Krempfielins J-M, New Eunicellin-Based Diterpenoids from the Soft Coral Cladiella krempfi
Abstract
:1. Introduction

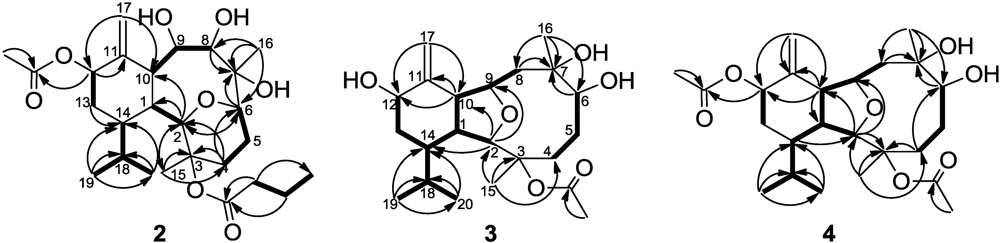
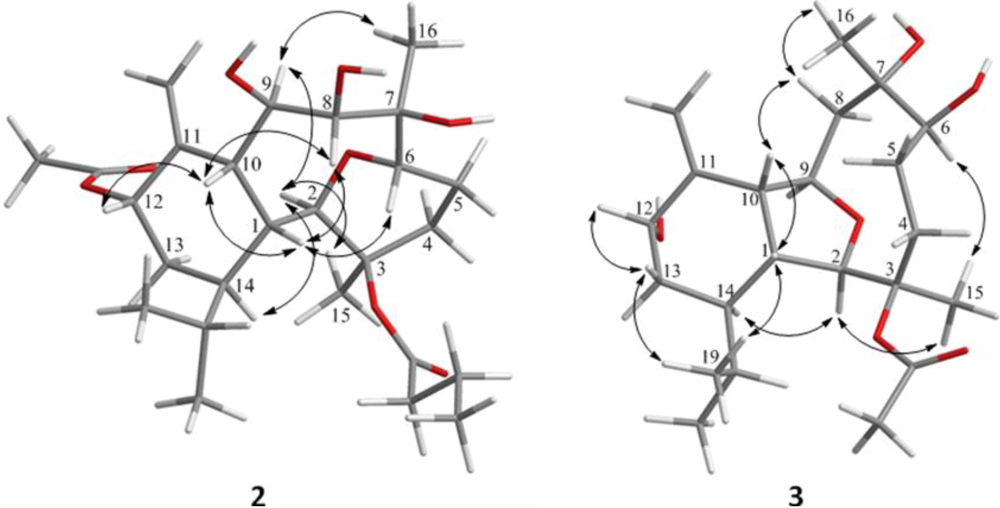
2. Results and Discussion
| 1 a | 2 b | 3 a | 4 a | |||||
|---|---|---|---|---|---|---|---|---|
| δC | δH | δC | δH | δC | δH | δC | δH | |
| 1 | 45.6, CH c | 2.20 dd | 37.8, CH | 3.24 m | 44.2, CH | 2.31 t (8.0) | 44.2, CH | 2.31 ddd |
| (10.8, 8.0) d | (8.0, 6.0, 2.0) | |||||||
| 2 | 92.1, CH | 3.59 br s | 80.9, CH | 3.58 m | 90.4, CH | 3.69 br s | 90.2, CH | 3.69 d (2.0) |
| 3 | 86.6, C | 83.8, C | 86.6, C | 86.6, C | ||||
| 4 | 36.8, CH2 | 1.75 m | 28.8, CH2 | 1.92 m | 34.6, CH2 | 1.94 m | 34.9, CH2 | 1.92 m |
| 2.61 dd | 2.90 dd | 2.48 m | 2.52 dd | |||||
| (14.4, 9.2) | (14.8, 7.0) | (14.8, 8.8) | ||||||
| 5 | 26.8, CH2 | 1.33 m | 19.9, CH2 | 1.50 m | 30.4, CH2 | 1.42 m | 30.5, CH2 | 1.45 m |
| 1.59 m | 1.71 m | 1.71 m | 1.68 m | |||||
| 6 | 90.6, CH | 4.12 d (6.0) | 70.1, CH | 3.68 m | 79.0, CH | 4.57 d (8.4) | 79.1, CH | 4.58 d (7.2) |
| 7 | 75.9, C | 75.5, C | 76.6, C | 76.5, C | ||||
| 8 | 45.0, CH2 | 1.82 m | 79.0, CH | 3.62 m | 46.0, CH2 | 1.85 m | 45.9, CH2 | 1.83 m |
| 9 | 78.5, CH | 4.18 dd | 69.9, CH | 4.39 t (8.0) | 79.4, CH | 4.53 m | 78.8, CH | 4.40 m |
| (14.8, 7.2) | ||||||||
| 10 | 53.9, CH | 2.95 t (7.2) | 50.0, CH | 2.51 br s | 51.0, CH | 2.86 m | 51.4, CH | 2.93 dd |
| (7.2, 6.0) | ||||||||
| 11 | 147.6, C | 145.3, C | 148.0, C | 143.0, C | ||||
| 12 | 31.5, CH2 | 2.04 m | 71.6, CH | 5.29 m | 70.7, CH | 4.37 s | 72.5, CH | 5.44 dd |
| 2.25 m | (5.6, 2.8) | |||||||
| 13 | 24.6, CH2 | 1.00 m | 31.9, CH2 | 1.58 m | 31.2, CH2 | 1.46 m | 29.0, CH2 | 1.42 m |
| 1.70 m | 2.17 m | 1.84 m | 1.90 m | |||||
| 14 | 44.0, CH | 1.26 m | 40.1, CH | 1.48 m | 36.5, CH | 1.82 m | 37.2, CH | 1.70 m |
| 15 | 23.1, CH3 | 1.41 s | 26.3, CH3 | 1.56 s | 23.2, CH3 | 1.48 s | 23.2, CH3 | 1.47 s |
| 16 | 23.6, CH3 | 1.13 s | 13.7, CH3 | 1.19 s | 22.6, CH3 | 1.17 s | 22.8, CH3 | 1.18 s |
| 17 | 109.4, CH2 | 4.64 s | 105.0, CH2 e | 5.04 s | 111.8, CH2 | 4.87 s | 114.9, CH2 | 4.97 s |
| 4.68 s | 5.23 s | 5.06 s | 5.14 s | |||||
| 18 | 29.1, CH | 1.72 m | 28.3, CH | 2.03 m | 29.0, CH | 1.80 m | 28.9, CH | 1.80 m |
| 19 | 15.6, CH3 | 0.79 d (6.8) | 21.5, CH3 | 1.06 d (6.5) | 16.4, CH3 | 0.85 d (6.4) | 16.2, CH3 | 0.83 d (6.8) |
