 5 | 10.00 | 25.00 | 6.00 | 30.84 | 31.36 |
| 6 | 40.00 | 25.00 | 6.00 | 32.81 | 33.14 |
| 7 | 10.00 | 45.00 | 6.00 | 30.35 | 30.67 |
| 8 | 40.00 | 45.00 | 6.00 | 32.51 | 33.00 |
| 9 | 2.50 | 35.00 | 4.00 | 27.73 | 27.60 |
| 10 | 47.50 | 35.00 | 4.00 | 31.51 | 31.41 |
| 11 | 25.00 | 20.00 | 4.00 | 31.90 | 31.76 |
| 12 | 25.00 | 50.00 | 4.00 | 31.96 | 31.88 |
| 13 | 25.00 | 35.00 | 1.00 | 17.97 | 18.79 |
| 14 | 25.00 | 35.00 | 7.00 | 32.01 | 30.96 |
| 15 | 25.00 | 35.00 | 4.00 | 31.29 | 30.98 |
| 16 | 25.00 | 35.00 | 4.00 | 30.11 | 30.98 |
| 17 | 25.00 | 35.00 | 4.00 | 30.56 | 30.98 |
| 18 | 25.00 | 35.00 | 4.00 | 31.28 | 30.98 |
| 19 | 25.00 | 35.00 | 4.00 | 31.64 | 30.98 |
| 20 | 25.00 | 35.00 | 4.00 | 30.83 | 30.98 |
| Source | Sum of squares | Df | Mean square | F value | p-Value |
|---|---|---|---|---|---|
| Model | 308.98 | 9 | 34.33 | 76.57 | < 0.0001 |
| Linear | |||||
| X1 | 20.16 | 1 | 20.16 | 44.97 | < 0.0001 |
| X2 | 0.022 | 1 | 0.022 | 0.049 | 0.8301 |
| X3 | 205.72 | 1 | 205.72 | 458.83 | < 0.0001 |
| Quadratic | |||||
| X12 | 0.15 | 1 | 0.15 | 0.34 | 0.0105 |
| X22 | 0.47 | 1 | 0.47 | 1.05 | 0.1041 |
| X32 | 0.50 | 1 | 0.50 | 1.12 | < 0.0001 |
| Interaction | |||||
| X1X2 | 4.43 | 1 | 4.43 | 9.87 | 0.5714 |
| X1X3 | 1.43 | 1 | 1.43 | 3.20 | 0.3301 |
| X2X3 | 75.84 | 1 | 75.84 | 169.16 | 0.3148 |
| Residual | 4.48 | 10 | 0.45 | ||
| Lack of fit | 2.91 | 5 | 0.58 | 1.85 | 0.2578 |
| Pure error | 1.57 | 5 | 0.31 | ||
| Cor total | 313.47 | 19 |
2.3. The Validation of the Model
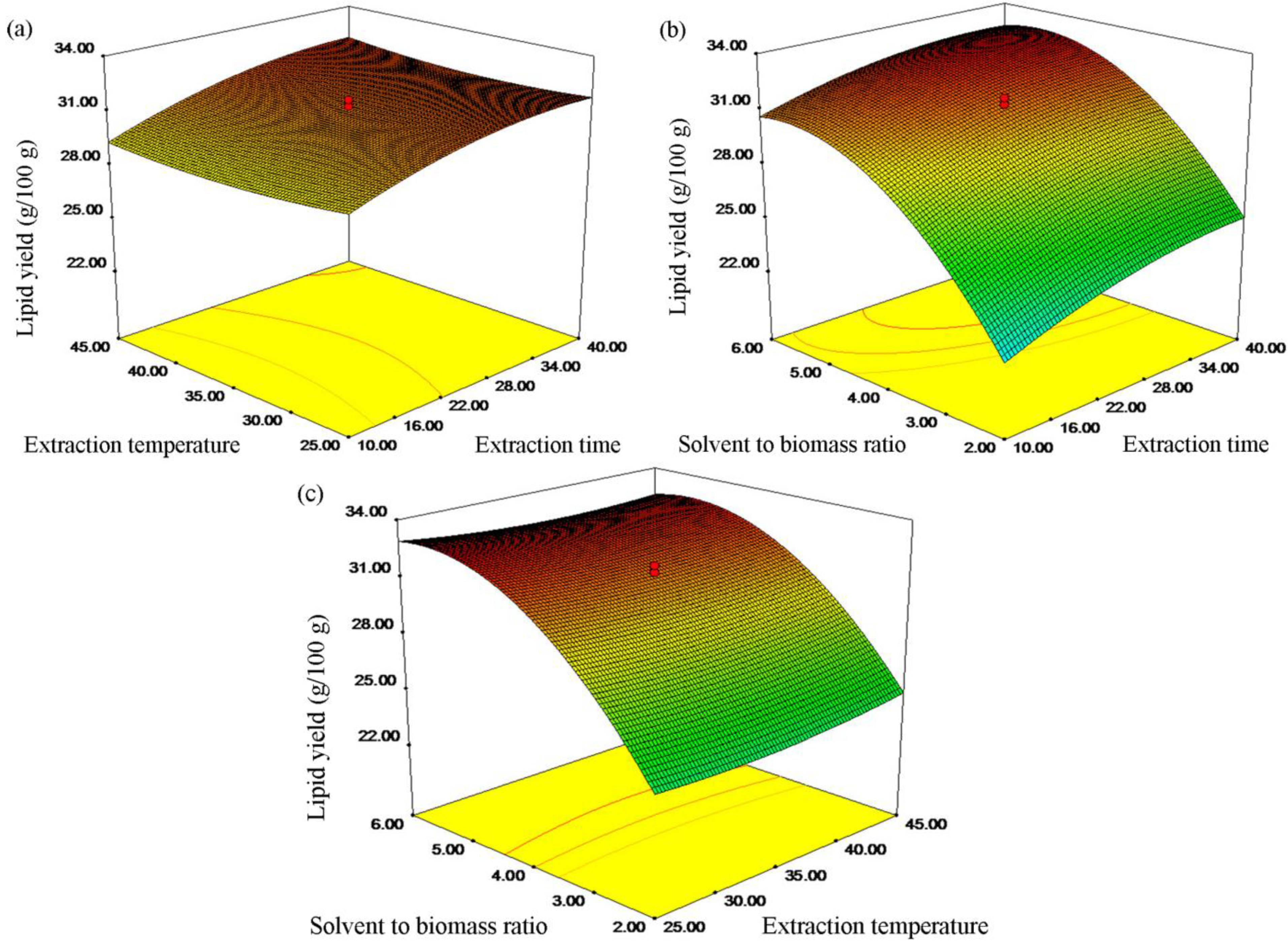
2.4. Lipid Analysis and Fatty Acid Composition Comparisons
| Extraction method | ||
|---|---|---|
| Bligh-Dyer | Ethanol | |
| Lipid extraction yield (% of the dry weight) | 33.18 ± 0.24 | 33.04 ± 0.16 |
| Fatty acid composition (% of FAME) | ||
| Saturated | ||
| C16:0 | 32.49 ± 1.54 | 29.48 ± 3.12 |
| C18:0 | 2.82 ± 0.43 | 6.00 ± 1.89 |
| Unsaturated | ||
| C16:1 | 2.57 ± 0.62 | 2.05 ± 0.38 |
| C16:2 | 5.76 ± 0.16 | 5.65 ± 0.58 |
| C16:3 | 6.62 ± 0.54 | 6.09 ± 0.11 |
| C18:1 | 8.42 ± 0.01 | 9.06 ± 0.28 |
| C18:2 | 22.25 ± 0.21 | 22.47 ± 2.63 |
| C18:3 | 17.37 ± 0.65 | 17.04 ± 0.93 |
| Others | 1.71 ± 0.35 | 2.16 ± 0.40 |