| 20 | 21.9, CH3 | 0.97 d (6.8) | 21.5, CH3 | 1.09 d (6.5) | 21.9, CH3 | 1.00 d (6.4) | 21.8, CH3 | 0.96 d (6.8) |
| 3-n-butyrate | 172.6, C | |||||||
| 37.5, CH2 | 2.22 m | |||||||
| 2.30 m | ||||||||
| 18.6, CH2 | 1.64 m | |||||||
| 13.8, CH3 | 0.95 t (7.5) | |||||||
| 3-OAc | 169.6, C | 169.5, C | 169.4, C | |||||
| 22.4, CH3 | 2.12 s | 22.4, CH3 | 2.07 s | 22.4, CH3 | 2.07 s | |||
| 6-OMe | 57.0, CH3 | 3.34 s | ||||||
| 6-OAc | ||||||||
| 8-OAc | ||||||||
| 12-OAc | 170.0, C | 170.3, C | ||||||
| 21.2, CH3 | 2.14 s | 21.5, CH3 | 2.06 s | |||||
| Compound | Elastase | ||
|---|---|---|---|
| Inh% | IC50 (μM) | ||
| 1 | 19.42 ± 7.89 | * | >10 |
| 2 | 45.51 ± 2.69 | *** | >10 |
| 3 | 18.67 ± 5.75 | * | >10 |
| 4 | 27.30 ± 5.42 | ** | >10 |
| 5 | −0.32 ± 0.81 | >10 | |
| 6 | 6.15 ± 3.42 | >10 | |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Separation
3.3.1. Krempfielin J (1)
3.3.2. Krempfielin K (2)
3.3.3. Krempfielin L (3)
3.3.4. Krempfielin M (4)
3.4. In Vitro Anti-Inflammatory Assay—Superoxide Anion Generation and Elastase Release by Human Neutrophils
4. Conclusions
Acknowledgements
References
- Ahmed, A.F.; Wu, M.-H.; Wang, G.-H.; Wu, Y.-C.; Sheu, J.-H. Eunicellin-based diterpenoids, australins A–D, isolated from the soft coral Cladiella australis. J. Nat. Prod. 2005, 68, 1051–1055. [Google Scholar] [CrossRef]
- Wu, S.-L.; Su, J.-H.; Wen, Z.-H.; Hsu, C.-H.; Chen, B.-W.; Dai, C.-F.; Kuo, Y.-H.; Sheu, J.-H. Simplexins A–I, eunicellin-based diterpenoids from the soft coral Klyxum simplex. J. Nat. Prod. 2009, 72, 994–1000. [Google Scholar] [CrossRef]
- Chen, B.-W.; Wu, Y.-C.; Chiang, M.Y.; Su, J.-H.; Wang, W.-H.; Fan, T.-Y.; Sheu, J.-H. Eunicellin-based diterpenoids from the cultured soft coral Klyxum simplex. Tetrahedron 2009, 65, 7016–7022. [Google Scholar]
- Chen, Y.-H.; Tai, C.-Y.; Hwang, T.-L.; Weng, C.-F.; Li, J.-J.; Fang, L.-S.; Wang, W.-H.; Wu, Y.-C.; Sung, P.-J. Cladielloides A and B: New eunicellin-type diterpenoids from an Indonesian octocoral Cladiella sp. Mar. Drugs 2010, 8, 2936–2945. [Google Scholar] [CrossRef]
- Chen, B.-W.; Chang, S.-M.; Huang, C.-Y.; Chao, C.-H.; Su, J.-H.; Wen, Z.-H.; Hsu, C.-H.; Dai, C.-F.; Wu, Y.-C.; Sheu, J.-H. Hirsutalins A–H, eunicellin-based diterpenoids from the soft coral Cladiella hirsuta. J. Nat. Prod. 2010, 73, 1785–1791. [Google Scholar] [CrossRef]
- Hassan, H.M.; Khanfar, M.A.; Elnagar, A.Y.; Mohammed, R.; Shaala, L.A.; Youssef, D.T.A.; Hifnawy, M.S.; El Sayed, K.A. Pachycladins A–E, prostate cancer invasion and migration inhibitory eunicellin-based diterpenoids from the Red Sea soft coral Cladiella pachyclados. J. Nat. Prod. 2010, 73, 848–853. [Google Scholar] [CrossRef]
- Chen, B.-W.; Chao, C.-H.; Su, J.-H.; Wen, Z.-H.; Sung, P.-J.; Sheu, J.-H. Anti-inflammatory eunicellin-based diterpenoids from the cultured soft coral Klyxum simplex. Org. Biomol. Chem. 2010, 8, 2363–2366. [Google Scholar] [CrossRef]
- Wu, S.-L.; Su, J.-H.; Lu, Y.; Chen, B.-W.; Huang, C.-Y.; Wen, Z.-H.; Kuo, Y.-H.; Sheu, J.-H. Simplexins J–O, eunicellin-based diterpenoids from a Dongsha Atoll soft coral Klyxum simplex. Bull. Chem. Soc. Jpn. 2011, 84, 626–632. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Tai, C.-Y.; Su, Y.-D.; Chang, Y.-C.; Lu, M.-C.; Weng, C.-F.; Su, J.-H.; Hwang, T.-L.; Wu, Y.-C.; Sung, P.-J. Discovery of new eunicellins from an Indonesian octocoral Cladiella sp. Mar. Drugs 2011, 9, 934–943. [Google Scholar] [CrossRef]
- Hsu, F.-J.; Chen, B.-W.; Wen, Z.-H.; Huang, C.-Y.; Dai, C.-F.; Su, J.-H.; Wu, Y.-C.; Sheu, J.-H. Klymollins A–H, bioactive eunicellin-based diterpenoids from the Formosan soft coral Klyxum molle. J. Nat. Prod. 2011, 74, 2467–2471. [Google Scholar] [CrossRef]
- Chen, B.-W.; Huang, C.-Y.; Wen, Z.-H.; Su, J.-H.; Wang, W.-H.; Sung, P.-J.; Wu, Y.-C.; Sheu, J.-H. Klysimplexins U–X, eunicellin-based diterpenoids from the cultured soft coral Klyxum simplex. Bull. Chem. Soc. Jpn. 2011, 84, 1237–1242. [Google Scholar] [CrossRef]
- Chen, B.-W.; Chao, C.-H.; Su, J.-H.; Tsai, C.-W.; Wang, W.-H.; Wen, Z.-H.; Huang, C.-Y.; Ung, P.-J.; Wu, Y.-C.; Sheu, J.-H. Klysimplexins I–T, eunicellin-based diterpenoids from the cultured soft coral Klyxum simplex. Org. Biomol. Chem. 2011, 9, 834–844. [Google Scholar] [CrossRef]
- Wu, S.-L.; Su, J.-H.; Huang, C.-Y.; Tai, C.-J.; Sung, P.-J.; Liaw, C.-C.; Sheu, J.-H. Simplexins P–S, eunicellin-based diterpenes from the soft coral Klyxum simplex. Mar. Drugs 2012, 10, 1203–1211. [Google Scholar] [CrossRef]
- Williams, D.E.; Amlani, A.; Dewi, A.S.; Patrict, B.O.; van Ofwegen, L.; Mui, A.L.-F.; Andersen, R.J. Australin E isolated from the soft coral Cladiella sp. collected in pohnpei activates the inositol 5-phosphatase SHIP1. Aust. J. Chem. 2010, 63, 895–900. [Google Scholar] [CrossRef]
- Ospina, C.A.; Rodríguez, A.D. Bioactive compounds from the gorgonian Briareum polyanthes. Correction of the structures of four asbestinane-type diterpenes. J. Nat. Prod. 2006, 69, 1721–1727. [Google Scholar] [CrossRef]