| Lipid class | Extraction method | |
|---|---|---|
| Bligh-Dyer | Ethanol | |
| Neutral lipid | 54.73 ± 1.47 | 53.49 ± 2.11 |
| Glycolipid | 16.46 ± 0.76 | 15.62 ± 0.54 |
| Phospholipid | 28.81 ± 0.71 | 30.89 ± 1.57 |
2.5. Lipid Extraction at Larger Scale and Ethanol Recycling

2.6. Lipid Extraction Methods Comparison
3. Materials and Methods
3.1. Strain, Culture Conditions and Harvesting Method
3.2. Conventional Lipid Extraction Method
3.3. Lipid Extraction using Ethanol as Extractant
3.4. Experimental Design and Data Analysis
3.5. Esterification and Analysis of Fatty Acids
3.6. Lipid Analysis Using Silica Gel Column Chromatography
3.7. Lipid Extraction at Larger Scale and Ethanol Recycling
4. Conclusions
Acknowledgements
Conflict of Interest
References
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Liam, B.; Philip, O. Biofuels from microalgae-a review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 2, 557–577. [Google Scholar]
- Mairet, F.; Bernard, O.; Masci, P.; Lacour, T.; Sciandra, A. Modelling neutral lipid production by the microalga Isochrysis aff. galbana under nitrogen limitation. Bioresour. Technol. 2011, 1, 142–149. [Google Scholar]
- Rawat, I.; Ranjith, K.R.; Mutanda, T.; Bux, F. Dual role of microalgae: Phycoremediation of domestic wasterwater and biomass production for sustainable biofuels production. Appl. Energy 2011, 88, 3411–3424. [Google Scholar] [CrossRef]
- Hende, S.V.D.; Vervaeren, H.; Boon, N. Flue gas compounds and microalgae: (Bio-) chemical interactions leading to biotechnological opportunities. Biotechnol. Adv. 2012, 30, 1405–1424. [Google Scholar] [CrossRef]
- Hu, Q.; Sommerfeld, M.; Jarvis, E.; Ghirardi, M.; Posewitz, M.; Seibert, M.; Darzins, A. Microalgal triacylglycerols as feedstocks for biofuel production: Perspectives and advances. Plant J. 2008, 54, 621–639. [Google Scholar] [CrossRef]
- Schenk, P.M.; Thomas-Hall, S.R.; Stephens, E.; Marx, U.C.; Mussgnug, J.H.; Posten, C.; Kruse, O.; Hankamer, B. Second generation biofuels: High-Efficiency microalgae for biodiesel production. BioEnergy Res. 2008, 1, 20–43. [Google Scholar] [CrossRef]
- Singh, J.; Gu, S. Commercialization potential of microalgae for biofuels production. Renew. Sustain. Energy Rev. 2010, 9, 2596–2610. [Google Scholar] [CrossRef]
- Lardon, L.; Helias, A.; Sialve, B.; Steyer, J.P.; Bernard, O. Life-Cycle assessment of biodiesel production from microalgae. Sci. Technol. 2009, 17, 6475–6481. [Google Scholar]
- Patil, P.D.; Gude, V.G.; Mannarswamy, A.; Cooke, P.; Nirmalakhandan, N.; Lammers, P.; Deng, S.G. Comparison of direct transesterification of algal biomass under supercritical methanol and microwave irradiation conditions. Fuel 2012, 97, 822–831. [Google Scholar] [CrossRef]
- Adam, F.; Vian, M.A.; Peltier, G.; Chemat, F. “Solvent-Free” ultrasound assisted extraction of lipids from fresh microalgae cells: A green, clean and scalable process. Bioresour. Technol. 2012, 114, 457–465. [Google Scholar] [CrossRef]
- Tanzi, C.D.; Vian, M.A.; Chemat, F. New procedure for extraction of algal lipids from wet biomass: A green clean and scalable process. Bioresour. Technol. 2013, 134, 271–275. [Google Scholar] [CrossRef]
- Cheng, J.; Yu, T.; Li, T.; Zhou, J.H.; Cen, K.F. Using wet microalgae for direct biodiesel production via microwave irradiation. Bioresour. Technol. 2013, 131, 531–535. [Google Scholar] [CrossRef]
- Halim, R.; Gladman, B.; Danquah, M.K.; Webley, P.A. Oil extraction from microalgae for biodiesel production. Bioresour. Technol. 2011, 1, 178–185. [Google Scholar]
- Fajardo, A.R.; Cerdán, L.E.; Medina, A.R.; Fernández, F.G.A.; Moreno, P.A.G.; Grima, E.M. Lipid extraction from the microalga Phaeodactylum tricornutum. Eur. J. Lipid Sci. Technol. 2007, 2, 120–126. [Google Scholar]
- Chen, M.; Chen, X.L.; Liu, T.Z.; Zhang, W. Subcritical ethanol extraction of lipid from wet microalgae paste of Nannochloropsis sp. J. Biobased Mater. Bioenergy 2011, 5, 385–389. [Google Scholar] [CrossRef]
- Halim, R.; Danquah, M.K.; Webley, P.A. Extraction of oil from microalgae for biodiesel production: A review. Biotechnol. Adv. 2012, 30, 709–732. [Google Scholar] [CrossRef]
- Ranjan, A.; Patil, C.; Moholkar, V.S. Mechanistic assessment of microalgal lipid extraction. Ind. Eng. Chem. Res 2010, 49, 2979–2985. [Google Scholar] [CrossRef]