- Sarma, N.S.; Chavakula, R.; Rao, I.N.; Kadirvelraj, R.; Row, T.N.G.; Saito, I. Crystal and molecular structure of sclerophytin F methyl ether from the soft coral Cladiella krempfi. J. Nat. Prod. 1993, 56, 1977–1980. [Google Scholar] [CrossRef]
- Cai, Y.-S.; Yao, L.-G.; Pascale, A.D.; Irace, C.; Mollo, E.; Taglialatela-Scafati, O.; Guo, Y.-W. Polyoxygenated diterpenoids of the eunicellin-type from the Chinese soft coral Cladiella krempfi. Tetrahedron 2013, 69, 2214–2219. [Google Scholar] [CrossRef]
- Lan, W.-J.; Lin, C.-W.; Su, J.-Y.; Zeng, L.-M. Two steroidal glycosides from the soft coral Cladiella krempfi. Chem. J. Chin. Univ. 2003, 24, 2019–2021. [Google Scholar]
- Huang, X.-P.; Deng, Z.-W.; Ofwegen, L.V.; Li, J.; Fu, H.-Z.; Zhu, X.-B.; Lin, W.-H. Two new pregnane glycosides from soft coral Cladiella krempfi. J. Asian Nat. Prod. Res. 2006, 8, 287–291. [Google Scholar] [CrossRef]
- Huang, X.-P.; Deng, Z.-W.; Zhu, X.-B.; Ofwegen, L.V.; Proksch, P.; Lin, W.-H. Krempenes A–D: A series of unprecedented pregnane-type steroids from the marine soft coral Cladiella krempfi. Helv. Chim. Acta 2006, 89, 2020–2026. [Google Scholar] [CrossRef]
- Tai, C.-J.; Su, J.-H.; Huang, M.-S.; Wen, Z.-H.; Dai, C.-F.; Sheu, J.-H. Bioactive eunicellin-based diterpenoids from the soft coral Cladiella krempfi. Mar. Drugs 2011, 9, 2036–2045. [Google Scholar] [CrossRef]
- Tai, C.-J.; Su, J.-H.; Huang, C.-Y.; Huang, M.-S.; Wen, Z.-H.; Dai, C.-F.; Sheu, J.-H. Cytotoxic and anti-inflammatory eunicellin-based diterpenoids from the soft coral Cladiella krempfi. Mar. Drugs 2013, 11, 788–799. [Google Scholar] [CrossRef]
- Alam, M.; Sharma, P.; Zektzer, A.S.; Martin, G.E.; Ji, X.; Helm, D. Sclerophytin C–F: Isolation and structures of four new diterpenes from the soft coral Sclerophytum capitalis. J. Org. Chem. 1989, 54, 1896–1900. [Google Scholar] [CrossRef]
- Miyamoto, T.; Yamada, K.; Ikeda, N.; Komori, T.; Higuchi, R. Bioactive terpenoids from octocorallia, I. Bioactive diterpenoids: Litophynols A and B from the mucus of the soft coral Litophyton sp. J. Nat. Prod. 1994, 57, 1212–1219. [Google Scholar] [CrossRef]
- Iwagawa, T.; Kusatsu, T.; Tsuha, K.; Hamada, T.; Okamura, H.; Furukawa, T.; Akiyama, S.; Doe, M.; Morimoto, Y.; Iwase, F.; Takemura, K. Cytotoxic eunicellin-type diterpenes from the soft coral Litophyton viscudium. Heterocycles 2011, 83, 2149–2155. [Google Scholar] [CrossRef]
- Roussis, V.; Fenical, W.; Vagias, C.; Kornprobst, J.-M.; Miralles, J. Labiatamides A, B, and other eunicellan diterpenoids from the Senegalese gorgonian Eunicella labiata. Tetrahedron 1996, 52, 2735–2742. [Google Scholar] [CrossRef]
- Rao, C.B.; Rao, D.S.; Satyanarayana, C.; Rao, D.V.; Kassühlke, K.E.; Faulkner, D.J. New cladiellane diterpenes from the soft coral Cladiella australis of the Andaman and Nicobar Islands. J. Nat. Prod. 1994, 57, 574–580. [Google Scholar] [CrossRef]
- Yu, H.-P.; Hsieh, P.-W.; Chang, Y.-J.; Chung, P.-J.; Kuo, L.-M.; Hwang, T.-L. 2-(2-Fluorobenzamido) benzoate ethyl ester (EFB-1) inhibits superoxide production by human neutrophils and attenuates hemorrhagic shock-induced organ dysfunction in rats. Free Radic. Biol. Med. 2011, 50, 1737–1748. [Google Scholar] [CrossRef]
- Hwang, T.-L.; Wang, C.-C.; Kuo, Y.-H.; Huang, H.-C.; Wu, Y.-C.; Kuo, L.-M.; Wu, Y.-H. The hederagenin saponin SMG-1 is a natural FMLP receptor inhibitor that suppresses human neutrophil activation. Biochem. Pharmacol. 2010, 80, 1190–1200. [Google Scholar] [CrossRef]
- Hwang, T.-L.; Leu, Y.-L.; Kao, S.-H.; Tang, M.-C.; Chang, H.-L. Viscolin, a new chalcone from Viscum coloratum, inhibits human neutrophil superoxide anion and elastase release via a cAMP-dependent pathway. Free Radic. Biol. Med. 2006, 41, 1433–1441. [Google Scholar] [CrossRef]
- Samples Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, Y.-N.; Tai, C.-J.; Hwang, T.-L.; Sheu, J.-H. Krempfielins J-M, New Eunicellin-Based Diterpenoids from the Soft Coral Cladiella krempfi. Mar. Drugs 2013, 11, 2741-2750. https://doi.org/10.3390/md11082741
Lee Y-N, Tai C-J, Hwang T-L, Sheu J-H. Krempfielins J-M, New Eunicellin-Based Diterpenoids from the Soft Coral Cladiella krempfi. Marine Drugs. 2013; 11(8):2741-2750. https://doi.org/10.3390/md11082741
Chicago/Turabian StyleLee, Yan-Ning, Chi-Jen Tai, Tsong-Long Hwang, and Jyh-Horng Sheu. 2013. "Krempfielins J-M, New Eunicellin-Based Diterpenoids from the Soft Coral Cladiella krempfi" Marine Drugs 11, no. 8: 2741-2750. https://doi.org/10.3390/md11082741
APA StyleLee, Y.-N., Tai, C.-J., Hwang, T.-L., & Sheu, J.-H. (2013). Krempfielins J-M, New Eunicellin-Based Diterpenoids from the Soft Coral Cladiella krempfi. Marine Drugs, 11(8), 2741-2750. https://doi.org/10.3390/md11082741