- Brian, M.C. Kinetics of Lipid Extraction from Microalgae; University of New Hampshire, University of New Hampshire Scholars’ Repository: Durham, NC, USA, 2013. [Google Scholar]
- Prommuak, C.; Pavasant, P.; Quitain, A.T.; Goto, M.; Shotipruk, A. Microalgal lipid extraction and evaluation of single-step biodiesel production. Eng. J. 2012, 5, 157–166. [Google Scholar]
- Knothe, G. Dependence of biodiesel fuel properties on the structure of fatty acid alkyl esters. Fuel Process Technol. 2005, 86, 1059–1070. [Google Scholar] [CrossRef]
- Hugo, P.; Luisa, B.; Filipe, F.; Luisa, C.; Catarina, V.D.; Cristina, P.; Eva, R.; Aschwin, E.; Joao, V. Polyunsaturated fatty acids of marine macroalgae: Potential for nutritional and pharmaceutical applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 913–917. [Google Scholar]
- Goldberg, K.I.; Shrestha, P.; Cohen, Z. Mobilization of arachidonyl moieties from triacylglycerols into chloroplastic lipids following recovery from nitrogen starvation of the microalga Parietochloris incisa. BBA Mol. Cell Biol. Lipids 2005, 1738, 63–71. [Google Scholar]
- Christie, W.W. Lipid Analysis: Isolation, Separation, Identification and Structural Analysis of Lipids., 3rd ed.; The Oily Press: Bridgewater, UK, 2003; pp. 373–387. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, F.; Xiang, W.; Sun, X.; Wu, H.; Li, T.; Long, L. A Novel Lipid Extraction Method from Wet Microalga Picochlorum sp. at Room Temperature. Mar. Drugs 2014, 12, 1258-1270. https://doi.org/10.3390/md12031258
Yang F, Xiang W, Sun X, Wu H, Li T, Long L. A Novel Lipid Extraction Method from Wet Microalga Picochlorum sp. at Room Temperature. Marine Drugs. 2014; 12(3):1258-1270. https://doi.org/10.3390/md12031258
Chicago/Turabian StyleYang, Fangfang, Wenzhou Xiang, Xiumei Sun, Hualian Wu, Tao Li, and Lijuan Long. 2014. "A Novel Lipid Extraction Method from Wet Microalga Picochlorum sp. at Room Temperature" Marine Drugs 12, no. 3: 1258-1270. https://doi.org/10.3390/md12031258
APA StyleYang, F., Xiang, W., Sun, X., Wu, H., Li, T., & Long, L. (2014). A Novel Lipid Extraction Method from Wet Microalga Picochlorum sp. at Room Temperature. Marine Drugs, 12(3), 1258-1270. https://doi.org/10.3390/md12031258






